Research Article, J Clin Exp Radiol Vol: 1 Issue: 1
Imaging of Recurrent Head and Neck Tumors in Patients with Prior Flap Reconstruction
Bobinski M1*, Lee SP1, Farwell G2, Shen YP1 and Glastonbury MC3
1Department of Radiology, University of California-Davis, Sacramento CA, USA
2Department of Otolaryngology, University of California-Davis, Sacramento CA, USA
3Department of Radiology, UCSF School of Medicine, San Francisco, USA
*Corresponding Author : Dr. Matthew Bobinski
Department of Radiology, University of California-Davis, Sacramento CA, USA
Tel: 916-734-3271
Fax: 916-734-8490
E-mail: mbobinski@ucdavis.edu
Received: March 30, 2017 Accepted: January 04, 2018 Published: January 11, 2018
Citation: Bobinski M, Lee SP, Farwell G, Shen YP, Glastonbury MC (2018) Imaging of Recurrent Head and Neck Tumors in Patients with Prior Flap Reconstruction. J Clin Exp Radiol 1:1.
Abstract
Objective
The purpose of this study is to provide a comprehensive description of the spectrum of imaging characteristics and distribution of recurrent disease in patients with head and neck cancers reconstructed with flaps, which may allow salvage surgery and contribute to improved survival.
Methods
Computed Tomography images and clinical data of all consecutive patients who underwent head and neck tumor resection, neck dissection and flap reconstruction over 7.5 year period were reviewed. Imaging findings were correlated with flap type, recurrence site, and time to recurrence and clinical suspicion.
Results
Recurrence occurred in 29 of the 82 patients, with average time to recurrence at 9 months. Recurrence was clinically suspected in 62% of patients by detecting a new neck mass. Almost 76% of recurrent tumors were solid enhancing masses and over 24% were necrotic masses. While most recurrent lesions occurred at the margin of free flap, 31% were found remotely from the flap. There was no statistical correlation between flap type, recurrence characteristics, recurrence site and either time to recurrence or clinical suspicion.
Conclusion
The majority of patients with recurrent disease following flap reconstruction developed their recurrence within the post-operative first eight months. Recurrent tumors typically present as infiltrating, enhancing masses which may have necrotic foci, and tend to occur along the flap margin and/or suture line. Approximately 38% of recurrences were not clinically suspected. Routine 3-month baseline imaging then further 3-month interval follow-up in the first year should be considered in patients with flap reconstruction for head and neck malignancy.
Keywords: Flap; Neck cancer; Recurrence; CT
Introduction
The imaging diagnosis of recurrent disease is challenging in the post-treatment neck, particularly with flap reconstruction of complex head and neck post-surgical cancer defects. These may be further complicated by post-operative radiation. Distortion of anatomy and loss of normal neck symmetry after surgical resection and from radiation fibrosis also limits clinical assessment, and despite the promise of imaging, many modalities are limited by false-positive results, particularly in the early post-surgical period [1-6]. Contrastenhanced cross-sectional imaging, particularly CT, remains the mainstay for evaluating patients early in the post-treatment period and for routine surveillance and patterns of disease recurrence can be readily apparent to the experienced eye [1,7].
The presence of non-native tissue dramatically alters imaging characteristics of the head and neck, and inflammation, fibrosis, and healing occur as a continuum over many months and are variable across different patients [1-6]. While a significant percentage of head and neck tumor patients will experience locoregional recurrence, the pattern and imaging characteristics of these recurrences in post-surgical and post-radiation patients have not been comprehensively described [4-7]. The purpose of this study is to provide a comprehensive description of the spectrum of imaging characteristics and distribution of recurrent disease in patients with cancers of the head and neck reconstructed with flaps, which may allow salvage surgery and contribute to improved survival.
Materials and Methods
Patients
The institutional review board approved this Health Insurance Portability and Accountability Act compliant retrospective study, and the requirement for informed consent was waived. All patients who underwent head and neck tumor resection, neck dissection and surgical flap reconstruction in our institution over a seven-year period were reviewed. Patients were initially identified through query of oncologic otolaryngology surgical database. Additional patient’s clinical and imaging information were obtained through Electronic Medical Record (EMR) and picture archiving and communication system (PACS).
Patients were excluded if they did not have post-surgical imaging or if the flap had been placed for reason other than carcinoma, such as penetrating trauma.
Clinical data
Patients’ demographics, clinical and surgical data collected included patient age, gender, site of primary tumor and nodal metastasis, clinical and imaging staging of primary tumor (TNM staging), type of surgical reconstruction (antero-lateral thigh flap- ALT, fibular, radial forearm, rectus, scapular and composite flaps), types of chemoradiaton received, time to tumor recurrence, site of tumor recurrence and clinical follow-up examinations. Information was obtained through review of EMR clinical notes, surgical pathology and operative reports by both radiologists and otolaryngologists. All patients with suspected local head and neck recurrence on clinical or imaging examinations underwent image-guided biopsy and the corresponding pathology reports were reviewed.
Image analysis
All CT studies have been performed on a 16-row scanner General Electric (Milwaukee, Wisconsin, USA) with a 2.5 mm slice thickness axial, coronal and sagittal reformats following intravenous contrast administration (150 ml of Omnipaque 350). The following imaging features were evaluated by one board certifiied neuroradiologist: type of the flap reconstruction, presence of abnormal enhancement, soft tissue nodularity, regional adenopathy, distant metastases and complications.
Statistical analysis
Statistical analysis was performed with chi square test.
Results
82 patients (59 Males, 23 Females) with ages ranging from 20 to 86 years (median of 63 years) were treated at our institution for head and neck cancer between 1/1/2002 and 6/30/2009 and satisfied the inclusion criteria. Seventy-four patients had squamous cell carcinoma (SCC) of the head and neck and eight patients had non-SCC diagnosis involving mandible, masticator space, parotid gland, and paranasal sinuses.
All of the patients were treated with primary resection and reconstruction using flap reconstruction: 28-antero-lateral thigh (ALT), 23-radial forearm, 14-fibular, 7-rectus, 4-scapular, and 6-composite flaps (2-fibular and pectoralis major, 2-ALT and pectoralis major, 1-ALT and fibular, and 1-scapular and latissimus dorsi).
All but four patients with recurrent disease received postoperative radiation therapy (RT): two patients did not follow-up for treatment (case 5, 12, Table 1), one refused RT (case 25), and one had already had previous RT (case 28). Recurrence occurred in 29 of the 82 patients (35.4%), with average time to recurrence at 9 months (median 5 months; Tables 1 and 2). Of the 29 patients with recurrence, 18 (62%) were clinically suspected by detecting a new neck mass. Nodal metastases were present in 6 of the 29 with locally recurrent tumor. Isolated nodal or distant tumor without local recurrence occurred in 7 and 3 patients, respectively (Table 2). Dermal tumor implants were present in 3 patients all of whom also had local recurrence and/or nodal disease.
| Case No | Age/Sex | Pathology | Flap Type | Recurrence Characteristics | Relation to Flap | Time to Recurrence(mo) | Clinically suspected |
|---|---|---|---|---|---|---|---|
| 1 | 48/M | Mucoepidermoid ethmoid air cells | ALT | E | R | 5 | N |
| 2 | 61/M | SCC BOT | ALT | E | M | 5 | N |
| 3 | 49/F | SCC larynx | ALT | E | M | 4 | Y |
| 4 | 46/F | SCC tongue | ALT | E | R | 5 | Y |
| 5 | 64/M | SCC scalp | ALT | E | M | 5 | Y |
| 6 | 50/M | SCC nasopharynx | ALT | E | M | 3 | Y |
| 7 | 45/F | SCC tongue | ALT | E | M | 5 | N |
| 8 | 62/F | SCC oral cavity | Fibular | N | M | 6 | Y |
| 9 | 66/M | SCC FOM | Fibular | N | M | 7 | Y |
| 10 | 58/M | SCC oropharynx | Fibular | N | R | 46 | N |
| 11 | 73/M | SCC hypopharynx | Fibular | E | R | 5 | Y |
| 12 | 65/F | SCC FOM | Radial | E/N | M | 22 | N |
| 13 | 61/M | SCC oral cavity | Radial | E | M | 6 | N |
| 14 | 58/M | SCC BOT | Radial | E | R | 6 | Y |
| 15 | 73/M | SCC FOM | Radial | E | M | 23 | Y |
| 16 | 57/M | SCC oropharynx | Radial | E | M | 13 | N |
| 17 | 53/M | SCC tongue | ALT | E | R | 7 | Y |
| 18 | 21/F | Synovial cell carcinoma TMJ | Rectus | E | M | 2 | N |
| 19 | 80/F | SCC oral cavity | Scapular | N | M | 5 | Y |
| 20 | 63/F | SCC FOM | Fibular | E | M | 31 | Y |
| 21 | 63/M | SCC FOM | Fibular/ALT | E | R | 8 | N |
| 22 | 54/M | SCC scalp | ALT | E | M | 4 | Y |
| 23 | 61/M | SCC tongue | Scapular/Latissimus dorsi | N | M | 5 | N |
| 24 | 43/M | SCC tongue | Radial | E | R | 5 | N |
| 25 | 73/F | SCC FOM | Scapular | E/N | M | 5 | Y |
| 26 | 61/M | SCC tongue | ALT | E | M | 4 | Y |
| 27 | 76/M | SCC scalp | Rectus | E | M | 3 | Y |
| 28 | 81/M | SCC tongue | ALT | E | M | 5 | Y |
| 29 | 75/M | SCC scalp | ALT | E | R | 10 | Y |
M = at margin of flap
N = necrotic mass
R = remotely from flap, deep to, no contact
ALT = antero-lateral thigh flap
Pathology: SCC = squamous cell carcinoma;
BOT = base of tongue;
FOM = floor of the mouth;
TMJ = temporomandibular joint
Table 1: CT imaging findings in patients with local recurrent tumor following flap reconstruction of the head and neck cancer.
| Recurrence site | Number of patients |
|---|---|
| None | 43 |
| Local only | 20 |
| Local + Nodal | 5 |
| Local + Distant | 3 |
| Local + Nodal + Distant | 1 |
| Nodal only | 5 |
| Nodal + Distant | 2 |
| Distant | 3 |
Table 2: Pattern of recurrent disease in patients following flap reconstruction in head and neck.
Local recurrences presented either as a solid enhancing mass, necrotic mass, or enhancing mass with small necrotic focus (Table 1). Almost 76% of recurrent tumors were solid enhancing masses and over 24% were either enhancing masses with a small necrotic focus or necrotic masses (7% and 17%, respectively) (Figure 1). Eight patients developed either one or several peripherally enhancing fluid collections, which were originally suspected to be abscesses. Three of these patients had local and systemic signs and symptoms of infection (fever, erythema, leukocytosis) 2-3 weeks following flap placement and were initially treated with antibiotics and drainage. Five patients developed collections within 5-8 months following flap placement and had no clinical signs of infection. They were followed up with imaging and tissue sampling, which showed delayed development of enhancing loculated fluid collection, which represented recurrent disease and not abscess.
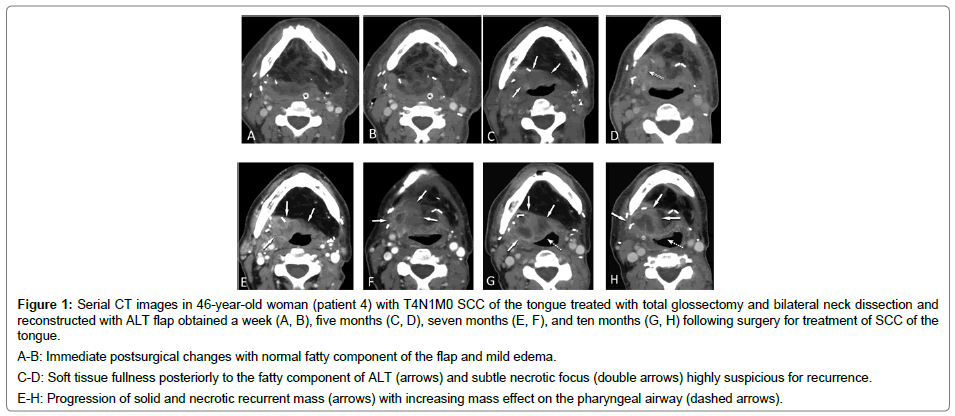
Figure 1: Serial CT images in 46-year-old woman (patient 4) with T4N1M0 SCC of the tongue treated with total glossectomy and bilateral neck dissection and reconstructed with ALT flap obtained a week (A, B), five months (C, D), seven months (E, F), and ten months (G, H) following surgery for treatment of SCC of the tongue.
A-B: Immediate postsurgical changes with normal fatty component of the flap and mild edema.
C-D: Soft tissue fullness posteriorly to the fatty component of ALT (arrows) and subtle necrotic focus (double arrows) highly suspicious for recurrence.
E-H: Progression of solid and necrotic recurrent mass (arrows) with increasing mass effect on the pharyngeal airway (dashed arrows).
While most recurrent lesions occurred at the margin of free flap (69%; Figure 2), 31% were found remotely from the flap, either deeper to the flap or within pharyngeal mucosa but not abutting the flap margin (Figure 3). There was no statistical correlation between flap type, recurrence characteristics, recurrence site and either time to recurrence or clinical suspicion.
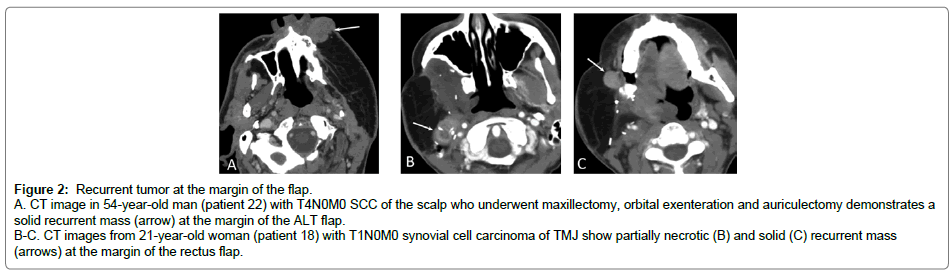
Figure 2: Recurrent tumor at the margin of the flap.
A. CT image in 54-year-old man (patient 22) with T4N0M0 SCC of the scalp who underwent maxillectomy, orbital exenteration and auriculectomy demonstrates a solid recurrent mass (arrow) at the margin of the ALT flap.
B-C. CT images from 21-year-old woman (patient 18) with T1N0M0 synovial cell carcinoma of TMJ show partially necrotic (B) and solid (C) recurrent mass (arrows) at the margin of the rectus flap.
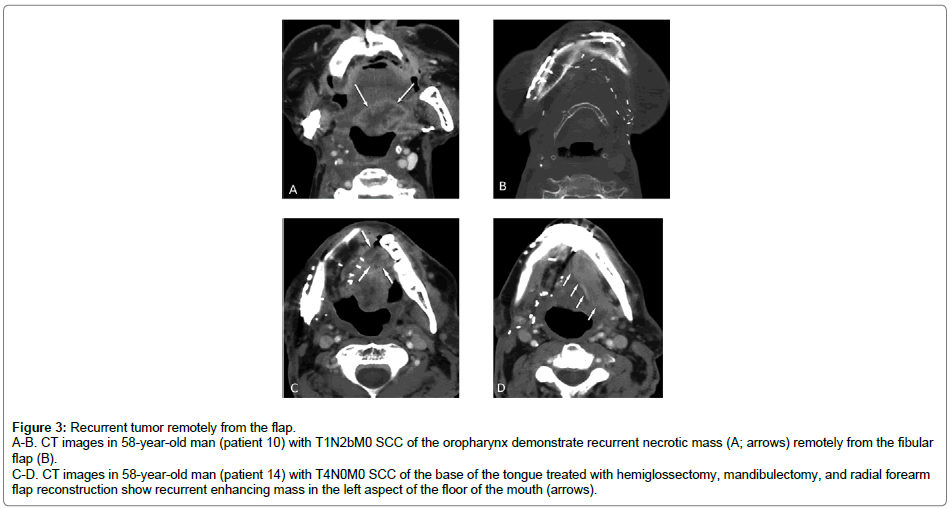
Figure 3: Recurrent tumor remotely from the flap.
A-B. CT images in 58-year-old man (patient 10) with T1N2bM0 SCC of the oropharynx demonstrate recurrent necrotic mass (A; arrows) remotely from the fibular flap (B).
C-D. CT images in 58-year-old man (patient 14) with T4N0M0 SCC of the base of the tongue treated with hemiglossectomy, mandibulectomy, and radial forearm flap reconstruction show recurrent enhancing mass in the left aspect of the floor of the mouth (arrows).
Discussion
Over 79% of patients in our study developed recurrent disease within the first eight months following the surgery (median 5 months). Only 17% of patients recurred more than a year after the treatment. This underscores the importance of early routine imaging of patients with flap reconstruction, and the importance of vigilance to imaging evidence of recurrence. While there are a number of recommendations for post-treatment follow-up and the site specific recommendations vary widely [8,9] our study suggests that the first imaging study in patients with advanced stage cancer should be obtained at 3 months then repeated every 3 months for the first year.
Regular imaging follow-up is especially important since almost 38% of the recurrences were not clinically suspected. These results are similar to those from a prior prospective study [10], which found that physical examination detected only 44% or recurrent disease among patients with stage III or IV head and neck cancer. This again emphasizes the importance of surveillance imaging in these patients.
Radiographic detection of recurrent tumor and its differentiation from post-surgical and post-RT changes is challenging. Most recurrent tumors in our study (69%) occurred at the flap margin and along the suture line, similarly to previous reports [3-6,11-13]. Surprisingly, however, we found a significant number of recurrences remotely from the flap and anastomosis site, either deep to it or away from its margin (31%). Tomura et al found only one recurrence distal to the flap among 25 cases and no additional study commented on such recurrences [12]. While the difference possibly results from case selection, all our cases with remote recurrence had no unifying feature; they varied in terms of pathology, type of flap and time to recurrence. It underscores the importance of careful examination of not only the surgical bed and flap margins but also tissues not in direct contact with flap.
Recurrent tumors typically present as infiltrating enhancing masses [3,7,12]. We found that almost 76% of recurrent tumors were solid enhancing masses and over 24% were either enhancing masses with small necrotic focus or necrotic masses (7% and 17%, respectively). While all necrotic recurrences occurred in patients with SCC, most recurrences in patients with SCC were solid (74%) rather than necrotic. Any new distortion, nodularity or local mass effect on adjacent structures, whether at the flap margin or remotely from it, should be viewed with suspicion. Importantly, necrotic recurrences should not be mistaken for abscesses. The latter occur within few days/ weeks after the surgery and are always accompanied by systemic signs/ symptoms of infection (fever, erythema, leukocytosis). In contrast, necrotic recurrences develop within months (5-8 months among our patients) and without symptoms of infection. While dermal tumor implants are infrequent (10%), they may not be clinically evident due to the initial edema that occurs following radiation followed by fibrotic changes that occur later.
We acknowledge several limitations of the current study. The retrospective nature of this study is associated with several inherent biases. Selection bias seems to be unavoidable and played a role in our study as well. By nature of the study, only patients with imaging were included in the cohort. The patients, who died prior to imaging, were lost to follow-up, and those with cutaneous lesions treated without imaging were not included in our study.
References
- Saito N, Nadgir RN, Nakahira M, Takahashi M, Uchino A, et al. (2012) Posttreatment CT and MR imaging in head and neck cancer: what the radiologist needs to know. Radiographics 32: 1261-1282.
- Chong J, Chan LL, Langstein HN, Ginsberg LE (2001) MR imaging of the muscular component of myocutaneous flaps in the head and neck. AJNR Am J Neuroradiol 22: 170-174.
- Hermans R (2008) Posttreatment imaging in head and neck cancer. Eur J Radiol 66: 501-511.
- Hudgins PA (2002a) Flap reconstruction in the head and neck: expected appearance, complications, and recurrent disease. Eur J Radiol 44: 130-138.
- Hudgins PA (2002b) Flap reconstruction in the head and neck: expected appearance, complications, and recurrent disease. Semin Ultrasound CT MR 23: 492-500.
- Hudgins PA, Burson JG, Gussack GS, Grist WJ (1994) CT and MR appearance of recurrent malignant head and neck neoplasms after resection and flap reconstruction. AJNR Am J Neuroradiol 15: 1689-1694.
- Lell M, Baum U, Greess H, Nomayr A, Nkenke E, et al. (2000) Head and neck tumors: imaging recurrent tumor and post-therapeutic changes with CT and MRI. Eur J Radiol 33: 239-247.
- Pfister DG, Spencer S, Brizel DM, Burtness B, Busse PM, et al. (2015) Head and Neck Cancers, Version 1.2015. J Natl Compr Canc Netw 13: 847-856.
- Manikantan K, Khode S, Dwivedi RC, Palav R, Nutting CM, et al. (2009) Making sense of post-treatment surveillance in head and neck cancer: when and what of follow-up. Cancer Treat Rev 35: 744-53.
- Lowe VJ, Boyd JH, Dunphy FR, Kim H, Dunleavy T, et al. (2000) Surveillance for recurrent head and neck cancer using positron emission tomography. J Clin Oncol 18: 651-658.
- Offiah C, Hall E (2011) Post-treatment imaging appearances in head and neck cancer patients. Clin Radiol 66: 13-24.
- Tomura N, Watanabe O, Hirano Y, Kato K, Takahashi S, et al. (2002) MR imaging of recurrent head and neck tumours following flap reconstructive surgery. Clin Radiol 57: 109-113.
- Som PM, Urken ML, Biller H, Lidov M (1993) Imaging the postoperative neck. Radiology 187: 593-603.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi