Case Report, Clin Oncol Case Rep Vol: 3 Issue: 5
Gemcitabine Cardiotoxicity: Two Case Report and Review of the Literature
Marie Valero1*, Thomas Gilbert1, Chauvenet2, Anissa Bouali3, Marc Bonnefoy1 and Claire Falandry1
1Geriatric Unit, Lyon-Sud University Hospital, Pierre-Bénite, France
2Gastroenterology Unit, Lyon-Sud University Hospital, Pierre-Bénite, France
3Cardiology Unit, Lyon-Sud University Hospital, Pierre-Bénite, France
*Corresponding Author: Marie Valero, Geriatric Unit
Lyon-Sud University
Hospital
Pierre-Benite, France
E-mail: marie.valero@chu-lyon.fr
Received: July 14, 2020 Accepted: September 28, 2020 Published: October 13, 2020
Citation: Valero M, Gilbert T, Chauvenet, Bouali A, Bonnefoy M, et al. (2020) Gemcitabine Cardiotoxicity: Two Case Report and Review of the Literature. Clin Oncol Case Rep 3:5. DOI: 10.37532/cocr.2020.3(5).146
Abstract
Gemcitabine is a chemotherapy treatment that is generally well tolerated, also in elderly patients. In Phases 1 and 2 clinical studies, there was no increased risk of cardiac toxicity. However, in our clinical practice, we have managed two patients in their nineties with cardiovascular events on gemcitabine : one patient with a heart attack and arrhythmia, the other patient in heart decompensation on a paroxysmal arrhythmia. In the literature, there are 15 cases of cardiovascular events on gemcitabine monotherapy and 6 cases on gemcitabine in combination with a platinum salt. The cardiotoxicity of gemcitabine in the elderly is underestimated due to comorbidities and multidrug therapy. Gemcitabine may be a potential cause although the exact mechanism remains unknown. At-risk patients could be targeted for adaptation of infusion modalities by reducing volumes, increasing surveillance and more systematic data reporting.
Keywords: Gemcitabine; Cardiovascular events; Elderly patients with cancer; Cardiotoxicity
Introduction
Gemcitabine is a nucleoside analogue that suppresses DNA synthesis and leads to apoptosis. This antimetabolite is generally well tolerated compared to other chemotherapies. It is therefore used widely in geriatric oncology due to a good safety profile, including in the most vulnerable patients. The most frequently seen side effects are hematological toxicity (thrombocytopenia), digestive disorders (nausea/vomiting) and even an influenza syndrome within 48 hours after infusion [1,2]. In phases 1 and 2 clinical studies with gemcitabine did not show an increased risk of cardiotoxicity [3]. However, in our clinical practice, we have managed two patients who experienced cardiovascular events on gemcitabine. The cardiotoxicity of gemcitabine is therefore potentially underestimated in elderly patients. A review of the literature highlights patient characteristics (age, gender, cardiovascular risk factors) and treatment (monochemotherapy or polychemotherapy with concomitant administration of platinum salts) that could contribute to the occurrence of cardiovascular events.
Case Report
The first clinical case involved an 89-year-old patient with cardiovascular risk factors (hypertension, diabetes on insulin, dyslipidemia, overweight) with a main history of marginal area lymphoma treated by splenectomy and chemotherapy (four R-FC). His active comorbidities included tritroncular heart disease with angioplasty and insertion of a stent eight years earlier. In 2018, discovery of diabetes leading to the diagnosis of localized adenocarcinoma of the pancreas T2N0M0. He was assessed by a geriatrician that found harmonious aging with good nutritional status, without neuro cognitive disorders, but with eight drugs. He was vulnerable on locomotor level with a short physical performance battery to 6/12 showing sarcopenia. In discussion with the patient, medical treatment was preferred to surgical treatment. He received five injections of gemcitabine at 100% dosing and then two injections with a reduction to 75% dosing due to the development of a thrombocytopenia, which is relatively classic with gemcitabine. About ten days after the start of the third cycle (7th injection), he was hospitalized in emergency for anginal pain with troponin positive, which suggested a myocardial attack without ST elevation. Coronary angiography showed tight stenosis of the right coronary artery for which a stent was implanted. A few days later, he was again hospitalized urgently for chest pain, this time revealing a cardiac arrhythmia, with initiation of antiarrhythmic therapy. Thereafter, chemotherapy was continued at 75% of the total dose without any particular incident for a total of four courses of treatment. Finally, he benefited from a final radiochemotherapy with a 60 Gy irradiation potentiated by capecitabine.
The second clinical case involved a 90-year-old patient with treated hypertension and type 2 diabetes on insulin. In the summer of 2018, discovery of a degenerated TIPMP showing three pancreatic lesions. The patient was inaccessible to a surgical treatment due to idiopathic hepatic cirrhosis. He was assessed by a geriatrician that found a patient in intermediate aging with only four drugs, without neurocognitive disorders and with preserved autonomy. On the other hand, he was highly vulnerable from then utritional viewpoint due to undernutrition criteria: weight loss of ten kg, body mass index=18.42kg/cm2, hypoalbuminemia to 24.5 g/L. The pretreatment heart ultrasound was normal. He received four cycles of gemcitabine in medical oncology on day hospital with good safety and efficacy. Decision to continue chemotherapy by home hospitalisation. During the fifth cycle, he developed grade IV dyspnea, which led to his admission to the emergency room and then to hospitalization in cardiology. Clinically, global cardiac decompensation in a context of paroxysmal arrhythmia around 100 bpm at the admission ECG. At that time, the cardiac ultrasound found a simple hypertrophic cardiopathy with a good ventricular ejection fraction. Biology showed a very high NT pro-BNP of 18,000 ng/L and a troponin of 44. At the therapeutic level, introduction of an oral anticoagulant, a betablocker and an antiarrhythmic. The etiologic assessment found no infectious cause, no hydroelectrolytic disorder (no dyskalemia) and no dysthyroidism. There was normocyte anemia with no argument for a hemolytic uremic syndrome. A potential link between gemcitabine and the development of this paroxysmal arrhythmia can therefore be established.
Discussion
Gemcitabine is generally not considered to be a cardiotoxic agent. However, in clinical practice, the cause-and-effect relationship of an anti-cancer agent in the development of acute coronary syndrome or arrhythmia is still difficult to establish due to the comorbidities of each and the complex treatment pathways of chemotherapies. On the one hand, there are factors attributable to patient characteristics (age and cardiovascular risk factors). On the other hand, there are factors attributable to the drug, such as anthracyclines [4] or the combined chemotherapy [5,6]. Early phase clinical trials with gemcitabine monotherapy doesn’t show a risk of increased cardiac toxicity. Indeed, in phase 1 clinical trials with gemcitabine treatment showed cardiac arrhythmia in 0.7% to 1.4% of patients and a significant reduction in VG ejection function in 0.2% of patients [7]. Aapro et al. reviewed 22 clinical trials in phase 2 that reported an incidence of 0.5% for acute coronary syndromes and 0.2% for rhythm disorders [8]. Patients with pre-existing cardiovascular disease may have had to be excluded from these early phase studies or may not have been subject to special intensive monitoring.
In the literature, there are only fifteen case reports (71.4%) with cardiovascular events on gemcitabine monotherapy and six cases in combination with platinum salt (28.6%). The concomitant use of a platinum salt with gemcitabine could therefore be an additional risk factor. Patients were between 43 and 82 years old with a median age of 64.5 years and 38% > 70 years. The acute cardiovascular events reported were : acute coronary syndrome [4] coronary vasospasm [2] atrial fibrillation [6] congestive heart failure [2] and heart failure [7] (Table 1).
| Age | Gender | Chemotherapy | Cardiovascular event | History cardiovascular risk factor |
|---|---|---|---|---|
| 54 | M | Gemcitabine | 1 Heart attack | 0 |
| 70 | M | Gemcitabine | 1 Arrhythmia | 0 |
| 78 | M | Gemcitabine | 1 Arrhythmia | 0 |
| >70 | M | Gemcitabine | 0 | 0 |
| >70 | M | Gemcitabine navelbine | 1 Tox grade 1 | Severe heart disease : |
| 1 Tox grade 2 | One heart attack history | |||
| 4 Tox grade 3 (heart failure or arrhythmia) | High blood pressure + heart attack | |||
| 82 | W | Gemcitabine | Heart failure | ? |
| Gemcitabine | 4 Pericarditis | Radiation history | ||
| 58 | M | Gemcitabine carboplatine | 1 Heart attack | ? |
| 43 | W | Gemcitabine | 1 Heart attack+ventricular tachycardia | Coronary history |
| 72 | W | Gemcitabine | 1 Arrhythmia | 0 |
| 73 | W | Gemcitabine | 1 Arrhythmia | 0 |
| 65 | M | Gemcitabine | 1 Arrhythmia | 0 |
| 59 | W | Gemcitabine | 1 Coronary vasospasm | Coronary history |
| 67 | W | Gemcitabine | 1 Supra ventricular tachycardia | 0 |
| 48 | W | Gemcitabine cisplatine | 1 Heart attack with death | 0 |
| 56 | M | Gemcitabine | 1 Congestive heart failure | 0 |
| 156 W | M | Gemcitabine | 7 Congestive heart failure | Coronary history+diabetes |
| 77 | M | Gemcitabine carboplatine | 1 Heart failure | History of high blood pressure, dyslipidemia, smoking |
| 64 | W | Gemcitabine carboplatine | 1 Tako-tsubo | 0 |
| 67 | W | Rituximab gemcitabine oxaliplatine | 1 Heart failure | 0 |
| 52 | M | Gemcitabine cisplatine | 1 Heart failure | History of high blood pressure, diabetes, arrhythmia |
| 62 | M | Gemcitabine | 1 Heart failure | History of coronary and high blood pressure |
| 63 | M | Gemcitabine | 1 Heart failure | 0 |
| 72 | W | Gemcitabine | 1 Heart failure | History of high blood pressure, diabetes, dyslipidemia |
| 73 | M | Gemcitabine | 1 Congestive heart failure | History of paroxysmal arrhythmia |
Table 1: Clinical cases identifying cardiovascular events in patients who have received gemcitabine as monotherapy or in combination.
Acute coronary syndrome: Kalapura et al. described the case of a 54-year-old patient with no cardiovascular history who developed an acute coronary syndrome six hours after the fifth cycle of gemcitabine [9]. Later, Bdair et al. described the case of a 43 year-old patient with a history of coronary artery disease who developed an acute coronary syndrome three days after the gemcitabine infusion [10].
There are several hypotheses to explain the pathophysiology: A coronary spasm as classically described for 5FU [11] thrombotic microangiopathies [12,13] pro-inflammatory factors damaging the endothelium.
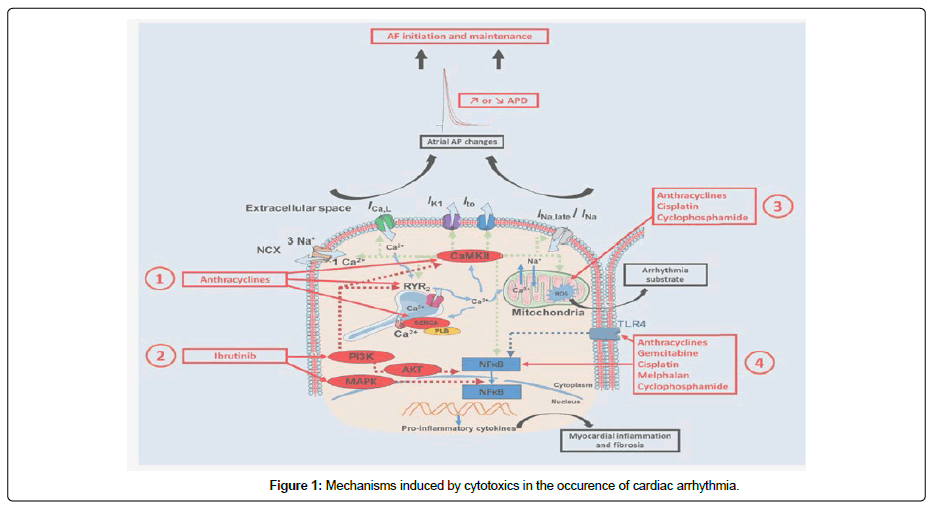
Cardiac arrhythmia: Ciotti et al. described the occurrence of atrial fibrillation in a 70-year-old man [14]. He proposed as an explanation an inflammatory pathogenic mechanism mediated by the release of cytokines that would lead to the proliferation of myofibroblasts and collagen in the left atrium [14]. Ferrari et al. described the occurrence of atrial fibrillation on gemcitabine in two women aged 72 and 73 years [15]. The deaminated metabolite of gemcitabine, di fluorodeoxy- uridine with a half-life of 24 hours on average, could have a direct effect on myocardial cells and the conduction system inducing oversimulation of the sino atrial node (Figure 1).
Finally, another hypothesis would be the activation of the nuclear factor kappa B which could induce long-term cardiac remodelling.
Conclusion
Gemcitabine should be considered as a possible cause of cardiovascular events, although its mechanism is still unknown. However, its use in elderly patients with cardiovascular risk factors should not be called into question but rather lead to increased interactions between oncologists, geriatricians and cardiologists. Despite a frequency of cardiovascular events being low on gemcitabine, high-risk patients could be subject to an adaptation of their infusion modality with volume reduction, and intensified monitoring of clinical and biological indicators (troponin and BNP) as well as a more systematic reporting of events.
References
- Tonato M, Mosconi AM, Martin C (1995) Safety profile of gemcitabine. Anticancer Drugs 6: 27-32.
- Green MR (1996) Gemcitabine safety overview. Semin Oncol 23: 32-35.
- Khan MF, Gottesman S, Boyella R, Juneman E (2014) Gemcitabine-induced cardiomyopathy: A case report and review of the literature. J Med Case Rep 8: 220
- Alexandre J, Moslehi JJ, Bersell KR, Brentano CF, Roden DM, et al. (2018) Anticancer drug-induced cardiac rhythm disorders: Current knowledge and basic underlying mechanisms. Pharmacol Ther 189: 89-103
- Gridelli C, Cigolari S, Gallo C, Manzione L, Ianniello GP (2001) Activity and toxicity of gemcitabine and gemcitabinevinorelbine in advanced non small-cell lung cancer elderly patients. Phase II data from the Multicenter Italian Lung Cancer in the Elderly Study (MILES) randomized trial. Lung Cancer 31: 277-284.
- Mohebali D, Matos J, Chang JD (2017) Gemcitabine induced cardiomyopathy: A case of multiple hit cardiotoxicity. ESC Heart Failure 4: 71-74.
- Storniolo AM, Allerheiligen SR, Pearce HL (1997) Preclinical, pharmacologic, and phase I studies of gemcitabine. Semin Oncol 24: s2-s7.
- Aapro MS, Martin C, Hatty S (1998) Gemcitabine-a safety review. Anticancer Drugs 9: 191-201.
- Kalapura T, Krishnamurthy M, Reddy CV (1999) Acute myocardial infarction following gemcitabine therapy. A case report. Angiologie 50: 1021-1025.
- Bdair FM, Graham SP, Smith PF, Javle MM (2006) Gemcitabine and acute myocardial infarction-a case report. Angiology 57: 367-371.
- Katircibasi MT, Eken A (2017) Gemcitabine-induced coronary vasospasm: A case report. Turk Kardiyol Dern Ars 45: 172-175.
- Duvic M, Talpur R, Wen S, Kurzrock R, David CL (2006) Phase II evaluation of gemcitabine monotherapy for cutaneous T-cell lymphoma. Clin Lymphoma Myeloma 7: 51-58.
- Dasanu CA (2008) Gemcitabine: Vascular toxicity and prothrombotic potential. Expert Opin Drug Saf 7: 703-716.
- Ciotti R, Belotti G, Facchi E, Cantu A, D'Amico A (1999) Sudden cardio-pulmonary toxicity following a single infusion of gemcitabine. Ann Oncol 10: 997.
- Ferrari D, Carbone C, Codecà C, Fumagalli L, Gilardi L, et al. (2006) Gemcitabine and atrial fibrillation: A rare manifestation of chemotherapy toxicity. Anticancer Drugs 17: 359-361.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi