Review Article, J Clin Exp Radiol Vol: 1 Issue: 1
Hibernoma: Imaging Review and Management
Bilal Mujtaba*, Sameh M. Nassar, Rizwan Aslam, Naveen Garg
Department of Diagnostic Imaging, University of Texas MD Anderson Cancer Center, USA
*Corresponding Author : Bilal Mujtaba
Department of Diagnostic Imaging, University of Texas MD Anderson Cancer Center, USA
E-mail: BMujtaba@mdanderson.org
Received: December 05, 2017 Accepted: March 13, 2018 Published: March 20, 2018
Citation: Mujtaba B, Nassar SM, Aslam R, Garg N (2018) Hibernoma: Imaging Review and Management. J Clin Exp Radiol 1:1.
Abstract
Hibernoma is a rare benign soft tissue tumor originating from brown fat cells being the only known tumor originating from brown fat. It was first described in 1906 by Merkel H and was initially called “Pseudolipoma “In 1914 it was named “Hibernoma “by Gery L who noted the similarity between tumor cells and normal brown fat cells found in hibernating animals and newborn humans. Hibernomas represent around 1.6% of benign lipomatous tumors and about 1.1% of all tumors arising from adipose tissues, most commonly occur in adults between 20-40 years old with a mean age of 38 years. Hibernoma present as small rubbery, painless, slowly growing soft tissue mass with rapid growth of the tumor can occur in minority of cases.
Keywords: Hibernoma; Pseudolipoma; Imaging review; X-Ray
Introduction
Definition
Hibernoma is a rare benign soft tissue tumor originating from brown fat cells being the only known tumor originating from brown fat [1]. It was first described in 1906 by Merkel H and was initially called “Pseudolipoma “In 1914 it was named “Hibernoma “by Gery L who noted the similarity between tumor cells and normal brown fat cells found in hibernating animals and newborn humans[2,3]. Although the exact function of brown fat remains unknown, it’s thought to be responsible for non-shivering thermogenesis in those species [4,5].
Hibernoma has also been called fetal lipoma and lipoma of embryonic fat due to close similarity between brown fat cells and immature white fat cells [6,7].
Incidence
Hibernomas represent around 1.6% of benign lipomatous tumors and about 1.1% of all tumors arising from adipose tissues, most commonly occur in adults between 20-40 years old with a mean age of 38 years, however they may occur in all age groups with slight predominance in males [8,9]. Brown fat cells present diffusely in human newborns but they regress gradually after 8 weeks of age in the majority of cases. However, residual deposits of brown fat cells may remain in the thigh, shoulder, back, neck, chest, arm and abdomen/retroperitoneum therefore being common locations for hibernomas in order of decreasing frequency of incidence with the thigh representing the location of about 30% of hibernomas. Injury to brown fat by trauma, infection or inflammation may be a precipitating factor for the development of hibernoma in those locations [3].
Presentation
Hibernoma present as small rubbery, painless, slowly growing soft tissue mass with rapid growth of the tumor can occur in minority of cases [3]. On examination, hibernoma will be non-tender, nonpulsatile mass; non adherent to adjacent structures with free/relative mobility and no erythema or overlying skin changes [10]. However, localized warmth may be present on the surface due to increased vascularity [11]. Hibernomas are usually painless at the time of presentation; however symptoms may develop from compression of adjacent structures which rarely occur despite the fact that hibernomas are usually large at presentation [3,12].
Pathology
Grossly, hibernomas appear as soft rubbery masses that are wellcircumscribed, well-encapsulated with thin capsules and lobulated with prominent feeding vessels [3]. On cut surface their color varies from light brown to gray depending on lipid concentration with rare areas of hemorrhage [10]. They are usually large in size at presentation ranging from 1 to 24 cm with a mean size of 9.4 cm; however compression of surrounding structures rarely occurs. These tumors may be intermuscular/intramuscular, subcutaneous or retroperitoneal with tendency to enlarge along facial planes causing displacement of adjacent structures [3]. Microscopically, hibernoma characteristically consists of what so-called “hibernoma cell” which is a large multi-vacuolated cell with small central vesicular nucleus, granular eosinophilic cytoplasm and single prominent nucleolus with no atypia “atypia is a rare finding that was previously described in a single case report in 1979”. These cytoplasmic vacuoles are small, rounded and uniform. These cells usually stain positive for S100; ranging from focal to diffuse and CD31, and negative for CD34 and p53 [3,9]. Histologically, hibernomas can be classified into four histological variants, in order of decreasing frequency: Typical (classic) variant: the most common variant with the thigh being the most common location for this variant, consists of varying mixture of brown multi-vacuolated and white uni-vacuolated fat cells with 3 different subtypes. Pale cell subtype; consists mainly of pale multivacuolated fat cells, mixed cell subtype; consists of approximately equal amount of both pale and eosinophilic multi-vacuolated cells and eosinophilic cell subtype; consists mainly of hibernoma cells with deeply eosinophilic granular cytoplasm. Myxoid variant: consists of multi-vacuolated brown fat cells with slightly eosinophilic cytoplasm separated by loose acellular myxoid stroma, occurs more commonly in males with the head and neck region being the most common location for this variant. Lipoma-like variant: consists of a few scattered hibernoma cells among abundant white fat cells with the thigh being the most common location for this variant. Spindle-cell variant: has combined features of both spindle-cell lipoma and hibernoma, consists of a mixture of multi-vacuolated fat cells and spindle cells together with thick collagen bands, mast cells and myxoid stroma, this variant most commonly occurs in the posterior neck and scalp [9].
Imaging Characteristics
X-ray shows radiolucent mass without underlying osseous involvement with characteristic absence of calcifications [3]. Sonography usually demonstrates a well-circumscribed hyper echoic mass with increased vascular flow on Doppler imaging [7,13]. Angiography demonstrates increased vascularity within the tumor, typical tumor blush and May even reveals internal arteriovenous shunting [3,14].
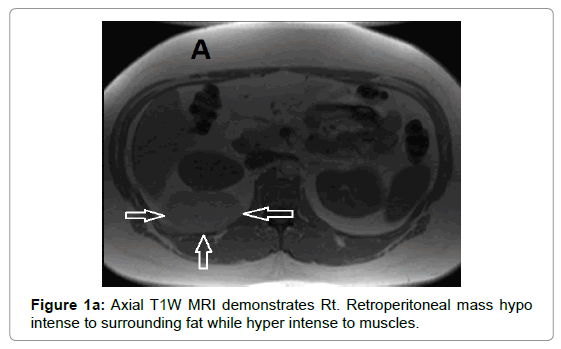
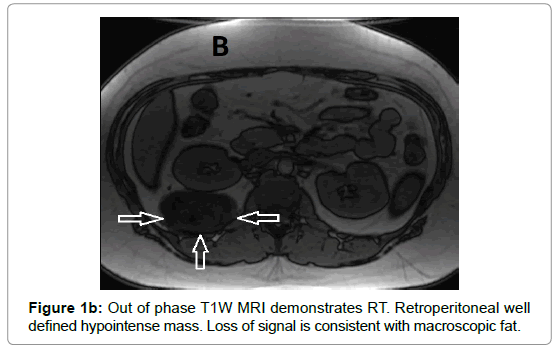
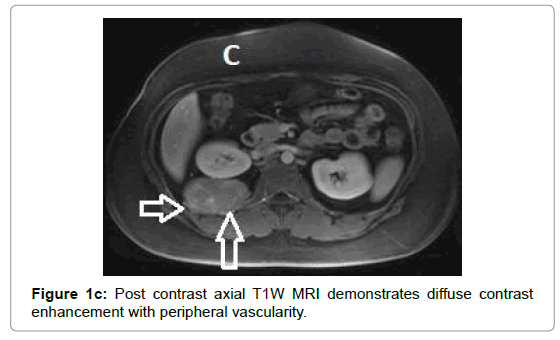
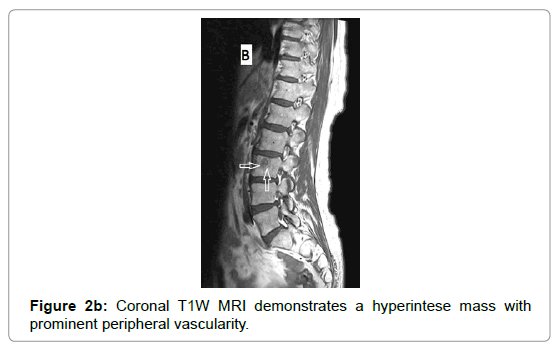
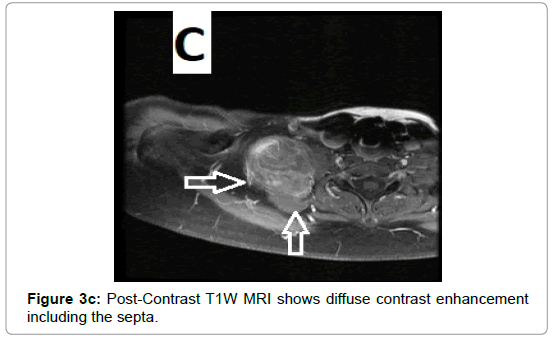
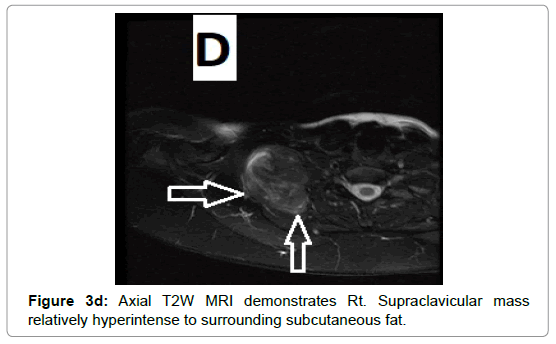
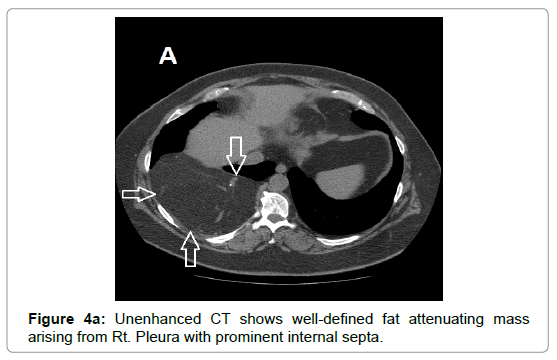
CT examination shows heterogeneous slightly hyper dense, compared with subcutaneous fat, low attenuation mass with internal septa. Intravenous contrast enhances those septa but enhancement varies according to internal tumor composition. Vascularity may be detected inside the tumor [15,16]. However, in the last two decades MRI has replaced both CT and angiography in the assessment of soft tissue tumors. On T1-weighted spin-echo SE images hibernoma appears as well-defined heterogeneous mass slightly/clearly hypo intense to surrounding subcutaneous fat while hyper intense to muscles with irregular low signal intensity bands. On contrastenhanced T1-weighted sequences hibernoma shows slight/significant contrast enhancement. On fat-saturated T2W fast SE or STIR MRI images hibernoma appears relatively hyper intense to surrounding subcutaneous fat with variable fat suppression. However, those MRI features may vary depending on histological variant of hibernoma [1,3,15,16]. The different proportions of variable cellular contents lead to different MRI signal characteristics. So if we want to correlate histological appearances of hibernomas with their MR characteristics they can be classified into: Lipoma-like hibernomas and non-lipomalike hibernomas. Lipoma-like group [<70% multi-vacuolated fat cells] demonstrates signal characteristics similar to subcutaneous fat and usually appears isointense with subcutaneous fat on T1-weighting images whereas non-lipoma-like group [>70% multi-vacuolated fat cells] demonstrates distinctive signal characteristics than subcutaneous fat and appears predominantly slightly hypo intense to subcutaneous fat on T1-weighting images and enhancement is typical. However, the appearances of non-lipoma-like hibernomas are not diagnostic and may resemble lipoma variants, atypical lipoma and well-differentiated lip sarcoma [17] (Figures 1a-c and 2a-c).
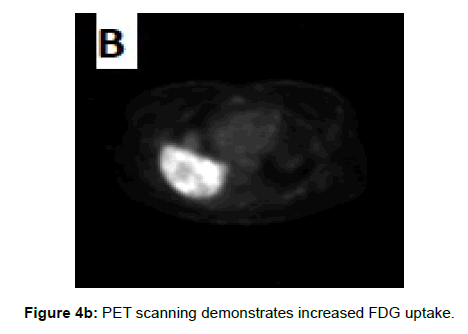
On Tc-99 m Tetrofosmin and FDG Positron Emission Tomographic Scanning hibernoma demonstrates increased uptake of tetrofosmin and fluorine-18 fluorodeoxyglucose on PET scanning, these scintigraphic features help to differentiate hibernoma from other adiogenic tumors as hibernoma may be a benign tumor but it is a metabolically active one and due to high levels of mitochondria in brown fat cells [3,18,19]. However, all adult humans have scattered areas of residual active brown fat that also demonstrate increased uptake on scintigraphy decreasing its importance as a diagnostic imaging tool [10].
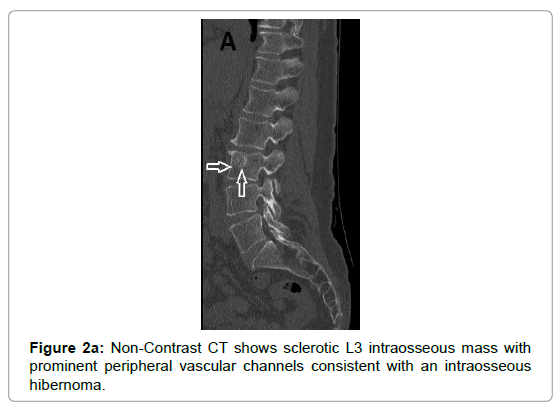
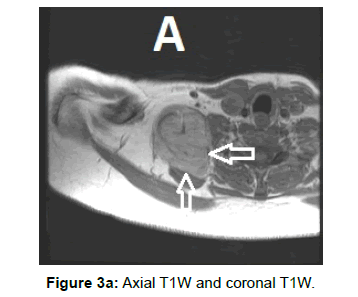
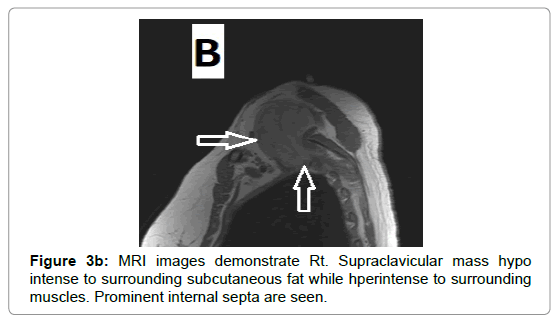
Due to vascular nature of the tumor indicated by large feeding vessels demonstrated by Doppler ultrasound, MR angiography plays an important role preoperatively allowing the surgeon to identify the feeding vessels clearly before surgery leading to more efficient procedure. Also, the use of intravascular embolization considered for the treatment of these tumors but still under study [20] (Figures 3a-d and 4a,b).
Differential Diagnosis
Preoperative diagnosis of hibernoma may be challenging due to its clinical and radiological overlap with other soft tissue masses including benign lesions e.g. lipoma, hemangioma and angiolipoma, as well as malignant lesions e.g. well-differentiated lip sarcoma [21,22].
Hibernomas, especially those with myxoid change, may be confused with myxoid liposarcoma. However, liposarcoma tends to occur deeper and histologically characterized by the presence of atypia, plexiform capillary pattern and particular molecular translocations including t (12; 16) [3,9].
Despite of variable imaging modalities, radiological diagnosis of hibernoma remains challenging even after biopsy [21]. However MRI plays an important role in preoperative presumptive diagnosis of hibernoma and differentiation from the more aggressive myxoid liposarcoma which significantly affects the clinical decision regarding type of operation and need for chemotherapy/radiotherapy [11,23].
Treatment
Percutaneous core-needle biopsy is not advised in suspected cases of hibernoma due to the possibility of massive bleeding owing to hyper vascular nature of the tumor [15].
Considering the benign behavior of hibernoma without potential for malignant transformation nor metastasis, complete marginal surgical excision is considered the treatment of choice for hibernoma, however local recurrence has been documented after incomplete excision. Intralesional excision may be considered for large hibernomas adjacent to important structures, nerves or vessels, however may lead to continued growth and recurrence [3,15].
As hibernomas are well-encapsulated and non-adherent to adjacent structures, they can be easily separated from surrounding soft tissues, however due to hyper vascular nature significant bleeding may occur during surgical excision [3,24].
Conclusion
Hibernoma is a rare benign soft tissue tumor of brown fat usually occurs in adults with slight male predominance. Presents as small soft mobile painless slowly growing mass with the thigh being the most common location. MRI plays an important role in initial diagnosis of hibernoma and differentiating it from other soft tissue masses. On T1W MRI images hibernoma appears as well-defined heterogeneous mass hypo intense to subcutaneous fat and hyper intense to muscles with contrast enhancement on contrast-enhanced images and failed to fully suppress on fat-saturated T2W or STIR images. Hibernoma has no potential for malignant transformation or metastasis, so complete marginal surgical excision is the treatment of choice with no documented recurrence after complete excision.
References
- Lee JC, Gupta A, Saifuddin A (2006) Hibernoma: MRI features in eight consecutive cases. Clin Radiol 61: 1029-1034.
- Merkel H (1906) On a pseudolipoma of the breast. Beitr Pathol Anat 39: 152-157.
- Klevos G, Jose J, Pretell-Mazzini J CS (2015) Hibernoma. Am J Orthop 44: 284-287.
- Minni A, Barbaro M, Vitolo D, Filipo R (2008) Hibernoma of the para-glottic space: an unusual tumour of the larynx. Acta Otorhinolaryngol. Ital 28: 141-143
- Gadea E, Thivat E, Paulon R, et al. (2014) Hibernoma: A Clinical Model for Exploring the Role of Brown Adipose Tissue in the Regulation of Body Weight? J Clin Endocrinol Metab 99: 1-6.
- Honoki K, Morita K, Kasai T (2010) Hibernoma of the axillary region: A rare benign adipocytic tumor. Rare Tumors 2: 20-21.
- Chen CL, Chen WC, Chiang JH, Ho CF (2011) Interscapular hibernoma—Case report and literature review. Kaohsiung J Med Sci 27: 348-352.
- Mavrogenis A, Coll-Mesa L (2013) Soft tissue: Hibernomas. Atlas Genet Cytogenet Oncol Haematol 17: 60-64.
- Furlong M, Fanburg-Smith JC, Miettinen M (2001) The morphologic spectrum of hibernoma: a clinicopathologic study of 170 cases. Am J Surg Pathol 25: 809-814.
- DeRosa DC, Lim RB, Lin-Hurtubise K, Johnson EA (2012) Symptomatic hibernoma: a rare soft tissue tumor. Hawaii J Med Public Health 71: 342-345.
- Kallas KM, Vaughan L, Haghighi P, Resnick D (2003) Hibernoma of the left axilla: a case report and review of MR imaging. Skeletal Radiol 32: 290-294.
- Vassos N, Lell M, Hohenberger W, Croner RS, Agaimy A (2013) Deep-seated huge hibernoma of soft tissue: A rare differential diagnosis of atypical lipomatous tumor/well differentiated liposarcoma. Int J Clin Exp Pathol 6: 2178-2184.
- Hardes J, Scheil-Bertram S, Hartwig E, Gebert C, Gosheger G, et al. (2005) Sonographic findings of hibernoma. A report of two cases. J Clin Ultrasound 33: 298-301.
- Pandya A, Wasnik AP (2011) Presacral hibernoma: Radiologic-pathologic correlation. Indian J Radiol Imaging 21: 270-273.
- Dursun M, Agayev A, Bakir B (2008) CT and MR characteristics of hibernoma: six cases. Clin Imaging 32: 42-47.
- Papathanassiou ZG, Alberghini M, Taieb S, Errani C, Picci P, et al. (2011) Imaging of hibernomas: A retrospective study on twelve cases. Clin Sarcoma Res 1: 3.
- Ritchie DA, Aniq H, Davies AM, Mangham DC, Helliwell TR (2006) Hibernoma - Correlation of histopathology and magnetic-resonance-imaging features in 10 cases. Skeletal Radiol 35: 579-589.
- Chatterton BE, Mensforth D, Coventry BJ, Cohen P (2002) Hibernoma: Intense uptake seen on Tc-99m tetrofosmin and FDG positron emission tomographic scanning. Clin Nucl Med 27: 369-370.
- Park JH, Ogura K, Fujiwara T (2015) The Values and Limitations of FDG-PET/CT for Diagnosis of Hibernoma. Case Rep Orthop.
- Tomihama RT, Lindskog DM, Ahrens W, Haims AH (2007) Hibernoma: A case report demonstrating usefulness of MR angiography in characterizing the tumor. Skeletal Radiol 36: 541-545.
- Daubner D, Spieth S, Pablik J, Zophel K, Paulus T, et al. (2015) Hibernoma--two patients with a rare lipoid soft-tissue tumour. BMC Med Imaging 15: 4.
- Murphey MD, Carroll JF, Flemming DJ, Pope TL, Gannon FH, et al. (2004) From the archives of the AFIP: benign musculoskeletal lipomatous lesions. Radiographics 24: 1433-1466.
- Han HH, Choi JY, Seo BF (2014) Treatment for intramuscular lipoma frequently confused with sarcoma: A 6-year restrospective study and literature review. Biomed Res Int.
- Ogilvie CM, Torbert JT, Hosalkar HS, Stopyra GA, Lackman RD (2005) Recurrence and bleeding in hibernomas. Clin Orthop Relat Res 438: 137-143.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi