Research Article, Clin Dermatol Res J Vol: 2 Issue: 1
Efficacy of a Topical Lotion Containing Lactoferricin, Glycerophosphoinositol Lysine and Verbascoside for External Otitis Due to Atopic Dermatitis
Lisa Accomasso1*, Immacolata Miano2, Tiziana Cocca2, Mauro Bigliati1, Silvia Arpicco3 and Alessandro Barge3
1Candioli Farmaceutici SpA, Via Manzoni 2, Beinasco, (TO) Italy
2Clinica Veterinaria Napolivet, Napoli, (NA) Italy
3Dipartimento di Scienza e Tecnologia del Farmaco, University of Turin, Italy
*Corresponding Author : Lisa Accomasso
Candioli Farmaceutici SpA., Via Manzoni 2, 10092-Beinasco, (TO) Italy
E-mail: lisa.accomasso@candioli.it
Received: June 06, 2017 Accepted: July 11, 2017 Published: July 17, 2017
Citation: Accomasso L, Miano I, Cocca T, Bigliati M, Arpicco S, et al. (2017) Efficacy of a Topical Lotion Containing Lactoferricin, Glycerophosphoinositol Lysine and Verbascoside for External Otitis Due to Atopic Dermatitis. Clin Dermatol Res J 2:1.
Abstract
Importance: Atopic dermatitis (AD) is a common skin disease in dogs with a great impact in quality of life, especially when ears are affected because of the difficult application of lotion due to the lesions localization.
Objective: This study is aimed to examine the effect of topical lotion containing lactoferricin, glycerophosphoinositol lysine and verbascoside on canine atopic dermatitis. The topical lotion must be easy to use in ears of dogs under treatment.
Methods: 21 dogs with external otitis due to atopic dermatitis were treated for 14 days and monitored from day 0 to day 14 to ascertain the effect of the treatment with different parameters such as CADESI score, lichenification, excoriation, alopecia, pruritus and cytological analysis.
Results: The lotion here prepared showed the correct density and viscosity to be used in the dogs’ ears. Thanks to the easy application, all the 21 dogs were treated for the entire period without any complain. Moreover, in all treated dogs a clear decreasing in the clinical symptoms was observed. The cytological analysis also confirms the decrease of bacteria and yeast number.
Conclusion: This study demonstrates the efficacy of a treatment based on the administration of a lotion containing the active ingredients over described in reducing clinical symptoms of canine atopic dermatitis in dogs ears.
Keywords: Atopic dermatitis; Ears; Lactoferricin; Glycerophosphoinositol lysine; Verbascoside
Introduction
The prevalence of the atopic dermatitis (AD) in the general dog population is 3-15% it represents the diagnosis for up to 58% of dogs affected with skin disease [1,2]. Canine AD has been defined by Halliwell in 2006, as a “genetically predisposed inflammatory and pruritic allergic skin disease with characteristic clinical features, associated with IgE antibodies most commonly directed against environmental allergens” [3]. Indeed, genetic factors and familiar predisposition to the disease have been found to play a major role in its pathogenesis, although the full pathogenesis is still unknown. It is certain that the immunological aberration is usually associated with skin barrier dysfunctions [4]; pro-inflammatory cytokines, neuronal itch stimuli and the animal’s pruritic behaviors establish a vicious cycle of itch that perpetuates and potentially exacerbates the skin lesions and defects in the skin barrier function [5]. The initial clinical feature of canine AD is pruritus, which at the beginning may be associated with no lesion or with primary skin lesions such as erythema and occasionally papules [6,7]. The mediators that elicit the sensation of pruritus have not been elucidated, but histamine does not appear to be a mediator in dogs in contrast to humans and mice [8]. It is usually a life-long pathology which can be controlled, but it can be seldom cured. The diagnosis of canine AD does not require any analytical test, such as IgE determination and/or intradermal skin test, and it can be done on the basis of pruritus associated with skin lesions [5,9]. The skin lesions are usually associated with the detachment of corneocytes from live skin. Furthermore, the skin lesions are usually associated with the cytological presence and overgrowth of bacteria (mainly Staphylococcus) [10,11] and fungal or yeast (mainly Malassezia Pachydermatis) [11] in the damaged region, and it has been purpose that this overgrowth could be also responsible of most of symptoms. Due to the skin infection, canine AD often develops otitis externa and usually the signs of atopic otitis were noticed by the owners before the other signs of AD [9].
The treatment of the disease involves, among others, the administration of systemic anti-pruritic drugs such as glucocorticosteroids or cyclosporin, specific immunotherapy and topical treatment improving skin condition of the disease. Antibiotic local treatment could represent an effective route to reduce the most relevant symptoms and to limit the skin infection. The progress of treatment and the severity of symptoms can be monitored by assessing the intensity of pruritus [12], the intensity of skin changes with the “Canine Atopic Dermatitis Extent and Severity Index” (CADESI) method [13] and the cytological analysis. The topical treatment is long-lasting and atopic patients could receive medical care throughout their lives. For this reason, monitoring the progress of treatment is therefore crucial in therapy.
Among all antibiotic agents, lactoferricin is a natural antimicrobial peptide which shows a wide range of activity against bacterial, fungal, viral, and parasitic pathogens [14,15]. It was discovered by a Japanese dairy company in 1992 which observed that bovin lactoferrin is cleaved by pepsin giving antimicrobial peptide [16]. Later, its structure has been elucidated: lactoferricin is a breakdown product in the gut of humans, which includes residues 17-41 of lactoferrin [17]. This is a very basic peptide which contains two tryptophan units and a disulfide bridge. The two tryptophan residues are very important structural motifs which confer the antimicrobial activity to the whole peptide [18,19], whereas the disulfide bridge is not essential. In vitro tests on lactoferrin and lactoferricin have demonstrated that the antimicrobial activity of the last is much higher than the first [16]. The different behavior could be explained in term of different structure, as determined by Hwang and coworkers in 1998 using NMR spectroscopy [20]. When the peptide is released from protein, its structure is converted from helix to beta turn structure, which is probably a more efficient structure to bind bacterial membranes. Moreover, the lactoferricin structure seems to be able to bind bacterial general domains [20], acting against a wide variety of bacteria (both Gram- positive and negative) [15-19]. An important role is played, of course, by the basic feature of this peptide, indeed, the positive charge on its surface can electrostatically interact with lipopolysaccharide (LPS) contained in the outer membrane of Gram-negative bacteria and with teichoic acid layer present on the membrane of Gram-positive bacteria [19,21]. Lactoferricin is therefore a promising candidate as antimicrobial agent in local treatment of canine dermatitis.
Glycerophosphoinositol lysine (GPI) is a sunflower derived active ingredient with anti-inflammatory effects [22]. It has been shown that GPI can be used in cream or emollient formulation in the treatment of irritative and allergic dermatitis [23].
Verbascoside is a caffeoyl phenylethanoid glycoside has been reported to have strong anti-inflammatory, antioxidant, ironchelating and glutathione-S-transferasi-inducing activity; thanks to its properties verbascoside is able to promote skin repair and ameliorates skin inflammation [24-26].
Here, we report a study in which a combination of lactoferricin with verbascoside and GPI is used as active ingredients of a dermatological lotion for topical ear treatment of canine dermatitis.
Materials and Methods
All reagents were purchased by ACEF, except glycerophosphoinositol Lysine and verbascoside (Croda), Lactoferricin (BICT) and oleic acid (Tradeco). HDPE bottles tube and dispenser were purchased by Cosmar.
Lotion preparation
A solution of polysorbate 80 (500 mg), benzyl alcohol (800 mg), oleic acid (200 mg), Lactoferricin (27 g), Glycerophosphoinositol Lysine (1.2 g), Ethylhexylglycerin (700 mg), maltodextrin (16 mg), Tocopherol (80 mg), Syringa Vulgaris extract (verbascoside) (4 mg) and Magnesium aluminium silicate (250 mg) was added to a solution of glycerine (7 g), arginine (1.2 g), Carbopol (0.8 g) in water (160.25 g). The mixture was stirred for 40 minutes, and then it was subjected to analytical control and packed.
Stability evaluation
In order to define product life and the proper storage conditions, stability tests were performed on the final lotion formulation. The tests were conducted on three different batches, packaged in corresponding 50 mL white bottles tubes made in high-density polyethylene (HDPE). All batches were stored under routine storage conditions at room temperature of 25 ± 2°C and relative humidity of 60% ± 5%. The study duration was set at 36 months and all parameters were checked at month 0, 12, 24 and 36. Each time the following parameters were evaluated: appearance (opaque, fluid, beige gel, with a characteristic odor), pH of 10% w/w water solution, density at 20°C, viscosity at 20°C, microbial contamination.
Dogs evaluation and treatment
21 dogs affected by allergic skin disease with lesions in the auricular zone have been included in the study. They were off antibiotics, antifungal and anti-inflammatory therapies for a minimum of 2 weeks before inclusion. During the first examination veterinarians had taught to owners how to apply the topical lotion which had been applied in the ears daily (two times per day) in affected areas.
Dogs were evaluated on days 0, 7 and 14. During the visit at day 0 and 14, the examination to ascertain different parameters was done. In particular, a score between 0 and 5 was assigned to each dog for different parameters: CADESI-03, lichenification, excoriation, alopecia. For the pruritus, a validated scale was used, ranging from 0 to 10 where higher score means more severe pruritus [27,28].
For the cytological analysis, the number of detached corneocytes, bacteria and Malassezia was evaluated, assigning a score as illustrated in Table 1.
| Score 0 | Score 1 | Score 2 | Score 3 | |
|---|---|---|---|---|
| Mean number of cocci per field | 0-1 | 2-5 | 6-9 | >10 |
| Mean number of yeasts per field | 0-1 | 2-4 | 5-7 | >8 |
Table 1: Cytological score assigned to cocci and Malassezia parameters.
During the entire duration of the study, possible adverse effected were recorded.
Statistical analysis
All the data were collected by the authors and analyzed using the Wilcoxon signed-rank test with Graph Pad Prism 6. The statistical significance was defined as p<0.05.
Results and Discussion
Lotion 36-months stability evaluation
Three dermatological lotion batches were prepared by dissolving in water all active ingredients (lactoferricin, verbacoside and GPI) together with all needed bulking agents and packed in high-density polyethylene bottles equipped with dispenser suitable for ear treatment. From the batches, some samples were stored to analyze stability during the first 36 months after production and the test results were summarized in Table 2.
| Test Specifications | Acceptance criteria | Time 0 | 12 months | 24 months | 36 months |
|---|---|---|---|---|---|
| Appearance | Opaque fluid gel | Conform | Conform | Conform | Conform |
| Colour | Beige | Beige | Beige | Beige | Beige |
| Odor | Characteristic | Characteristic | Characteristic | Characteristic | Characteristic |
| pH | 5.80-6.30 | 5.82-5.84-5.90 | 5.85-5.86-5.92 | 5.87-5.89-5.96 | 5.89-5.90-5.98 |
| Density (mg/mL) | 1.018-1.026 | 1.024-1.024-1.022 | 1.024-1.024-1.022 | 1.024-1.024-1.021 | 1.024-1023-1.021 |
| Viscosity (cP) | 6.000-10.000 | 6.500-6.511-6.821 | 6.488-6.464-6.788 | 6.412-6.385-6.741 | 6.335-6.348-6.628 |
| Total bacterial count | <100 ufc/mL | <100 | <100 | <100 | <100 |
| Total yeasts and molds count (TYMC) | <10 ufc/mL | <10 | <10 | <10 | <10 |
| E. coli | Absent in 1 g | Absent | Absent | Absent | Absent |
Table 2: Results of stability tests of three lotion batches, each cell reports the parameters values of the three batches; when it is reported a single value, it means that the value is identical for all batches.
The desired density and viscosity were obtained to allow an easy application of the dermatological lotion. Appearance and organoleptic properties practically remained unvaried regarding initial values. Density and pH values did not significantly change even at expiration time. On the basis of these observations, we can conclude that all parameters changes occurred during 36 months do not exceed the acceptability threshold. The product maintains satisfactory chemical, physical and organoleptic properties for a minimum period of 36 months.
The evaluation of microbial contamination, carried out at the end of the observation period, shows that the defined microbiological requirements are respected after 36 months of storage at room temperature.
Based on what has been previously discussed, it is possible to conclude that tested dermatological lotion has good long-term conservation stability at room temperature, without any significant alterations in terms of organoleptic and physic-chemical properties.
Clinical results
Samples from different batches of tested dermatological lotion were distributed to veterinarians. Total number 21 dogs, coming from different contexts, were selected for the study and dogs’ owners signed an informed consensus form. During the study period, no additional systemic or topical treatments were allowed, as well as in the 2 weeks before the start of the study. No protocol violation or lack in the compliance were reported for the entire time of this study, without any complain from dogs’ owner regarding the usage of the lotion, highlighting the realization of the goal to obtain a product easy to be used.
All dogs were accepted and included in the present study at the first visit (day 0), during which all clinical scores were recorded and the first local ear treatment has been administered. Owners repeated the treatment twice a day for two week. An intermediate visit, at day 7, was organized with the aim to evaluate the adverse reaction and to follow the adherence to the therapy. At day 14 all dogs were re-evaluated and final clinical scores were collected. The data collected during day 0 and day 14 was: CADESI score, lichenification, excoriation, alopecia, pruritus and cytological analysis. The data were collected, organized and statistically evaluated by authors.
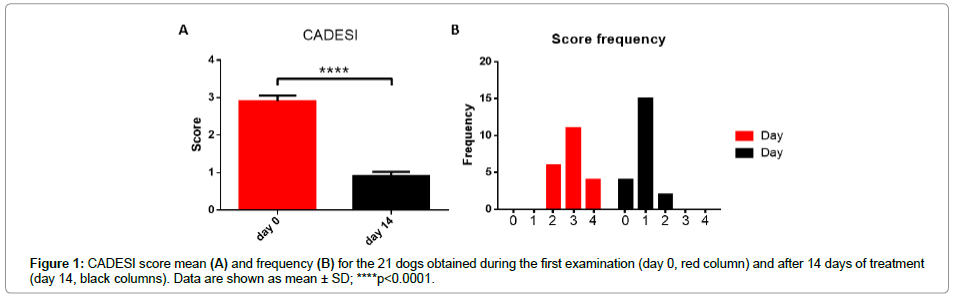
As shown in Figure 1A, the variation of the CADESI score mean obtained from the 21 dogs at day 0 and day 14 was significantly different. The score frequency (Figure 1B) underlines the different distribution of the score between the different time points. It is clearly showed as CADESI score shifts, after treatments, towards low values also for those dogs affected by a severe disease. The most frequent CADESI value passed from three to one
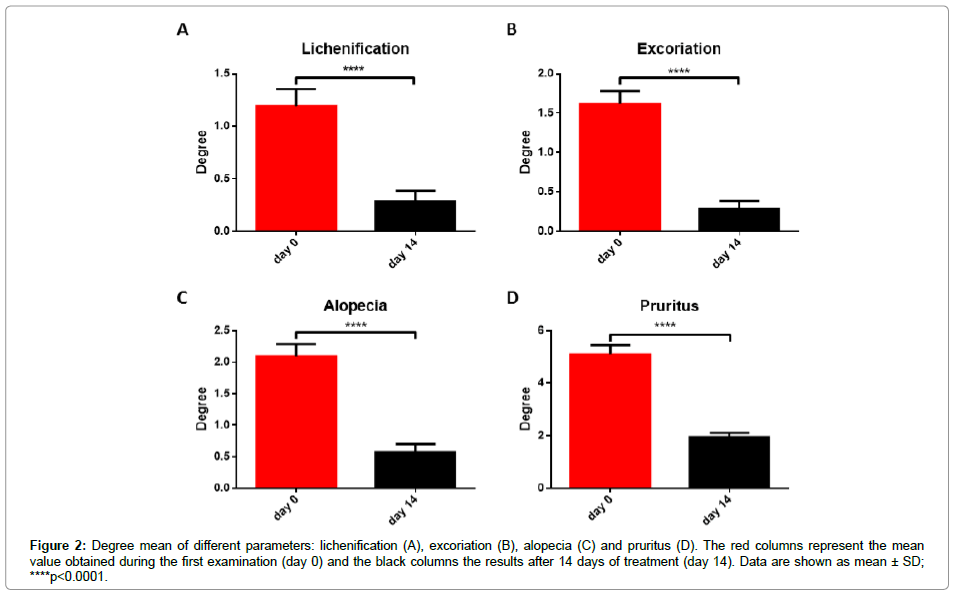
However, CADESI score is an objective parameter able to summarize the severity of the skin lesion and it cannot be used to deeply investigate the action of this formulation. The severity of dermatitis has been therefore investigated more in detail analyzing also other concurrent symptoms such as lichenification, excoriation, alopecia and pruritus. The first three parameters have been assigned a degree between 0 and 5, whereas in the case of pruritus the degree ranged from 0 to 10. As shown in Figure 2, the treatment of the damaged skin with the lotion significantly reduces the mean degree value of all the considered parameters. The reduction in CADESI score is then due to a general reduction of all specific parameters.
Figure 2: Degree mean of different parameters: lichenification (A), excoriation (B), alopecia (C) and pruritus (D). The red columns represent the mean value obtained during the first examination (day 0) and the black columns the results after 14 days of treatment (day 14). Data are shown as mean ± SD; ****p<0.0001.
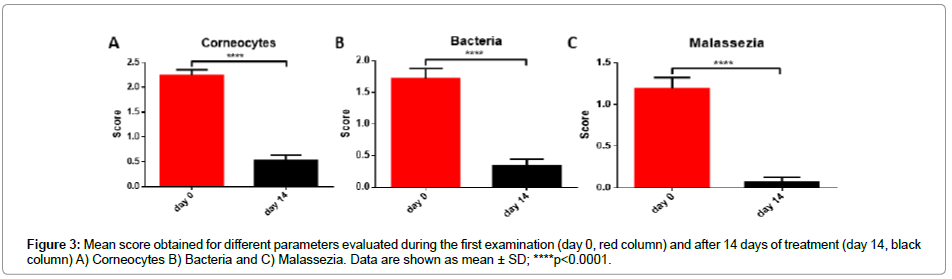
For a completely objective analysis, the progress of treatment has been also monitored by assessing the cytological analysis and to evaluate the epidermis integrity, the number of detached corneocytes has been determined to monitor the evolution of the skin lesions. The 14-days treatment with the dermatological lotion, results in a significant decrease of the corneocytes number (Figure 3A) produced by the desquamation processes.
The presence of lactoferricin, GPI and verbascoside as active ingredients of lotion formulation, could be the reason of the observed reduction also in bacteria and Malassezia number in the damaged zone; the restoring action of the skin barrier function, exerted by active ingredients, could be at the base of this effect. During the first examination, both bacteria and Malassezia were present because of the epidermal barrier defect (Figures 3B and 3C); the treatment results in a significant decrease of the score assigned, in particular after 14 days, only in one case Malassezia yeasts were still present, whereas in all other dogs they completely disappeared.
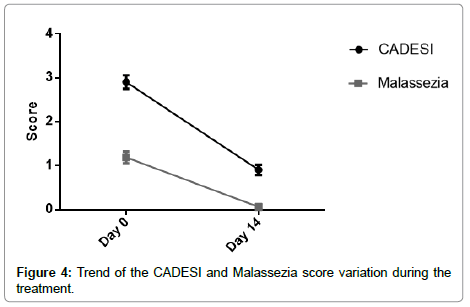
The reduction of Malassezia seems to be correlated, as expected, with the reduction of all symptoms summarized with CADESI score (Figure 4), confirming the involvement of these microorganism in the evolution of AD and the beneficial effect obtained with the active components present in this lotion formulation.
Figure 5 shows an example of the efficacy of this treatment.
Conclusion
The present study demonstrates the efficacy of a formulation based on lactoferricin, glycerophosphoinositol lysine and verbascoside in AD treatment. The three active ingredients seem to exert their favorable action through the reduction of microbial and yeast contamination of the lesion. Moreover, the rheological characteristics and the simplicity of its use, make this formulation a good solution to reduce clinical symptoms of the external otitis due to atopic dermatitis.
References
- Hillier A, Griffin CE (2001) The ACVD task force on canine atopic dermatitis (I): incidence and prevalence. Vet Immunol Immunopathol 81: 147-151.
- Hill PB, Lo A, Eden CAN, Huntley S, Morey V, et al. (2006) Survey of the prevalence, diagnosis and treatment of dermatological conditions in small animals in general practice. Vet Rec 158: 533-539.
- Halliwell R (2006) Revised nomenclature for veterinary allergy. Vet Immunol Immunopathol 114: 207-208.
- Marsella R, Olivry T, Nicklin C, Lopez J (2006) Pilot investigation of a model for canine atopic dermatitis: environmental house dust mite challenge of high-IgE-producing beagles, mite hypersensitive dogs with atopic dermatitis and normal dogs. Vet Dermatol 17: 24-35.
- Olivry T, DeBoer DJ, Favrot C, Jackson HA, Mueller RS, et al. (2010) Treatment of canine atopic dermatitis: 2010 clinical practice guidelines from the International Task Force on Canine Atopic Dermatitis. Vet Dermatol 21: 233-248.
- Zur G, Ihrke PJ, White SD, Kass PH (2002) Canine atopic dermatitis: a retrospective study of 266 cases examined at the University of California, Davis, 1992-1998. Part I. Clinical features and allergy testing results. Vet Dermatol 13: 89-102.
- Bensignor E, Marignac G, Crosaz O, Cavana P (2013) Pruritus in dogs. Vet Dermatol 24: 292.
- Lourenço-Martins AM, Delgado E, Neto I, Peleteiro MC, Morais-Almeida M, et al. (2011) Allergic conjunctivitis and conjunctival provocation tests in atopic dogs. Vet Ophthalmol 14: 248-256.
- Favrot C, Steffan J, Seewald W, Picco F (2010) A prospective study on the clinical features of chronic canine atopic dermatitis and its diagnosis. Vet Dermatol 21: 23-31.
- Bradley CW, Morris DO, Rankin SC, Cain CL, Misic AM, et al. (2016) Longitudinal evaluation of the skin microbiome and association with microenvironment and treatment in canine atopic dermatitis. J Invest Dermatol 136: 1182-1190
- Saridomichelakis MN, Olivry T (2016) An update on the treatment of canine atopic dermatitis. Vet J 207: 29-37.
- Olivry T, Rivierre C, Jackson HA, Murphy KM, Davidson G, et al. (2002) Cyclosporine decreases skin lesions and pruritus in dogs with atopic dermatitis: A blinded randomized prednisolone-controlled trial. Vet dermatol 13: 77-87.
- Olivry T, Marsella R, Iwasaki T, Mueller R (2007) Validation of CADESI-03, a severity scale for clinical trials enrolling dogs with atopic dermatitis. Vet Dermatol 18: 78-86.
- González-Chávez SA, Arévalo-Gallegos S, Rascón-Cruz Q (2009) Lactoferrin: structure, function and applications. Int J Antimicrob Agents 33: 301 e1-8.
- Vogel HJ (2012) Lactoferrin, a bird's eye view. Biochem Cell Biol 90: 233-244.
- Bellamy W, Takase M, Yamauchi K, Wakabayashi H, Kawase K, et al. (1992) Identification of the bactericidal domain of lactoferrin. Biochim Biophys Acta 1121: 130-136.
- Kuwata H, Yip TT, Tomita M, Hutchens TW (1998) Direct evidence of the generation in human stomach of an antimicrobial peptide domain (lactoferricin) from ingested lactoferrin. Biochim Biophys Act 1429: 129-141.
- Strom MB, Haug BE, Rekdal O, Skar ML, Stensen W, et al. (2002) Important structural features of 15-residue lactoferricin derivatives and methods for improvement of antimicrobial activity. Biochem Cell Biol 80: 65-74.
- Gifford JL, Hunter HN, Vogel HJ (2005) Lactoferricin: a lactoferrin-derived peptide with antimicrobial, antiviral, antitumor and immunological properties. Cell Mol Life Sci 62: 2588-2598.
- Hwang PM, Zhou N, Shan X, Arrowsmith CH, Vogel HJ (1998) Three-dimensional solution structure of lactoferricin B, an antimicrobial peptide derived from bovine lactoferrin. Biochemistry 37: 4288-4298.
- Bruni N, Capucchio MT, Biasibetti E, Pessione E, Cirrincione S, et al. (2016) Antimicrobial activity of lactoferrin-related peptides and applications in human and veterinary medicine. Molecules 21: e752.
- Corda D, Zizza P, Varone A, Bruzik KS, Mariggiò S (2012) The glycerophosphoinositols and their cellular functions. Portland Press Limited.
- Motolese A, Simonelli M, Effect of glycerophosphoinositol salt of choline 1% cream on mild-to-moderate inflammatory and allergic dermatitis. J Istituto Dermatologico San Gallicano 10: 135-140.
- Pardo F, Perich F, Villarroel L, Torres R (1993) Isolation of verbascoside, an antimicrobial constituent of Buddleja globosa leaves. J Ethnopharmacol 39: 221-222.
- Avila JG, de Liverant JG, Martínez A, Martínez G, Muñoz JL, et al. (1999) Mode of action of Buddleja cordata verbascoside against Staphylococcus aureus. J Ethnopharmacol 66: 75-78.
- Pesce M, Franceschelli S, Ferrone A, De Lutiis MA, Patruno A, et al. (2015) Verbascoside down-regulates some pro-inflammatory signal transduction pathways by increasing the activity of tyrosine phosphatase SHP-1 in the U937 cell line. J Cell Mol Med 19: 1548-1556.
- Hill PB, Lau P, Rybnicek J (2007) Development of an owner-assessed scale to measure the severity of pruritus in dogs. Vet Dermatol 18: 301-308.
- Rybnícek J, Lau-Gillard PJ, Harvey R, Hill PB (2009) Further validation of a pruritus severity scale for use in dogs. Vet Dermatol 20: 115-122.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi