Research Article, Jftte Vol: 14 Issue: 2
Green Extraction of Cellulose from Waste Olive Wood Using Microwave Assisted Alkali Treatment
Mohammad Mahbubul Alam1,2*, Antonio Greco2 and Carola Esposito Corcione2
1Department of Textile Engineering, Ahsanullah University of Science and Technology, Dhaka, Bangladesh
2Department of Innovation Engineering, University of Salento, Lecce, Italy
*Corresponding Author: Mohammad Mahbubul Alam
Department of Textile Engineering, Ahsanullah University of Science and Technology, Dhaka, Bangladesh
E-mail: mahbub_tex@aust.edu
Received: 02-Mar-2026, Manuscript No. jftte-26-186275
Editor assigned: 04-Mar-2026, PreQC No. jftte-26-186275 (PQ)
Reviewed: 18-Mar-2026, QC No.jftte-26-186275
Revised: 24-Mar-2026, Manuscript No. jftte-26-186275 (R),
Published: 30-Mar-2026, DOI: 10.4172/2329-9568.1000402
Citation: Alam M, Greco A, Corcione CE (2026) Green Extraction of Cellulose from Waste Olive Wood Using Microwave Assisted Alkali Treatment. J Fashion Technol Textile 14: 402
Abstract
There has been a growing emphasis placed on the development of environmentally friendly methods for extracting cellulose from natural sources over the course of the past several years. Waste olive wood is a resource that is both abundant and sustainable, and it has the potential to be utilized for the extraction of cellulose through the utilization of a microwave assisted extraction (MAE) technique that is also environmentally friendly. Obtaining cellulose from waste olive wood in a manner that is both environmentally acceptable and effective through the utilization of alkali as a primary component is the purpose of this effort, which aims to develop an MAE process. The extraction process was carried out at 90°C and 100°C for 20 minutes. Where the maximum amount of cellulose obtained yield of 48.64% at 100°C, and the crystallinity index was found to be 62.58%. The microwave with alkali was able to successfully remove the cellulose from waste olive wood, as demonstrated by the scanning electron microscopy (SEM) image, the Fourier transform infrared (FT-IR) analysis, and the thermogravimetric analysis (TGA). In compared to more traditional extraction methods, microwave technology has significantly reduced the amount of time required for extraction as well as the amount of energy that is consumed. As a result, the process has become more environmentally friendly and efficient. The cellulose that has been extracted has the potential to be utilized in a variety of industries, such as the textile industry, the packaging industry, and the biomedical industry. The findings of this study indicate that it is feasible to obtain cellulose of superior quality from olive wood that has been discarded
Keywords: Extraction; Microwave; Olive wood; Cellulose; Alkali; Sustainable
Introduction
Cellulose is the most common biopolymer on Earth and is very important in many industries, such as textiles, paper, medicines, and biofuels. Cellulose is a nearly limitless polymeric raw material since it is the most abundant component in most plants. The availability, renewability and biodegradability of this material, as well as its low cost, are the major benefits, but it has two major drawbacks when compared to synthetic polymers: high hygroscopicity, owing to hydroxyl groups, and limited processing due to rapid disintegration [1,2]. Cellulose can be derived from a variety of natural sources, including wood (which is classified as lignocellulosic biomass), agricultural trash, cotton, flax, hemp, sisal, and vegetable leftovers in particular. Wood pulp is the most widely used raw material for the processing of cellulose, particularly for the production of paper and cardboard [3]. It is commonly known that cellulose, extracted from spruce, pine, and other plants, is used to make viscose, lyocell, modal, cellulose acetate, and cellulose triacetate. The main industrial uses of cellulose derivatives are cellulose esters and ethers, which are used in molding, extrusion, and films and in construction materials, food, personal care items, paints, and pharmaceuticals. This form of cellulose was difficult to prepare and use in biodegradable plastic sheets until solvent methods were developed. Plasticization and mixing with other polymers change cellulose's mechanical and chemical properties [4]. Cellulose fibers and derivatives are utilized to manufacture biopolymer composite fillers and matrices. Recent biocomposite research has replaced petroleum-based polymers (PE and PP) with biopolymers like cellulose and starch, and glass fibers with cellulose [5]. Therefore, it became necessary to identify appropriate uses for the leftover olive wood coming from damaged trees. Olive tree pruning (OTP) biomass contains cellulose, hemicelluloses, lignin, extractives, and ash like other lignocellulosic biomasses. The chemical composition of OTP biomass depends on tree age, soil type, and climate. These biomaterials have several uses due to their characteristics. As is known, cellulose can be harvested and used to make biodegradable polymers, textiles, and paper. However, current cellulose extraction methods involve harmful chemicals and high temperatures, polluting the environment [6].
The conventional cellulose extraction procedure involves a 4-–6-hour alkaline treatment (NaOH/KOH) and a 1-–2-hour bleaching (NaClO/NaOCl2). As hemicelluloses bound to cellulose are separated during bleaching, the result is whiter. An alkaline procedure that entirely breaks down lignin is usually best bleached with acidified sodium chlorite (NaClO2) [7]. In addition to the conventional way of heating, microwave heating is an additional method that can speed up chemical reactions. This is achieved by the item that is being heated by microwaves coming into direct contact with the electromagnetic field that is being used. This results in a heating process that is both more fast and more volumetric than that of conventional heating methods. In addition to this, it provides further advantages, such as improved selectivity and homogeneity, while simultaneously using less energy for the processing of biomass [8]. Microwave-assisted alkaline action and decolorization cause cellulose to swell, as well as the breakdown of hemicellulose, lignin, and a variety of non-cellulosic and non-crystalline impurities that are present in olive wood. Chemical treatments that are supported by microwaves, such as bleaching and alkali treatment, bring about a reduction in the quantities of hemicellulose and lignin, which ultimately leads to an increase in crystallinity and an improvement in thermal properties. The utilization of microwave irradiation in conjunction with alkali treatment has the potential to quickly accelerate the chemical reactions that are responsible for the removal of lignin and the degradation of hemicellulose [9]. Microwave heating raises molecules' temperatures quickly by electric dipole movement and ion conductivity. Microwaving works by rapidly moving hydrophilic molecules, which collide to generate heat. Higher power levels increase energy and temperature, which damages fibers and degrades lignin [10].
Due to its vast natural sources and unique microstructure and characteristics, cellulose has great promise for food packaging. However, the cellulose network's complex hydrogen connections between and within molecules make dissolving and processing difficult. Many eco-friendly solvents have been developed to address this issue. Cellulose-based food packaging films made with these new solvent solutions have excellent mechanical properties and barrier properties [11,12]. The current research was conducted with the intention of isolating cellulose from waste olive wood tree by means of a chemically applied microwave treatment. In addition, the investigation encompassed the technological-functional qualities and characterization of the recovered cellulose.
Experimental
Materials and Methods
Raw materials
The waste olive wood came from Puglia, Italy, where “Xylella” bacterium killed olive trees.
The collected wood was initially cut into small pieces in a local factory. The wood was ground using a milling machine (Retsch, Type ZM 100, F.Kurt Retsch GmbH & Co. KG, Germany) to achieve a particle size of 0.25 mm and preserve in airtight polyethylene bags at room temperature until ready for use.
The chemicals utilized in the experiment were analytical grade and used without undergoing any prior purification. These chemicals include sodium hydroxide pellets (≥ 98.00%, Honeywell/Fluka, Germany), sodium chlorite (80.00%, Honeywell/Fluka, Germany), sulfuric acid (95–97%, Honeywell/Fluka, Germany), and acetic acid (≥99.80%, Honeywell/Fluka, Germany).
Method
Alkaline treatment
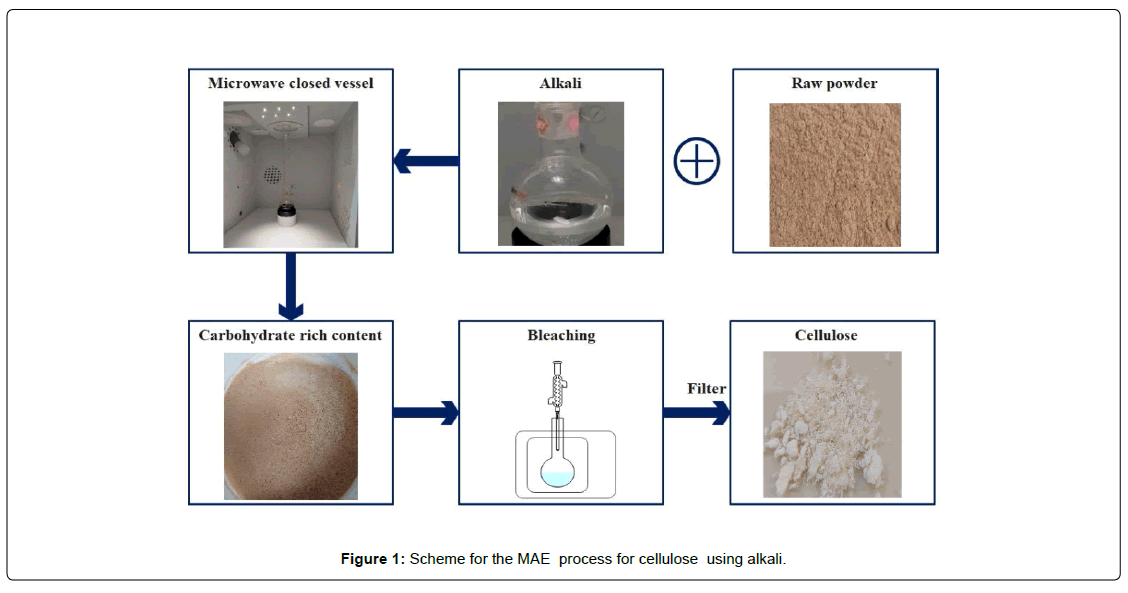
The olive wood powder was first subjected to treatment in NaOH alkaline solution (7% w/v) by using a microwave with open vessel configuration in a round bottom flask. The waste OWP was approximately 1.50 grams and placed into the sealed flask. The reaction was conducted for 20 minutes at temperature of 90 0C and 100 0C, where the temperature was increased in 100C/min. The maximum operating power of the microwave was 200 W. The duration was constant for both extraction techniques. After the extraction procedure, the vessel was cooled down to a lower temperature. The extracted components were passed through a glass microfiber filter (MFV3; filter lab) to separate the solid component. The remaining liquid (black liquor), was further separated to collect the lignin. The solid component underwent three rounds of washing with distilled water in order to get a neutral pH level. The material was thereafter subjected to dehydration in a 60 °C oven for a period of 24 hours, followed by storage at room temperature for the subsequent procedures. Figure 1 shows the Scheme for the MAE of cellulose using alkali. [Figure: 1]
Bleaching Treatment
The alkaline treated samples were subjected to bleaching using 1.5% (w/v) sodium chlorite and 0.5% (w/v) acetic acid in a microwave. The pH of the solution was adjusted to a range 4–5 by using acetic acid. The reaction mixture was exposed to microwave irradiation at 70 °C for 15 min, using a maximum power of 200 W. At the end of the irradiation, the resultant solid material was filtered under vacuum and then washed three times with distilled water until it reached a neutral pH. The cellulose product was then dried and stored for the further steps.
Characterization
Scanning Electron Microscopy (SEM)
The surface profiles of extracted cellulose were assessed using a JEOL JSM-840 scanning electron microscope (Peabody, MA, USA) at an acceleration voltage of 5.00 kV. Prior to assessment, the samples were coated with a coating of gold to enhance their electrical conductivity. This was achieved using a SCD 004 Balzers sputter coater (manufactured by Bal Tec. AG, Fürstentum, Lichtenstein) in a vacuum environment. The other samples including lignin were analyzed by using Zeiss E Evo 40 (Oberkochen, Germany). The images were recorded at a magnification of 100x.
Fourier Transform Infra-Red (FTIR)
The extracted cellulose and lignin samples were analyzed using a Bruker Analitik IFS 66 FTIR spectrometer (Ettlingen, Germany) with an ATR adapter to get ATR-FTIR spectra. The spectra were obtained by measuring absorbance mode from 4000 to 500 cm-1, using 64 scans and a resolution of 4 cm-1.
X-ray Diffraction (XRD)
The X-ray diffraction (XRD) patterns of the extracted cellulose were determined by using a Bruker (Billerica, MA, USA) D8-Advance diffractometer. The diffractometer was fitted with a Goebel mirror to handle samples that are not flat, a high-temperature chamber capable of achieving temperatures of up to 900 °C, a KRISTALLOFLEX K760–80F X-ray generator with a power output of 3000 W, a voltage range of 20–60 kV, and a current range of 5–80 mA. The arrangement used an X-ray tube with a copper anode. The data were collected using Cu-Kα radiation with a wavelength of 1.5406 Å at room temperature. The scattering angles (2θ) ranged from 2.5° to 80°, with a step size of 0.05° per minute. The crystallinity index of cellulose was calculated using the Segal technique by the following equation:
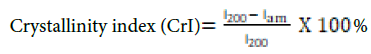
Where I200 represent the highest intensity of the X-ray diffraction peak at an angle of 2θ = 22–23°, and Iam represent the lowest intensity of the peak at an angle of 2θ = 18°, which indicates the presence of the amorphous area.
Thermogravimetric Analysis
The TGA/SDTA 851 Mettler Toledo equipment was used to conduct thermogravimetric analysis (TGA). The dynamic tests were conducted across a temperature range of 25 to 700 0C, with a heating rate of 10 0C/min. Experiments were conducted under nitrogen atmosphere with a flow rate 50 ml/min. Approximately 3 mg of sample were used to measure.
Results and discussion
Cellulose Content
The microwave extraction process was carried out at two different temperatures: 90 0C and 100 0C. At these temperatures, the yield of cellulose was obtained 37.34% and 42.36% respectively. The fact that this is the case shows that there is a positive connection between temperature and cellulose yield, which means that the quantity of cellulose yield increases in proportion to the temperature that was being raised. Following the extraction of cellulose, it was subjected to bleaching using sodium chlorite at a constant temperature of 70 0C for all the samples. The cellulose yield after bleaching was 34.23% and 40.16% at temperatures of 90 0C and 100 0C, respectively. There was a little reduction in the quantity of cellulose recovered post-bleaching, in comparison to the amount collected during the extraction procedure. One possible explanation for the drop in yield is that the bleaching process, which involves removing color, residual lignin and impurities from the extracted cellulose, is responsible for the reduction in yield [13] [Table: 1].
| Microwave Extraction | Alkali Concentration | Solid residue (Cellulose) Yield (%) | Bleaching | Cellulose (%) |
|---|---|---|---|---|
| Temp. 90 °C Time: 20 min Ratio: 1:20 Ramp: 10 min |
7 wt% NaOH (w/v) | 37.34 | Sodium Chlorite: 1.5 % Temp. 70 °C Time: 15 min |
34.23 |
| Temp. 100 °C Time: 20 min Ratio: 1:20 Ramp: 10 min |
7 wt% NaOH (w/v) | 42.36 | 40.16 |
Table 1: Cellulose content by using MAE with alkali
SEM Analysis
The SEM micrographs reveal substantial morphological alterations between the raw olive wood powder and the cellulose derived from microwave-assisted alkali treatment. Figure (a) shows that the untreated olive powder has a compact, irregular, and stiff structure with a reasonably smooth and dense surface. This means that the cellulose fibers are tightly embedded in the lignin and hemicellulose matrix of the lignocellulosic biomass. Because of this continuous matrix, it's harder to see individual fibrils, and the shape is less porous [14,15]. In contrast, Figure (b) demonstrates a more fibrillated, rough, and porous structure after microwave-assisted alkaline extraction. The breakdown of the compact structure and the formation of long fiber bundles and visible microfibrils show that lignin and hemicellulose have been effectively removed [16]. Heating with a microwave makes it easier for alkali to get into the biomass, which breaks down the cell walls and makes it easier to separate the cellulose fibers. Similar morphological changes have been documented in the literature after the alkaline pretreatment of lignocellulosic biomass, wherein the elimination of non-cellulosic components results in the exposure of cellulose fibrils and an enhancement of surface roughness [17]. These changes in structure show that cellulose was successfully taken out of olive wood powder using microwave-assisted alkaline treatment [Figure: 2].
FTIR Analysis
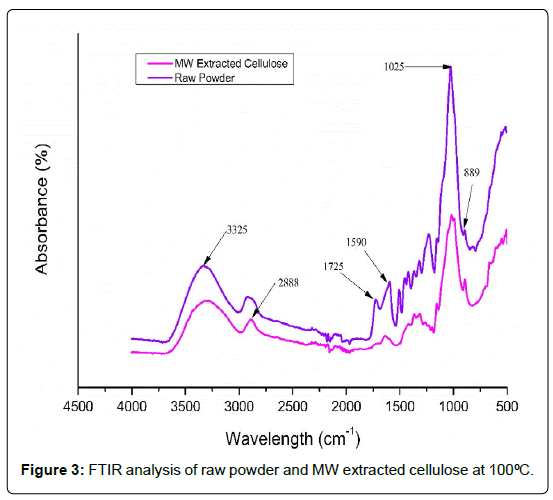
The FTIR spectra of raw olive wood powder and cellulose extracted using microwaves exhibit distinct chemical changes that suggest that non-cellulosic parts have been removed. The wide absorption band at 3325 cm-1 is due to the O–H stretching vibration of hydroxyl groups in cellulose, hemicellulose, and lignin [18,19]. This band gets a little sharper in the cellulose that was taken out, which means that the hydrogen bonds in the cellulose structure are stronger after the surrounding matrix elements have been taken away [20]. The peak around 2888 cm-1 is due to C–H stretching vibrations of aliphatic groups in polysaccharides [21]. In the raw powder, there is a clear peak around 1725 cm-1. This peak is linked to the C=O stretching of acetyl and uronic ester groups found in hemicellulose and the ester linkages of lignin. The fact that this peak is lower or gone in the treated sample suggests that hemicellulose was effectively removed during alkaline microwave treatment [6]. The band at 1590 cm-1 is due to aromatic skeletal vibrations of lignin. Its lower intensity in the extracted cellulose shows that some of the lignin has been removed [19,22]. The strong peak at 1025 cm-1 is due to the C–O–C and C–O stretching vibrations of the β-1,4-glycosidic bonds in cellulose [23,24]. This peak gets stronger following extraction, which shows that cellulose has been enriched. The band about 889 cm-1, which is typical of β-glycosidic connections in cellulose, further supports the idea that the structure of cellulose is still there after treatment [25] [Figure: 3].
XRD Analysis
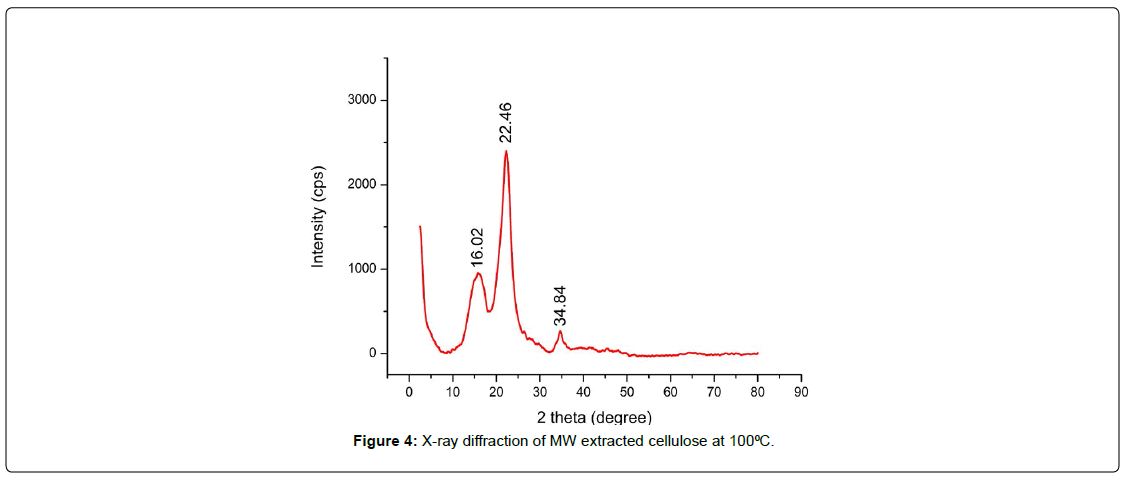
The X-ray diffraction (XRD) pattern reveals clear peaks at 2θ values of 16.02°, 22.46°, and 34.84°. These peaks are typical of the crystalline regions of cellulose I, which is the native crystalline form of cellulose found in plants [26]. The prominent peak at 22.46°, which corresponds to the (200) plane, shows that the sample is quite crystalline. The peaks at 16.02° and 34.84°, on the other hand, relate to the (110) and (004) planes, respectively [27]. The crystallinity index was obtained by MAE using the alkali is 62.58%. This effect can be caused by the greater structural changes caused by microwaves during cellulose extraction, joined to the presence of some amounts of lignin and hemicellulose in the cellulose structure , resulting in overall lower crystallinity [28]. Moreover, microwave with alkali treatment can lead to the breakage of ester linkage bonds between lignin and polyuronic, resulting in partial dissolution of the binding material. This process enhances the exposure of cellulose, subsequently promoting the crystallization of the treated fibers [29]. The fact that these peaks are there and crisp means that the cellulose has been successfully extracted and purified, with most of the amorphous parts, including lignin and hemicellulose, being eliminated during processing. This XRD pattern matches what has been reported in the literature for cellulose I, which shows that the extraction and purification process used worked [30] [Figure: 4].
Thermal Analysis
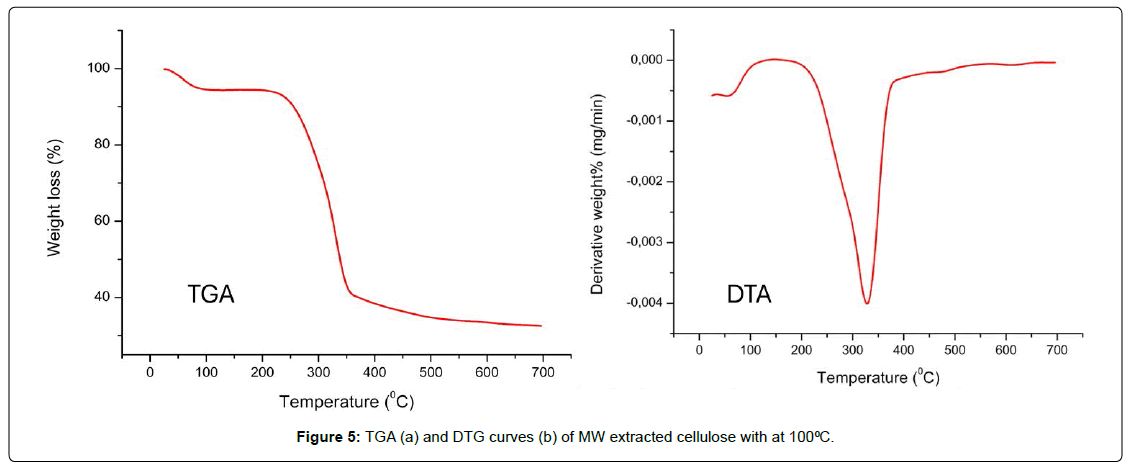
The TGA curve shows a minor loss of mass at temperatures below about 100–120 °C. This is because the physically adsorbed moisture and bound water molecules in the cellulose structure are evaporating [31]. After this point, the curve stays relatively constant until about 260–280 °C. This means that the cellulose that was taken out is thermally stable in this range [32]. The principal degradation stage starts at about 280 °C, when the mass drops quickly and sharply. This continues until about 360–380 °C [33]. This stage is the heat depolymerization of cellulose, which breaks β-1,4-glycosidic bonds, causes dehydration processes, and makes volatile compounds such levoglucosan. The DTA curve has a clear peak at about 340–350 °C, which is the maximum decomposition temperature (Tmax) and demonstrates the fastest rate of thermal breakdown of the cellulose backbone [34]. When the temperature goes beyond 400 °C, the rate of weight loss slows down, which means that carbonization and the generation of char residue are happening slowly. At 700 °C, a significant portion of residual mass persists, likely due to carbonaceous char and potential inorganic or mineral residues either from the alkali extraction process or the initial biomass [35]. In general, the fact that there is only one primary degradation peak and one distinct main decomposition zone shows that the sample is mostly cellulose with little hemicellulose and lignin [26] [Figure: 5].
Conclusion
Microwave assisted alkali extraction is a very efficient method for isolating and improving the quality of cellulose extracted from waste olive wood powder. In the raw material, the structure was dense and uneven; however, following extraction, the cellulose fibers were well separated and fibrillar. This transformation is revealed by the SEM photographs. The elimination of non-cellulosic components, such as lignin and hemicellulose, as well as the enrichment of cellulose-specific functional groups, are both confirmed by FTIR spectra. Further evidence that these findings are accurate comes from X-ray diffraction examination, which reveals unique crystalline peaks that are characteristic of cellulose I and indicate great crystallinity and purity. Thermal analysis using TGA and DTA demonstrates that the extracted cellulose is thermally stable. The extracted cellulose has a sharp breakdown profile, which is typical of purified cellulose, and extremely little residue, which indicates that there are no contaminants present. These data, taken as a whole, provide evidence that the microwave-assisted alkali technique is capable of producing cellulose that is of a high purity, crystallinity, and thermal stability, which makes it appropriate for a variety of advanced applications.
Microwave assisted extraction has downsides despite its efficacy. Uneven heating throughout the sample can reduce extraction effectiveness and degrade sensitive chemicals. Microwave penetration depth and the necessity for specialized equipment make scaling MAE for industrial applications difficult. MAE solvents and reagents may not work with microwave energy, limiting extraction conditions. The rapid and intense heating might also produce undesirable byproducts or partially degrade the target material. These characteristics emphasize the need of optimizing MAE for cellulose extraction and other processes.
References
- Vinhas S, Sarraguça M, Moniz T, Reis S, Rangel M (2024). A New Microwave-Assisted Protocol for Cellulose Extraction from Eucalyptus and Pine Tree Wood Waste. Polymers. 16-20.
- Klunklin W, Hinmo S, Thipchai P, Rachtanapun P (2023). Effect of Bleaching Processes on Physicochemical and Functional Properties of Cellulose and Carboxymethyl Cellulose from Young and Mature Coconut Coir. Polymers. 15-3376.
- Ferrari F, Striani R, Fico D, Alam M M, Greco A, et al (2022). An Overview on Wood Waste Valorization as Biopolymers and Biocomposites: Definition, Classification, Production, Properties and Applications. Polymers. 14-5519.
- Tanase-Opedal M, Espinosa E, Rodríguez A, Chinga-Carrasco G (2019). Lignin: A Biopolymer from Forestry Biomass for Biocomposites and 3D Printing. Materials. 12-3006.
- Shaghaleh H, Xu X, Wang S (2018). Current progress in production of biopolymeric materials based on cellulose, cellulose nanofibers, and cellulose derivatives. RSC Adv. 8: 825–842.
- Alam M M, Greco A, Corcione C E, Jiménez A, Garrigós M C (2024). Microwave-Assisted Extraction of Cellulose from Waste Olive Wood Powder Using Deep Eutectic Solvents.
- Akhlaq M, Uroos M (2023). Evaluating the Impact of Cellulose Extraction via Traditional and Ionosolv Pretreatments from Domestic Matchstick Waste on the Properties of Carboxymethyl Cellulose. ACS Omega. 8: 8722–8731.
- Bahndral, Shams R, Dash K K, Ali N A, Shaikh A M, et al (2024). Microwave assisted extraction of cellulose from lemon grass: Effect on techno-functional and microstructural properties. Journal of Agriculture and Food Research. 16-101170.
- Sweygers N, Harrer J, Dewil R, Appels L (2018). A microwave-assisted process for the in-situ production of 5-hydroxymethylfurfural and furfural from lignocellulosic polysaccharides in a biphasic reaction system. Journal of Cleaner Production. 187: 1014–1024.
- Sudiana I N, Mitsudo S, Nishiwaki T, Susilowati P E, Lestari L, et al (2016). Synthesis and Characterization of Microwave Sintered Silica Xerogel Produced from Rice Husk Ash. J Phys: Conf Ser. 739-012059.
- Liu X, Qin Z, Ma Y, Liu H, Wang X (2023). Cellulose-Based Films for Food Packaging Applications: Review of Preparation, Properties, and Prospects. JRM. 11: 3203–3225.
- Alam M M, Greco A, Rajabimashhadi Z, Esposito Corcione C (2024). Efficient and environmentally friendly techniques for extracting lignin from lignocellulose biomass and subsequent uses: A review. Cleaner Materials. 13-100253.
- Klunklin W, Hinmo S, Thipchai P, Rachtanapun P (2023). Effect of Bleaching Processes on Physicochemical and Functional Properties of Cellulose and Carboxymethyl Cellulose from Young and Mature Coconut Coir. Polymers (Basel). 15-3376.
- Alam M M, Greco A, Corcione C E, Jiménez A, Garrigós M C (2024). Microwave-Assisted Extraction of Cellulose from Waste Olive Wood Powder Using Deep Eutectic Solvents.
- Jonoobi M, Oladi R, Davoudpour Y, Oksman K, Dufresne A, et al (2015). Different preparation methods and properties of nanostructured cellulose from various natural resources and residues: a review. Cellulose. 22: 935–969.
- Chen W, Yu H, Liu Y, Chen P, Zhang M, et al (2011). Individualization of cellulose nanofibers from wood using high-intensity ultrasonication combined with chemical pretreatments. Carbohydrate Polymers. 83: 1804–1811.
- Han W, Geng Y (2022). Optimization and characterization of cellulose extraction from olive pomace.
- Javier-Astete R, Jimenez-Davalos J, Zolla G (2021). Determination of hemicellulose, cellulose, holocellulose and lignin content using FTIR in Calycophyllum spruceanum (Benth.) K. Schum. and Guazuma crinita Lam. PLOS ONE. 16-e0256559.
- Souissi S, Lachtar F, Elloumi A, Bergeret A (2022). Properties of wood polymer composites based on polypropylene/olive wood flour: effects of fiber treatment and compatibilizer. Iran Polym J. 31: 1511–1521.
- Zhuang J, Li M, Pu Y, Ragauskas A J, Yoo C G (2020). Observation of Potential Contaminants in Processed Biomass Using Fourier Transform Infrared Spectroscopy. Applied Sciences. 10-4345.
- Traoré M, Kaal J, Martínez Cortizas A (2018). Differentiation between pine woods according to species and growing location using FTIR-ATR. Wood Sci Technol. 52: 487–504.
- Hachaichi, Kouini B, Kian L K, Asim M, Jawaid M. (2021) Extraction and Characterization of Microcrystalline Cellulose from Date Palm Fibers using Successive Chemical Treatments. J Polym Environ. 29: 1990–1999.
- Sánchez-Gutiérrez M, Espinosa E, Bascón-Villegas I, Pérez-Rodríguez F, Carrasco E (2020). Production of Cellulose Nanofibers from Olive Tree Harvest—A Residue with Wide Applications. Agronomy. 10-696.
- Ndruru S T C L, Wahyuningrum D, Bundjali B, Arcana I M (2019). Green simple microwave-assisted extraction (MAE) of cellulose from Theobroma cacao L (TCL) husk. IOP Conf Ser: Mater Sci Eng. 541-012017.
- Singh S, Gaikwad K K, Park S I, Lee Y S(2017). Microwave-assisted step reduced extraction of seaweed (Gelidiella aceroso) cellulose nanocrystals. Int J of Bio Macromolecules. 99: 506–510.
- Kian L K, Saba N, Jawaid M, Alothman O Y, Fouad H (2020). Properties and characteristics of nanocrystalline cellulose isolated from olive fiber. Carbohydrate Polymers. 241-116423.
- Kale R D, Bansal P S, Gorade V G (2018). Extraction of Microcrystalline Cellulose from Cotton Sliver and Its Comparison with Commercial Microcrystalline Cellulose. J Polym Environ. 26: 355–364.
- Isci, Erdem G M, Bagder Elmaci S, Sakiyan O, Lamp A, et al (2020). Effect of microwave-assisted deep eutectic solvent pretreatment on lignocellulosic structure and bioconversion of wheat straw. Cellulose. 27: 8949–8962.
- Djalal M, Nafissa M, Mansour R, Jawaid M, Hocine M, et al (2024). Effect of alkali treatment on new lignocellulosic fibres from the stem of the Aster squamatus plant. J Mater Res Technol. 32: 2882–2890.
- Morán J I, Alvarez V A, Cyras V P, Vázquez A (2008). Extraction of cellulose and preparation of nanocellulose from sisal fibers. Cellulose. 15: 149–159.
- Garcia-Maraver, Salvachúa D, Martínez M J, Diaz L F, Zamorano M (2013). Analysis of the relation between the cellulose, hemicellulose and lignin content and the thermal behavior of residual biomass from olive trees. Waste Management. 33: 2245–2249.
- Nurazzi N M, Asyraf M R M, Rayung M, Norrrahim M N F, Shazleen S S, et al (2021). Thermogravimetric Analysis Properties of Cellulosic Natural Fiber Polymer Composites: A Review on Influence of Chemical Treatments. Polymers. 13-2710.
- Thomason J L, Rudeiros-Fernández J L (2021). Thermal degradation behaviour of natural fibres at thermoplastic composite processing temperatures. Polym Degrad Stab. 188-109594.
- Burnham A K, Zhou X, Broadbelt L J (2015). Critical Review of the Global Chemical Kinetics of Cellulose Thermal Decomposition. Energy Fuels. 29-2906–2918.
- Taflick T, Schwendler L A, Rosa S M L, Bica C I D, Nachtigall S M B (2017). Cellulose nanocrystals from acacia bark–Influence of solvent extraction. Int J Biol Macromol. 101: 553–561.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi