Research Article, J Fashion Technol Textile Eng S Vol: 0 Issue: 3
Hydrophilic-Impermeable Modified Polyethylene Terephthalate for Selective Endothelialization
Chetouane D1*, Fafet JF1, Barbet R2 and Dieval F1
1Université de Haute Alsace, ENSISA, Laboratoire de Physique et Mécanique Textiles, 11 rue Alfred Werner, 68093 Mulhouse, France
2Institut de Recherche en Hématologie et Transplantation, 87 avenue d’Altkirch, 68100 Mulhouse, France
*Corresponding Author : Donia Chetouane
Université de Haute Alsace, ENSISA, Laboratoire de Physique et Mécanique Textiles, 11 rue Alfred Werner, 68093 Mulhouse, France
E-mail: donia.chetouane@uha.fr
Received: September 22, 2017 Accepted: October 12, 2017 Published: October 16, 2017
Citation: Chetouane D, Fafet JF, Barbet R, Dieval F (2017) Hydrophilic-Impermeable Modified Polyethylene Terephthalate for Selective Endothelialization. J Fashion Technol Textile Eng S3:003. doi:10.4172/2329-9568.S3-003
Abstract
The aim of this study is to create a modified polyethylene terephthalate (PET) responding to vascular implants’ requirements, mainly with a surface promoting selective endothelialization. The surface alteration was carried out by hydrophilic functionalization in an alkaline solution with the presence of specific surfactant (TA). The carboxylic groups resulting from this reaction were quantified by colorimetric titration using bleu toluidine O dye (TBO). A singlesided coating process was then optimized to cover the PET surface by micro spherical structures’ polymeric layer. This coating provided to the PET surface high impermeability to the water under a pressure of 120 mmHg and enhanced its hydrophilic property. This spherical topography reduced the adhesion of Mesenchymal Stem Cells (MSC) by 37% and inhibited their proliferation after 3 days by 50%. The hydrophilic functionalized PET (PET-TA) surface decreased the MSC adhesion by 50% and promoted HUVEC attachment with a number twice more important than the number of HUVEC adhered onto non treated-PET
Keywords: Vascular prosthesis; Polyethylene terephthalate; Hydrophilic properties; Endothelialization
Introduction
Several premature complications might occur after vascular prostheses implantation, especially those replacing small diameter arteries (< 6 mm), causing thus serious vascular problems such as dilation, rupture, bleeding and thrombosis formation. However, it has been reported that endothelialization of blood substitutes’ surface could be a promising alternative to reduce the risk of these deadly incidents. For this purpose, several authors have been interested in coating the intimal wall of prosthesis by nanofibers layer since they closely mimic the nanoscale fibrous architecture of ECM. In fact, it has been demonstrated that electrospun pullulan/dextran nanofibers substrate enabled a stable confluent monolayer attachment over 14 days, but it also contributed to Smooth Muscle Cells (SMC) recruitment [1]. Moreover, Sabatier [2] activated the polyethylene terephthalate surface by plasma treatment and coated it with poly lactic acid (PLA) air-spun nanofibers for their slowly degradation properties. The NH2-functionalized PET definitely improved the nanofibers’ adhesion but it was showed that the nanofibers layer began to delaminate in the flow direction in only one hour in flow chamber. Besides, nanofibers production, their deposit and adhesion on PET fabric remain a delicate step, difficult to be industrially implemented.
These literature findings allowed us to consolidate our approach to produce a modified-PET substrate suitable for blood vessel replacements by promoting endothelial cells’ adhesion and inhibiting Mesenchymal Stem Cells’ (MSC) proliferation in internal wall of vascular substitute. To reach this goal, we decided, at the first stage, to alter the chemistry surface of the polyethylene terephthalate fabric by hydrophilic functionalization. Beside this crucial criterion of cell selectivity’s surface, the PET fabric must meet several important requirements to be more inclusive. Among them, patency is the property about which scientific views might differ: some researchers prefer increasing surface’s permeability rate to ensure transmural capillary ingrowth and therefore a good endothelialization while others prefer poor patency to create a gate to cell infiltration into the graft lumen and avoid blood leakage. In our case, we chose to provide a great tightness to the hydrophilic modified PET by lining the textile pattern with a non-cytotoxic polymeric layer. For this, a single-sided coating process, denominated MB process, were developed to obtain micro spherical structures layer homogenizing the surface status of the fabric. These microspheres form as well a permanent porous shelter to regulate cell colonization.
Materials and Methods
Materials
The fabric used during this study was provided by Concordia Textile. It is 100% polyethylene terephthalate white cross twill woven. It contains 37 threads in warp direction of 35 tex and 38 threads of 19 tex in weft direction. Its surface mass is 196 g/m2. For the cell culture, Human Umbilical Vein Endothelial Cells (HUVECs) were used to evaluate the affinity of PET-TA surface with Endothelial Cells (EC). In addition, Mesenchymal Stem Cells (MSCs) were tested to assess the selective endothelial adhesion’s property of this substrate, as they are known for their strong adhesive abilities.
Methods
Hydrophilic functionalization: The polyethylene terephthalate surface was modified by alkaline hydrolysis, using a solution of 2% w/v of hydroxide sodium during one hour at 115°C in the dyeing machine AHIBA TurboColor. A specific surfactant was added to be covalently grafted onto PET macromolecules. This surface will be called PETTA. Two different surfactants TA1 and TA2 were assessed.
MB process: It involves a single-sided coating process using a perforated flat bed and a textile printing machine. The printing past is transferred to fabric, thanks to a scraper, through the frame’s micro pores. The polymeric coating is, then, fixed by thermic treatment letting the microspherical structures grafting onto the textile material surface. This surface will be called PET-MB or PET-TA-MB if the coating is respectively applied on reference PET or functionalized PET (PET-TA). In order to develop this technique, several instrumental parameters were investigated. Among them, the influence of the temperature of thermic treatment on coating aspect was studied. The chemical formulation of the MB product was also optimized to achieve a structure meeting the surgical needs (softness, thickness).
Substrates characterization: To assess the hydrophilic property of the PET-TA substrate, carboxylic groups’ number was quantified by colorimetric titration using the toluidine blue O (TBO) dye. The COOH functions number is expressed in μmol/g. The wettability of MB coated surfaces was determined by the drop contact angle method. Five microliters of water drop were deposited on the substrate and a monochrome camera (5 MPx) permitted to take a photo or a video of the spreading drop. Thanks to Virtual Dub software, images were then extracted and the contact angle was measured by the Dropsnake Module of Image J software. Concerning the water permeability test, an area of 1cm2 of each substrate was tested under a constant water pressure of 120 mmHg and the flowrate of water passing through the sample during 60 s was measured according to international standard ISO-FDIS 7189 [3]. The result is expressed in ml/cm2.min
In vitro cell culture: All scaffolds were cut in circular shape of 16 mm diameter by ultrasound gun to avoid fraying. Then, they were sterilized in 70% ethanol and passed under UV for 30 minutes. After this, they were seeded by 104 cells in well culture containing 400 μL of media. Cultures were conducted for each substrate in an incubator containing 5% of CO2 at 37°C for 1, 2 and 3 days. Before absorbance lecture, 20 μl of the cell counting kit-8 (CCK-8) was added to each well and immediately returned to the incubator for 4 hours. It allowed us to determine the number of living cells.
Results and Discussion
Hydrophilic functionalization
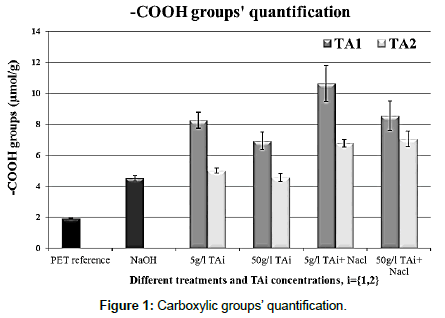
The influence of two concentrations (0.5% w/v and 5% w/v) of each surfactant on -COOH functions number, determined by TBO method, was studied (Figure 1). On both PET-TA1 and PET-TA2, it was found a higher carboxylic groups amount than the existing and/or created ones by hydrolysis on PET substrate. This result is attributed to the capacity of the surfactants to reduce the surface tension of the fabric. Moreover, it was pointed out that the potential barrier between the solution and the substrate were lowered by the addition of an electrolyte leading consequently to a larger number of carboxylic groups. Furthermore, it was noted that the surfactants’ concentration of 0.5% w/v provided a greater carboxylic groups’ amount than 5% w/v concentration. This could be explained by a transesterification reaction between surfactants and carboxylic groups created onto the PET surface bringing thereby a complementary hydrophilicity to the material: In case of an excess of surfactant (5% w/v), a higher number of hydrophilic groups’ surfactant could be grafted onto PET surface reducing thus the carboxylic groups amount. Finally, it seemed that the surfactant TA1 gave rise to the best result with 11μmol/g of – COOH functions, so it was chosen to functionalize the PET substrate in an alkaline solution in the presence of sodium chloride.
MB process
The third magnetic force of the machine was chosen to apply the printing paste, containing 60/40 MB product/ thickening agent, on the Fabric (Figure 2a). The thermic treatment investigation allowed us to sit the optimal thermic duration at 2 minutes and a temperature of 110°C to obtain micro spherical structures covering all the surface fibers. Below this temperature, the MB coating had a grainy aspect (Figure 2b).
Finally, a plasticizer (PEG) and a binding agent (CLAR) were incorporated in the printing paste to provide greater flexibility and homogeneity to the MB coating by decreasing the glass transition temperature (Tg) of the copolymer MB from 0.13°C to -3.67°C (Figure 2c). In fact, as observed in the SEM image (Figure 2d), the microspheres are more dispersed in monolayer reducing thus the MB treatment’s thickness.
The process MB was used to cover the surface of the hydrophilic functionalized PET (PET-TA) by a layer of micro spherical structures at 110°C. We noted that there is a high affinity between the TAtreated fibers and the polyvinyl ester copolymer (MB product), that the space inter fibers was filled by polymeric microspheres (Figure 3a). Another application of the process MB consists in applying the product MB into the non-treated PET at 200°C to coat the whole of the surface by a MB thin layer (Figure 3b). The appearance of this coating resembles nanofibers mat’s aspect. It was tested only with cell culture to investigate the influence of the surface morphology on cell colonization.
Wettability
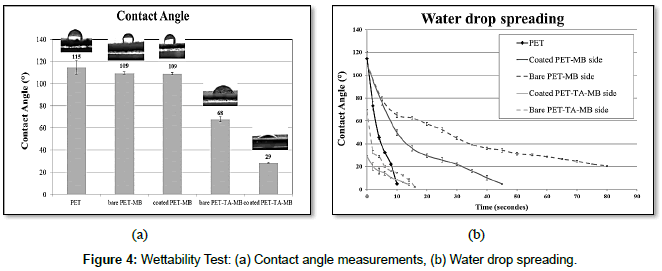
The water drop contact angle measurements allowed us to assess the wettability of our substrates. The Figure 4a highlights the initial hydrophobicity of the non-treated PET (115°) before hydrophilic functionalization. The MB coating didn’t significantly influence this hydrophobic property on both the two sides of the fabric PET-MB but it accelerates the drop water spreading which disappears onto the coated face after 50s against 80s with a contact angle of 20° onto the bare face (Figure 4b).
However, the hydrophilic functionalization has highly decreased the hydrophobicity of the non-treated-PET before MB coating: In fact the bare side of PET-TA-MB substrate exhibits a contact angle of 68° which, in turn, diminish to achieve 29° onto its coated face. This result proves that the presence of microspheres on the functionalized-PET surface intensify hydrophilic property of the initial surface PET-TA. This is consistent with Wenzel model [4] according to the equation:
cosθw = r.cosθ
Where θ represents contact angle onto a flat surface, θW is the contact angle onto rough surface of the same material and r is the roughness.
Water permeability
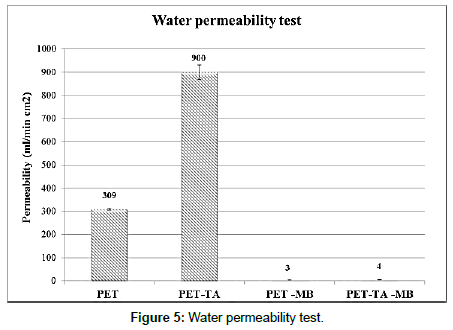
The test of water permeability (Figure 5) demonstrates that the MB coated fabrics became extremely impermeable to the water for both PET-MB and PET-MB-TA. The water permeability decreases from 900 to 4 ml/min.cm2. This impermeability property is necessary to avoid bleeding through the prosthesis wall.
Cell culture
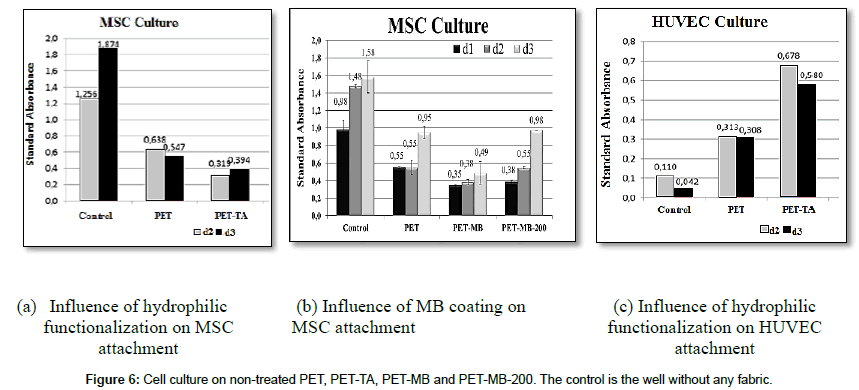
The cell culture graphs (Figure 6a and b) display the number of seeded Mesenchymal Stem Cells (MSC) onto non-treated PET, hydrophilic functionalized PET (PET-TA), PET-MB and fabric lined by thin MB layer at 200°C (PET-MB-200) during 2 and 3 days.
A decrease of 50% and 28% of adhered MSC onto PET-TA comparing to non-treated PET was noted after respectively 2 and 3 days of culture. Concerning the MB coating, the thin MB layer covering the PET surface (PET-MB-200), which resembles to a polymeric nanofibers mat, promoted the same MSC proliferation than the reference PET surface: no significant difference has been observed after 3 days of cell seeding. However, the micro spherical structures’ coating of PET-MB reduced the MSC adhesion by 37% and their proliferation after 3 days by almost 50% comparing to the flat PET. Regarding to the HUVEC (Figure 6c), the PET-TA substrate enhanced their proliferation by 47% after 3 days of culture. These results allowed us to infer the influence of hydrophilic property to enable a fast and selective endothelialization and the importance of rough surface to foster controlled MSC proliferation.
Conclusion
Hydrophilic functionalization of the polyethylene terephthalate was carried out by alkaline hydrolysis in the presence of an electrolyte and 0.5% w/v of specific surfactant TA1 at 110°C. This treatment brought to the PET surface an important carboxylic functions’ amount of 11 μmol/g that made it hydrophilic. It was shown that this chemical surface alteration improved the endothelial cells’ proliferation by 47%.
The MB process was optimized to cover the PET surface with micro spherical structures’ polymeric layer. This MB coating provided to the PET substrate high water impermeability to ensure a great tightness of the textile material. The roughness created by the microspheres onto the PET-TA surface enhanced its hydrophilic property. This curvilinear topography reduced the MSC adhesion and regulated their proliferation. Therefore, we obtained a hydrophilic impermeable-modified polyethylene terephthalate (PET-TA-MB) able to promote endothelialization and inhibit unwanted cells recruitment. In pursuing our work, micro spherical structures will be exploited to incorporate peptides inhibiting platelet aggregation. Such a coating could be interestingly applied in the field of the functionalization of internal walls of vascular substitutes.
References
- Shi L, Aid R, Le Visage C, Chew SY (2012) Biomimicking polysaccharide nanofibers promote vascular phenotypes: A potential application for vascular tissue engineering. Macromol Biosci 12: 395-401
- Sabatier G (2015) Conception et élaboration d’échafaudages de nanofibres à dégradation contrôlée pour des applications en médecine régénratrice vasculaire.
- ISO (1998) Cardiovascular implants – Tubular vascular prostheses.
- Wenzel RN (1979) Surface roughness and contact angle. J Phys Chem 53: 1466-1467.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi