Case Report, Clin Oncol Case Rep Vol: 5 Issue: 9
Primary Angiosarcoma Of The Spleen With Spontaneous Rupture: A Case Report
Severino Rey Nodar1*, Adriana P. Ortíz Barbosa2, Beatriz Espejo García2, Carlos Cárdenas Aguirre3, José Tomás Cárdenas Gómez2
1Department of Pathology, Hospital of Mérida, Spain
2Department of Radiology, Hospital of Mérida Mérida, Spain
3Foundation for Research and Sciences (FORESC), USA
*Corresponding Author: Severino Rey Nodar
Department of Pathology, Hospital of Mérida, Spain
E-mail: sevrey@yahoo.es
Received: September 05, 2022; Manuscript No: COCR-22-76276;
Editor Assigned: September 07, 2022; PreQC Id: COCR-22-76276 (PQ);
Reviewed: September 21, 2022; QC No: COCR-22-76276 (Q);
Revised: September 23, 2022; Manuscript No: COCR-22-76276 (R);
Published: September 30, 2022; DOI: 10.4172/cocr.5(9).249
Citation: Nodar SR, Ortíz Barbosa AP, García BE, Aguirre CC, Cárdenas Gómez JT (2022) Primary Angiosarcoma of the Spleen with Spontaneous Rupture: A Case Report. Clin Oncol Case Rep 5:9
Abstract
Primary Splenic Angiosarcoma (PSA), is a rare neoplasm of vascular origin associated with aggressive behavior and poor prognosis. Primary splenic angiosarcoma is the most frequent non-lymphoid primary neoplasm of the spleen. The clinical presentation is usually non-specific and is mostly characterized by a wasting disease with anemia and splenomegaly, mimicking a wide range of entities. We present a case of a 68-year-old man who consulted for acute abdominal pain, fever, and acute anemia secondary to hemoperitoneum and splenic mass with capsular rupture. The literature will be determined on radiological and pathological findings for this rare tumor.
Keywords: Angiosarcoma; Splenic diseases; Splenomegaly; Hemangiosarcoma; Splenic rupture, Case report
Introduction
Primary Splenic Angiosarcoma (PSA) is an extremely rare neoplasm, but it is the most common non hematolymphoid malignancy of the spleen [1]. Morphologically, it demonstrates conventional blood vessel differentiation. This aggressive tumor usually presents in adults in their sixth to seventh decade with a slight predilection for males [1- 4]. However, it can occur at any age, with cases reported in pediatric patients [5].
Reviewing the literature, we find some common characteristics of splenic angiosarcoma such as splenomegaly and abdominal pain (usually the main form of presentation in up to 83% in some series) [2,4]. It is common that at the time of diagnosis patients have distant metastases, more often liver metastases. Patients may present with hemoperitoneum secondary to splenic rupture (25%-30%), as well as bleeding from liver metastases [1,3,6, 7]. Other forms of presentation are anemia, fever, and weight loss [8].
Malignant splenic vascular tumors may be mistaken for benign vascular or non-vascular malignant tumors because of their frequently highly variable morphology.
Histologically, PSA is a heterogeneous tumor, showing a predominant vasoformative component lined by atypical endothelial cells. Other patterns, such as solid, sarcomatous, or papillary, can be recognized in most cases. Immunohistochemically, PSA shows reactivity for markers of vascular differentiation such as CD31, CD34, Factor VIII and Vascular Endothelial Growth Factor Receptor-2 (VEGFR2) Flk1/KDR.
Case Report
68-year-old man was admitted in Merida hospital emergency room suffering from abdominal pain 36 hours of evolution, palpable abdominal mass in left hypochondrium and fever (38.4ºC). Initially the pain was accompanied by vegetative symptoms (profuse sweating, tremor, blurred vision). No vomiting or changes in bowel habits were reported.
The day before the patient had consulted the emergency department for nonspecific abdominal pain without admission criteria and was discharged.
Physical examination revealed pain on deep abdominal palpation, more pronounced over the left flank. Peripheral blood count revealed anemia (hemoglobin 11.2 g/dl) and thrombocytopenia (platelets 98/ L×109/L). Previous Hb (one day before was 14.2 g/dL). Leucocytes 12.52 (N 77%). LDH 815. PCR 109. Biochemistry showed no abnormalities.
An abdominal ultrasound showed hemoperitoneum and splenomegaly (14.4cm) with a large mixed echogenicity mass with poorly defined and irregular contours. Multiple focal hepatic lesions with a "bull's eye" sign (echoic center with hypoechoic halo in the periphery), suggestive of metastasis were described (Figure 1 and 2).
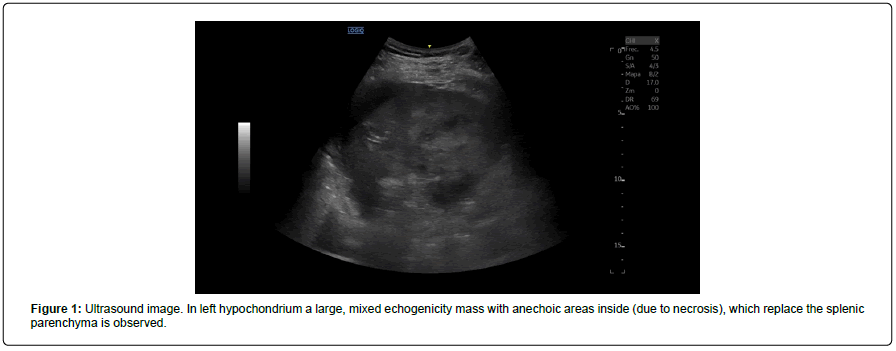
Figure 1: Ultrasound image. In left hypochondrium a large, mixed echogenicity mass with anechoic areas inside (due to necrosis), which replace the splenic parenchyma is observed.
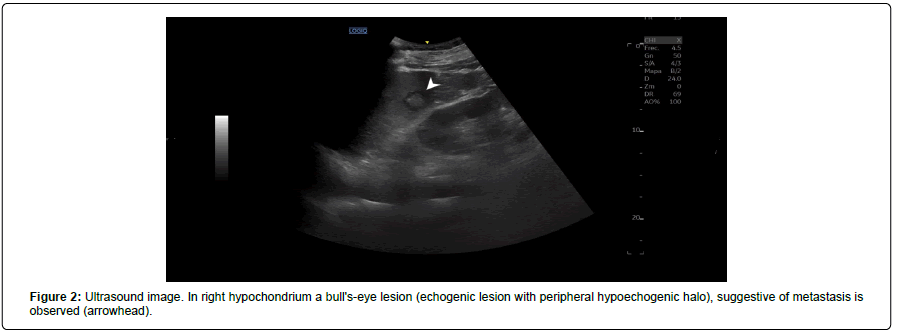
Figure 2: Ultrasound image. In right hypochondrium a bull's-eye lesion (echogenic lesion with peripheral hypoechogenic halo), suggestive of metastasis is observed (arrowhead).
A simple CT and an intravenous contrast CT were performed, corroborating the hemoperitoneum, multiple hepatic lesions with nonhypervascular heterogeneous enhancement suggestive of metastasis and a large 10.5 cm in size splenic tumor with heterogeneous soft tissue density. The enhancement is also heterogeneous, highlighting hypodense areas in its interior suggestive of necrosis and a small subcapsular hematoma. No other lesions suggestive of a primary neoplastic process are observed in the remaining abdominal organs (Figure 3).
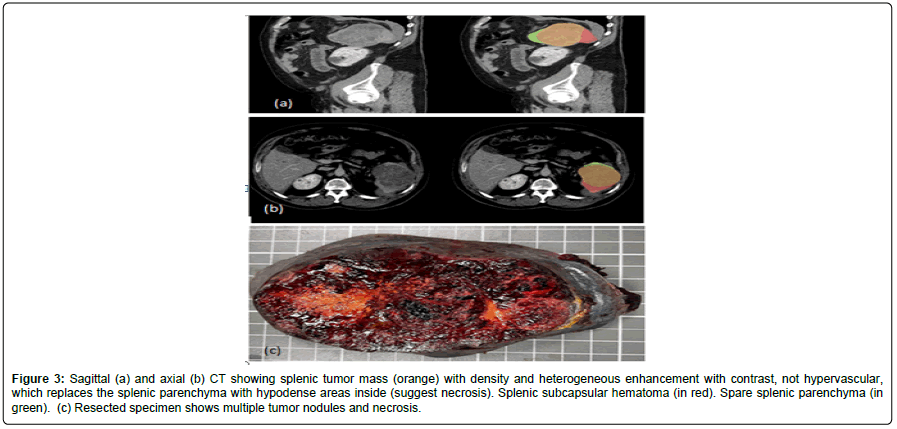
Figure 3: Sagittal (a) and axial (b) CT showing splenic tumor mass (orange) with density and heterogeneous enhancement with contrast, not hypervascular, which replaces the splenic parenchyma with hypodense areas inside (suggest necrosis). Splenic subcapsular hematoma (in red). Spare splenic parenchyma (in green). (c) Resected specimen shows multiple tumor nodules and necrosis.
Considering these findings, the diagnosis of splenic angiosarcoma with secondary hemoperitoneum is proposed.
An emergency splenectomy was performed, finding 1000cc of free blood in the peritoneal cavity, a splenic tumor with capsular rupture, and multiple liver metastases (Figure 4).
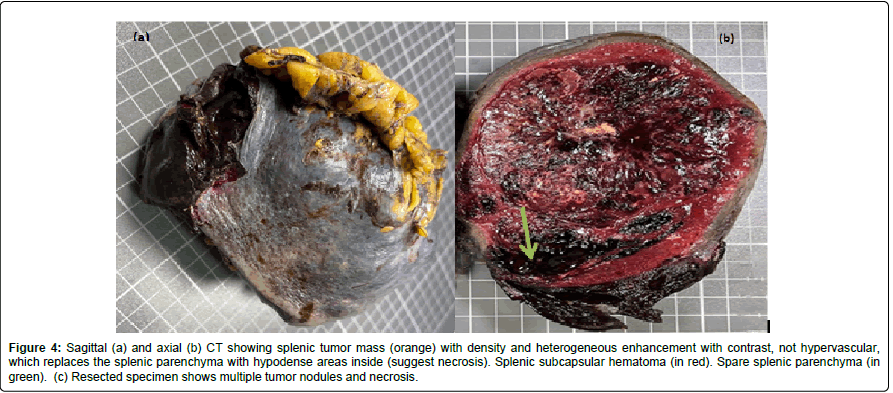
Figure 4: Sagittal (a) and axial (b) CT showing splenic tumor mass (orange) with density and heterogeneous enhancement with contrast, not hypervascular, which replaces the splenic parenchyma with hypodense areas inside (suggest necrosis). Splenic subcapsular hematoma (in red). Spare splenic parenchyma (in green). (c) Resected specimen shows multiple tumor nodules and necrosis.
Histologically, the splenic parenchyma was congested and replaced by a tumor formed by a rich network of vascular channels lined by pleomorphic endothelial cells, with a malignant appearance. The tumor cells were mostly spindle-shaped, with nuclear atypia. Some vessels are dilated and contain blood in the lumen. Moderate amount of extravasated erythrocytes (Figures 5 and 6).
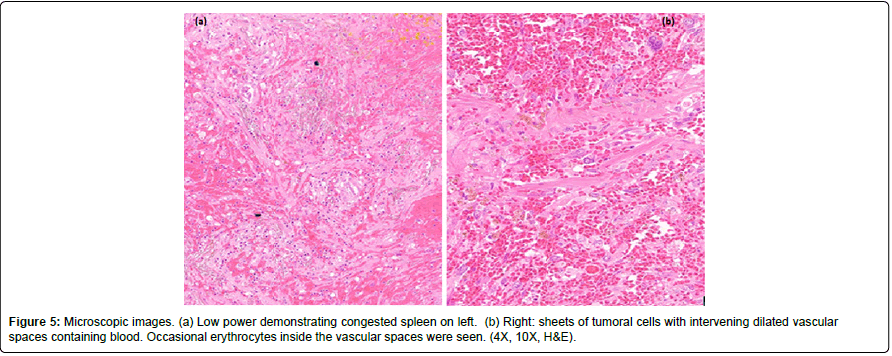
Figure 5: Microscopic images. (a) Low power demonstrating congested spleen on left. (b) Right: sheets of tumoral cells with intervening dilated vascular spaces containing blood. Occasional erythrocytes inside the vascular spaces were seen. (4X, 10X, H&E).
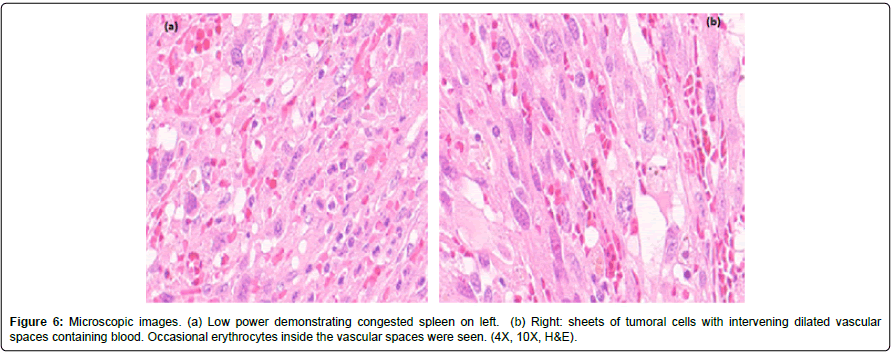
Figure 6: Microscopic images. (a) Low power demonstrating congested spleen on left. (b) Right: sheets of tumoral cells with intervening dilated vascular spaces containing blood. Occasional erythrocytes inside the vascular spaces were seen. (4X, 10X, H&E).
One month later, a control CT scan is performed, showing a marked increase in metastases, which replace almost all the liver parenchyma. Additionally, the appearance of multiple nodules in the lung parenchyma with “halo sign” (solid nodules with ground glass areas in the periphery), highly suggestive of pulmonary metastases were reported (Figures 7 and 8).
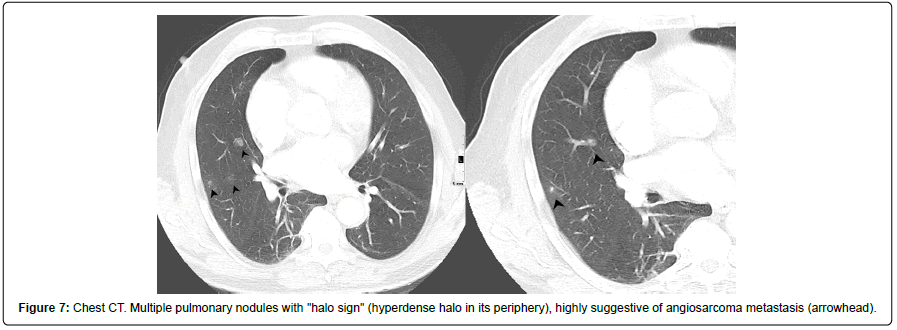
Figure 7: Chest CT. Multiple pulmonary nodules with "halo sign" (hyperdense halo in its periphery), highly suggestive of angiosarcoma metastasis (arrowhead).
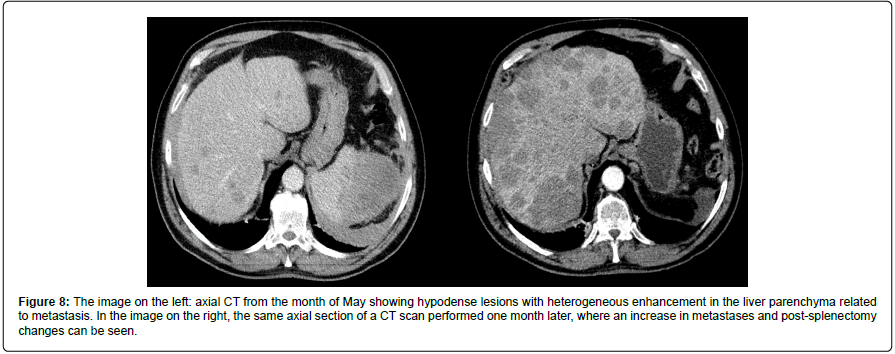
Figure 8: The image on the left: axial CT from the month of May showing hypodense lesions with heterogeneous enhancement in the liver parenchyma related to metastasis. In the image on the right, the same axial section of a CT scan performed one month later, where an increase in metastases and post-splenectomy changes can be seen.
The patient died two months after surgery. His past medical history was unremarkable, and he did not have any known risk factors for angiosarcoma.
Discussion and Conclusion
Angiosarcoma is a malignant tumor that occur in the subcutaneous tissue of many locations in the body, typically head and neck, or breast and other uncommon locations like the spleen; showing morphological or immunophenotypic evidence of endothelial differentiation. Originally described by Theodor Langhans in 1879, the PSA is one of the rarest human malignancies, which affects 0.14 per million to 0.25 per million people [1]. Etiology is unknown in most cases. Angiosarcomas in general are associated with the exposure to Polyvinyl chloride, arsenic, thorium dioxide (Thorotrast) a radiocontrast agent used historically between 1930 to 1950 on clinical radiography, and a small number of cases that occur in association with: implanted foreign material, in pre-existing hemangioma/vascular malformation, in regions of prior trauma or surgery and produced by therapeutic radiation, generally after management of Hodgkin lymphoma or breast carcinomas, with a median time of progression from about 8 years to 10 years.
Clinical presentation of splenic angiosarcoma is variable. Left upper abdominal pain is the most common symptom. Splenomegaly is the most common physical examination finding. Hepatomegaly secondary to metastasis is not an uncommon finding. Patients with angiosarcoma of the spleen should undergo early splenectomy whenever possible to avoid splenic rupture. The most severe manifestation is splenic rupture, which often leads to fatal hemorrhage [9].
PSA is an extremely aggressive neoplasm with a very poor prognosis (most patients die one year after diagnosis) [1]. Metastatic spread at presentation is common, especially to the liver (up to 67% in some series) [2,8]. PSA can also cause lung, lymph node, bone, and brain metastases [8]. Lung metastases are described as halo sign nodules (soft-tissue dense nodules with ground-glass halo) [10,11].
Ultrasound and CT scan show splenomegaly secondary to the presence of one or multiples heterogeneous masses with poorly defined margins and irregular contours, with features of recent or old bleeding inside. It can cause extracapsular or perisplenic blood collections (hemoperitoneum) at the time of presentation [1-3 ,12,13].
On ultrasound, this tumor usually has heterogeneous echogenicity, predominantly echogenic, with increased vascularization on Doppler [2]. On CT scan, single or multiple masses, heterogeneous in density, can be seen. Not all angiosarcomas are hipervascular [5]. The enhancement of these lesions is variable; In some cases, intense enhancement has been described in the periphery with the use of IV contrast in both the primary tumor and in metastases. But in others (such as our patient) the enhancement is minimal and hypodense areas predominate inside the tumor due to the presence of necrosis. Calcifications have been reported in some cases [2].
Despite the infrequency of this neoplasm, angiosarcoma should be considered as the main differential diagnosis of non-hypervascular solid-cystic splenic lesions with a malignant appearance [5]. The authors believe that this entity should be considered in the differential diagnosis of spontaneous non-traumatic hemoperitoneum highlighting that we must keep this pathology in mind in patients who come to the emergency room at the time of diagnosis, as it happened with this patient.
The biopsy is contraindicated in splenic angiosarcoma because of high risk of bleeding. Histologic studies can therefore only be made after splenectomy.
Histologically, numerous irregularly shaped anastomosing vascular channels lined by atypical endothelial cells with a highly infiltrative architecture and poor demarcation are described. Solid areas and necrosis are usual. Sometimes, the cells have an epithelioid appearance with an increased nuclear-cytoplasmic ratio and the presence of atypical mitoses.
The differential diagnosis includes th e sp lenic ha martomas and littoral cell angiomas [14]. These lesions are both considered primary splenic lesions, most likely derived from the splenic red pulp. Like the primary angiosarcomas, these tumors show “biphasic” immunoreactivity for vascular and histiocytic markers [15,16].
References
- Falk S, Krishnan J, Meis JM (1993) Primary angiosarcoma of the spleen. A clinicopathologic study of 40 cases. Am J Surg Pathol 17: 959-970. [Google Scholar] [Cross Ref]
- Thompson WM, Levy AD, Aguilera NS, Gorospe L, Abbott RM (2005) Angiosarcoma of the spleen: Imaging characteristics in 12 patients. Radiology 235: 106-115. [Google Scholar] [Cross Ref]
- Neuhauser TS, Derringer GA, Thompson LD, Fanburg-Smith JC, Miettinen M, et al. (2000) Splenic angiosarcoma: A clinicopathologic and immunophenotypic study of 28 cases. Modern Pathol 13: 978-987. [Google Scholar] [Cross Ref]
- Badiani R, Schaller G, Jain K, Swamy R, Gupta S (2013) Angiosarcoma of the spleen presenting as spontaneous splenic rupture: A rare case report and review of the literature. Int J Surg Case Rep 4: 765-767. [Google Scholar] [Cross Ref]
- Lee HJ, Kim JW, Hong JH, Kim GS, Shin SS, et al. (2018) Cross-sectional imaging of splenic lesions: RadioGraphics fundamentals online presentation. RadioGraphics 38: 435-436. [Google Scholar] [Cross Ref]
- Vrachliotis TG, Bennett WF, Vaswani KK, Niemann TH, Bova JG (2000) Primary angiosarcoma of the spleen-CT, MR, and sonographic characteristics: Report of two cases. Abdom Imag 25: 283-285. [Google Scholar] [Cross Ref]
- Linan-Padilla A, Suárez-Grau JM, Valera Sánchez Z, Vázquez-Medina A, Docobo-Durántez F (2008) Spontaneous hemoperitoneum due to hemangiosarcoma of the spleen. Cirugia Espanola 84: 171-172. [Google Scholar] [Cross Ref]
- Chen X, Li H, Wang F, Liu H (2018) Early detection and integral resection are keys to extend survival in patients suffered from primary angiosarcoma of the spleen: A care-compliant case report and literature review. Medicine 97: e9718. [Google Scholar] [Cross Ref]
- Coppola S, Leva A, Pagni F, Famularo S, Gianotti L (2017) Demanding diagnosis of splenic angiosarcoma as cause of delayed treatment of spontaneous splenic rupture: A case report and literature review. Case Rep Surg 2017: 6256102. [Google Scholar] [Cross Ref]
- Parron M, Torres I, Pardo M, Morales C, Navarro M, et al. (2008) The halo sign in computed tomography images: Differential diagnosis and correlation with pathology findings. Arch Bronconeumol 44: 386-392. [Google Scholar] [Cross Ref]
- Tateishi U, Hasegawa T, Kusumoto M, Yamazaki N, Iinuma G, et al. (2003) Metastatic angiosarcoma of the lung: Spectrum of CT findings. AJR Am J Roentgenol 180: 1671-1674. [Google Scholar] [Cross Ref]
- Hamid KS, Rodriguez JA, Lairmore TC. (2010) Primary splenic angiosarcoma. JSLS. 14: 431-435. [Google Scholar] [Cross Ref]
- Ferrozzi F, Bova D, Draghi F, Garlaschi G (1996) CT findings in primary vascular tumors of the spleen. AJR Am J Roentgenol 166: 1097-1101. [Google Scholar] [Cross Ref]
- Maier A, Bataille F, Krenz D, Anthuber M. (2004) Angiosarcoma as a rare differential diagnosis in spontaneous rupture of the spleen. Chirurg 75: 70-74. [Google Scholar] [Cross Ref]
- Duan YF, Jiang Y, Wu CX, Zhu F (2013) Spontaneous rupture of primary splenic angiosarcoma: A case report and literature review. World J Surg Oncol 11: 53. [Google Scholar] [Cross Ref]
- Li R, Li M, Zhang LF, Liu XM, Hu TZ, et al. (2018) Clinical characteristics and prognostic factors of primary splenic angiosarcoma: A retrospective clinical analysis from china. Cell Physiol Biochem 49: 1959-1969. [Google Scholar] [Cross Ref]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 