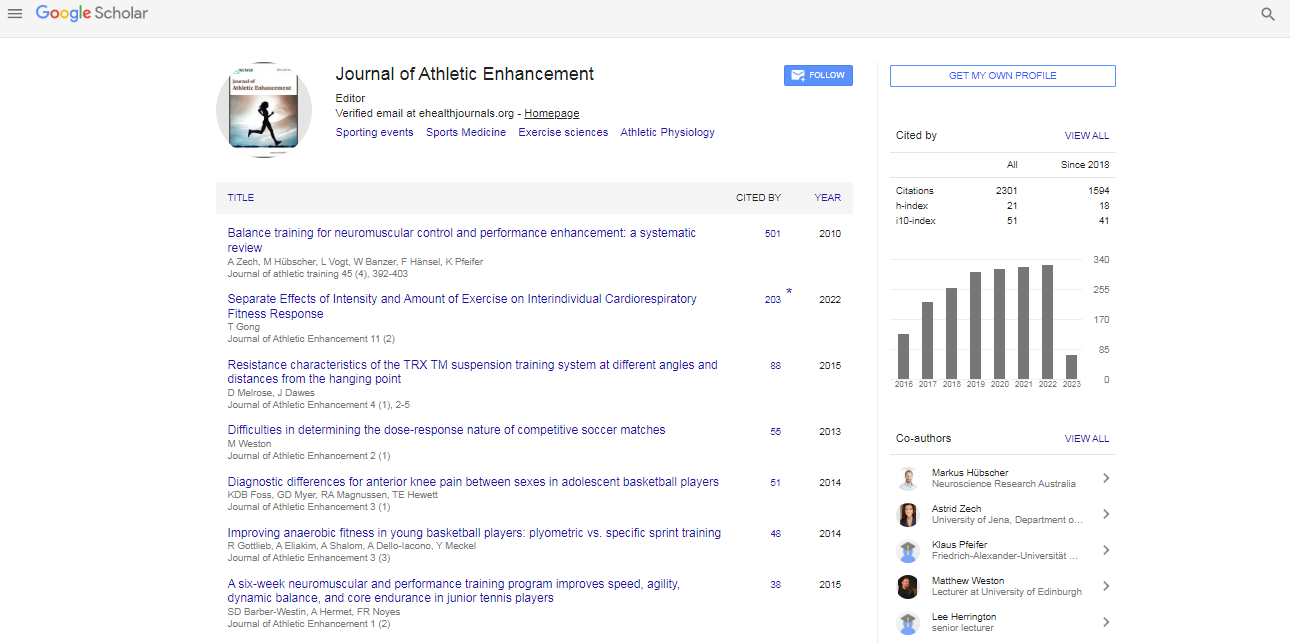
Research Article, J Athl Enhancement Vol: 4 Issue: 4
Efficacy of Adaptogenic Supplements on Adapting to Stress: A Randomized, Controlled Trial
| Areg Hovhannisyan1, Magnus Nylander2, Georg Wikman2 and Alexander Panossian2* | |
| 1Republican Centre of Sport Medicine and Anti-doping Service of Armenia, Armenia | |
| 2Swedish Herbal Institute Research & Development, Vallberga, Sweden | |
| Corresponding author : Prof. Alexander Panossian, PhD Swedish Herbal Institute Research & Development, Kövlingevägen 21, SE-312 50 Vallberga, Sweden Tel: +46-702818171, Fax +46-43023723 E-mail: ap@shi.se |
|
| Received: May 04, 2015 Accepted: September 09, 2015 Published: September 16, 2015 | |
| Citation: Hovhannisyan A, Nylander M, Wikman G, Panossian A (2015) Efficacy of Adaptogenic Supplements on Adapting to Stress: A Randomized, Controlled Trial. J Athl Enhancement 4:4. doi:10.4172/2324-9080.1000205 |
Abstract
Efficacy of Adaptogenic Supplements on Adapting to Stress: A Randomized, Controlled Trial
Objective: The aim of this randomized, double-blinded, placebocontrolled trial was to assess the efficacy of adaptogens on the physical and emotional stress-induced loss of concentration, coordination, fatigue and hormonal changes in 215 elite athletes. This study explores two formulations, ADAPT-232S and ADAPT-S, that had standardized contents of salidroside, schisandrin, eleutherosides B and E, ecdisterone and pantothenic acid. Methods: Standard psychological scales and computerized neurophysiological and blood tests were used to assess the efficacy of the treatments at the baseline and then after 7, 8, 28 and 29 days of daily oral administration. Fatigue was chosen as one of the primary outcomes and was measured as selective inattention and impulsivity in the Conners’ computerized continuous performance test (CCPT) and three standard psychological rating scales, the fatigue severity score (FSS), the perceived stress score (PSS) and the Shirom-Melamed burnout score (SMBS). The anabolic index (blood testosterone/cortisol ratio) and blood lactate were selected as primary outcomes in the phase of recovery of athletes on the second day after heavy physical exercise or competitions. Results: Significant differences (p<0.01) between the placebo and verum groups in favor of ADAPT-232S and ADAPT-S were found in the primary and some secondary outcomes: Conners’ computerized continuous performance test parameters of selective attention and impulsivity, anabolic index, cortisol and lactate levels in blood, fatigue severity score, perceived stress score, and physical performance tests. The sport achievements of athletes were in favor of the ADAPT groups. All of the treatments were well tolerated. The total numbers of adverse events did not differ among the three groups. Conclusions: The adaptogenic preparations, ADAPT-232S or ADAPT-S, are safe and effective supplements for enhancing tolerance to physical and emotional stress as well as for the recovery of athletes during and after high–intensity exercise and competitions.
Keywords: Sport medicine; ADAPT; Fatigue; Randomized controlled trial
Keywords |
|
| Sport medicine; ADAPT; Fatigue; Randomized controlled trial | |
Introduction |
|
| Overtraining syndrome (OTS) is a stress-related disorder that results in fatigue, decreased performance and impaired recovery [1-3]. Loss of concentration, decreased coordination and fatigue are some of the many unintended consequences of the heavy physical and emotional loads of competitive athletes. The use of natural health products to enhance recovery and reduce fatigue in athletes has become a topic of discussion and importance in sport medicine. In this context, adaptogens are considered to be the most suitable supplements, which are safe by definition and enhance “nonspecific resistance to stress” [4-9]. At the cellular and molecular levels, adaptogens initiate the activation of adaptive stress response pathways, resulting in increased expression of heat-shock proteins, neuropeptide Y, antioxidant enzymes and anti-apoptotic proteins [10,11]. ADAPT-232, a combination of natural compounds of plant origin standardized for the contents of p-hydroxyphenethyl-glucopyranoside and several lignans, has been shown to significantly improve attention and the ability to concentrate, expressed as a decrease in errors made and an increase in the speed and accuracy when performing stressful cognitive tasks on various psychometric tests, both in ordinary healthy subjects [12] and in cosmonauts [7,13]. | |
| In a placebo-controlled clinical study with pneumonia patients, the average decrease in the time of recovery and duration of treatment with antibiotics was 25% or about two days shorter in the group of patients who received antibiotics in combination with ADAPT-232 compared with the placebo group, who only received the standard treatment with antibiotics. This indicates the beneficial health effects of this combination of adaptogens in the recovery of pneumoniainfected patients [14]. | |
| Recently, it was shown that the compounds in ADAPT-232 contribute to the efficacy of the product [10]. This is due to synergistic reactions on the molecular networks in the targeted cells that induce the deregulation of certain genes. This is a specific effect of the combination of compounds in ADAPT-232 and does not appear when the individual compounds in ADAPT-232 are tested separately [10]. | |
| The main purpose of the present study was to investigate the effects of two similar combinations of natural compounds, ADAPT-232S and ADAPT-S, which were standardized for the content of salidroside, schizandrin, eleutherosides B and E, 20-hydroxyecdysterone and pantothenic acid, on attention, the ability to concentrate and the recovery of the hormonal system of athletes after physical and emotional stress experienced during the course of intense training sessions and competitions. | |
Materials and Methods |
|
| Trial design | |
| A double-blind, placebo-controlled, randomized, parallel-group trial was carried out in Yerevan, Armenia, with the approval of the Health Research Ethics Board at the Yerevan Medical State University of Armenia. The study meets the ethical standards of the International Journal of Sports [15]. All participants signed written consent forms. | |
| Participants | |
| This study was conducted in several separate groups of athletes in various time of the year, sometimes with intervals of several months. The athletes were recruited among available groups of athletes from various sport disciplines included in national Olympic teams during training sessions in training camps. The choice of sport disciplines depends mainly on four issues: availability of sufficient amount of high-level athletes, availability of training sessions and competitions during the study period, popularity of particular sport discipline in Armenia (wrestling and judo are the most popular sport in Armenia) and inclusion a sport in Olympic disciplines. | |
| A group of 215 healthy athletes (208 male and 7 female Caucasian volunteers, ages 18-35) with a background of at least four years of systematic-specific athletic exercise were enrolled at the Sport Medicine and Anti-Doping Service Republican Center for training sessions and sports competitions in six different sport disciplines. The following numbers of athletes from each sport-track and field: 18, judo: 31, wrestling: 35, ice hockey: 47, basketball: 34, and football: 50-were eligible for participation in this trial. All of the subjects were in good health and were not taking any medication, nutritional supplements (i.e., amino acids, etc.) or drugs, including anabolic-doping agents that could influence the experimental protocol. Exclusion criteria were: any known abuse of medication, narcotics or alcohol; any known allergies to cut flowers, herbal spices, or bitter seeds; pregnancy or breast-feeding; infections requiring medical treatment; consumption of any type of medications and/or mood-elevating substances (such as B and E vitamins, ginkgo extracts, homeopathic preparations or food supplements); and illness lasting more than 36 hours, including the common cold or fever. All of the subjects also underwent a careful examination to avoid potential interference from any teeth, mouth, upper gastrointestinal or respiratory problems. None of the subjects was a smoker. | |
| Interventions | |
| The ADAPT-232S formulation evaluated in this study is a combination of phenethyl- and phenylpropenyl-glycosides, lignans, epigallocatechingallates, and mono-, sesqui- and triterpene glycosides isolated from plants belonging to the Crassulaceae, Araliaceae and Schisandraceae families [11,16]. ADAPT-S is a variation of ADAPT- 232S with the addition of ecdysterones and calcium panthotenate. The quality of all botanicals and pharmaceutical-grade compounds used in this study were tested by TLC and HPLC in accordance with the specifications and using appropriate reference standards. All of the analytical methods were validated for selectivity, accuracy and precision. Samples from all of the products were retained. Microbiological analyses of the herbal substances and herbal compounds were performed in accordance with the specifications of the European Pharmacopoeia, 6th Ed. The analyses of pesticides, aflatoxins and heavy metals, residual solvents and radioactivity in botanicals were performed in accordance with specifications of the European Pharmacopoeia, 6th Ed. | |
| ADAPT-232S and APADT-S were administered orally as hard gelatin capsules manufactured at the Swedish Herbal Institute in accordance with ICH guidelines for guidance alignment process (GAP) validation and good manufacturing practices (GMP) for active pharmaceutical ingredients (APIs). The APIs were standardized for the contents of tyrosol, p-hydroxyphenethyl-glucopyranoside, rosavin, schizandrin, γ-schisandrin, eleutherosides B and E and 20-hydroxyecdysterone [11]. Each capsule of ADAPT-232S contained 0.5 mg of salidroside, 1.0 mg pf Schizandrin and 0.35 mg of Eleutherosides B and E, while each capsule of ADAPT-S contains 1.5 mg of salidroside, 1.5 mg of 20-hydroxyecdisterone and the same amounts of schizandrin and eleutherosides B and E as the ADAPT- 232S capsules. The placebo group was given the same type of hard gelatin capsules containing microcrystalline cellulose, corn starch and magnesium stearate. The placebo capsules had the same appearance, volume, weight, odor and taste as the verum capsules that contained ADAPT-232S and ADAPT-S. Daily dose - 4 capsules (2 capsules two times a day, in two hours after the meal. | |
| Measurements of compliance: The compliance of individual subjects was ensured by recording the daily consumption of capsules. This was performed on special forms provided to the subjects together with the self-assessment questionnaires. | |
| Randomization and blinding | |
| Sequence generation: The study supplements (packages of placebo and ADAPT capsules) were labeled in accordance to the Treatment Randomization Code (TRC) generated by the Excel Random Number Generator (Add-On). The table contained three columns (A, B and C) filled with randomly distributed unique numbers from 1 to 240. Column A (100 unique numbers) corresponded to ADAPT-232S, column B (80 unique numbers) to placebo and column C (60 unique numbers) to ADAPT-S. The assignment of ADAPT and placebo to A, B or C groups/sets of packages - treatment code (TC) was encoded by the qualified pharmacist (QP) in the course of the study supplement (treatment) randomization procedure at the manufacturing site. | |
| Allocation concealment: This Treatment Randomization Code was kept by the QP at the investigational product manufacturing site (at sponsor) until the study was finalized, before statistical evaluation of the data was performed. | |
| The treatment code (TC), identifying the assignment of patients to the placebo and the two investigational ADAPT products , was kept by the Qualified Pharmacist at the investigational product manufacturing site until the study was finalized. | |
| Implementation: The participants were assigned to a study number, which was actually the same number as the study supplement number (printed on the label of each individual package): each athlete voluntarily chose among all of the available boxes at the team club on day 1 of treatment. The participants’ names were recorded on the labels of the packages as well as in the list of participants and the CRF, where their treatment/study numbers were recorded by the principal investigator of the study. Once the athlete had chosen a treatment number, the team doctor handed the athlete the corresponding bib number, which corresponded to the number on the study supplement packages. The participants wore the numbers, which were the same as their treatment numbers in the study, during allocation of the randomized study supplements. | |
| The Study Participants list identifying the patients and the study supplement packages (treatment numbers) was generated by Principal Investigator. This list was used for statistical analysis at the end of the study together with the Treatment Randomization Code received from the QP. The treatment code providing the information about actual assignment of groups A, B and C to ADAPT and the placebo was broken by the QP after the statistical analysis of the results of the study on the basis of comparisons among groups A, B and C was complete. | |
| Blinding | |
| Blinding of the trial subjects was performed by using labeled packages containing blisters with capsules of identical appearance. The study supplements were delivered to the clinic pre-labeled and coded according the randomization list. The randomization code was not revealed to the clinic or the participating investigators in Sweden until the code was broken after termination of the study. In this way, the study supplement was also blinded to the investigators, and the study was double-blind. | |
| The Treatment Randomization Code was provided to the Principal Investigator after all of the patients had completed the treatment, but before the statistical evaluation of results of the study. It contained information about the content of each package, that is, how the placebo and ADAPT packages/containers were encoded. The patients were assigned to study groups A, B and C by the principal investigator at the end of the study. | |
| The randomization codes, including one verified copy, were kept in a sealed envelope marked “Trial Treatment Randomization Codes. They have not been broken until the study has been finalized” by the Qualified Person at the investigational product manufacturing site. | |
| Follow up | |
| At visit #1 (pretreatment), the athletes were assessed to be included or excluded from the study, and those who met the inclusion criteria were recruited, informed about the details of the pending study, and provided with study medication and self-assessment questionnaires (Supplement 2). Informed consent forms in Armenian and English were given to every included subject. After the signed informed consent was obtained, all athletes were subjected to medical and physical examinations and blood and urine samples were taken. | |
| On the Day 1, Day 7 and Day 28 of treatment (Visits 2, 3 and 4), the athletes were randomized to take either ADAPT-232S, ADAPTS or identical placebo capsules as described above (Table 1, Supplement 1). The athletes performed intense exercises for 1 h at work load level III (Table 2, Supplement 1), during which time the heart rates of athletes were in the range from 160 to 180 min- 1. The blood lactate concentrations after exercise were in the range of 5-8 mmol/l. Twenty sec after the completion of the exercises, in accordance with the training plan, the coach or doctor checked the heart rates of the athletes. Fifteen min after the completion of the exercises, the participants donated blood for an analysis of the blood lactate, cortisol and testosterone levels. On Day 1 and Day 28 of the treatment, after completion of blood sampling, the participants were invited to undergo a special test for physical performance, attention and motor coordination, and the results were recorded by the sport club doctor. Then, the participants completed the BMS, FSS and PSS Questionnaires and proceeded with Connors computerized test. After completion of the test, the athletes took the designated study medication and continue to take it regularly for 30 days. To eliminate any circadian rhythm, nutrition and climate-related bias factors, all of the experimental evaluations were performed in similar conditions in the morning from 9:30 to 11:30 am, two hours after a standardized breakfast. On the 29th day after initiation of treatment, the athletes arrived at the therapeutic department of CRO for physical examinations, and blood and urine samples for laboratory analyses were taken for final analysis (Table 1, Supplement 1). | |
| Table 1: Schedule of assessment. | |
| Table 2: Work load scale [18] used for the assessment of the intensity of exercise in trained athletes. | |
| Training and competition schedule: In the course of the treatment, the athletes followed training and competition schedule with: | |
| • high intensity strenuous exercise at work load level III on the first and fourth weeks of treatment (days 1-7 and days 21-28 of treatment) in which the heart rates of athletes were in the range of 160 to 180 min-1 and their blood lactate concentrations after the exercise were in the range of 5-8 mM (Table 2, Supplement 1). | |
| • moderate intensity strenuous exercise at work load level II on the second and third weeks (7th -21st days 7-21 of treatment) in which the heart rates of athletes were in the range of 140 to 160 min-1 and their blood lactate concentrations after the exercise were in the range of 2 to 4 mM. | |
| • maximal intensity of strenuous exercise (work load levels IV-V) on the days 26-28 of the treatment, during competitions. | |
| The heart rate monitoring and blood lactate concentration tests were performed according to the schedule ( 1 and 2). These two variables are the most important for the assessment of the work load level and intensity of exercise [17,18]. | |
| Outcomes | |
| • Efficacy | |
| • Primary outcome measures: Sustained and selective attention and impulsivity were measured by using a computerized neuropsychological test (CCPT, Conners’ Continuous Performance Test) [19,20]. | |
| • Psychological outcome measures – Perceived stress Score (PSS) [21], Fatigue Severity Score (FSS) [22], Shirom-Melamed Burnout Questionnaire (SMBQ) [23], athletic achievement and competition results. | |
| • Anabolic index (testosterone/cortisol ratio) [24,25]. | |
| • Secondary outcome measures: Physiological outcome measures - blood lactate [17,18], Physical performance tests and athletic achievements. | |
| • Safety: Adverse events, heart rate variability, and blood biochemical markers | |
| Primary outcomes: Validated psychological scales, physiological tests and bioassays were used for the assessment of the efficacy of treatments at the baseline and after 7, 8, 28 and 29 days of repeated daily oral administration. | |
| Fatigue was chosen as one of the primary outcomes. However, because there is no widelyagreed- upon tool for measuring fatigue in healthy populations, two measurement tools were used to assess this outcome, and the correlation between them was assessed later. One tool is a neuropsychological test-Conners’ computerized continuous performance test (CCPT), which provides objective information about the cognitive performance of participants, their selective attention and impulsivity (i.e., - the ability to distinguish and detect a target, errors made in responses to target signal, speed of correct responses to target signal) . | |
| The CCPT presents target letters on a computer screen. The examinee's task is to press the space bar or click the mouse whenever any letter other than "X" appears. Letters are displayed for 250 milliseconds at 1-, 2-, and 4-second intervals. The administration time is 14 minutes. | |
| Test Scoring | |
| • Response times (Hit Reaction time) - the average speed of correct responses for the entire test. | |
| • Response times (Hit Reaction time) - the average speed of correct responses for the entire test. | |
| • Changes in the reaction time speed and consistency | |
| • Signal detection theory statistics/detectability - the ability to distinguish and detect the target and non-target stimuli. Higher rates of correct detections indicate better attentional capacity. | |
| • Omission errors - the failure to respond to the target. High omission rates indicate that the subject is either not paying attention to the stimuli (distractibility) or has a sluggish response. | |
| • Commission errors - errors are made when responses are given to non-targets. A fast reaction time and high commission error rate points to difficulties with impulsivity. A slow reaction time with high commission and omission errors indicates inattention in general. | |
| • Confidence index | |
| The program generates three types of reports: one summarizing the results of the individual tests; one comparing the results from several tests of the same subject; and the third comparing the results to the scores from the Conners' Rating Scales. T-scores and percentile ranks are also provided. | |
| This test is validated for the assessment of selective attention and impulsivity and can assist health professionals with objective information regarding inattention in fatigue [19]. | |
| Three other tools used for assessment of fatigue were validated psychological scales: Fatigue Severity Score (FSS) [22], Perceived Stress Score (PSS-10) and the Shirom-Melamed Burnout Questionnaire (SMBQ) score [23,26]. | |
| These scales comprises 9, 10 and 12 items that are collectively aimed at measuring fatigue/energy, the perception of stress, and burnout – a mental condition defined as a result of continuous and long-term stress exposure, particularly related to psychosocial factors. | |
| The Fatigue Severity Scale is one of the best known and most used validated fatigue scales [22,27], particularly among patients with Multiple Sclerosis (MS) and Systemic Lupus Erythematosus (SLE). However, the FSS is now widely used in various fields. The questionnaire consists of 9 items with a Likert scale ranging from 1 (completely disagree) to 7 (completely agree). A mean score above 4 indicates elevated levels of fatigue. Krupp and colleagues found that only 5% of healthy subjects have a mean score above 4, while up to 90 % of patients with medical disorders experienced fatigue at or above this level. The questionnaire has good test-retest reliability, high internal consistency (0.81- 0.94) and is sensitive to changes over time and after treatment. The athletes completed the FSS-10 on Visits 2, 3 and 4. | |
| The 12-item version of the burnout scale include three subscales: "physical fatigue," "emotional exhaustion" and "cognitive weariness." These measures stress the three levels that comprise the burnout condition, emotional exhaustion, physical fatigue, and cognitive weariness. The results consisted of the responses to the following: Answer each of the statements below by indicating how often you have the feeling during working hours. “Almost always” =1 point; “very frequently” =2 points; “quite frequently” =3; “sometimes” =4; “quite infrequently” =5; “very infrequently” =6; “almost never” =7. Male participants whose scores average 3.0 to 3.75 and women whose scores average 3.6 to 4.0 are at the high end of the burnout range and should seek expert help for preventative measures. | |
| The Perceived Stress Scale (PSS) is a 10-item self-report questionnaire that measures a person’s evaluation of the stressfulness of the situations in the past month of their lives [21]. PSS is the most widely used validated psychological instrument for measuring the perception of stress and the degree to which situations in one’s life are appraised as stressful. The items were designed to test how unpredictable, uncontrollable, and overloaded the respondents find their lives. The scale also includes a number of direct queries about current levels of experienced stress. The PSS was designed for use in community samples with at least a junior high school education. The questions in the PSS ask about feelings and thoughts during the last month. In each case, the respondents are asked how often they felt a certain way. The Perceived Stress Scale is the only empirically established index of general stress appraisal. “The PSS measures the degree to which situations in one’s life are appraised as stressful.” The final PSS-10 scores were obtained by reversing the scores on the four positive items, e.g., 0=4, 1=3, 2=2, and so on and then summing across all 10 items. Items 4, 5, 7, and 8 are the positively stated items. The scores can range from 0 to 40, with higher scores indicating greater stress. | |
| Physiological stress parameters, including endocrine (cortisol, testosterone, insulin-like growth factor, etc.) biochemical (e.g., lactate) and hematological parameters that characterize the homeostasis and recovery of the neuroendocrine system in stress were measured using bioassays of blood plasma and urine samples. | |
| Bioassays: The blood samples collected for the biochemical and hematological analyses were analyzed immediately after collection. The samples for the analyses of endocrine functions were stored in a refrigerator for no more than 2 hours before analysis in the Clinical Laboratory of CRO. | |
| General blood analysis includes Hemoglobin (g/l), Hematocrit (%), RBC (10*12/L), WBC (10*9/L), PLT (10*9/L), Lymphocytes (10*9/L), NEUT(10*9/L), MXD, (10*9/L), MCH, (pg/cell), ESR (mm/h), Se-K (mmol/l), Se-Na (mmol/l), Protein (g/l), Urea (mmol/l), Se-creatinine (μmol/l), Se-bilirubin (μmol/l), Glucose (mmol/l), Cholesterol (mmol/l), BUN (mmol/l),Triglycerides (mmol/l), ALT (U/l) AST (U/l) GGT (U/l). | |
| Insulin (μIU/mL), T3 (ng/dL), T4 (μg/dL), and TSH (mIU/L). | |
| Blood Cortisol and Testosterone analysis Cortisol and testosterone in blood were analyzed in accordance with the instructions described in the DRG Cortisol ELISA kits and DRG Testosterone ELISA kits (DRG Instrument GmbH, Germany, web: http://www.drgdiagnostics. de). | |
| Blood samples (5 ml) were drawn from the antecubital vein. The serum samples were immediately stored in plastic Eppendorf tubes and frozen at -20ºC. The frozen serum samples were thawed and centrifuged at 2000 rpm for 10 min, resulting in a clear supernatant of low viscosity. A 20-μL aliquot of each Standard, Control and sample was dispensed into the appropriate wells. Then, 200 μL of the Enzyme Conjugate was dispensed into each well. The solution was thoroughly mixed for 10 seconds. The contents of the wells were incubated for 60 minutes at room temperature (without covering the plate) and then briskly shaken out. The wells were subsequently rinsed 3 times with diluted Wash Solution (400 μL per well). Then, 100 μL of Substrate Solution was added to each well and incubated for 15 minutes at room temperature. The enzymatic reaction was stopped by adding 100 μL of Stop Solution to each well. The cortisol and testosterone concentrations were measured at 450 ± 10 nm and 630 ± 10 nm, respectively, using a microtiter plate reader within 10 minutes after adding the Stop Solution. | |
| The standard curves were generated using a computer-controlled program, and the cortisol and testosterone concentration of each sample was calculated. | |
| With respect to intra-assay variation, samples with a normal concentration of cortisol had a CV of 2.1% and testosterone of 1.8%, whereas samples with a high concentration had a CV of 3.5%. | |
| The Urine analyses included specific gravity, pH, urobilinogen (mg/L), glucose, ketones, bilirubin, Erythrocytes and leukocytes. | |
| The standard analytical equipment used for the routine blood analyses included: | |
| • Lactometer «Accutrend Lactate», («Roche Diagnostics »); | |
| • Enzyme immune assay EIA microplate reader «StatFax303Plus» with a microprocessor controlled bichromatic photometer system, six filters and an incubator (“Awareness Technology Inc” (USA); | |
| • Microprocessor controlled photometer «StatFax3300», “Awareness Technology Inc” (USA); Sysmex. Automated Hematology Analyzer pocH-100i “Sysmex Corporation, Cobe, Japan” | |
| Secondary outcomes: Sport-specific psychophysical measures. Field tests for attention and motor coordination: | |
| a) Football: Test # 1-the athletes ran in a circle around the ball for 10 seconds and shot at the goal. If the ball entered the goal, 1 (one) point was awarded. Fouls (the ball did not enter the goal) were not recorded [28]. | |
| b) Basketball: Test #2 - immediately after a full time basketball game and without any time to rest, the athletes attempted five free throws. If the ball entered the basket, 1 (one) point was assigned. Fouls (the ball did not enter the basket) were not recorded. | |
| c) Ice hockey: Test 3# - after a hockey game (20 min), the participants skated around the ice hockey arena and attempted five shots at the goal from 5 meters away from the goal, which was closed by a special shield that had four holes on the upper and lower corners. If the hockey puck entered the goal, 1 (one) point was awarded. Fouls (the ball did not enter the goal) were not recorded. | |
| d) Track and field athletic and wrestling: Test #4-the athletes were divided into groups of 6 people and ran around the stadium. At a signal from the coach (one, two or three whistles), the athletes were to immediately change their running motion, sprint, stop or drop to a starting position. For athletes who properly carried out the commands, 1 (one) points was awarded. Fouls (the ball does not enter the goal) were not recorded. | |
| e) Judo: Test #5 -the participants were divided into groups of 4 athletes within "conventional" weight categories (the difference between judo was no more than 2-3 kg). Immediately after the primary competition, the athletes competed within the category of a round system in accordance with the rules of Judo competition. For every win, the athlete accrued two points, whereas 1 point was credited for a draw. No points were awarded for a defeat (Figure 1 & 2). | |
| Figure 1: Schematic diagram of the trial. | |
| Figure 2: Participant flowchart. | |
| Competitive achievements results: Sports doctors recorded the results of the athletes’ participation in the competitions in the last week of the treatment course or during one month after the treatment. | |
| The relative number of winners in each study group was calculated as the percent of the group size (100%) by the equation: W (%) = (A/n)100, were: | |
| A – number of athletes whose results in the sport improved or who won the international competition n – number of athletes in the study group | |
| Urine analysis includes Specific gravity, pH, Urobilinogen (mg/L), Glucose, Ketones, Bilirubin, Erythrocytes and Leukocytes. | |
| The Data and Safety Monitoring Board reviewed the adverse event data and assessed their causality in a blinded fashion. | |
| Sample size | |
| In a previous study of ADAPT-232, we used fatigue as an outcome measure in the physical and psychological domains of quality-of-life (WHOQOL-Brief questionnaires) [14]. The sample size for the present study was calculated using an effect size of 1.42 from a comparable study of ADAPT-232 on fatigue [14]. Using a two-sided, two-sample t-test, a sample size of 30 participants in each group was required to achieve 95% power to detect an absolute mean difference of 1.42 on the FSS subscale between the ADAPT-232S and placebo at the 0.05 level of significance (Figure 3A; Stat-Mate, version 2.00, 2004, GraphPad software Inc.). We originally planned for a 10% drop-out rate, which would have increased our intended sample size to 100 participants (33 in each group). However, instead of using equal number of subjects in each group, the decision was made to use unequal numbers of participants: 60 subjects in ADAPT-S, 80 subjects in placebo and 100 subjects in ADAPT-232S groups, for a total of 240 participants. The total sample size was increased because of the various treatment "costs" in each group. The power to detect a difference of 0.28 between means with a significance level (alpha) of 0.05 (two-tailed) was determined to be more than 99% in the completed trial with ADAPT-S (n=55, SD=0.43) ADAPT-232S (n=92, SD=0.40) and placebo (n=68, SD=0.28). The results calculated for the FSS test are shown in Figure 3B. | |
| Figure 3: A - The Sample size for he unpaired t-test: the trade-off between the sample size, the difference between means can be detected and power. B - power (%) – difference between means of placebo and ADAPT-232S/ADAPT-S plots. FSS-test. Stat-Mate, version 2.00, 2004, GraphPad software, Inc. | |
| No interim analyses were planned or performed, and there were no stopping rules set up for this trial. | |
| Statistical methods | |
| The data were analyzed using GraphPad (San Diego, CA, USA) Prism software (version 3.03 for Windows). Continuous variables were presented as the means ± standard deviation (SD) and categorical variables as counts (percentages). | |
| The statistical analyses involved evaluating the subject's change in scores from the initial visit (baseline) to the intermediate and the final visits and at each scheduled visit of the study. All of the data were checked for normality. Depending on the results of the normality test, the comparative assessment of the baseline characteristics between groups (ADAPT232S and ADAPT- S vs Placebo) was made using: | |
| • Kruskal-Wallis (KW) non-parametric one-way ANOVA rankorder test, with post hoc Dunn's Multiple Comparison Test or | |
| • parametric one-way independent measures ANOVA with Dunnett's Multiple Comparison Test. | |
| • The analyses of changes within treatment groups in the course of the study (before versus after) were performed using: | |
| • paired t-test (parametric data - variables with normal distribution) or/and | |
| • Wilcoxon signed rank test (nonparametric data), or Friedman test for several repeated measures (nonparametric data). | |
| • Assessment of the efficacy of the study supplements was achieved by comparing the mean changes from the baseline (differences before and after treatment of every single patient) for each group (ADAPT232S and ADAPT- S vs. Placebo) using: | |
| • Kruskal-Wallis (KW) non-parametric one-way ANOVA rankorder test, with post hoc Dunn's Multiple Comparison Test, and/ or parametric one-way independent measures ANOVA with Dunnett's Multiple Comparison Test (variables with normal distribution). The statistical significance was set with alpha at=0.05 | |
Results |
|
| Study participants/disposition of participants and baseline variables | |
| A total of 220 subjects were screened, and 215 were randomly allocated to three treatment groups: 92 to ADAPT-232S, 55 to ADAPT-S and 68 to placebo. One recruited subject was excluded from the study because the person’s blood analysis did not meet the requirements for inclusion, and four subjects dropped out before the second visit because they were not able to come to the randomization venue for various reasons. Thus, a total of 215 athletes were randomized, completed the study and were included in the analyses. Of this total, n=92 in the ADAPT-232S group; n=55 in the ADAPT-S group and n=68 in the placebo group were included in the analyses. The proportion of elite athletes (participants in European and World Championships) was 37% of the total number of athletes included in the study. The elite athletes were randomly distributed in each group: 25 athletes in the placebo group (36.76%), 21 in the ADAPT-S (38.18%) and 33 in the ADAPT-232S (35.87%). | |
| The flow of participants through the trial is described in Figure 1. | |
| The baseline demographic data for participants in each group are provided in Table 1. | |
| Between-groups comparisons (Tables 3-5, Supplement 1) showed no significant difference between the placebo group and either treatment group (ADAPT-S and ADAPT-232S) at the baseline. Between groups, the comparisons showed no significant difference between the placebo group and both treatment groups (ADAPT-S and ADAPT-232S) at the baseline. No statistically significant differences in the mean values of blood pressure, heart rate or body temperature of the athletes among the three groups were observed at the beginning of the study, and no statistically significant differences in the mean values of blood cells and other blood parameters among the three groups of athletes were observed at the beginning of the study. See Tables 3-5 in Supplement 1. | |
| Table 3: Comparison of the athletes’ baseline data. Hematology characteristics. | |
| Table 4: Comparison of athletes’ baseline blood biochemistry data. | |
| Table 5: Between and within group comparisons of Conner’s test scores. | |
| See Tables 3 and 4 in Supplement 1. | |
| Efficacy of treatment | |
| Significant differences (p<0.01) between the placebo and the verum groups in favor of ADAPT-232S and ADAPT-S were found in primary and secondary outcomes: | |
| • CCPT parameters of selective attention and impulsivity (the ability to distinguish and detect a target, errors made in responses to target signal, speed of correct responses to target signal) | |
| • Anabolic index (testosterone/cortisol ratio) | |
| • Cortisol level in the blood | |
| • Fatigue severity score | |
| • Perceived stress score | |
| • Lactate level in blood | |
| • Physical performance tests | |
| All participants in the trial were involved in competitions at the end of the treatment course. Among them, 12.7% of the athletes in the ADAPT-S group and 5.4% of the athletes in the ADAPT-232S group demonstrated significantly improved sports results or won competitions during or within 10 days after the end of the treatment. In comparison, only 1.5% of the athletes in the placebo group significantly improved their sports results or won competitions. | |
| All treatments were well tolerated. Few adverse effects were reported, and the total number of adverse events did not differ between groups. | |
| • Primary outcomes | |
| • Sustained and selective attention and impulsivity: Sustained and selective attention and the impulsivity of athletes were evaluated to assess the level of fatigue. The pretreatment (day 1; 15 min after exercise) and post-treatment (day 28; 15 min after competitions) data were compared. Between-groups comparison showed significant differences between the placebo group and both treatment groups (ADAPT232S or ADAPT-S) in: | |
| • Response Times | |
| • Response Times Errors | |
| • Changes in Reaction Time Speed and Consistency | |
| • Signal Detection | |
| • Confidence Index | |
| • Commission Errors | |
| • Detectability | |
| • Perseverance | |
| The results of Conner`s Continuous Performance Test showed (Figure 4) that sustained selective attention and impulsivity were significantly higher in the ADAPT-S or ADAPT232S groups than in the Placebo group (Table 5 in Supplement 1). | |
| Figure 4: Effect of the treatments on attention measures in Conners’ computerized performance test. The mean change of the scores from the baseline tot he end of the study in three independent groups. Between groups comparison, Kruskal-Wallis non-parametric one-way ANOVA rank order test, with post hoc Dunn’s Multiple Comparison Test. | |
| • Psychological outcome measures: The Shirom-Melamed Burnout Questionnaire (SMBQ), Perceived Stress Scale (PSS), and Fatigue Severity Scale (FSS) were evaluated to assess the level of perceived stress and fatigue, Figure 5. Pretreatment (day 1) and post-treatment (day 28) data were compared. The between-groups comparisons showed significant differences between the placebo group and both treatment groups (ADAPT-232S or ADAPT-S) in the change of Perceived Stress Scale and Fatigue Severity Scale. After treatment, there was a significant decrease in the Perceived Stress and Fatigue Severity of athletes in both groups (ADAPT-232S or ADAPT-S), which was not seen in the placebo group (Table 6 in Supplement 1). | |
| Figure 5: Effect of treatment on the FSS and PSS questionnaire scores compared to the placebo group (ADAPT vs Placebo). Comparisons of difference between pre- and post-treatment within each treatment group. | |
| Table 6: Summary table of the effect of treatment with on BMS, FSS and PSS questionnaire scores compared to placebo groups. | |
| • Anabolic index | |
| It has been suggested that the T/C ratio is a valid biological marker that athletes and coaches can utilize within their training regimes. The anabolic index (testosterone/cortisol ratio) of the athletes was evaluated to assess the metabolic state after physical exercise/ competition (days 1, 7 and 28) as well as the efficacy of recovery of athletes on the next days after a heavy physical load (day 8 and 29). The pretreatment (day 1, before an exercise) and post-treatment (days 7, 8, and 28) data were used for comparison. | |
| Figure 6A summarizes the results of the post-exercise testosterone/ cortisol ratio at the baseline (before treatment – day 0) and at the end of treatment (day 29). There were no significant differences in the blood T/C ratio among the groups at the beginning of the study (day 0 – before treatment). This ratio decreased non-significantly in the placebo group at the end of the training season (day 29), while in ADAPT-232S and ADAPT-S groups, it was significantly higher than at the beginning of study (Figure 6A). The between-groups comparison of the changes from the baseline to day 29 showed a highly significant difference between the Placebo and ADAPT groups (Figure 6B). | |
| Figure 6: (A) – The anabolic index (testosterone/cortisol ratio) before and after treatment, within group comparisons (day 29 vs baseline) in each treatment group, paired t-test. (B) - Effect of the treatment on the anabolic index compared to the placebo group (ADAPT vs Placebo). Between-groups comparison of the changes from baseline to day 29. | |
| The between-groups comparison showed significant differences between the placebo group and both treatment groups (ADAPT- 232S and ADAPT-S) after the treatment on days 7, 8 and 28, while no statistically significant differences were observed on day 1. Both ADAPT-232S and ADAPT-S increased the anabolic index after exercise as well as in the recovery phase compared with the Placebo group. (Tables 7,8 in Supplement 1) | |
| Table 7: Within and between group comparisons of the anabolic index (testosterone/cortisol ratio) levels. | |
| Table 8: Within and between group comparisons of cortisol, testosterone and anabolic index (testosterone/cortisol ratio) levels during the recovery phase. | |
| The cortisol level in the blood of athletes was evaluated to assess the stress response after physical exercise/competition (days 7 and 28) as well as the efficacy of the recovery of athletes on the day following a heavy physical load (day 8 and 29). The pre-treatment (day 1, 15 min after an exercise and post-treatment (days 7, and 28) data were used for comparison. Figure 7A summarizes the results of the post-exercise cortisol levels in blood at the baseline (before treatment – day 0) and at the end of treatment (day 29). There were no significant differences in the blood cortisol levels among the groups at the beginning of the study (day 0 – before treatment). Cortisol significantly increased in the placebo group at the end of training season (day 29), while in the ADAPT-232S and ADAPT-S groups, it was significantly lower that at the beginning of study (Figure 6A). The between-groups comparison of the changes from the baseline to day 29 showed highly significant differences between the Placebo and ADAPT groups (Figure 7B). | |
| Figure 7: (A) - Blood cortisol levels before and after treatment, within group comparisons (day 29 vs baseline) in each treatment group, and paired t-test.(B) - Effect of treatment on blood cortisol compared to placebo group (ADAPT vs Placebo). Between-groups comparison of changes from baseline to day 29. | |
| The between-groups comparison showed significant differences between the placebo group and both treatment groups (ADAPT232S or ADAPT-S) after the treatment on day 7, whereas no statistically significant differences were observed at the baseline (day 1). Between day 7 and day 28, the cortisol level was not significantly changed in the ADAPT-232S group compared with the Placebo group. Both ADAPT232S and ADAPT-S decreased the level of Cortisol during the recovery phase compared with the Placebo group. After physical exercise, the level of Cortisol was increased to a lesser extent in the treatment groups than in the placebo group (Table 9 in Supplement 1). | |
| Table 9: Within and between group comparisons of the Cortisol levels. | |
| The testosterone level in the blood of athletes was evaluated to assess the anabolic activity after physical exercise/competition (days 1, 7 and 28) as well as the efficacy of the recovery of athletes on the next days after a heavy physical load (day 8 and 29). The pretreatment (day 0, before an exercise) and post-treatment (days 29) data were used for comparison (Figure 8). | |
| Figure 8: Post-exercise recovery levels of blood (a) testosterone, (b) cortisol, (c) anabolic index, (d) lactate. | |
| Figure 9A summarizes the results of the post-exercise testosterone levels at the baseline (before treatment – day 0) and at the end of treatment (day 29). The between-groups comparison of changes from the baseline to day 29 showed a significant difference between the Placebo and ADAPT-S group (Figure 9B) (Table 10 in Supplement 1). | |
| Figure 9: (A)-Blood testosterone levels before and after treatment, within-group comparisons (day 29 vs baseline) in each treatment group, and paired t-test.(B) - Effect of treatment on blood testosterone changes from baseline to day 29. | |
| Figure 10: Effect of treatment on physical performance test compared to placebo group (ADAPT vs Placebo). Between-groups comparisons of changes from baseline to day 29. | |
| Table 10: Within and between group comparisons of the testosterone levels. | |
| • Secondary outcomes | |
| • Physical performance tests: In comparison with the Placebo group, ADAPT-S and ADAPT232S significantly increased the results of the physical performance tests ((Figure 10, Table 11 in Supplement 1). | |
| Table 11: Within and between group comparisons of the scores of the physical performance test. | |
| • Athletic achievement and competition results: All of the participants in the trial were involved in competitions at the end of the treatment course. Among them, 12.7% of athletes of the ADAPT-S group and 5.4% athletes of the ADAPT232S group won the competitions during or within 10 days after the end of the treatment. For comparison, only 1.5% of athletes of the Placebo group won the competitions. It is noteworthy that these records were mainly observed in wrestling and the long jump. | |
| It is interesting that both long jumpers achieved their best results on the 6th attempt. (Tables 12-13 in Supplement 1). | |
| Table 12: Individual results of the highest sport achievements of the study participants. | |
| Table 13: The relative percent of competitive achievements for each group. | |
| • The lactate levels in the blood of the athletes were measured to assess athletes’ recovery rate after heavy physical exercise and the competition. The supplementation with ADAPT-S or ADAPT232S significantly reduced the post-exercise plasma lactate level compared to the placebo. (Figure 9, Table 14 in Supplement 1). | |
| Table 14: Within and between group comparisons of the blood lactate level. | |
| Risk of harm | |
| There was one adverse event (sleeplessness) reported by one participant in the ADAPT-232S group and one event (nausea) reported by 2 participants in the placebo group. These adverse events were categorized as mild. It was concluded that there were no issues of safety of the study products (active or placebo arms). | |
Discussion |
|
| The major symptoms and signs of overtraining and OTS can be classified into four categories [1,2]: | |
| • Physiological: decreased performance, decreased body fat, decreased muscular strength, increased VO2 at submaximal loads, muscle soreness, changes in the heart rate, prolonged recovery periods, loss of appetite, and chronic fatigue. | |
| • Psychological: feelings of depression, general apathy, difficulty concentrating, emotional instability, fear of competition, excitation, and restlessness. | |
| • Immunological: increased susceptibility to infection, increased severity of minor infections, decreased functional activity of neutrophils, decreased total lymphocyte counts, reduced response to mitogens, and decreased production of immunoglobulins. | |
| • Biochemical: decreased hemoglobin, increased lactate, elevated cortisol levels, decreased free testosterone levels, and a decrease of more than 30% in the ratio of free testosterone to cortisol. | |
| Among these symptoms and signs, the testosterone to cortisol ratio (T/C) is known to be a reliable marker that can be used to monitor an athlete’s training and possibly diagnose overtraining and/ or OTS [29-32]. | |
| The purpose of this study was to determine the effects of 30 days of treatment with ADAPT-232S or ADAPT-S on the basis of the results of the following analyses/tests: | |
| • Evaluation of concentration difficulties, emotional instability, excitation, and restlessness using Conners’ computerized continuous performance test (CCPT) | |
| • Evaluation of fatigue, general apathy, and feelings of depression using three standard psychological rating scales: FSS, PSS and SMBS. | |
| • Evaluation of performance tests, heart rate, and chronic fatigue using sport-specific tests for attention and motor coordination as well as the competitive achievements results. | |
| • Evaluation of immunological dysfunction in white blood cells counts, immunoglobulins and cytokines. | |
| • Evaluation of elevated cortisol levels, decreased free testosterone levels, decreased ratio of free testosterone to cortisol, decreased hemoglobin and increased lactate. | |
| The results of this investigation indicate that supplementation with ADAPT-232S and ADAPT-S has a significant, beneficial effect on these symptoms of overtraining in athletes in the course of training sessions and in competitions/tournaments. | |
| The majority of athletes who significantly improved their sports results or won competitions were athletes from the two ADAPT groups. This overall outcome is one of the most important results for athletes. | |
| The T/C ratio is a biological marker of exercise that indicates whether an athlete’s training is effective or not [33]. It has been proposed that the T/C ratio might be a reflection of the anabolic and catabolic hormonal balance in the skeletal muscles. Furthermore, it was proposed that this ratio might change in response to exercise [24,30,34,35]. Thus, a prolonged increase in catabolism and decrease in anabolism may lead to a decrease in athletic performance and perhaps result in the athlete overtraining [2,29]. This study was specifically designed to help determine the effects of ADAPT-232S and ADAPT-S on athletes’ testosterone and cortisol levels as well as their ratio during the recovery phase; that is, whether the ratio would return to “normal” levels within 20 hours after each exercise/ competition session. Thus, the efficacy of recovery in athletes was assessed by analyzing the testosterone/cortisol ratios 20 hours after acute, intense exercise or upon completion of tournaments. This index increased to normal values in the two ADAPTS treatment groups, in which it was significantly higher than that of the placebo group. Similarly, we determined that 30 days of supplementation with ADAPT would be sufficient to cause the athletes to enter an excessive anabolic hormonal (i.e., adaptive) state. These results are consistent with our observations in isolated neuroglia cells, where ADAPT-232S down-regulates the adenylate cyclase gene ADC2Y and up-regulates the phosphodiesterase gene PDE4D, suggesting a down-regulation of cAMP that is essential for energy homeostasis as well as for switching from catabolic to anabolic states and vice versa. Down-regulation of cAMP by ADAPT-232S may decrease cAMP-dependent protein kinase A (PKA) activity in various cells, resulting in the inhibition of stress-induced catabolic transformations [10]. | |
| Among well-trained and elite athletes in competitions, a fourweek course of ADAPT-232S or ADAPT-S significantly increased the adaptation to physical and emotional stress compared with placebo. Both compounds significantly decreased inattention, impulsivity, and the perception of stress; reduced fatigue; and increased the anabolic index. Both compounds have good tolerability profiles. Therefore, ADAPT-23S2 and ADAPT-S can be used to increase tolerance for physical and emotional stress and decrease fatigue during the course of exercise and in competitions. | |
| Both compounds have revealed adaptogenic activities. The mechanism of action of adaptogens is known to be associated with mechanisms of sensory and physiological adaptation, defined as the following: | |
| • A change in the excitability of a sense organ during continuous stimulation, resulting in the need for increasingly intense stimuli to produce the same response. | |
| • A biochemical change in an organism that results from exposure to certain environmental conditions or stressors and generates a more effective response to them. | |
| Such adaptive changes alter the organism from its normal steady state (homeostasis) to a heightened level of dynamic equilibrium (heterostasis) or to the so-called state of non-specific resistance (SNSR) [4] of the stress system (a function of the neuroendocrineimmune complex). The general adaptation syndrome has three stages: the alarm reaction, the point at which the body detects the external stimulus; adaptation, during which the body engages defensive countermeasures against the stressor; and exhaustion, where the body begins to run out of defenses [36]. Stress, including both eustress and distress (roughly meaning challenge and overload, respectively) may be the result of negative or positive events. While eustress is essential to life (in the sense, for example, that exercise is required to avoid muscular atrophy), distress can cause disease. | |
| In the mid 1900s, it was suggested that SNSR could be achieved either by gradually training an organism to withstand the effects of the stressor or by introducing biological response modifiers, which are chemicals that could directly raise the organism to a state of heterostasis. Such compounds were given the name “adaptogens” [37]. As originally defined, an adaptogen is a substance that: (a) showed some non-specific effect, such as increasing bodily resistance to physically, chemically, or biologically noxious agents or factors; (b) had a normalizing influence on a pathological state, independent of the nature of that state; and (c) was innocuous and did not disturb body functions at a normal level [4]. | |
| The repeated administration of adaptogens gives rise to an adaptogenic, or stress-protective, effect in a manner analogous to that produced by repeated physical exercise, leading to prolonged SNSR and increased endurance and stamina [38-41]. Such repeat doses of adaptogens have been shown to be of particular value in sports medicine, where the anti-fatigue effects can lead, for example, to increased endurance for long-distance runners during competition and, more importantly, to a more rapid recovery from a strenuous event [42-45]. | |
| A characteristic feature of adaptogens is that they act as eustressors. Thus, a single administration of an adaptogen mainly produces a challenging (stimulating or stress-agonizing) effect [46-49], a finding that is used in sports medicine, where a single dose of an adaptogen can increase the performance of sprinters by making them more alert [50-53]. It follows that the stress-protective effect achieved by multiple administrations of adaptogens is not the result of the inhibition of the stress response of an organism but rather causes adaptive changes in the organism as a response to the repeated stress-agonistic effects of the drug. In other words, using pharmacological terminology, adaptogens are stress-agonists and not stress-antagonists [49]. At the cellular and molecular level adaptogens, specifically ADAPT-232, involve activation of adaptive stress response pathways that result in increased expression of heat-shock proteins, antioxidant enzymes and anti-apoptotic proteins [10,54]. | |
Conclusions |
|
| The results of this study show that supplementation with adaptogenic preparations, ADAPT-S and ADAPT-232S, increases physical performance and the recovery of athletes after heavy physical and emotional loads. Both dietary supplements improve the adaptation to physical and emotional stress. Both ADAPT-232S and ADAPT-S are effective, but the effect of ADAPT-S were superior in respect of anabolic index (Figure 9), blood testosterone (Figure 8) and physical performance index (Figure 10). They significantly decrease inattention, impulsivity and the perception of stress, reduce fatigue, increase the anabolic index and have very good tolerability profiles. The results of this study suggest that ADAPT-232S and ADAPT-S might be useful for recovery of athletes after exercising and for prevention of the symptoms of overtraining. | |
Acknowledgments |
|
| This work was supported in part by the Swedish Herbal Institute Research & Development. | |
| Role of funder: This project was funded by the Swedish Herbal Institute AB. The funder developed and prepared the trial intervention, but had no role in the design, conduct, analysis or preparation of the manuscript of this study. | |
| Trial Registration: This trial was registered at the Yerevan State Medical University (Protocol No 01-2011-10-13) on 13 October, 2011. | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi