Research Article, Vector Biol J Vol: 3 Issue: 2
Relation of Entomological Parameters and Dengue Transmission to Develop the Optimal Model and Threshold for Dengue Epidemics
Mohd Ayoub Bhat1* and K Krishnamurthy2
1Division of Epidemiology and Operational Research, Vector Control Research Centre, India
2Council of Medical Research, Indira Nagar, Pondicherry-605006, India
*Corresponding Author : Mohd. Ayoub Bhat
Division of Epidemiology and Operational Research, Vector Control Research Centre, Indian Council of Medical Research, Indira Nagar, Pondicherry-605006, India
E-mail: ayoub.sajad@gmail.com
Received: July 03, 2018 Accepted: November 13, 2018 Published: December 29, 2018
Citation: Bhat MA, Krishnamurthy K (2018) Relation of Entomological Parameters and Dengue Transmission to Develop the Optimal Model and Threshold for Dengue Epidemics. Vector Biol J 3:2. doi: 10.4172/2473-4810.1000132
Abstract
An observational analytical study, year-long entomological surveys were carried out in selected villages in Tirunelveli district. The vector breeding indices were compared with the incidence of cases in each of the selected sites. Concurrent epidemiological data were used to relate the entomological risk. The values at lower 95% confidence limit of 1.34% of St. aegypti positive water storage containers, 7.85 St. aegypti positive water storage containers per 100 houses and 18.02 St. aegypti pupae per 100 houses in water storage breeding sources have been determined to be the threshold level above which the chances in dengue transmission would be higher. The lower 95% confidence limit of 19.9 St. aegypti pupae per 100 houses can also be considered as threshold level when the estimates were made irrespective of the type of breeding habitats. Proof-of-concept of how to search for the optimal model and threshold for dengue epidemics is determined.
Keywords: Tirunelveli; St. aegypti; Larval indices; Dengue cases; Threshold values
Introduction
Vector borne diseases (VBDs) continues to be the major public health concern worldwide and pose potential threats to the mankind, accounting for about 17% of the estimated global burden of all infectious diseases. Dengue, a mosquito borne disease affects millions of people worldwide and is considered an emergent mosquito-borne viral disease in terms of mortality and morbidity [1] in both developing and developed countries. The disease may cause hemorrhages and plasma leakage giving birth to fatal diseases and causes massive infant mortality [2].
The transmission, incidence and severity of dengue are associated with the geographic expansion and distribution of dengue vectors and viruses [3,4]. Increased population growth rate, global warming, unplanned urbanization, inefficient mosquito control, frequent air travel, and lack of health care facilities are the reasons for the present status of dengue [3] and are associated with the expansion of Aedine mosquitoes due to which there is increase in the prevalence of the diseases and virus proliferation. Poor sanitation, improper water and solid managements make it difficult to control the explosion of dengue of dengue vectors [5-7].
Materials and Methods
Based on the incidence of dengue in 2012, the villages/wards in Tirunelveli were stratified in three strata (Low, medium and high). From each stratum 2 villages were selected (Figure 1) and there were a cohort of six study sites for longitudinal survey. Manur and Valliyur represent low stratum (with no confirmed cases), Palayamkottai and VK-Puthur (confirmed cases above 1 and below 10) represent medium and Vadakarrai and Kadayanalur represent high incidence stratum (confirmed cases above 10). As many as 180 houses (30 houses per village) were selected by systematic sampling with a random start for entomological monitoring every month.
Three species (Stegomyia aegypti, Stegomyia albopicta and Fredwardsius vittatus) were encountered in this study and the vector breeding indices has been obtained on the basis of types of habitat. The term “Aedine mosquitoes or Aedine species” has been used to include all the three species of mosquitoes encountered. Habitat analysis has shown that the vector breeding indices based on only water storage breeding sources of only St. aegypti were significantly associated with the dengue cases.
Immature surveys
The basic sampling unit was the house or premise. Water holding habitats were considered as breeding sources and regarded as a wet container and the wet container with immatures of mosquito were regarded as the positive breeding source. Each of the selected houses was thoroughly searched for the presence of water storage containersand other containers that hold water. The search was continued till all containers have been examined. Since it is difficult to identify the mosquito larva/pupa up to species level, specimens (late instars and pupae) were collected for laboratory examination and emergence to adults to confirm the species. The collection of Aedes immatures was carried out by followed the specific protocols [8,9]. The premises where breeding was found was considered as positive house. All the water filled containers from all the selected premises were screened for the presence of immatures. The study consisted of two types of breeding containers; rain dependent discarded breeding sources and water storage breeding sources. The containers which were regularly used for prolonged water storage throughout the year were regarded as “water storage breeding sources” whereas the other categories of containers included discarded breeding sources which were found positive during the rainy seasons and were regarded as “rain dependent breeding sources/containers.
During sampling, a dipper of 20 cm wide and 10 cm deep was dipped ten times at a sampling point of each of the selected systems/ sites for collection of pupae and late instar larvae. The contents were passed through a strainer of 20 meshes/cm mosquito nylon netting to retain the immatures. The immatures were then transferred to the plastic containers (half-filled with breeding site water) by using pipette. The plastic containers containing the immatures were labeled with breeding source code, locality code, house identification code and date of collection. The samples were carried to the laboratory in Vector Control Research Centre (VCRC), Pondicherry. In the laboratory the samples were transferred into plastic containers (100 ml capacity) and labeled. The containers were covered with nylon net secured with a rubber band. The immatures were reared to reach the adult stages. Larval food was added and was held for the adults to emerge. Every day the emerging adults were collected using oral aspirator and were transferred into test tubes. The adults were immobilized by tapping and were transferred to petridishes for examination. The adults were examined using binocular stereo-dissection microscope. Using the taxonomic key the adults were identified up to species level and recorded.
The larval indices were used for monitoring infestation. The indices are useful for determining general distribution, seasonal changes and principal larval habitats, as well as for evaluating environmental sanitation programs. They have direct relevance to the dynamics of disease transmission. However, the threshold levels of vector infestation that constitute a trigger for dengue transmission are influenced by many factors, including mosquito longevity and immunological status of the human population. These larval indices were calculated as given below.
House Index (HI): Percentage of houses infested with Aedine larvae or pupae.
HI=(Number of houses infested)/(Number of houses inspected) × 100
Container Index (CI): Percentage of water-holding containers infested with Aedine larvae or pupae.
CI=(Number of containers with Aedine larvae or pupae)/ (Number of containers examined) × 100
Breateau Index (BI): Number of positive containers per 100 houses inspected.
BI=(Number of containers with Aedine larvae or pupae)/ (Number of houses inspected) × 100
Pupal Index (PI) is the number of pupae per 100 houses.
PI=(Number of pupae)/(Number of houses inspected) ×100
Epidemiological interpretation of vector surveillance
The vector breeding indices were compared with the incidence of cases in each of the selected sites. The analysis was done with the help of Excel spreadsheet 2013 and SPSS. Regression analysis was carried out using monthly data and the incidence in different sites to examine the correlation. The confidence interval was also calculated for its significance. Concurrent epidemiological data were used to relate the entomological risk. Dengue cases recorded in Tirunelveli district during 2013-14 for different months and the cases recorded for the other districts were obtained from the “Directorate of Public Health and Preventive Medicine, Chennai, Govt. of Tamil Nadu”. The association between entomological parameters recorded in the corresponding month and incidence of dengue was assessed by pooling the data from the six study sites representing the district.
Results
Vector breeding sources
Two types of breeding sources were observed in this study; rain-dependent discarded breeding sources and water storage breeding sources.
Water storage breeding sources
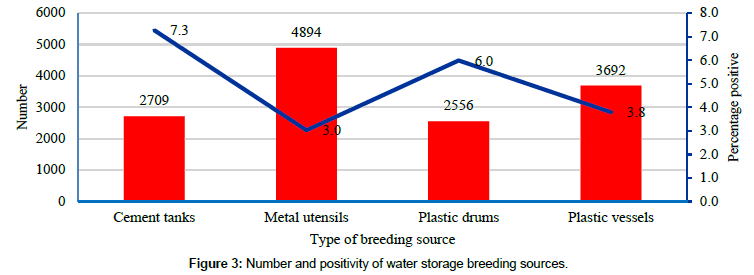
Cement tanks, plastic drums, metal utensils and plastic vessels were used to store water for domestic purpose in the study area (Figure 2). On an average 7.3% of cement tanks, 6.0% of plastic drums, 3.8% of plastic vessels and 3.0% of metal utensils were found supporting vector breeding (Figure 3). The proportion of habitats supporting vector breeding was significantly higher (p<0.05) in cement tanks and plastic drums (almost two times) when compared to plastic and metal utensils. From the control point of view the more risk habitats are larger water storage sources, requiring adequate attention in preventing vector breeding.
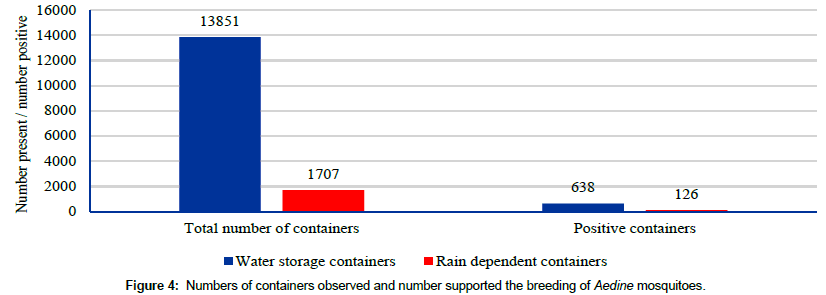
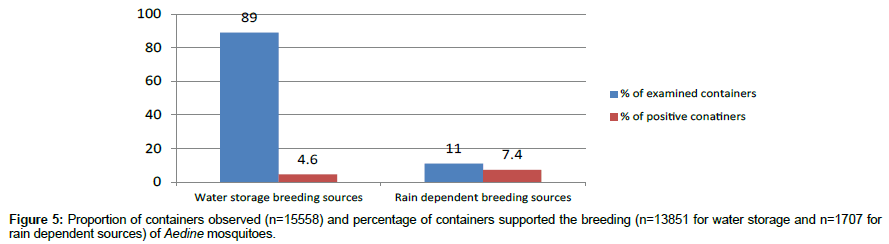
A total of 13851 water storage sources were found and 638 containers were found positive during the yearlong survey through 2160 household visits (Figure 4). On an average 6 breeding sources are present in every house. The percentage of water storage sources supporting vector breeding was about 4.6% (Figure 5). These are the perennial habitats and are manmade which can be easily modifiable so as to maintain the sources free from vector breeding.
Rain dependent sources
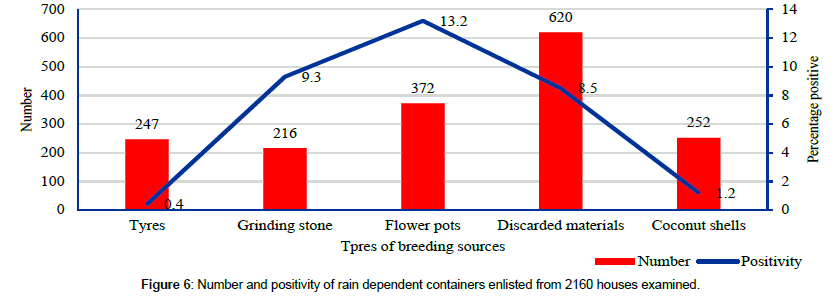
Discarded pots (discarded plastic and metal containers), unused tyres, flower pots, grinding stones and coconut shells are the common sources of habitats prone for water collection during rain (Figure 2). The positivity in different rain dependent breeding sources varied from 0.4% to 13.2% (Figure 6).
As many as 1707 rain dependent containers were encountered, with an average of 0.8 per house visit. With 126 containers with immature stages of vector mosquito, the positivity rate was 7.4% (Figure 5). These are the seasonal habitats and were found during the rainy seasons.
Dengue cases were reported from the study sites in Tirunelveli district during the period of May 2013 – April 2014 during which monthly entomological data was generated. A total of 168 confirmed cases of dengue were reported. Cases were recorded in all the months of study period of one year indicating that transmission was perennial. Cases vary from 3 (November 2013) to 36 (May 2014) in the study area. The number of cases during monsoon (September to December) were the least (below 10 per month) (Table 1). The cases started showing an increasing trend during post monsoon months, reaching its peak in May (pre monsoon). The same trend was observed for the district as a whole during 2012 and the outbreak was in May 2012.
| Seasons | No. of dengue cases | Dengue cases per month (Average) |
|---|---|---|
| Pre-monsoon | 88 | 22 |
| Monsoon | 28 | 7 |
| Post -monsoon | 52 | 13 |
Table 1: Dengue cases in different seasons form May 2013 to April 2014.
Larval indices of Aedine mosquitoes, as well as the larval indices of Stegomyia aegypti, were analyzed separately. Larval indices irrespective of breeding sources as well as larval indices with respect to water storage breeding sources were analyzed separately.
Analysis of cases in relation to season viz., monsoon (September to December), post-monsoon (January – April) and pre-monsoon (May-August) showed significantly (χ2 58.1; p<0.05) higher house index (34.3 houses per 100 with Aedine mosquito breeding sources) irrespective of breeding type in monsoon months compared to other seasons (Table 2). During post-monsoon months the dengue cases were moderate but the house index of Aedine mosquitoes irrespective of habitat type is almost comparable with that of pre-monsoon months (Table 2) and did not show any association with dengue cases.
| Seasons | Houses examined | Positive houses | Containers examined | Positive containers | Pupae collected | Larval indices of Aedine mosquitoes irrespective of breeding sources | |||
|---|---|---|---|---|---|---|---|---|---|
| HI | CI | BI | PI | ||||||
| Pre-monsoon | 720 | 145 | 4981 | 213 | 758 | 20.1 | 4.3 | 29.6 | 105.3 |
| Monsoon | 720 | 247 | 5610 | 365 | 619 | 34.3 | 6.5 | 50.7 | 86.0 |
| Post-monsoon | 720 | 134 | 4967 | 186 | 587 | 18.6 | 3.7 | 25.8 | 81.5 |
Table 2: Larval indices of Aedine mosquitoes irrespective of breeding type in different seasons.
House index of Aedine mosquitoes with regard to water storage containers also did not show any relationship with dengue cases (Table 3) in different seasons, as the house index did not show any market seasonal variation as it is present throughout the year, independent of rain. A similar trend with St. aegypti was observed with house index irrespective of breeding type (Table 4) as well as with respect to water storage breeding sources (Table 5) and dengue cases.
| Seasons | Houses examined | Positive houses | Containers examined | Positive containers | Pupae collected | Larval indices of Aedine mosquitoes in water storage breeding sources | |||
|---|---|---|---|---|---|---|---|---|---|
| HI | CI | BI | PI | ||||||
| Pre-monsoon | 720 | 145 | 4706 | 213 | 758 | 20.1 | 4.5 | 29.6 | 105.3 |
| Monsoon | 720 | 148 | 4625 | 239 | 570 | 20.6 | 5.2 | 33.2 | 79.2 |
| Post monsoon | 720 | 134 | 4520 | 186 | 587 | 18.6 | 4.1 | 25.8 | 81.5 |
Table 3: Larval indices of Aedine mosquitoes in water storage breeding sources in different seasons.
| Seasons | Houses examined | Positive houses | Containers examined | Positive containers | Pupae collected | Larval indices of St. aegypti irrespective of breeding sources | |||
|---|---|---|---|---|---|---|---|---|---|
| HI | CI | BI | PI | ||||||
| Pre-monsoon | 720 | 68 | 4981 | 94 | 392 | 9.4 | 1.9 | 13.1 | 54.4 |
| Monsoon | 720 | 85 | 5610 | 110 | 188 | 11.8 | 2.0 | 15.3 | 26.1 |
| Post monsoon | 720 | 60 | 4967 | 81 | 204 | 8.3 | 1.6 | 11.3 | 28.3 |
Table 4: Larval indices of St. aegypti irrespective of breeding sources in different seasons.
| Seasons | Houses examined | Positive houses | Containers examined | Positive containers | Pupae collected | Larval indices of St. aegypti in water storage breeding sources | |||
|---|---|---|---|---|---|---|---|---|---|
| HI | CI | BI | PI | ||||||
| Pre-monsoon | 720 | 68 | 4706 | 94 | 392 | 9.4 | 2.0 | 13.1 | 54.4 |
| Monsoon | 720 | 37 | 4625 | 58 | 147 | 5.1 | 1.3 | 8.1 | 20.4 |
| Post monsoon | 720 | 60 | 4520 | 81 | 204 | 8.3 | 1.8 | 11.3 | 28.3 |
Table 5: Larval indices of St. aegypti in relation to water storage breeding sources in different seasons.
The proportion of positive containers irrespective of breeding sources as well as proportion of positive water storage containers of Aedine mosquitoes did not differ significantly in different seasons which indicates container index irrespective of breeding sources as well as container index in water storage breeding sources of Aedine mosquitoes also did not show any relationship with dengue cases.
Proportion of positive containers for St. aegypti irrespective of type of breeding sources did not differ significantly (χ2 0.81; p=0.36) among seasons which indicates that the container index of St. aegypti irrespective of breeding sources also did not show any significant relationship with dengue cases.
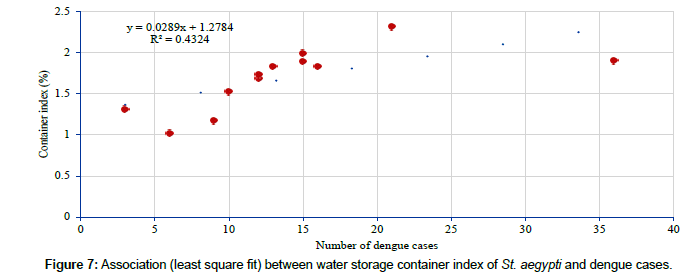
The container index of St. aegypti (2.0%) in water storage breeding sources was higher during pre-monsoon seasons which indicates the positive association with the dengue cases in different seasons. Analysis has shown that the container index of St. aegypti in water storage breeding sources varied from 1.0 (December 2013) – 2.3 (July 2013) (Table 6) and had shown significantly positive correlation (r=0.658, p=0.020) with the monthly dengue cases (Figure 7).
| Month | BI irrespective of type of breeding sources | CI in water storage containers | BI in water storage containers | PI in water storage containers | Dengue cases |
|---|---|---|---|---|---|
| May (2013) | 46.7 | 1.9 | 14.4 | 46.7 | 36 |
| Jun (2013) | 55.0 | 2.0 | 12.2 | 55.0 | 15 |
| Jul (2013) | 65.6 | 2.3 | 13.9 | 65.6 | 21 |
| Aug (2013) | 50.6 | 1.8 | 11.7 | 50.6 | 16 |
| Sep (2013) | 38.3 | 1.2 | 7.2 | 30.0 | 9 |
| Oct (2013) | 35.6 | 1.5 | 10.0 | 32.2 | 10 |
| Nov (2013) | 21.7 | 1.3 | 8.3 | 15.6 | 3 |
| Dec (2013) | 8.9 | 1.0 | 6.7 | 3.9 | 6 |
| Jan (2014) | 23.9 | 1.7 | 10.0 | 23.9 | 12 |
| Feb (2014) | 35.0 | 1.9 | 13.3 | 35.0 | 15 |
| Mar (2014) | 31.7 | 1.8 | 11.7 | 31.7 | 13 |
| Apr (2014) | 22.8 | 1.7 | 10.0 | 22.8 | 12 |
Table 6: Month wise entomological indicators of St. aegypti that showed significant association with dengue cases.
Breteau index of Aedine mosquitoes irrespective of type of breeding sources during monsoon months was about almost two times (50.7%) of that of Post monsoon season (25.8%) and pre-monsoon season (29.6) (Table 2) and it is significant (χ2 110.8; p<0.05). Similarly, BI of Aedine mosquitoes with respect to water storage containers showed that during monsoon months it was (33.2%) significantly (χ2 6.94.8; p<0.05) higher than the other seasons (27.7%) (Table 3).
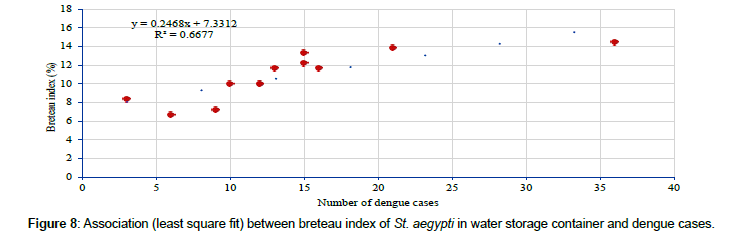
Relatively a higher level of BI of St. aegypti in water storage breeding sources (13.1 positive container per 100 houses i.e. BI) during pre-monsoon months when compared to other seasons (Monsoon=8.1 and post monsoon 11.3%) (Table 5), matches with the trend in dengue cases. Analysis has shown that the BI of St. aegypti in water storage breeding sources varied from 6.7 (December 2013) – 14.4 (May 2013) (Table 6) and has shown significantly positive correlation (r=0.817, p=0.001) with the monthly dengue cases (Figure 8).
The breteau index of St. aegypti irrespective of type of breeding sources and BI of Aedine species irrespective of breeding sources as well as in water storage breeding sources is not related to the trend in dengue cases.
Pupal index of Aedine mosquitoes irrespective of type of breeding sources was 105.3 during pre-monsoon season while it was 86.0 and 81.5 during monsoon and post monsoon seasons respectively. Though the mean number was relatively higher during pre-monsoon season it was not significantly different (pre-monsoon vs. monsoon=t:1.36; p=0.22; pre-monsoon vs. post monsoon=t:1.79; p=0.12) and did not show significant relation with the dengue cases. Pupal index of Aedine mosquitoes with respect to water storage containers also showed a similar trend.
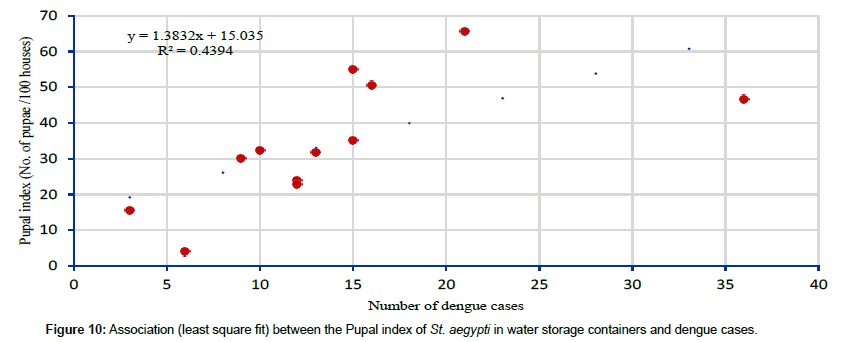
Pupal index of Stegomyia aegypti irrespective of breeding sources and with respect to water storage breeding sources varied from 26.1 to 54.4 and 20.4 to 54.4 respectively. Lowest number of St. aegypti pupae were recorded during monsoon, moderate during post monsoon and high during pre-monsoon. This indicates that pupal indices of St. aegypti showed a positive relationship with dengue cases. Analysis has shown that the pupal index of St. aegypti irrespective of breeding sources had shown the significantly positive correlation with the dengue cases (r=0.612, p=0.034) (Figure 9) and it varied from 8.9 (December 2013) to 65.6 (July 2013) (Table 6). Similarly the pupal index of St. aegypti in water storage breeding sources had also shown significantly positive correlation with the dengue cases (r=0.663, p=0.019) (Figure 10) and it varied from 3.9 (December 2013) – 65.6 (July 2013) (Table 6).
Thus, the season wise analysis of dengue cases in relation to various entomological indicators showed that container index and breteau index of water storage containers supporting St. aegypti, and pupal index irrespective of type of habitats as well as in water storage containers supporting St. aegypti showed positive trend with dengue cases. These four parameters were analyzed further by using regression (least square fit) model.
The values at lower 95% confidence limit of 1.34% of St. aegypti positive water storage containers, 7.85 St. aegypti positive water storage containers per 100 houses and 18.02 St. aegypti pupae per 100 houses in water storage breeding sources have been determined to be the threshold level above which the changes of dengue transmission would be higher. The lower 95% confidence limit of 19.9 St. aegypti pupae per 100 houses can also be considered as threshold level when the estimates were made irrespective of the type of breeding habitats.
Discussion
The association between the risk factors with dengue cases in 6 different sites was analyzed and found that all the parameters except house index can be used for monitoring the situation and identify the areas that require intervention when the values exceed the threshold level. The significant association was found with water storage and with respect to vector species. The results indicate that in such situation, survey should cover water storage containers and the samples from the positive houses are to be identified to species after emergence in order to estimate container, breteau and pupal indices of St. aegypti. Since it is specific to type of breeding sources and the vector species which can be considered as minimum when compared to the values when other habitats and Aedine species are included. The implication of higher rates of container index with respect to rain dependent container due to least disturbances unlike the water storage containers is that it may reduce the threshold level. However, the lower value of 95% confidence limit is likely to take care of this elevated level of container index when both the types of breeding sources are included.
The threshold values of percentage of water storage containers supporting St. aegypti (CI) and number of positive water storage containers with St. aegypti in 100 houses (BI) were compared in different months (irrespective of the sites) (Table 7) and different sites (irrespective of months) (Table 8).
| Indices | Container index | Breteau index |
|---|---|---|
| Threshold | 1.34 | 7.85 |
| May (2013) | 1.9 | 14.4 |
| Jun (2013) | 2.0 | 12.2 |
| Jul (2013) | 2.3 | 13.9 |
| Aug (2013) | 1.8 | 11.7 |
| Sep (2013) | 1.2 | 7.2 |
| Oct (2013) | 1.5 | 10.0 |
| Nov (2013) | 1.3 | 8.3 |
| Dec (2013) | 1.0 | 6.7 |
| Jan (2014) | 1.7 | 10.0 |
| Feb (2014) | 1.9 | 13.3 |
| Mar (2014) | 1.8 | 11.7 |
| Apr (2014) | 1.7 | 10.0 |
Table 7: Container index and Breteau index of St. aegypti in water storage breeding sources in different month compared to their respective threshold.
| Sites | Container index | Breteau index |
|---|---|---|
| Threshold values | 1.34 | 7.85 |
| Manur | 1.8 | 11.4 |
| Vadakarai | 2.2 | 14.7 |
| Kadayanalur | 2.0 | 13.9 |
| Palayamkottai | 1.3 | 7.2 |
| Valliyur | 1.3 | 9.7 |
| VK-Puthur | 1.5 | 7.8 |
| Total | 1.7 | 10.8 |
Table 8: Container Index and breteau index of St. aegypti in water storage breeding sources in different sites compared to their respective threshold.
During the months of September and December (monsoon months), the values were below the threshold. All the sites except Palaymkottai (urban) were at risk having above the threshold levels. The analysis showed that risk exists in all the months and in all the sites except the urban areas.
Lee et al. [10] using a seasonal cycle subseries (SCS) plot and univariate negative binomial regression (NBR) model and generalized estimating equation (GEE) to assess the relationship between monthly incidence rates and weather factors (temperature and precipitation) showed that during the rainy season there was an increase in dengue incidence. Climatic factors were shown to have significant impacts on dengue in Guangzhou and lag effects of temperature on dengue lasted the local whole epidemic season [11]. Limper et al. [12] and Xuan et al. [13] also showed that climate factor was an important factor for increasing dengue cases, rainfall being the major determinant. However, present study did not show rainfall as a determinant.
With the objectives of evaluating the temporal relationship between meteorological variables, entomological surveillance indices and confirmed dengue cases; and establishing the threshold for entomological surveillance indices including three mosquito larval indices [Breteau (BI), Container (CI) and House indices (HI)] and one adult index (AI) as an early warning tool for dengue epidemic [14]. Chang [14] showed that Model-AI, BI, CI and HI predicted the occurrence of dengue cases with 83.8, 87.8, 88.3 and 88.4% accuracy, respectively. The predicting threshold based on individual Model-AI, BI, CI and HI was 0.97, 1.16, 1.79 and 0.997, respectively. However, it was concluded that there was little evidence of quantifiable association among vector indices, meteorological factors and dengue transmission that could reliably be used for outbreak prediction. Other studies was carried out to determine the larval indices but was not related with the dengue cases [15,16]. Our study here provided the proof-of-concept of how to search for the optimal model and determine the threshold for dengue epidemics. Since those factors used for prediction varied, depending on the ecology and herd immunity level under different geological areas, different thresholds may be developed for different countries using a similar structure of the two-stage model. The present study results showed a higher threshold of 7.8 BI but lower CI threshold of 1.3 compared with the above findings. The difference could be that the CI and BI were calculated for water storage containers, the key breeding sources in the study area and St. aegpti which was the second predominant species.
Conclusion
Dengue infection is emerging with high proportion with extensive geographical spread. Vector species St. aegypti has been recorded in all the surveyed districts. Association between entomological risk factors indicated that there has been a strong and significant relationship between various breeding indices with respect to St. aegypti. Threshold levels have been derived which can be used as indicator to predict outbreaks. Source reduction targeting water storage breeding sources through community participation is required to prevent the outbreak. Disowning the practice of indiscriminate disposal of unusable containers by the community should be carried out as a permanent measure, besides proper disposal of solid wastes could result in preventing vector breeding in rain dependent habitats.
As the risks exist throughout the year due to water storage practices, it is important to monitor vector breeding indices in sentinel sites. When container, breteau and pupal indices are recorded above the critical level determined for this district, vector management measures needs to be initiated in order to prevent transmission and outbreak of dengue.
Acknowledgements
The authors are very thankful to Pondicherry University for providing the financial support. The Director, Vector Control Research Centre (ICMR) also provided great support by making the availability of various institutional facilities.
References
- Gubler DJ (2002) The global emergence/resurgence of arboviral diseases as public health problems. Arch Med Res 33: 330-342.
- Monath TP (1994) Dengue: the risk to developed and developing countries. Proc Natl Acad Sci U S A 91: 2395-2400.
- Gubler DJ (1998) Dengue and dengue hemorrhagic fever. Clin Microbiol Rew 11: 480-496
- Gubler DJ (2004) The changing epidemiology of yellow fever and dengue, 1900 to 2003: full circle? Comp Immunol Microbiol Infect Dis 27: 319-330
- Ooi EE, Gubler DJ (2009) Dengue in Southeast Asia: epidemiological characteristics and strategic challenges in disease prevention. Cadernos de Saúde Pública 25: 115-124.
- Torres JR, Castro J (2007) The health and economic impact of dengue in Latin America. Cad Saude Publica 23: 23-31.
- Jacobs M (2000) Dengue: emergence as a global public health problem and prospects for control. Trans R Soc Trop Med Hyg 94: 7-8.
- Balakrishnan N, Venkatesh S, Lal S (2006) An entomological Study on the Dengue vectors during outbreak of Dengue in Tiruppur town and its surroundings, Tamil Nadu, India. J Commun Dis 38: 164.
- Balaya S, Paul SD, D'Lima LV, Pavri KM (1969) Investigations on an outbreak of dengue in Delhi in 1967. Indian J Med Res 57: 767-774.
- Lee HS, Nguyen-Viet H, Nam VS, Lee M, Won S (2017) Seasonal patterns of dengue fever and associated climate factors in 4 provinces in Vietnam from 1994 to 2013. BMC Infect Dis 17: 218.
- Xiang J, Hansen A, Liu Q, Liu X, Tong MX (2017) Association between dengue fever incidence and meteorological factors in Guangzhou, China, 2005-2014. Environ Res 153: 17-26.
- Limper M, Thai KTD, Gerstenbluth I, Osterhaus ADME, Duits AJ, et al. (2016) Climate factors as important determinants of dengue incidence in Curaçao. Zoonoses Public Health 63: 129-137.
- Xuan LTT, Van Hau P, Thu DT, Toan DTT (2014) Estimates of meteorological variability in association with dengue cases in a coastal city in northern Vietnam an ecological study. Glob Health Action 7: 23119.
- Chang FS, Tseng YT, Hsu PS, Chen CD, Lian IB, et al. (2015) Re-assess vector indices threshold as an early warning tool for predicting dengue epidemic in a dengue non-endemic country. PLoS Neglected Tropical Diseases 9: e0004043.
- Bhat MA, Krishnamoorthy K, Khan AB (2014) Entomological surveillance of dengue vectors in Tamil Nadu, India. J Entomol Zoo Studies 2: 158-164.
- Bhat MA, Krishnamoorthy K (2014) Entomological investigation and distribution of Aedes mosquitoes in Tirunelveli, Tamil Nadu, India. Int J Curr Microbiol App Sci 3: 253-260.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi