Research Article, Vector Biol J Vol: 2 Issue: 2
Effect of Aqueous Extract of Protium heptaphyllum (Burseraceae) on Lutzomyia longipalpis (Diptera: Psychodidae), a Proven Vector of Visceral Leishmaniasis in Brazil
Yrllan Ribeiro Sincurá1, Amanda Oliveira Baracho1, Cristiane Fernanda Fuzer Grael2, Emerson Cotta Bodevan3 and Ricardo Andrade Barata1*
1Laboratório de Parasitologia, Departamento de Ciências Biológicas,Universidade Federal dos Vales dos Jequitinhonha e Mucuri, Diamantina, MG, Brazil
2Laboratório de Farmacognosia, departamento de Farmácia, Universidade Federal dos Vales dos Jequitinhonha e Mucuri, Diamantina, MG, Brazil
3Laboratório de Matemática, Departamento de Matemática e Estatística, Universidade Federal dos Vales dos Jequitinhonha e Mucuri, Diamantina, Minas Gerais, Brazil
*Corresponding Author : Ricardo Andrade Barata
Universidade Federal dos Vales do Jequitinhonha e Mucuri, Laboratório de Parasitologia, Departamento de Ciências Biológicas, Campus JK, Rodovia MGT-367, Km 583, 5000, Alto da Jacuba, 39100-000, Diamantina, MG, Tel: 0-000 Diamantina, MG, Brazil
Tel: 55 38 3532-1236
E-mail: ricbarata@hotmail.com
Received: September 23, 2017 Accepted: October 03, 2017 Published: October 10, 2017
Citation: Sincurá YB, Baracho AO, Grael CFF, Bodevan EC, Barata RA (2017) Effect of Aqueous Extract of Protium heptaphyllum (Burseraceae) on Lutzomyia longipalpis (Diptera: Psychodidae), a Proven Vector of Visceral Leishmaniasis in Brazil. Vector Biol J 2:2. doi: 10.4172/2473-4810.1000120
Abstract
Objective: To evaluate the toxicity of aqueous extract of Protium heptaphyllum on Lutzomyia longipalpis a proven vector of visceral leishmaniasis (VL).
Methods: Aqueous extract of the leaves of P. heptaphyllum were prepared at 0.25, 0.10,0.05 and 0.025 mg/mL concentrations, and 100 μL was applied to adults of Lu. longipalpis (n=20). Controls included 20 adults that were treated with 100 μL distilled water (negative control) and 20 adults that were treated with 100 μL alpha-cypermethrin at 196 µg/mL concentration (positive control). The percentage of dead insects in each group was observed at 1, 2, 4, 8, 12, 24, 48 and 72h after application. Results: The mortality rate of Lu. longipalpis after 2h of exposure to the extract at a concentration of 0.1 mg/mL was 52%, behaving similarly to the positive controls (cypermethrin), and the extract at a concentration of 0.25 mg/ml was able to kill 56% of insects within the first hour of exposure. After 72h, the aqueous extract of P. heptaphyllum showed a mortality rate of about 81% and 83%, at concentrations of 0.1 and 0.25 mg/mL, respectively. Conclusion: The extract of P. heptaphyllum contains terpenes, a molecule with recognized toxicity in insects, and which may be responsible for killing the phlebotomines. These results demonstrate the potential use of this extract in the control of this vector
Keywords: Phlebotominae; Sand flies; Insecticidal activity; Bioinsecticide; Vector control
Introduction
Visceral leishmaniasis (VL) is an infectious parasitic disease that mainly affects hematopoietic organs such as liver, spleen and bone marrow causing hepatosplenomegaly, malnutrition, jaundice and can lead the individual to death, if not treated in time [1]. In the Americas, about 90 % of human cases have been recorded in Brazil [2], where the disease is distributed in all regions and is present in 26 of the 27 federative units [3-5].
In the Americas, the protozoan Leishmania infantum is transmitted to humans primarily through blood feeding of female Lutzomyia longipalpis, a sand fly that has shown high ecological plasticity and adaptability to various environments [6-8], and therefore, it has been incriminated as one of those vectors responsible for the urbanization phenomenon of the disease in Brazil [9,10].
The use of indoor residual insecticides has been an action used by the Ministry of Health to control VL. Currently, synthetic pyrethroids such as cypermethrin and deltamethrin are the most commonly used insecticides in Brazil. However, low efficiency in vector control was observed and some studies have pointed to resistance of Lu. longipalpis for these compounds, which increases the interest in new chemical products [11-14].
Popularly known as pitch-black, mastic, wild musk, among others, Protium hetaphillum is an evergreen tree species found in areas of forest and savannah (“cerrado”), highly very widespread in Brazil and South America, present from the Guianas to Argentina [15]. Studies show their sedative, anxiolytic, antidepressant, anti-inflammatory, antimicrobial and acaricidale activities [16-19]. Considering so many positive effects, this study aimed to investigate the insecticidal activity of the aqueous extract of P. heptaphyllum leaves on Lutzomyia longipalpis, a vector of vsceral leishmaniasis (VL) in Brazil.
Methods
Collection, identification, and preparation of plant material
In June 2015, Protium heptaphyllum leaves were collected in rock field area in the city of Diamantina (Minas Gerais, Brazil) (18° 11’S - 43°34’O). The stuff was deposited in the DIAM / UFVJM herbarium in Diamantina under no. DIAM nº 5878 and its identification made based on morphological characters [15].
Obtaining the plant extract
The collected material was subjected to drying in a forced circulation oven and air renewal to 60ºC and milled in a knife mill to obtain a fine powder of uniform granulation. To prepare the aqueous extract was added to the glass beaker, the powder obtained after grinding and distilled water at room temperature, keeping at rest for 48h.
Collection and identification of phlebotomine sand flies
The phlebotomine sand flies were collected in Diamantina / MG in the town of Aroeira (18° 88’ S - 43° 38’ W), using light traps HP [20] type exposed for two nights. The specimens were transported to the Laboratório de Parasitologia of Universidade Federal dos Vales do Jequitinhonha e Mucuri (UFVJM) and put in cages. From this material, a sample was taken for identification according to the classification proposed by Young & Duncan [21]. The sand flies remained at rest for 24h until perform the test, being offered to them sugar solution of distilled water and honey (1:1).
Biological tests on Lutzomyia longipalpis
The aqueous extract of P. heptaphyllum was applied on the filter paper in the following concentrations: 0.025 mg/mL; 0.05 mg/mL; 0.1 mg/mL; 0.25 mg/mL and introduced inside translucent plastic pots. Controls included 20 adults that were treated with 100 μL distilled water (negative control) and 20 adults that were treated with 100 μL alpha-cypermethrin at 196 μg/mL concentration (positive control). Tests, including controls, were performed in duplicate, with 15 males and 15 females in each pot. After 1h, 2h, 4h, 8h, 12h, 24h, 48h and 72h the dead sand flies were counted. The overall temperature of the laboratory was 28ºC.
Statistical analysis
The statistical test used to compare proportions between groups at each time point was the proportion of equality test (chi-square), with continuity correction or Fisher’s exact test. The software used for statistical analysis was the R (version 2015) [22], considering the 0.05 significance level.
Results
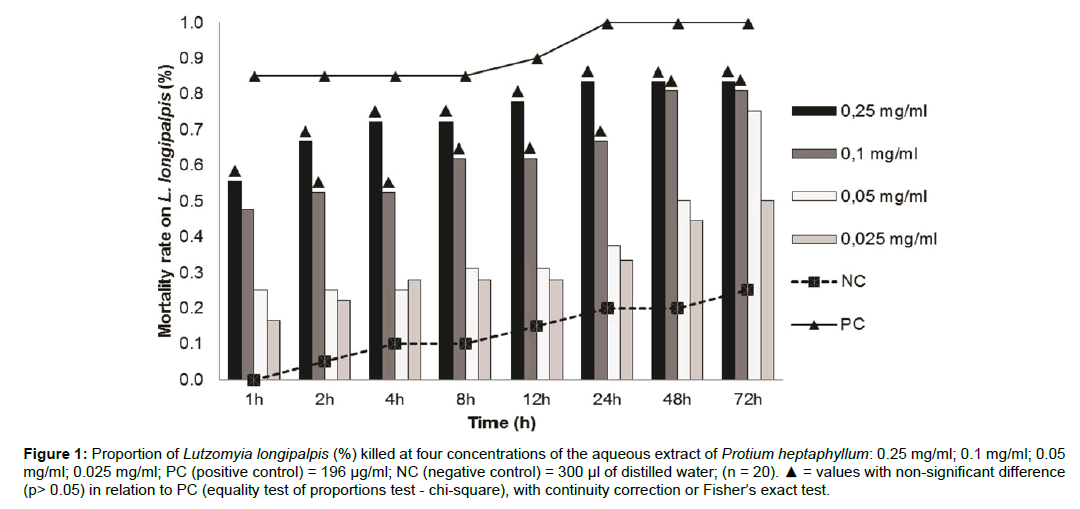
The aqueous extract of Protium heptaphyllum demonstrated toxic properties in all concentrations tested. The mortality rate of Lu. longipalpis after 2h of exposure to the extract at a concentration of 0.1 mg/mL was 52%, behaving similarly to the positive controls (cypermethrin), and the extract at a concentration of 0.25mg / ml was able to kill 56% of insects within the first hour of exposure. The aqueous extract of P. heptaphyllum showed a mortality rate of about 81% and 83% after 72 h, at concentrations of 0.1 and 0.25 mg/mL, respectively (Figure 1).
Figure 1: Proportion of Lutzomyia longipalpis (%) killed at four concentrations of the aqueous extract of Protium heptaphyllum: 0.25 mg/ml; 0.1 mg/ml; 0.05 mg/ml; 0.025 mg/ml; PC (positive control) = 196 μg/ml; NC (negative control) = 300 μl of distilled water; (n = 20). ▲ = values with non-significant difference (p> 0.05) in relation to PC (equality test of proportions test - chi-square), with continuity correction or Fisher’s exact test.
Discussion
The use of pyrethoids insecticides, mainly deltamethrin and alphacypermethrin, to eliminate phlebotomines constitutes a strategy of visceral leishmaniasis control. Deltamethrin has shown low efficacy on Lu. longipalpis [23-25]. Similarly, the alpha- cypermethrin had low mortality rates in the wall, which led the authors to assume the possibility of resistance of Lu. longipalpis strain used or even the low efficacy of this insecticide [26].
Studies of plants with insecticidal action effects on Lu. longipalpis have been performed [14]. It showed the capacity of the neem seed oil (Azadirachta indica) on the development of Lu. longipalpis. In the same way, Antonia ovata and Derris amazonica displayed significant insecticide effect against Lu. longipalpis [27]. At the same time, studies have demonstrated the effects of plant extracts on other organisms. Tests with P. heptaphyllum on Candida krusei, Cryptococcus neoformans, Staphylococcus aureus and Tetranychus urticae demonstrate a fungicidal, bactericidal, acaricidal action, antiinflammatory and healing activity [12,16-19,28,29-32]. In these works, the action effect found was associated with the presence of terpenes, components that are found in plants, used in defense against natural enemies [5]. Terpenes have been identified in large numbers in leaves of P. heptaphyllum, with β- caryophyllene being the most abundant component [33,34]. In view of the above, the presence of terpenes in extracts of P. heptaphyllum leaves, a molecule with recognized toxicity in insects, has been shown to be responsible for killing the phlebotomines and that the significant mortality rates observed in the present study show that P. heptaphyllum merits further study as to its potential use as an bioinsecticide.
References
- Brasil (2014) Manual de Vigilância e Controle da Leishmaniose Visceral (Ministério da Saúde). Ministério da Saúde, Brasília.
- Grimaldi G, Tesh RB (1993) Leishmaniases of the New World: current concepts and implications for future research. Clin Microbiol Rev 6: 230-250.
- Gontijo CMF, Melo MN (2004) Leishmaniose visceral no Brasil: quadro atual, desafios e perspectivas. Rev Bras Epidemiol 7: 338-349
- Dantas-Torres F, Brandão-Filho SP (2006) Expansão geográfica da leishmaniose visceral no Estado de Pernambuco. Rev Soc Bras Med Trop 39: 352-356.
- Cavalcante GM, Moreira AFC, Vasconcelos SD (2006) Potencialidade inseticida de extratos aquosos de essências florestais sobre mosca-branca. Pesq Agropec Bras 41: 9-14.
- Jeronimo SMB, Oliveira RM, Mackay S, Costa RM, Sweet J, et al. (1994) An urban outbreak of visceral leishmaniasis in Natal, Brazil. Trans R Soc Trop Med Hyg 88: 386-388
- Ximenes MFFM, Silva VPME, Queiroz PVS, Rego MM, Cortez AM, et al. (2007) Flebotomíneos (Diptera: Psychodidae) e leishmanioses no Rio Grande do Norte, Nordeste do Brasil: reflexos do ambiente antrópico. Neotrop Entomol 36: 128-137.
- Rangel EF, Vilela ML (2008) Lutzomyia longipalpis (Diptera, Psychodidae, Phlebotominae) and urbanization of visceral leishmaniasis in Brazil. Cad Saúde Pública 24: 2948-2952.
- Costa CHN, Pereira HF, Araújo MV (1990) Epidemia de leishmaniose visceral no nordeste do Piauí, Brasil, 1980-1986. Rev Saúde Pública 24: 361-372.
- Lainson R, Rangel EF (2005) Lutzomyia longipalpis and the eco-epidemiology of American visceral leishmaniasis, with particular reference to Brazil - a review. Mem Inst Oswaldo Cruz 100: 881-827.
- Mazzarri MB, Feliciangeli MD, Maroli M, Hernandez A, Bravo A (1997) Susceptibility of Lutzomyia longipalpis (Diptera: Psychodidae) to selected insecticides in an endemic focus of visceral leishmaniasis in Venezuela. J Am Mosq Control Assoc 13: 335-341.
- Viegas-Júnior C (2003) Terpenos com atividade inseticida: uma alternativa para o controle químico de insetos. Quim. Nova 26: 390-400.
- Amóra SS, Bevilaqua CML, Feijó FMC, Alves ND, Maciel M (2009) Control of phlebotomine (Diptera: Psychodidae) leishmaniasis vectors. Neotrop Entomol 38: 303-310.
- Maciel MV, Morais SM, Bevilaqua CML, Silva RA, Barros RS, et al. (2010) Atividade inseticida in vitro do óleo de sementes de nim sobre Lutzomyia longipalpis (Diptera: Psychodidae). Rev Bras Parasitol Vet 19: 7-11.
- Domene VD, Mattos PP, Salis SM (2010) Fenologia e crescimento de almécega no Pantanal da Nhecolândia, Mato Grosso do Sul. Comun. Técnico Embrapa Florestas 263: 5.
- Júnior GMV, Souza CML, Chaves MH (2005) Resina deProtium heptaphyllum: isolamento, caracterização estrutural e avaliação das propriedades térmicas. Quim Nova 28: 183-187.
- Aragão GF, Carneiro LM V, Junior PF, Vieira LC, Bandeira PN, et al. (2006) A possible mechanism for anxiolytic and antidepressant effects of alpha- and beta- amyrin from Protium heptaphyllum (Aubl.) March. Pharmacol Biochem Behav 85: 827-834.
- Holanda-Pinto SA, Pinto LMS, Cunha GMA, Chaves MH, Santos FA, et al. (2007) Anti-inflammatory effect of alpha, beta-amyrin, a pentacyclic triterpene fromProtium heptaphylluminrat model of acute periodontitis. Inflammo Pharmacol 15: 1-5
- Oliveira FA, Vieira-Junior GM, Chaves MH, Almeida FRC, Florencio MG, et al. (2004) Gastroprotective and anti-inflmmatory effects of resin from Protium heptaphyllum in mice and rats. Pharmacol Res 49: 105-111
- Pugedo H, Barata RA, França-Silva JC, Silva JC, Dias ES (2005) HP : um modelo aprimorado de armadilha luminosa de sucção para a captura de pequenos insetos. Rev Soc Bras Med Trop 38: 70-72.
- Young DG, Duncan MA (1994) Guide to the identification and geographic distribution of Lutzomyia sand flies in Mexico, the West Indies, central and south america (Diptera: Psychodidae). Mem Am Entomol Inst 54: 1-881.
- Team RC (2015) R: A Language and Environment for Statistical Computing.
- Seyedi-Rashi MA, Yezdan PH, Shah H, Jeradi M (1975) Susceptibility of Phlebotomus papatasi (Diptera: Psychodidae) to DDT in some foci of cutaneous leishmaniasis in Iran. J Am Mosq Control Assoc 8: 99-100
- Rahman SJ, Wattal BL, Mathur KK, Joshi GC, Kumar K (1982) Susceptibility of laboratory reared strain of Phlebotomus papatasi (Scopoli) to organochlorine insecticides. J Commun Dis 14: 122-124.
- Alexander B, Usma MC, Cadena H, Quesada BL, Solarte Y, et al. (1995) Phlebotomine sandflies associated with a focus of cutaneous leishmaniasis in Valle del Cauca. Colombia Med. Vet Entomol 9: 273-278.
- Nery-Guimarães F, Bustamante FM (1953) A aplicação domiciliária de DDT como base da profilaxia das leishmanioses. Estudo de um foco de leishmaniose muco-cutânea cinco anos depois da aspersão periódica com aquele inseticida. Rev Bras Malariol Doenças Trop 6: 127-130.
- Luitgards-Moura JF, Bermudez EGC, Rocha AFI, Tsouris P, Rosa-Freitas MG (2002) Preliminary assays indicate that Antonia ovata (Loganiaceae) and Derris amazonica (Papilionaceae), ichthyotoxic plants used for fishing in Roraima, Brazil, have an insecticide effect on Lutzomyia longipalpis (Diptera: Psychodidae: Phlebotominae). Mem Inst Oswaldo Cruz 97: 737-742
- Vinaud MC, Souza R, Junior L (2008) Activity of Stryphnodendron polyphyllum, a plant from the Brazilian savannah, against hemocytes of Biomphalaria glabrata, an intermediate host of Schistosoma mansoni. Rev Patol Trop 37: 237-246.
- Siani AC, Ramos MF, Menezes-de-Lima O, Ribeiro-dos-Santos R, Fernadez- Ferreira E, et al.(1999) Evaluation of anti-inflammatory-related activity of essential oils from the leaves and resin of species of Protium. J Ethnopharmacol 66: 57-69.
- Susunaga GS, Siani AC, Pizzolatti MG, Yunes RA, DelleMonache F (2001) Triterpenes from the resin of Protium heptaphyllum. Fitoterapia 72: 709-711.
- Pohlit AM, Quinard ELJ, Nunomura SM, Tadei WP, Hidalgo ADF, et al. (2004) Screening of plants found in the State of Amazonas, Brazil for activity against Aedes aegypti larvae. Acta Amaz 34: 97-105.
- Lima-Júnior RC, Oliveira FA, Gurgel LA, Cavalcante IJ, Santos KA, et al. (2006) Attenuation of visceral nociception by α- and β-amyrin, a triterpenoid mixture isolated from the resin of Protium heptaphyllum, in mice. Planta Med 72: 34-39.
- Citó A, Costa F, Lopes J (2006) Identificação dos constituintes voláteis de frutos e folhas de Protium heptaphyllum Aubl (March). Rev Bras PI Med 8: 4-7.
- Bandeira PN, Pessoa ODL, Trevisan MTS, Lemos TLG (2002) Metabólitos secundários de Protium heptaphyllum March. Quim Nova 25: 1078-1080.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi