Research Article, J Athl Enhanc Vol: 8 Issue: 1
Muscular Preconditioning Using Phototherapy Improves the Physical Work Capacity of the Quadriceps when Applied between Repeated Bouts of Resistance Exercise
Borsa PA1*, Dale RB2, Levine D3 and Crow JA4
1Department of Applied Physiology & Kinesiology, University of Florida, Gainesville, FL, USA
2Department of Physical Therapy, University of South Alabama, Mobile, AL, USA
3Department of Physical Therapy, University of Tennessee at Chattanooga, Chattanooga, TN, USA
4Department of Applied Physiology & Kinesiology, University of Florida, Gainesville, FL, USA
*Corresponding Author : Paul A Borsa
PhD, ATC, FACSM, Associate Professor, Department of Applied Physiology and Kinesiology University of Florida, 1864 Stadium Rd., 100 FLG Gainesville, FL 32611-8205, USA
Tel: 352-294-1726
Fax: 352-392-5262
E-mail: pborsa@hhp.ufl.edu
Received: October 15, 2018 Accepted: February 01, 2019 Published: February 08, 2019
Citation: Borsa PA, Dale RB, Levine D, Crow JA (2019) Muscular Preconditioning Using Phototherapy Improves the Physical Work Capacity of the Quadriceps when Applied between Repeated Bouts of Resistance Exercise. J Athl Enhanc 8:1.
Abstract
Preconditioning skeletal muscle with phototherapy is anexperimental technique for producing resistance against muscularfatigue during and after strenuous exercise. This study examinedthe ergogenic effects of phototherapy when applied intermittentlyduring passive recovery between repeated bouts of isokineticquadriceps exercise. A triple-blind, repeated measures, placebocontrolled,cross-over design was used in this study to comparethe effects of phototherapy on torque/work and power outputof the knee extensors in 20 healthy individuals (10 men, 10women). The exercise protocol consisted of four repeated boutsof isokinetic exercise. Each bout consisted of 30 continuousknee extension concentric contractions at an angular velocityof 75°/s with a 4min recovery interval between bouts. Subjectsreceived active and sham phototherapy in random order duringpassive recovery between exercise bouts. Phototherapy wasadministered using a near-infrared laser to the quadricepsfemoris muscle group at 10 J/cm2 (total dose of 1600-2400 J).Performance measures included normalized peak torque [Nm/kg], average peak torque [Nm], total work [Nm], and averagepower [W]. During the second, third and fourth exercise bouts,subjects produced significantly more torque (normalized andaverage) and total work/average power output during the activephototherapy condition compared to the sham phototherapycondition. Phototherapy applied as a preconditioning treatmentduring passive recovery intervals between bouts high of intensityand high volume resistance exercise significantly attenuatedfatigue and maintained muscular performance of the quadricepsfemoris muscle group. Preconditioning skeletal muscle withphototherapy intermittently during recovery intervals with fatiguingexercise may be a beneficial, non-invasive and safe ergogenic aidfor athletes that require high levels of muscular endurance.
Keywords: Near-infrared; Ergogenic; Isokinetic; Skeletal muscle; Fatigue; Preconditioning
Introduction
Exercise and sports scientists are constantly searching for ways to optimize the body’s response to strenuous physical activity by prolonging the ability of skeletal muscle to perform work while resisting exertional fatigue. Skeletal muscle fatigue is a rate-limiting factor for athletes who participate and compete in sports that require muscular endurance and sustained levels of strength [1]. Fatigue impairs the ability of skeletal muscle to generate force, and reduces a muscle’s capacity to perform work over an extended time period [2,3]. The decrease in muscle function associated with fatigue is believed to be a result of metabolic alterations such as substrate depletion (lack of ATP and glycogen), oxidative stress, tissue ischemia/hypoxia and blood acidification [2-4]. One ergogenic aid that has garnered interest in the sports and exercise community is the use of phototherapy or light therapy as a preconditioning treatment for skeletal muscle.
Preconditioning skeletal muscle with phototherapy or “muscular preconditioning” is an experimental technique for producing resistance against muscular fatigue and biochemical stress during and after strenuous exercise [5]. This pre-emptive technique exposes skeletal muscle to light energy in order to prepare the muscle for the rigors of strenuous exercise or physical activity. The concept for muscular preconditioning was adapted from ischemic preconditioning (IPC) for the heart [6,7]. This novel technique exposes cardiac muscle to periods of sub-lethal ischemia in order for the tissue to build a resistance against damage from future ischemic events such as stroke or myocardial infarction [8]. Muscular preconditioning using near-infrared (NIR) lasers or light-emitting diodes (LEDs) has been adopted by athletic trainers and other sports medicine professionals as an essential performance enhancing and injury prevention technique in amateur and professional sports such as baseball, rugby, soccer and track and field [9]. Research addressing the ability of phototherapy to modulate biological processes associated with skeletal muscle contractile function has yielded promising results [9-19]. Such modulatory processes associated with phototherapy are referred to as photobiomodulation. Photobiomodulation involves the use of narrow bands of non-ionizing light usually in the near-infrared spectrum (800 nm – 1000 nm) to induce biochemical changes in tissue in a stimulatory manner [20]. Phototherapy has been found to be a viable treatment modality for skeletal muscle because it is safe, easy to administer, non-invasive and has no known side effects and few reported contraindications [21].
During phototherapy, light energy is delivered to cells by penetrating the skin producing both photo-physical and photochemical events in the underlying tissues. The mechanisms associated with phototherapy have been studied at the cellular level by numerous basic science research groups [22-25]. The current understanding of the primary mechanism of action is that scattering and transmitting photons are absorbed by chromophores located in cytochrome c oxidase (CCO) and other heme complexes (hemoglobin/myoglobin) [5,22]. Cytochrome c oxidase is located in the inner membrane of the mitochondria and is the terminal enzyme of the electron-transport chain in eukaryotic cells, mediating the transfer of electrons from CCO to molecular oxygen [22]. The photon absorption dissociates inhibitory nitric oxide (NO) from CCO, leading to an increase in electron transport, increased mitochondrial membrane potential, and increased adenosine triphosphate (ATP) production [26-28]. Similar to CCO, hemoglobin (Hb) and myoglobin (Mb) are photoreceptors that absorb NIR light. When Hb and Mb are exposed to light, the molecules experience a change in redox state, thus releasing oxygen to the cytosol [19,20]. This increase in molecular oxygen can be used by the mitochondria for cellular respiration, thereby facilitating the production of ATP. Collectively, these intracellular changes lead to macroscopic effects including cyto-protection, increased oxidative capacity, and enhanced skeletal muscle contractile function [13,27,28].
Numerous muscular preconditioning studies have been performed that compared active phototherapy to sham phototherapy for the biceps brachii and quadriceps femoris muscle groups using isometric and isoinertial resistance exercise, as well as isokinetic dynamometry to measure muscle performance. Most studies to date have assessed the effects of phototherapy administered before strenuous exercise while the muscle tissue is at rest and not under physiological stress [10,11,15-19]. Several studies have investigated the effects of phototherapy after strenuous exercise or muscular postconditioning. This therapeutic approach is designed to administer phototherapy after skeletal muscle has been placed under physiologic stress as a means of improving functional and biochemical recovery [29]. Tissue cells that have been placed under metabolic or mechanical stress (e.g. injury or exercise) have been found to be more responsive to phototherapy [30]. More recent studies have investigated the effects of phototherapy applied intermittently between repeated bouts of physical exercise after muscle has been placed under physiologic stress [31-33]. This form of preconditioning occurs when phototherapy is administered to skeletal muscle during passive recovery after being placed under physiological stress and is in preparation for a repeated bout of exercise. Muscular preconditioning using phototherapy has direct applications to sports activity and clinical care where improvements in physical work capacity of the athlete or patient would be helpful for performance enhancement, injury prevention, functional progression, and/or improving functional recovery. Our objective was to examine the ergogenic effects of phototherapy when applied intermittently during passive recovery between repeated bouts of isokinetic quadriceps exercise. In this experimental model, the quadriceps has been placed under physiological stress (isokinetic exercise) and is in preparation mode for a repeated bout of isokinetic exercise. We hypothesize that skeletal muscle exposed to phototherapy between repeated bouts of high intensity and high volume resistance exercise would be able to delay the onset and extent of muscular fatigue and maintain higher levels of muscular torque and work output compared to sham phototherapy.
Materials and Methods
Study design
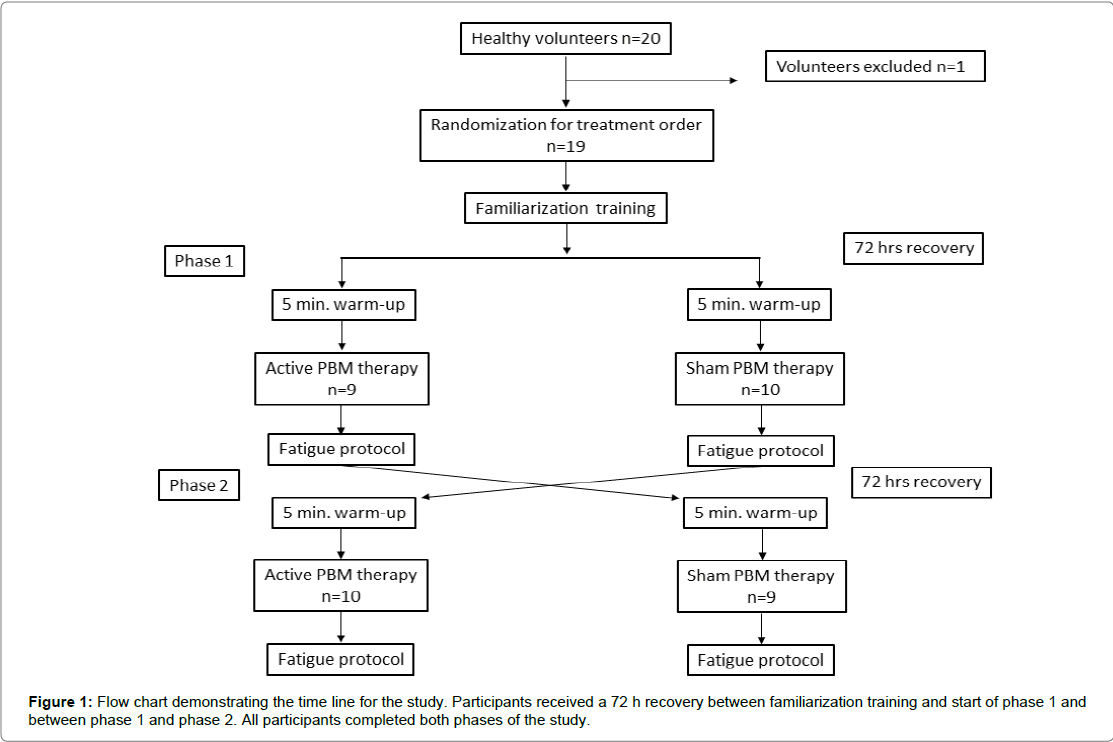
A triple-blind, repeated measures, sham-controlled, cross-over design was used in this study to compare the effects of phototherapy on torque/work and power output of the knee extensors. Each subject served as his/her own control and received an active and a sham phototherapy session at least 3 days apart. The treatment order was randomized, and the sham phototherapy served as the placebocontrol (Figure 1). The therapist administering phototherapy was separate from the experimenter who conducted and supervised the fatigue protocol. The subjects and experimenter were blinded to the treatment order (active or sham phototherapy). The therapist was aware of the treatment order, but was blinded to the outcome of the performance measures.
Participants
Twenty individuals (10 women, 10 men; age=23 ± 4.4 years, height=1.7 ± 0.08 m, mass=72.2 ± 5.5 kg) volunteered to participate in the study. Volunteers were eligible for inclusion if they were healthy and between the ages of 18 and 30 years.
Exclusion criteria were as follows: current or past musculoskeletal injury to their dominant knee, history of neuromuscular or cardiovascular disorder, or use of dietary supplements or pharmacological agents for the purpose of building muscle mass. All participants provided signed informed consent, and the study was approved by the Institutional Review Board of the University. All experimental procedures followed in this study conformed to the Code of Ethics of the World Medical Association (Helsinki Declaration of 1975).
Fatigue Protocol/Familiarization Training
Participants were instructed to refrain from engaging in any lower extremity exercise (running, cycling or lifting weights) in the 48 h before testing.
Participants were required to complete a fatigue protocol for the quadriceps muscle group. The BIODEX System 3 Pro isokinetic dynamometer (Biodex Medical Systems, Shirley, NY, USA) was used for all neuromuscular strength and endurance exercise testing.
Participants initially completed a series of familiarization training sessions prior to completing the fatigue protocol to eliminate a possible training effect. The familiarization sessions were identical in design to the fatigue protocol. Subjects were entered into the exercise testing phase of the study when peak torque values had less than a 10% difference between training sessions. Subjects averaged 2.6 training sessions and all participants had at least 2 training sessions. All familiarization training and exercise testing sessions were held a minimum of 72 h apart, and if the participant self-reported symptoms of muscle soreness (≥ 2 out of 10) they received an additional day of rest until their score dropped below 2.
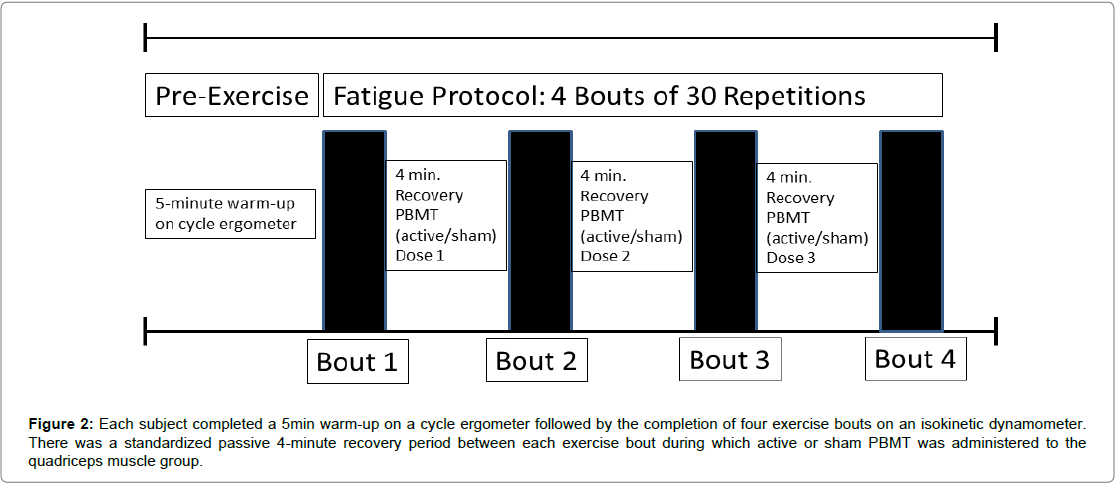
Participants prepared themselves for the fatigue protocol with a 5 min warm-up on a Schwinn® Airdyne® cycle ergometer at an intensity of 3 (moderate) out of 10 (maximal) using the modified Borg category-ratio scale of perceived exertion. The fatigue protocol consisted of 4 repeated bouts of 30 isokinetically controlled, concentric knee extensions. Each repetition was performed through a 75 range of motion (15-90°) at an angular velocity of 75°/s. There was a 4 min standardized passive recovery period between each exercise bout during which active or sham phototherapy was Administered to the quadriceps muscle group (Figure 2). Each subject was instructed to provide maximal effort during knee extension while submaximal effort was used during knee flexion to return to the start position. Torque and work/power output values were recorded for the knee extension component of the fatigue protocol since the quadriceps was the target muscle group for the study.
Figure 2: Each subject completed a 5min warm-up on a cycle ergometer followed by the completion of four exercise bouts on an isokinetic dynamometer. There was a standardized passive 4-minute recovery period between each exercise bout during which active or sham PBMT was administered to the quadriceps muscle group.
Performance/Fatigue resistance measures
The following isokinetic performance measures were recorded during each exercise bout: Normalized Peak Torque, Average Torque, Total Work, and Average Power. A definition for each measure is provided [34].
Normalized peak torque
The maximum torque produced during a set of resistance exercise that is normalized to body mass (Nm/Kg).
Average torque
The peak torque from every repetition averaged across the number of repetitions per set.
Total work
The cumulative amount of work (or torque) produced by the participant during a set of resistance exercise (Nm), also referred to as the “area under the torque curve”.
Average power
The total work divided by time (W). This measure represents how quickly a muscle can produce torque. The four isokinetic performance measures were used to characterize the fatigue response of the quadriceps muscles as a function of the intervention (active or sham phototherapy) and exercise bout (1, 2, 3 or 4).
Phototherapy
Phototherapy was administered using a commercially available FDA approved class IV high power therapeutic laser. The therapeutic laser (LCT-1000™, LiteCure Medical, LLC, Newark, DE) emitted photonic wavelengths at 810 nm and 980 nm. These wavelengths were chosen for their ability to penetrate deeply into tissue to maximize the photon flux (number of photons per unit area per second) [35]. The laser had a combined output power of 10 W (20% 810 nm, 80% 980 nm at 1.8 W/cm2 irradiance).
To deliver a uniform energy density of 10 J/cm2 to the quadriceps muscle group, a template was placed over the muscles to assure even distribution of light energy applied in a continuous scanning mode with a trackball in contact with the skin. Total dosages per treatment ranged from 1600 J to 2400 J. The treatment times ranged from 160 s to 240 s. The sham therapy was identical in treatment time and application technique with the exception that the output power of the laser was disabled and only the aiming beam (red light) was active. The aiming beam delivered a single wavelength of 650 nm with an output power of 4 mW. After completion of each phototherapy session subjects were asked what type of treatment they thought they received (active or sham treatment). A chi-square analysis of their responses indicated that the sham treatment was not distinguishable from the active treatment (χ2=8.2; p=0.63).
Statistical analysis
Statistical analyses were performed using SPSS v. 23.0 (IBM, Armonk, NY) for Windows. A 2 (treatment) × 4 (time) repeated measures ANOVA with between-subject factors of treatment (active vs. sham) and time (Bout 1 – 4) was performed. The a priori alpha significance level was set at p<0.05, and all significant interactions were followed by post hoc analyses. Paired comparisons in the form of multiple paired t-tests were performed to identify significant betweencondition and within-time differences. To control for inflated alpha levels resulting from repeated comparisons we used the Bonferroni procedure to adjust the level of significance by dividing the original alpha by the number of comparisons (C=4) per dependent variable (0.05/4) with the adjusted alpha set at=0.013.
Results
Treatment × time interaction effects were statistically significant for each performance measure except average power: Normalized Peak Torque [F(3,54)=7.43; p<0.001], Average Torque [F(3,54)=4.65; p=0.006], Total Work [F(3,54)=3.09; p=0.035] and Average Power [F(3,54)=2.69; p=0.055]. Paired comparison results are listed in Table 1.
| Bout 1 | Bout 2 | Bout 3 | Bout 4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Active | Sham | P value | Active | Sham | P value | Active | Sham | P value | Active | Sham | P value | |
| Variable | ||||||||||||
| Normalized PT (N/m) | 76.2 ± 15 (69.6,82.9) |
76.3 ± 16 (69.4,83.7) |
0.97* d=0.008 |
81.6 ± 14 (75.8,88.0) |
74.1 ± 14 (67.9,80.8) |
0.002 d=0.84 |
78.4 ± 15 (72.2,85.1) |
71.8 ± 13 (66.2,78.2) |
<0.001 d=1.0 |
75.5 ± 15 (69.3,82.3) |
69.1 ± 12 (63.9,74.8) |
<0.001 d=1.0 |
| Average PT (N/m) |
97.232 (83.8,112.6) |
95.1 ± 31 (82.5,109.8) |
0.40* d=0.19 |
98.3 ± 32 (85.4,113.3) |
88.6 ± 25 (78.0,101.0) |
0.004 d=0.75 |
91.5 ± 25 (81.0,103.1) |
85.4 ± 25 (74.7,97.5) |
<0.001 d=1.0 |
87.4 ± 23 (77.5,98.1) |
83.3 ± 22 (74.1,93.3) |
0.022* d=0.58 |
| Total Work (J) |
2553.7 ± 774 (2224.5,2926.3) |
2532.6 ± 864 (2175.2,2947.7) |
0.82* d=0.05 |
2514.1 ± 757 (2200.4,2896.3) |
2255.8 ± 614 (1985.0,2521.3) |
0.009 d=0.67 |
2316.5 ± 583 (2078.7,2600.1) |
2110.0 ± 582 (1863.1,2376.8) |
<0.001 d=1.0 |
2192.1 ± 524 (1968.4,2435.4) |
2015.2 ± 446 (1824.3,2212.2) |
0.001 d=0.90 |
| Average Power (W) | 111.3 ± 36 (94.8,127.2) |
109.3 ± 37 (92.9,126.3) |
0.49* d=0.16 |
111.2 ± 34 (96.5,127.3) |
100.4 ± 29 (88.1,113.5) |
0.009 d=0.68 |
102.3 ± 27 (90.0,114.0) | 94.6 ± 27 (83.3,106.8) |
<0.001 d=1.0 |
99.5 ± 28 (87.9,112.0) |
91.4 ± 23 (81.8,100.9) |
0.001 d=0.86 |
PT-peak torque
*NSD-no significant difference between conditions per exercise bout
Table 1: Performance measures for exercise bouts 1-4 and treatment conditions (Active v. Sham), Mean ± SD (95% Confidence Intervals), P value and effect size (Cohen’s d).
There was no significant difference in muscle performance between treatment conditions (active v. sham) during the first exercise bout. However, significant muscle performance decrements were observed between and within treatment conditions during bouts 2, 3 and 4.
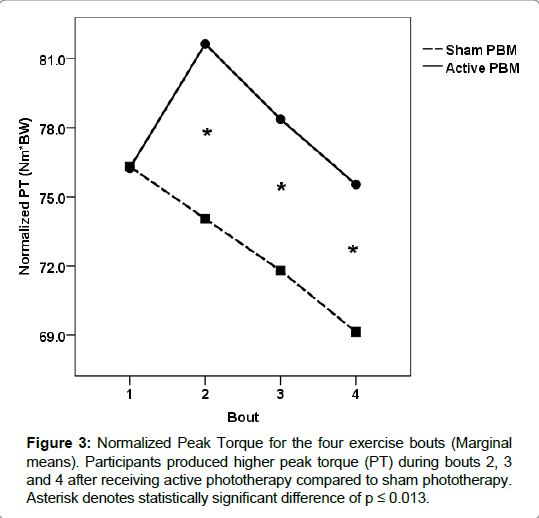
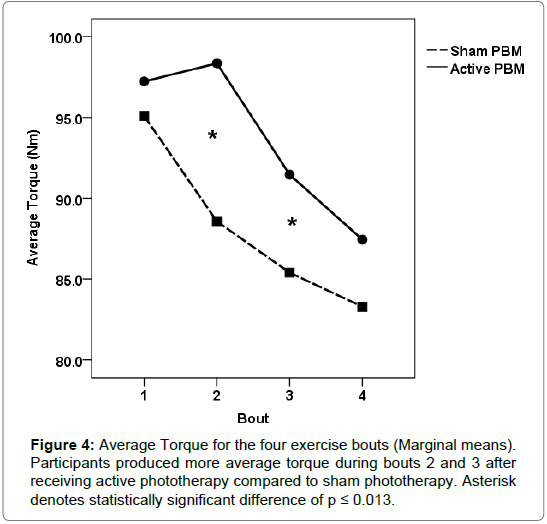
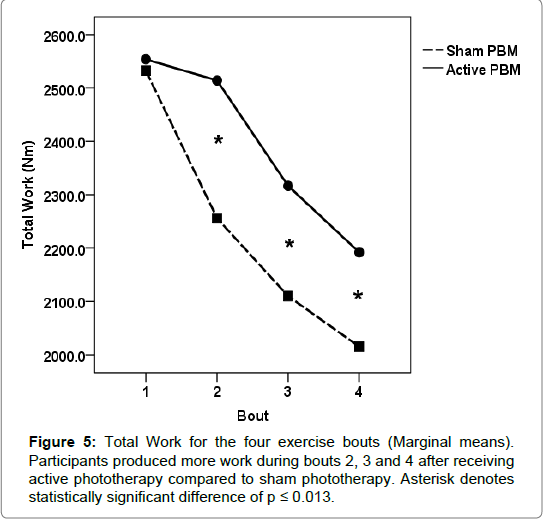
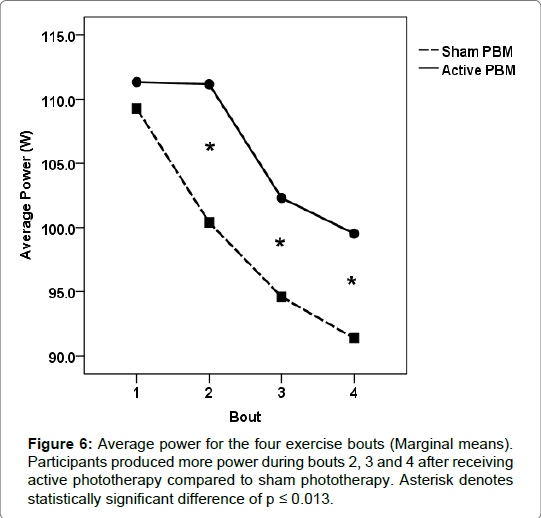
For the active treatment condition, the onset of fatigue was significantly delayed during the second bout of exercise compared to the sham condition for each performance measure (torque, work and power). During the second exercise bout, participants produced significantly more torque (normalized and average) and work/power output during the active treatment condition compared to the sham condition. Torque values (normalized peak and average) increased from the first exercise bout by 6.6%, and 1%, respectively for the active treatment condition, while the sham condition values decreased by 3%, and 6.8%, respectively (Figures 3 and 4). Total work and average power values decreased dramatically during the sham condition, 11% and 8%, respectively, compared to minimal decrements during the active treatment condition, 1% and <1%, respectively (Figures 5 and 6).
After the second bout of exercise, fatigue-induced performance decrements occurred for both treatment conditions and continued through the third and fourth bouts of exercise. The rate of decline in torque and work/power output from exercise bout 2 through 4 were similar between treatment conditions, although the active treatment condition continued to produce significantly higher torque and work/ power outputs than the sham treatment condition.
For the active treatment condition, torque values (NPT and average) declined from the second exercise bout by 4 and 7%, respectively, and both total work and average power output declined by 8%. For the sham treatment condition, torque values declined by 3 and 4%, respectively, and total work and average power values both dropped from the second exercise bout by 6%. For the active treatment condition, torque values (NPT and average) declined from the third exercise bout by 4 and 4%, respectively, and both total work and average power output declined by 5 and 3%, respectively. For the sham phototherapy condition, torque values declined by 4 and 2%, respectively, and total work and average power values both dropped by 4 and 3%, respectively.
Discussion
Our objective was to examine the ergogenic (performance enhancing) and fatigue resistance effects of phototherapy (active or sham) using a high volume/high intensity muscle fatiguing resistance exercise protocol. The main findings of this study were that (1) after receiving active phototherapy a significant increase in torque output for the quadriceps during the second bout of exercise was observed compared to the decline in torque output for the sham condition, (2) there were similar rates of decline in muscle performance during bouts 2, 3 and 4 for both treatment conditions, and (3)
Additional intermittent active phototherapy treatments between bouts 2 and 3 and 3 and 4 had no discernable ergogenic effects even though torque and work/power output by the quadriceps was significantly greater than the sham treatment condition.
The spike in torque and work/power output during the second bout of exercise for the active treatment condition indicates that the active phototherapy treatment enhanced muscle performance in the short-term, and additional preconditioning treatments between the 2nd and 3rd bouts and the 3rd and 4th bouts did not provide the same ergogenic effect that it did between bouts 1 and 2.
Our results indicate that phototherapy administered intermittently between exercise bouts was able to significantly improve and maintain the physical work capacity for the quadriceps femoris muscle group compared to the sham condition. The most dramatic ergogenic effect occurred between bouts one and two. Torque (normalized peak and average) output increased considerably during the second bout of exercise after participants received the active treatment, while conversely torque output declined significantly after sham treatment. Total work and average power were maintained between bouts 1 and 2 after receiving the active treatment, but also declined significantly during the sham condition.
After the second bout of exercise, muscular performance declined incrementally within each treatment condition over the course of bouts 3 and 4, however, for the active treatment condition, the quadriceps muscle group was able to conserve torque, work and power output during the last two exercise bouts.
Most studies to date have assessed the effects of phototherapy administered before exercise while the muscle tissue is in a resting, homeostatic state, and has not been stressed by physical exertion. Previous investigations of phototherapy’s pre-conditioning effect on skeletal muscle prior to high intensity resistance exercise have found significant increases in the number of repetitions or elapsed time to volitional fatigue (18, 19), and pre- to post-exercise maximum voluntary contraction (MVC) compared to a sham-control condition [10,11,16]; indicating a fatigue resistance effect of phototherapy.
More recently, animal and human studies have investigated the ergogenic and prophylactic effects of phototherapy applied between repeated bouts of resistance exercise when the muscle is in a state of passive recovery [31-33] de Brito Vieira et al. [33]. Used the number of knee extension repetitions to volitional fatigue as their criterion measure comparing the number of repetitions at baseline (pre-exercise) to the number of repetitions at 48 h post-exercise. In this study [33], subjects performed 3 sets of 20 repetitions of knee extensions using an isokinetic dynamometer at a controlled angular Velocity of 60/s. phototherapy (active or sham) was administered to their quadriceps femoris muscle between sets (1-2, 2-3, and again after set 3). Subjects were retested for their ability to perform repeated knee extensions until volitional fatigue at 48 h post-exercise to assess functional recovery. The group that received the active phototherapy between sets produced significantly more repetitions to failure at 48 h post-exercise/treatment compared to the sham condition. They speculated that phototherapy was able to facilitate the post-exercise recovery of the quadriceps femoris muscle over the 48 h time interval by limiting the extent of primary and secondary muscle damage, thus minimizing intramuscular levels of inflammatory cytokines, oxidative (ROS), and ischemic stress [33].
In our fatigue protocol, we measured the amount of torque, work and power output produced during exercise for our betweencondition (active vs. sham) comparisons. Consistent with the findings of de Brito Vieira et al. [33], we were able to show that phototherapy applied between bouts of resistance exercise produced positive ergogenic effects for the quadriceps femoris muscle group.
Electromyography does not quantify force production or work output, but rather EMG is a measure of muscle activation over time using signals from the electrical output of motor units within muscle tissue. The improved neuromuscular response of the motor units to sustained and repeated muscle contractions after phototherapy indicates a lack of ‘neural’ fatigue. De Brito Vieira et al. [33] speculated that neural drive to the muscle membrane was maintained after phototherapy likely due to improved metabolic conditions within the muscle.
An important issue to address when using a high intensity/high volume resistance exercise model is the disposition of the muscle at the time of phototherapy. In our experimental exercise model as well as others, administering the preconditioning treatment intermittently during passive recovery periods between exercise bouts exposes stressed skeletal muscle to phototherapy. Researchers have speculated that cells are more responsive to phototherapy when under physiological distress such as with mechanical injury (muscle damage) or metabolic insufficiency (elevated levels of lactate, ROS and inflammatory mediators; or decreased intracellular energy/ATP stores) as opposed to cells that have not been under physiological stress or in a state of homeostasis [30]. Researchers have further speculated that the mitochondria are more sensitive to light energy during periods of cell stress, and therefore exposing skeletal muscle to phototherapy during periods of stress restores cellular energy stores as well as protects tissue from mechanical and metabolic stress [14,30,33].
Previous studies have also provided evidence for the ability of phototherapy to enhance blood flow and microcirculation [36,37]. Increasing blood flow and microcirculation in muscle tissue exposed to phototherapy would lead to an increase in the delivery of oxygen and other energy substrates to exercising muscles, thus, leading to an increased ability for a muscle to perform work. High intensity and high volume endurance exercise uses primarily anaerobic or glycolytic pathways to supply energy to working muscles. A metabolic by-product of this form of exercise is lactic acid, a known inducer of muscle fatigue. Skeletal muscle treated with phototherapy and working primarily under anaerobic conditions may be better able to oxidize lactic acid to pyruvate, which may be used by the mitochondria to produce ATP via cellular respiration [38,39].
Overall, research findings have consistently shown a performance enhancing effect of phototherapy for enhancing skeletal muscle contractile function beyond that of placebo-control [9,11,13-19]. The mechanistic underpinnings of phototherapy are primarily linked to a vasodilatory effect for increasing local blood flow and elevated mitochondrial and respiratory chain activity for providing energy for optimizing muscle work during the exercise protocol and a more expedient recovery post-exercise. The important next steps need to be directed at a more in-depth analysis of the skeletal muscle’s myoelectrical as well as vascular and metabolic dynamics that occur in conjunction with receiving phototherapy.
Limitations
A limitation of this study is that we did not directly evaluate blood flow or tissue oxygenation levels, EMG activation, or blood lactate levels as other studies have done. Other limitations to this study include measuring the ergogenic effects in a controlled laboratory environment rather than in a sport-specific or clinical practice setting. Our fatigue protocol only measured torque and work output during the concentric phase of muscle contractions. Future studies may consider including torque and work output measurements from both phases (concentric and eccentric) of muscle actions as a means to capture a more comprehensive view of muscle contractile function. In addition, future studies should be designed to better replicate real sports performance settings in highly competitive and/or elite athletes or utilized during actual clinical cases.
Practical applications
Preconditioning skeletal muscle with phototherapy prior to fatiguing exercise is a rapidly growing area of investigation in the areas of sports medicine and exercise science. In our study, phototherapy (active or sham) was administered during passive recovery between bouts 2, 3 and 4. Therefore, the improvement in torque as well as the delay in the fatigue response for total work and average power indicates that phototherapy can provide an initial boost in energy to the muscle and help to conserve torque and work output during subsequent bouts.
Phototherapy administered between bouts of strenuous physical activity would be beneficial to an athlete by delaying the onset and extent of fatigue and prolonging muscular endurance. The quadriceps muscle group is vulnerable to exertional fatigue due to the high demands of the muscle group in sports and activities that require protracted periods of running, cycling, jumping, landing and cutting. From an injury prevention and performance enhancement perspective, phototherapy administered to the quadriceps intermittently during repeated bouts of high intensity and high volume exercise has the capability of producing an ergogenic effect for that muscle group. This ergogenic effect would be beneficial for an athlete by improving the muscles capacity to perform work while reducing the risk of fatigue-induced injury. Furthermore, our results have direct application to clinical practice for patients who are completing physical therapy where the quadriceps may be vulnerable to fatigue such as during recovery from Anterior Cruciate Ligament (ACL) reconstructive surgery or post-operative rehabilitation for a total knee replacement.
Conclusion
In conclusion, phototherapy applied as a preconditioning treatment during passive recovery intervals between bouts of high intensity and high volume resistance exercise significantly attenuated fatigue and maintained muscular performance of the quadriceps femoris muscle group. Preconditioning skeletal muscle with phototherapy intermittently during recovery intervals with fatiguing exercise may be a beneficial, noninvasive and safe ergogenic aid for athletes that require high levels of muscular endurance.
Declarations
Ethics approval and consent to participate
The study was approved by the Institutional Review Board (IRB) of the University of Tennessee at Chattanooga and complied with ethical standards of the latest Declaration of Helsinki (WMA, Oct. 2013).
Acknowledgements
We would like to thank LiteCure Medical, LLC who provided technical and equipment support for the study. LiteCure provided 2 lasers for the study and calibrated them prior to use.
References
- Allen DG (2009) Fatigue in working muscles. J Appl Physiol 106: 358-359
- Allen DG, Lamb GD, Westerblad H (2008) Skeletal muscle fatigue: cellular mechanisms. Physiol Reviews 88: 287-332.
- Enoka RM, Duchateau J (2008) Muscle fatigue: what, why and how it influences muscle function. J Physiol 586: 11-23.
- Westerblad H, Bruton JD, Katz A (2010) Skeletal muscle: energy metabolism, fiber types, fatigue and adaptability. Exp Cell Res 316: 3093-3099.
- Agrawal T, Gupta GK, Rai V, Carroll JD, Hamblin MR (2014) Pre-conditioning with low-level laser (light) therapy: light before the storm. Dose-Response 12: 619-649.
- Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 74: 1124-1136.
- Das M, Das DK (2008) Molecular mechanisms of preconditioning. IUBMB Life 60: 199-203.
- Koch S, Della-Morte D, Dave KR, Sacco RL, Perez-Pinson MA (2014) Biomarkers for ischemic preconditioning: finding the responders. J Cereb Blood Flow Metab 34: 933-941.
- Pinto HD, Vanin AA, Miranda EF, Tomazoni SS, Johnson DS, et al. (2016) Photobiomodulation therapy improves performance and accelerates recovery of high-level rugby players in field test: a randomized, cross-over, double-blind, placebo-controlled clinical study. J Strength Cond Res 30: 3329-3338.
- Baroni BM, Leal Junior EC, De Marchi T, Lopes AL, Salvador M, et al. (2010) Low level laser therapy before eccentric exercise reduces muscle damage markers in humans. Eur J Appl Physiol 110: 789-796.
- Baroni BM, Leal Junior EC, Geremia JM, Diefenthaeler F, Vaz MA (2010) Effect of light-emitting diodes therapy (LEDT) on knee extensor muscle fatigue. Photomed Laser Surg 28: 653-658.
- Baroni BM, Rodriques R, Freire BB, de Azevedo Franke R, Geremia JM, et al. (2015) Effect of low-level laser therapy on muscle adaptation to knee extensor eccentric training. Eur J Appl Physiol 115: 639-647.
- de Almeida P, Lopes-Martins RA, Tomazoni SS, Silva JA, Jr de Carvalho Pde T, et al. (2011) Low-level laser therapy improves skeletal muscle performance, decreases skeletal muscle damage and modulates mRNA expression of COX-1 and COX-2 in a dose-dependent manner. Photochem photobiol 87: 1159-1163.
- Ferraresi C, de Brito Oliveira T, de Oliveira Zafalon L, de Menezes Reiff RB, Baldissera V, et al. (2011) Effects of low level laser therapy (808 nm) on physical strength training in humans. Lasers Med Sci 26: 349-358.
- Larkin-Kaiser KA, Borsa PA, Baweja HS, Moore MA, Tillman MD, et al. (2016) Photobiomodulation delays the onset of skeletal muscle fatigue in a dose-dependent manner. Lasers Med Sci 31: 1325-1332.
- Larkin-Kaiser KA, Christou EA, Tillman MD, George SZ, Borsa PA (2015) Near-infrared light therapy to attenuate strength loss after strenuous resistance exercise. J Athl Train 50: 45-50.
- Leal Junior EC, Lopes-Martins RA, de Almeida P, Ramos L, Iversen VV, et al. (2010) Effect of low-level laser therapy (GaAs 904 nm) in skeletal muscle fatigue and biochemical markers of muscle damage in rats. Eur J Appl Physiol 108: 1083-1088.
- Leal Junior EC, Lopes-Martis RA, Frigo L, De Marchi T, Rossi RP, et al. (2010) Effects of low-level laser therapy (LLLT) in the development of exercise-induced skeletal muscle fatigue and changes in biochemical markers related to postexercise recovery. J Orthop Sports Phys Ther 40: 524-532.
- Leal Junior EC, Lopes-Martins RA, Vanin AA, Baroni BM, Grosselli D, et al. (2009). Effect of 830 nm low-level laser therapy in exercise-induced skeletal muscle fatigue in humans. Lasers Med Sci 24: 425-431.
- Anders JJ, Lanzafame RJ, Arany PR (2015) Low-level/laser therapy versus photobiomodulation therapy. Photomed Laser Surg 33: 183-184.
- Borsa PA, Larkin KA, True JM (2013) Does phototherapy enhance skeletal muscle contractile function and postexercise recovery? A systematic review. J Athl Train 48: 57-67.
- Karu TI (2010) Multiple roles of cytochrome c oxidase in mammalian cells under action of red and IR-A radiation. IUBMB Life 62: 607-610
- Silveira PC, Silva LA, Fraga DB, Freitas TP, Streck EL, et al. (2009) Evaluation of mitochondrial respiratory chain activity in muscle healing by low-level laser therapy. J Photochem Photobiol B 95: 89-92.
- Hayworth CR, Rojas JC, Padilla E, Holmes GM, Sheridan EC, et al. (2010) In vivo low-level light therapy increases cytochrome oxidase in skeletal muscle. Photochem Photobiol 86: 673-680.
- Morimoto Y, Arai T, Kikuchi M, Nakajima S, Nakamura H (1994) Effect of low- intensity argon laser irradiation on mitochondrial respiration. Lasers Surg Med 15: 191-199.
- Ferraresi C, Kaippert B, Avci P, Huang YY, Victor M, et al. (2015) Low-level laser (light) therapy increases mitochondrial membrane potential and ATP synthesis in C2C12 myotubes with a peak response at 3-6 hours. Photochem Photobiol 91: 411-416.
- Ferraresi C, Pires de Sousa MV, Huang YY, Bagnato VS, Parizotto NA, et al. (2015) Time response of increases in ATP and muscle resistance to fatigue after low-level laser (light) therapy (LLLT) in mice. Lasers Med Sci 30: 1259-1267.
- Albuquerque-Pontes GM, Vieira RP, Tomazoni SS, Caires CO, Nemeth V, et al. (2015) Effect of pre-irradiation with different doses, wavelengths, and application intervals of low-level laser therapy on cytrochrome c oxidase activity in intact skeletal muscle in rats. Lasers Med Sci 30: 59-66.
- dos Reis FA, da Silva BA, Laraia EM, de Melo RM, Silva PH, et al. (2014) Effects of pre- or post-exercise low-level laser therapy (830nm) on skeletal muscle fatigue and biochemical markers of recovery in humans: double-blind placebo- controlled trial. Photomed Laser Surg 32: 106-112.
- Karu T (2013) Is it time to consider Photobiomodulation as a drug equivalent? Photomed Laser Surg 31: 189-191.
- Felismino AS, Costa EC, Aoki MS, Ferraresi C, de Araujo Moura Lemos TM, et al. (2014) Effect of low-level laser therapy (808nm) on markers of muscle damage: a randomized double-blind placebo-controlled trial. Lasers Med Sci 29: 933-938.
- da Costa Santos VB, de Paula Ramos S, Milanez VF, Correa JC (2014) LED therapy or cryotherapy between exercise intervals in Wistar rats: anti-inflammatory and ergogenic effects. Lasers Med Sci 29: 599-605.
- de Brito Vieira WH, Bezerra RM, Queiroz RA, Maciel NF, Parizotto NA, et al. (2014) Use of low-level laser therapy (808nm) to muscle fatigue resistance: a randomized double-blind cross-over trial. Photomed Laser Surg 32: 678-685.
- Drouin JM, Valovich-McLeod TC, Shultz SJ, Gansneder BM, Perrin DH (2004) Reliability and validity of the Biodex system 3 pro isokinetic dynamometer velocity, torque, and position measurements. Eur J Appl Physiol 91: 22-29.
- Huang YY, Chen AC, Carroll JD, Hamblin MR (2009) Biphasic dose response in low level light therapy. Dose Response 7: 358-383.
- Ihsan FR (2005) Low-level laser therapy accelerates collateral circulation and enhances microcirculation. Photomed Laser Surg 23: 289-294.
- Larkin KA, Martin JS, Zeanah EH, True JM, Braith RW, et al. (2012) Limb blood flow after class 4 laser therapy. J Athl Train 47: 178-183.
- Brooks GA, Dubouchaud H, Brown M, Sicurello JP, Butz CE (1999) Role of mitochondrial lactate dehydrogenase and lactate oxidation in the intracellular lactate shuttle. Proc Natl Acad Sci USA 96: 1129-1134.
- Tonkonogi M, Sahlin K (2002) Physical exercise and mitochondrial function in human skeletal muscle. Exerc Sport Sci Rev 30: 129-137.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi