Research Article, Clin Res Orthop Vol: 1 Issue: 1
Intraarticular Bupivacaine and Epinephrine Do Not Save Blood in Primary Total Knee Arthroplasty without Drains
Wang JH1, Chang CW1-3*, Yang CY1, Chen YN1, Lai KA1 and Chang CH1
1Department of Orthopedics, National Cheng Kung University Hospital, College of Medicine, National Cheng Kung University, Tainan City, Taiwan
2Department of Biomedical Engineering, National Cheng Kung University, Tainan City, Taiwan
3Department of Orthopedics, College of Medicine, National Cheng Kung University, Tainan City, Taiwan
*Corresponding Author : Chang CW
Department of Biomedical Engineering No. 138, Sheng-Li Rd, North District, 70428, Tainan City, Taiwan
Tel: +886-6-2766689
E-mail: u7901064@yahoo.com.tw
Received: March 17, 2017 Accepted: April 11, 2017 Published: April 18, 2017
Citation: Wang JH, Chang CW, Yang CY, Chen YN, Lai KA, et al. (2017) Intraarticular Bupivacaine and Epinephrine Do Not Save Blood in Primary Total Knee Arthroplasty without Drains. Clin Res Orthop 1:1.
Abstract
Objective: After knee surgeries, the intra-articular administration of a solution mixing bupivacaine and epinephrine is a common regimen. It is logic to expect the potential in blood conservation using this regimen in total knee arthroplasty (TKA); however, there is inconsistent literature. The study was to explore the hemostatic efficacy of this regimen in our primary TKAs without drains.
Methods: A retrospective, case controlled review of our primary TKAs between 2009 and 2010 was conducted. After closure of arthrotomy, a solution containing a 40 ml vial of 0.5% bupivacaine with epinephrine 1: 200000 was given prior to the deflation of tourniquet, and two groups were simply divided according to the intra-articular injection or not. Bleeding parameters of each group, including the maximum hemoglobin reduction during stay, calculated blood loss, and transfusion requirement, were used to determine the hemostatic effect of this regimen.
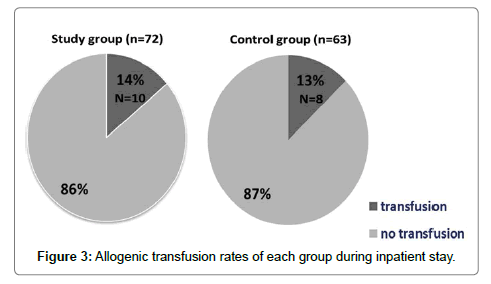
Results: Over a period of 12 months, 135 eligible patients comprised a control group (N=63) and a treatment group (N=72). On a comparative basis, there was no significant different hemoglobin drop or the mean calculated blood loss of both groups (650.4 vs. 648.8 ml, p=0.90; 2.5 vs. 2.4 g/dl; p=0.63). Also, similar allogenic transfusion rates were observed between groups. [13.9% vs. 12.7%, respectively]
Conclusion: Our results suggest little hemostatic benefit of this analgesic solution containing epinephrine in non-drained primary TKA except temporary pain relief. Newer and potent regimens are still required and to be explored.
Keywords: Total knee Arthroplasty; Non-drained; Epinephrine; Blood loss;Intra-articular; Analgesics; Hemostasis
Introduction
With the rapid increase of aging population, more and more arthroplasties are performed to alleviate accompanying painful disabilities of the aging joints. For these elective procedures, surgeons have to manage certain clinical challenges like acute postoperative pain and substantial blood loss.
In current practice, many protocols using topical analgesics have shown a good pain control following arthroplasty [1-3]. To our knowledge, local epinephrine- containing analgesics had been long applied to relieve pain after TKAs for decades [1-9]. In addition to pain control, this simple formula was supposed to be blood-saving as well for the integrating vasoconstrictor, epinephrine. However, we noted the inclusive results in the literature [2,4-10]. Anderson et al. [6], Gasparini et al. [5] and Lombardi et al. [2] reported the positive impacts on reduction of blood loss after TKA using diluted epinephrine for peri-articular injections or irrigation. However, conflicting results were reported by Malone et al. [7], Mesa-Ramos et al. [8] and Joo et al. [9], who observed no significant different postoperative blood loss after the intra-articular bupivacaine and epinephrine lavage.
Therefore, the aim of this study was to determine whether local bupivacaine and epinephrine can reduce postoperative blood loss in our primary TKAs without drains. Based on prior supportive literature [2,4-7], these non-drained TKAs receiving the intra-articular mixture were expected to be less blood-consuming.
Materials and Methods
A retrospective case-controlled review was conducted to identify the adult patients who had undergone elective unilateral primary TKAs by the same surgeon at our institution between 2009 and 2010.
Patients with known coagulopathy without correction or control (INR>1.4), active medical conditions compromising hemostasis (hepatic or renal dysfunction), concurrent use of anticoagulant, preoperative anemia (hemoglobin (Hb) <10.0), prior major surgeries over the operate limb, morbid obesity or leanness (BMI >35 or <15) and incomplete medical records, were excluded. Eligible patients were simply divided into two groups according to the application of intra-articular injection or not: the former index procedures between April and September 2009 were performed without any intra-articular injection comprised the control group; while the remaining TKRs receiving the intra-articular injection comprised the study group. The approval of the institutional review board was sought and obtained [NCKUH-10103045/ BR-100-098].
In this study, all the surgeries were performed by the same senior surgeon and consistent surgical techniques, including intraoperative use of tourniquet, a limited medial parapatellar arthrotomy, standard intra-medullary jigging techniques for femur and tibia, resurfaced patellar button, and even the sequence of bone cuts and soft tissue balancing. During the phase of wound closure, the extensor mechanism and capsule were closed in a watertight fashion without drains. Early release of tourniquet after arthrotomy closure to save ischemic duration was taken in all surgeries [11].
After surgery, all patients received twice daily thrombo-prophylaxis with 500 mg intravenous lysine acetylsalicylic acid for three days and mechanical calf compression whilst immobile. Also, the postoperative nursing and rehabilitation protocol, even the strategy to pain control were the same for the identical clinical pathway established.
Regular oral pain killers (acetaminophen 500 mg every 6 hours and meloxicam 15mg per day) were prescribed, and the rescue medication for breakthrough pain (visual analog scale >3) was consisted of intramuscular or intravenous meperidine 50 mg every 4-6 hours as needed.
During the inpatient stay, once patients could perform active straight leg raise smoothly, bend their knee at least 90 degrees and ambulate steadily with frames in addition to the stable surgical wounds, they were to be discharged and scheduled for regular follow-ups in the outpatient department.
Assessment and blood loss estimation
To determine the effect of this local injection on blood conservation, related bleeding parameters, including serial changes in Hb and Hct, postoperative blood loss, rate of allogenic blood transfusions, and reported complications specific to this intervention [5,12] were retrieved and compared.
In our clinical practice, the Hb as well as Hct level was measured at admission and on postoperative days 1, 2 and 4. During the assessment period, no patients underwent preoperative hemodilution, but sufficient intravenous crystalloid fluid. We evaluate blood loss by measuring maximum Hb drop, defined as the greatest difference between the postop Hb levels and the level at admission, and adding blood units to be transfused.
Hb loss=pre-operative Hb – (post-operative Hb – number of units transfused), assuming that 1U=1 g/dl.
In the absence of postoperative drains as well as recordable drainage, the estimation of blood loss was determined by the method described by Meunier et al. [13]. The Hct and the Hb concentrations of the patients were measured preoperatively. Other factors that were measured at the pre-operative assessment include sex, height, and weight. The post-operative Hct and Hb were measured up to 96 h after the operation, and before any blood transfusion products were given. The calculation for blood loss was made as follows below.
Blood volume (BV) estimation in men and women according to Nadler’s formula [13]:
Men: BV (ml) =0.0003669×height3 (cm) + [32.19×body weight (kg) + 604]
Women: BV (ml) =0.0003561×height3 (cm) + [33.08×bodyweight (kg) + 183]
Hb dilution method for estimating blood loss:
Blood loss (ml)=BV(Hbi−Hba) /Hbi
where BV is the calculated blood volume (ml); Hbi (g/dl)=the Hb concentration before blood operation; Hba (g/dl)=the Hb concentration after the operation.
Blood loss (ml) =the estimated lost blood volume according to the Hb dilution method.
The need for transfusion was guided by the level of hemoglobin (Hb). Patients were transfused only if their Hb levels decreased to <8.0 g/dL or they showed symptomatic anemia. Times of transfusion and the number of units of transfused red blood cell concentrates were recorded.
Statistical analysis
A power calculation was conducted based on previous work conducted by Kalairajah et al., showing analysis of 60 cases per treatment method would be required to detect a 10% difference in blood volume loss between groups, at a level of 95% confidence [14]. The data were tested for normality and analyzed using an F test to determine the most appropriate two-sided unpaired Student’s t test to utilize. A two tailed p value of 0.05 or less was considered to be significant with a 95% confidence level.
To analyze data statistically, SPSS version 14.0 was used (SPSS Inc., Chicago, IL, USA). Student’s t tests were used to analyze interval data, whereas the Chi-square or Fisher’s exact test was used for analyses of categorical variables wherever appropriate. Values of P <0.05 were deemed to be statistical significant.
Results
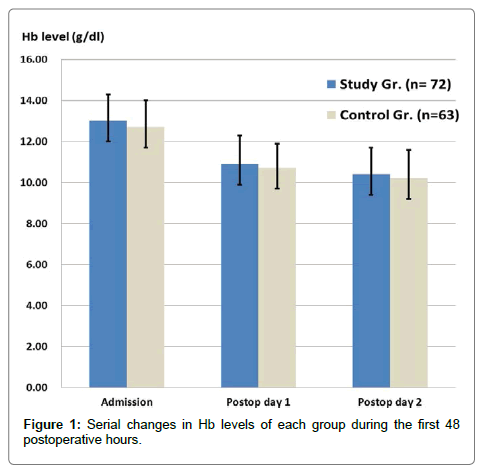
Over a period of 12 months, we retrospectively reviewed 152 patients who underwent unilateral primary cemented TKA at our hospital and 135 eligible patients (27 men) were enrolled in this study and all their charts were completely reviewed. In demographics (Table 1), both the control group and study group were similar with respect to age, proportion of gender, weight, height, type of anesthesia, preoperative hemoglobin level and side of deformity (Figure 1).
| Variables | Study Group (n=72) | Control Group (n=63) | p-value |
|---|---|---|---|
| Age (Yr) | 70.9 ± 7.5 | 70.4 ± 7.3 | n.s. |
| Side (Left /Right) | 34 / 38 | 32 / 31 | n.s. |
| Body Height (cm) | 155.8 ± 9.2 | 154.0 ± 6.8 | n.s. |
| Body weight (Kg) | 63.5 ± 11.6 | 64.7 ± 9.4 | n.s. |
| Body mass index (kg/m2 ) | 26.7 ± 3.8 | 27.3 ± 3.4 | n.s. |
| Hb level at admission (g/dL) | 13.0 ± 1.3 | 12.7 ± 1.3 | n.s. |
Table 1: Demographics.
Table 2 showed the peri-operative details. Under consistent surgical techniques by the same surgeon and operation setting, the mean operation time and ischemic time were similar between the 2 groups (p=0.31). After surgery, both group consumed similar dosages of rescue narcotics to relieve their breakthrough pain. Also, there was no different in inpatient stay between the 2 groups (p=0.678).
| Variables | Study Group (n=72) | Control Group (n=63) | p-value |
|---|---|---|---|
| Operative time (min) | 77.0 ± 10.7 | 79.8 ± 12.6 | n.s. |
| Ischemia time (min) | 50.5 ± 7.3 | 52.9 ± 8.7 | n.s. |
| Rescue Analgesics (mg)* | 20.6 ± 18.2 | 20.6 ± 16.8 | n.s. |
| Inpatient stay (days) | 6.3 ± 1.2 | 6.2 ± 1.1 | n.s. |
*in morphine equivalent
Table 2: Perioperative results.
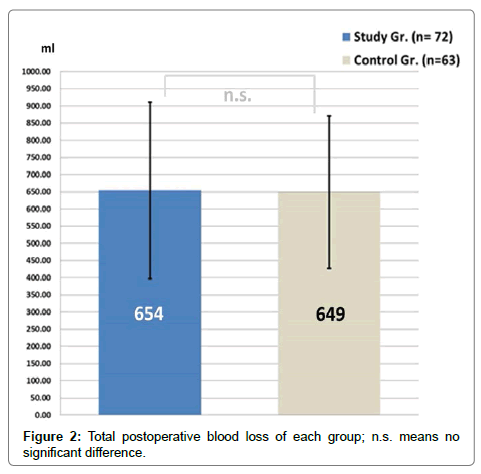
After surgery, the minimum postoperative Hb level, the maximum Hb reduction during inpatient stay and the mean estimated blood loss were similar in both groups in spite of the intervention of epinephrine-containing analgesics (Figure 2) (p =0.286, 0.634, and 0.902 respectively; Table 3).
| Variables | Study Group (n= 72) |
Control Group (n=63) |
p -value | |
|---|---|---|---|---|
| Hb level | Postop day 1 | 10.9 ± 1.4 | 10.7 ± 1.2 | n.s. |
| Postop day 2 | 10.4 ± 1.3 | 10.2 ± 1.4 | n.s. | |
| Reduction in Hb | At 24 hours | 2.1 ± 0.8 | 2.0 ± 0.7 | n.s. |
| At 48 hours | 2.5 ± 0.9 | 2.4 ± 0.8 | n.s. | |
| Calculated blood loss (mL) | 654.0 ± 257.1 | 648.8 ± 222.0 | n.s. | |
| Allogenic Transfusion rate (%) | 13.9 | 12.7 | n.s. | |
| Units of red cell transfused | 23 | 22 | n.s. | |
n.s. means no significant difference.
Table 3: Perioperative blood loss between groups.
During the study period, a total of 45 units of packed red blood cell were transfused to alleviate acute postoperative anemia. Under the same trigger, 10 patients in the study group and 8 patients in the control group underwent transfusion (Figure 3). There were no significant differences in transfusion rates between the 2 groups (Table 3).
In this series, no clinical significant DVT events developed after surgery, and no related wound complications [5,12] like surrounding skin damage, infection or dehiscence was observed in both groups at their discharge and first follow-ups. Certain problems associated with non-drained surgeries like ecchymosis and thigh swelling were transient and experienced in a similar proportion of each group (not listed here).
Discussion
Total knee arthroplasty (TKA) frequently involves substantial blood loss [15,16] and moderate to severe postoperative pain [17,18]. Thus, it is quite attractive to simultaneously alleviate postoperative pain as well as blood loss with a simple measure, local application of long-acting analgesic mixing vasoconstrictor in the surgical field. This study investigated whether there would be a difference in blood loss after the local application of epinephrine-containing analgesic in our primary non-drained TKAs. Various bleeding-related indices had been measured in the present study; however, on a comparative basis, we could not demonstrate any significant reduction in blood loss after the intervention of this simple formula.
For better pain management after primary TKAs, the authors decided to implement this infused analgesic solution since 2008 according to the supportive literature [2,4-7]. Meanwhile, further blood conservation was therefore anticipated not only by the infused vasoconstricting epinephrine but also by an enhanced tamponade by the added volume within the non-drained space. Similar concepts had been proposed by Yamada et al. [12] and Ryu et al. [19], but the augment solutions were infused through the drains in their studies rather than the direct bolus injection in the present study.
To our knowledge, local epinephrine-containing analgesics had been long applied to relieve pain after TKAs for decades [1-9]; however, related literature explored the efficacy of epinephrine- containing analgesics in reducing postoperative blood loss is inconsistent [2,4-9]. Great diversities between studies, including the study designs, use of drain or not, timing and location of injection, and even DVT prophylaxis [2,4-9], were found. Unlike wound lavage or staged per articular injections in other studies, we adopted a different method and timing to administer the epinephrine- containing analgesic with a bolus injection after closure of arthrotomy in order to save ischemic time and potential wound complications. Hence, the little hemostatic effect of the intra-articular analgesic might be explained either by the insufficient infused volume to further enhance an established tamponade or by a pharmacologic rebound phenomenon [9] balancing the initial vasoconstriction by epinephrine. Nevertheless, our results are in keeping with the authors who reported on the lack of efficacy of these vasoconstrictor agents [7-9].
In current practice of primary TKAs, limited use of drainage is to be expected based on its little benefits [20-22]. As a result, the clinical relevance of this study was that a simple but common intervention with intra-articular epinephrine and bupivacaine could not further provide hemostatic benefit in the setting of primary TKA without drainage. Further studies exploring newer potent regimens for topical hemostasis with a simple administering cocktail are still needed.
Since 2010, more and more trials demonstrated the application of tranexamic acid (TXA), an antifibrinolytic agent effectively reduce postoperative blood loss as well as requirements of blood transfusion [23,24]. So was the same experience of our clinical practice using TXA after we failed to demonstrate any hemostatic benefits using simple epinephrine-containing analgesics in non-drained TKAs. Thus, regarding to the clinical relevance, we believe the present study helps to clarify the blood-saving effect from the TXA rather than the remaining epinephrine-containing analgesics in the various cocktail regimens.
The use of intraarticular analgesics had been recommended to enhance control of postoperative pain. [1,3,25] Badner et al. [1] studied patients with TKA receiving intra-articular injections of bupivacaine and epinephrine and reported that injections performed after wound closure decreased the need for narcotics and increased the range of motion in the knee. Busch et al. [3] demonstrated that a periarticular intraoperative injection containing ropivacaine, ketorolac, epimorphine, and epinephrine decreased visual analog scores for pain and increased patient satisfaction after TKA. In clinical practice, we did observe some mentioned benefits after the intervention of intra-articular analgesic, including early trends in lower pain level, longer duration before the first call for rescue narcotics and improved range of motion. However, in this study, the equal consumption of rescue narcotics of each group during the similar inpatient stay implies a later rebound requirement for rescue narcotics in the following period in spite of the initial analgesic benefit. Our result supports the findings of the aforementioned studies suggested a relatively short-term benefit in postoperative pain management using similar formulae [2,3,26-28]. Lombardi et al. [2] described higher pain levels experienced on postoperative days 1 and 2 after higher activities levels obtainable on the day of surgery. Ritter et al. [27] investigated the effectiveness of intra-articular morphine and/ or bupivacaine injections after TKA, finding no significant difference or superior analgesic effects in the first 24 hours postoperatively.
Summary and Limitations
This study has a number of limitations, including the lack of randomization, retrospective nature and suboptimal estimation of blood loss. However, for a single surgeon, it is more practical to consistently execute all the consecutive procedures without technical disturbances by randomization. Also, the well-matched demographic features of each group, consistent surgical techniques and operation settings, standardized nursing and rehabilitation program, identical transfusion trigger and even the postoperative pain control protocol all enabled us to look at the intervention of intra-articular epinephrine-containing analgesic as the only variable in the comparison, and our data were reliable for clinical interpretation.
In summary, our series suggests that analgesic solution mixing epinephrine offer little blood-saving benefits in the primary total knee arthroplasty without drains. Neither improved hemostasis nor favorable early clinical outcomes were obtained by the study regimen; new and potent agents are still required and to be explored. Although this local regimen has been shown to be blood-saving in some studies, in non-drained primary TKAs, analgesic mixing epinephrine does not help save blood or reduce transfusion requirement.
References
- Badner NH, Bourne RB, Rorabeck CH (1996) Intra-articular injection of bupivacaine in knee-replacement operations. Results of use for analgesia and for preemptive blockade. J Bone Joint Surg Am 78: 734-378.
- Lombardi AV, Jr., Berend KR, Mallory TH (2004) Soft tissue and intra-articular injection of bupivacaine, epinephrine, and morphine has a beneficial effect after total knee arthroplasty. Clin Orthop Relat Res 125-30.
- Busch CA, Shore BJ, Bhandari R (2006) Efficacy of periarticular multimodal drug injection in total knee arthroplasty, a randomized trial. J Bone Joint Surg Am 88: 959-963.
- Padala PR, Rouholamin E, Mehta RL (2004) The role of drains and tourniquets in primary total knee replacement: A comparative study of TKR performed with drains and tourniquet versus no drains and adrenaline and saline in¬filtration. J Knee Surg 17: 24-27.
- Gasparini G, Papaleo P, Pola P (2006) Local infusion of norepinephrine reduces blood losses and need of transfusion in total knee arthroplasty. Int Orthop 30: 253-256.
- Anderson LA, Engel GM, Bruckner JD (2009) Reduced blood loss after total knee arthroplasty with local injection of bupivacaine and epinephrine. J Knee Surg 22: 130-136.
- Malone KJ, Matuszak S, Mayo D (2009) The effect of intra-articular epinephrine lavage on blood loss following total knee arthroplasty. Orthopedics 32: 100-103.
- Mesa-Ramos F, Mesa-Ramos M, Maquieira-Canosa C (2008) Predictors for blood transfusion following total knee arthroplasty: a prospective randomised study. Acta Orthop Belg 74: 83-89.
- Joo JH, Park JW, Kim JS (2011) Is intra-articular multimodal drug injection effective in pain management after total knee arthroplasty? A randomized, double-blinded, prospective study. J Arthroplasty 26: 1095-1099.
- Banerjee S, Issa K, Pivec R, McElroy MJ (2013) Intraoperative pharmacotherapeutic blood management strategies in total knee arthroplasty. J Knee Surg 26: 379-385.
- Chang CW, Lan SM, Tai TW (2012) An effective method to reduce ischemia time during total knee arthroplasty. J Formos Med Assoc 111: 19-23.
- Yamada K, Imaizumi T, Uemura M (2001) Comparison between 1-hour and 24-hour drain clamping using diluted epinephrine solution after total knee arthroplasty. J Arthroplasty 16: 458-462.
- Gibon E, Courpied JP, Hamadouche M (2013) Total joint replacement and blood loss: what is the best equation? Int Orthop 37: 735-739.
- Kalairajah Y, Simpson D, Cossey AJ (2005) Blood loss after total knee replacement: effects of computer assisted surgery. J Bone Joint Surg Br 87: 1480-1482.
- Bong MR, Patel V, Chang E, Issack PS, Hebert R, Di Cesare PE (2004) Risks associated with blood transfusion after total knee arthroplasty. J Arthroplasty 19: 281-287.
- Zhang W, Li N, Chen S, Tan Y, Al-Aidaros M, et al. (2014) The effects of a tourniquet used in total knee arthroplasty: a meta- analysis. J Orthop Surg Res 9:13.
- Kelley TC, Adams MJ, Mulliken BD (2013) Efficacy of multimodal perioperative analgesia protocol with periarticular medication injection in total knee arthroplasty: a randomized, double-blinded study. J Arthroplasty 28: 1274-1277.
- Goyal N, McKenzie J, Sharkey PF (2013) The 2012 Chitranjan Ranawat award: intraarticular analgesia after TKA reduces pain: a randomized, double-blinded, placebo-controlled, prospective study. Clin Orthopaed Related Res 471: 64-75.
- Ryu J, Sakamoto A, Honda T, Saito S (1997) The postoperative drain clamping method for hemostasis in total knee arthroplasty. Reducing postoperative bleeding in total knee arthroplasty. Bull Hosp Jt Dis 56: 251-254.
- Tai TW, Jou IM, Chang CW (2010) Non-drainage is better than 4-hour clamping drainage in total knee arthroplasty. Orthopedics 10: 156-160.
- Kumar S, Penematsa S, Parekh S (2007) Are drains required following a routine primary total joint arthroplasty? Int Orthop 31: 593-596.
- Kęska R, Paradowski TP, Witoński D (2014) Outcome in primary cemented total knee arthroplasty with or without drain: A prospective comparative study. Indian J Orthop 48: 404-409.
- Alshryda S, Sarda P, Sukeik M (2011) Tranexamic acid in total knee replacement: a systematic review and meta-analysis. J Bone Joint Surg Br 93: 1577-1585.
- Yang ZG, Chen WP, Wu LD (2012) Effectiveness and safety of tranexamic acid in reducing blood loss in total knee arthroplasty: a meta-analysis. J Bone Joint Surg Am 94: 1153-1159.
- Tanaka N, Sakahashi H, Sato E (2001) The efficacy of intra-articular analgesia after total knee arthroplasty in patients with rheumatoid arthritis and in patients with osteoarthritis. J Arthroplasty 16: 306-311.
- Browne C, Copp S, Reden L (2004) Bupivacaine bolus injection versus placebo for pain management following total knee arthroplasty. J Arthroplasty 19: 377-380.
- Ritter MA, Koehler M, Keating EM (1999) Intra-articular morphine and/or bupivacaine after total knee replacement. J Bone Joint Surg Br 81: 301-303.
- Rosen AS, Colwell CW Jr, Pulido PA (2010) A randomized controlled trial of intraarticular ropivacaine for pain management immediately following total knee arthroplasty. HSS J 6: 155-159.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi