Case Report, Clin Oncol Case Rep Vol: 3 Issue: 2
Primary Squamous Cell Carcinoma of the Liver
Paul Guzik1*, Andrew Gdowski1, Christopher Kim1, Amanda Rivera-Begeman2, Hannah Kay3, Declan Fleming4,5 and Gail Eckhardt1,2,6
1The University of Texas at Austin, Dell Medical School, Department of Internal Medicine, U.S.A
2The University of Texas at Austin, Dell Medical School, Department of Diagnostic Medicine, U.S.A
3The University of Texas at Austin, Dell Medical School, U.S.A
4The University of Texas at Austin, Dell Medical School, Department of Surgery and Perioperative Care, U.S.A
5LIVESTRONG Cancer Institutes, U.S.A
6The University of Texas at Austin, Dell Medical School, Department of Oncology, U.S.A
*Corresponding Author : Paul Guzik, The University of Texas at Austin
Dell
Medical School, Department of Internal Medicine, U.S.A
Tel: 7632349009
E-mail: paul.r.guzik@gmail.com
Received: March 06, 2020 Accepted: March 16, 2020 Published: April 03, 2020
Citation: Guzik P, Gdowski A, Kim C, Rivera-Begeman A, Kay H, et al. (2020) Primary Squamous Cell Carcinoma of the Liver. Clin Oncol Case Rep 3:2.
Abstract
Primary Squamous Cell carcinoma (SCC) of the liver is a very rare diagnosis with limited reported cases in the current literature. The pathogenesis of the disease is unclear, but is generally considered to be correlated with the long-term inflammation or metaplasia of biliary epithelial cells and congenital cyst of the liver. The current viewpoint is that surgical resection of the tumor, preoperative or postoperative subsidiary chemoembolization, radiotherapy and generalized chemotherapy may extend a patient’s survival.The prognosis is extremely poor with few patients surviving after 12 months, irrespective of treatment.Here, we report a case of hepatic primary squamous cell carcinoma.
Keywords: Liver cancer; Rare cancer; Rare liver cancer; Primary squamous cell carcinoma of the liver; Case report; Liver tumor; Liver mass
Case Report
A 60-year old woman presented to our hospital with ongoing abdominal pain and anorexia. She was evaluated one month before an outside hospital where she was diagnosed with poorly differentiated SCC by liver biopsy [1]. A palpable, tender abdominal mass in her right upper quadrant was noted on the exam. Her liver function tests comprised of an Alkaline Phosphatase (ALP) 188 μ/L (Upper Limit of Normal range (ULN) 121 μ/L), Aspartate Transferase (AST) 71 μ/L (ULN 34 μ/L), and Alanine Transferase (ALT) 86 μ/L (ULN 60 μ/L).
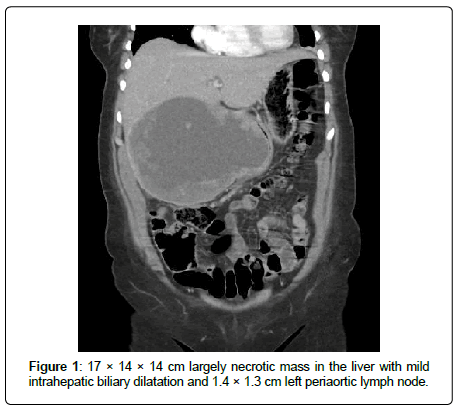
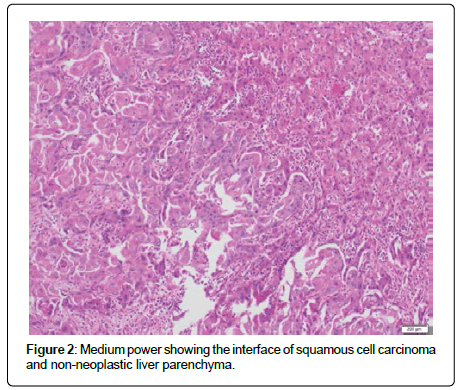
In this case total bilirubin was within normal range. Tumor markers revealed an elevated Carbohydrate Antigen (CA) 19-9 of 56.7 U/mL (ULN 3 U/mL) and CA-125 of 77.7 U/mL (ULN 35 U/mL). Alpha-fetoprotein (AFP), Carcinoembryonic Antigen (CEA) and CA 15-3 were all within normal limits. Computed Tomography (CT) scan of her abdomen revealed a 17 cm necrotic liver mass with mild intrahepatic biliary dilatation and a 1.4 cm left periaortic lymph node (Figure 1). Repeat liver biopsy revealed large areas of invasive SCC in solid nests and interconnecting cores with areas of necrosis (Figure 2).
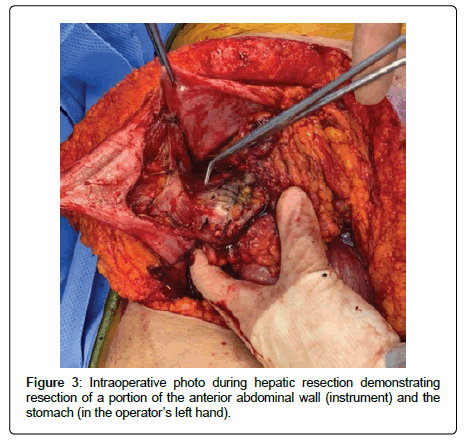
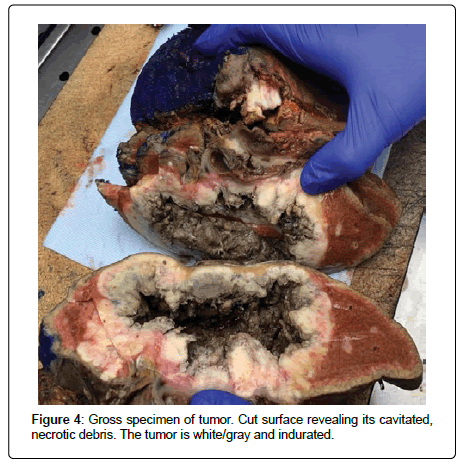
She underwent a right hepatectomy with resection of the common hepatic and common bile ducts as well as porta hepatis lymph nodes. The distal stomach and duodenum, a segment of the transverse colon, and a portion of the abdominal wall adjacent to the mass were also resected (Figures 3 and 4). Pathologic evaluation revealed moderately differentiated SCC of the liver with direct extension into the abdominal wall, stomach, and colon. There was evidence of lymphovascular invasion as one of five resected peri-hepatic lymph nodes were positive for metastatic malignancy. All resected surgical margins were free of malignancy. One month after hospital discharge, a PET scan revealed multifocal hypermetabolic metastatic liver lesions with hypermetabolic peritoneal metastases and ascites. She was started on palliative chemotherapy with carboplatin and paclitaxel but unfortunately passed away six months after her initial diagnosis.
Discussion
The pathogenesis of primary SCC of the liver is not completely understood. It is postulated that hepatic cysts undergo a metaplasiadysplasia transformation progressing from benign to malignant [2]. It is also suggested that inflammation in the biliary tract, possibly from stones or infection, may be the inciting step in the development of primary SCC [2]. Additional associations with liver cirrhosis and Caroli’s disease (a rare congenital liver disordercharacterized by cystic dilation of intrahepatic bile ducts and renal cysts) have been proposed [3,4]. This patient did not have known cirrhosis, Caroli’s disease, biliary stones or hepatic cysts.
In patients with localized disease, primary management should be aimed towards margin-negative surgical resection of the tumor. Patients who undergo surgical resection have longer overall survival than those who do not (17 vs 5 months) [1]. Due to the rare nature of this condition, there are no standardized guidelines to direct adjuvant therapy. The presence of the nodal metastatic disease may be considered to be an indicator of higher risk for the development of systemic metastasis, and thus a platinum-based chemotherapy regimen was started. Other treatment modalities that have been reported for patients with SCC of the liver include systemic chemotherapy with 5-fluorouracil (FU), hepatic artery infusion chemotherapy, and radiation therapy [1,5].
Conclusion
We presented a rare case of primary SCC of the liver in a patient with no known history of hepatic or biliary disease. She underwent extensive evaluation by multiple consultants while inpatient where no primary tumor outside the liver was identified. Due to the large tumor size and local invasion of adjacent organs with persistent infection secondary to a fistula between the necrotic center of cancer and the stomach, the patient was treated with radical surgical resection. Unfortunately, she developed early metastatic recurrence of her cancer in the peritoneum. She was treated with palliative chemotherapy until her eventual death four months after her surgery and approximately six months from her initial diagnosis.
References
- Zhang XF (2015) Primary squamous cell carcinoma of liver: Case series and review of literatures. Medicine 94: e868.
- Nakajima T, Kondo Y (1990) A clinicopathologic study of intrahepatic cholangiocarcinoma containing a component of squamous cell carcinoma. Cancer 65: 1401-1404.
- Spaggiari M (2011) Primary squamous cell carcinoma of the liver associated with Caroli's disease: Case Rep Onkologie 34: 193-195.
- Kaji R (2003) A case report of primary hepatic squamous cell carcinoma that remarkably responded to low dose arterial injection of anti-cancer drugs. Kurume Med J 50: 71-75.
- Takahashi H (1997) Primary adenosquamous carcinoma of liver resected by right trisegmentectomy: Report of a case and review of the literature. J Gastroenterol 32: 843-847.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi