Case Report, Clin Oncol Case Rep Vol: 3 Issue: 3
Remarkable Effect of Atezolizumab in Advanced Non-Small Cell Lung Cancer with PD-L1 Negative but Genome Instability is Increased: Case Report
Nozomu Motono1*, Yo Niida2, Sohsuke Yamada3 and Hidetaka Uramoto1
1Department of Thoracic Surgery, Kanazawa Medical University, Ishikawa, Japan
2Division of Genomic Medicine, Department of Advanced Medicine, Medical Research Institute, Kanazawa Medical University, Ishikawa, Japan
3Department of Clinical Pathology, Kanazawa Medical University, Ishikawa, Japan
*Corresponding Author : Nozomu Motono, Department of Thoracic Surgery
Kanazawa Medical University, 1-1 Daigaku, Uchinada, Ishikawa, 920-0293,
Japan
E-mail: motono@kanazawa-med.ac.jp
Received: March 26, 2020 Accepted: April 08, 2020 Published: April 20, 2020
Citation: Motono N, Niida Y, Yamada S, Uramoto H, et al. (2020) Remarkable Effect of Atezolizumab in Advanced Non-Small Cell Lung Cancer with PD-L1 Negative but Genome Instability is Increased: Case Report. Clin Oncol Case Rep 3:3.
Abstract
Immune checkpoint inhibitors have become important paradigms for treating non-small cell lung cancer patients. Although several biomarkers indicating the efficacy of immune checkpoint inhibitors have been reported, these markers sometimes are not accurate concerning the therapeutic effect. A 63-year-old man without a smoking history was diagnosed with lung adenocarcinoma pathological stage IVB without an epidermal growth factor receptor mutation or anaplastic lymphoma kinase translocation, rearrangement of ROS1, or expression of programmed deathligand 1 (PD-L1). Although cytotoxic anticancer therapies were administered, carcinoembryonic antigen (CEA) gradually became elevated, and the lesions progressed. Atezolizumab was administered as third-line chemotherapy, after which CEA normalized. After atezolizumab therapy was started, the Tumor Mutation Burden (TMB) and Microsatellite Instability (MSI) were analyzed in this patient’s samples. The TMB was identified at 16.1 mutations per megabase by next-generation sequencing. MSI was analyzed by a Bethesda panel assay, and three microsatellite loci (D2S123, D5S346, D17S250) were positive. Therefore, this patient was defined as TMB-high and MSI-high (MSI-H).Although this patient had negative PD-L1 expression, atezolizumab showed remarkable efficacy. PD-L1, TMB, and MSI are considered new predictive biomarkers for selecting patients that benefit from immune checkpoint inhibitors.
Keywords: Programmed death-ligand 1; Tumor mutation burden; Microsatellite instability; Immune checkpoint inhibitor; Non-small cell lung cancer; Prognosis
Introduction
Lung cancer remains the main cause of cancer-related mortality worldwide [1], and outcomes for patients diagnosed with advanced Non-small Cell Lung Cancer (NSCLC) are poor [2]. Patients with previously treated lesions or advanced or metastatic NSCLC are difficult to treat. Docetaxel has been the gold standard for secondor third-line treatment. The new development of antibodies that target the Programmed Death 1 (PD-1) and Programmed Deathligand 1 (PD-L1) pathways resulted in an important advance in the management of advanced NSCLC, and PD-1 inhibitors showed overall survival benefits compared to docetaxel. The POPLAR and OAK trials revealed that atezolizumab significantly improved survival compared to docetaxel in patients with previously treated NSCLC and showed prognostic improvement correlated with the PD-L1 expression on the tumor cells and tumor-infiltrating immune cells [3,4]. However, while a prognostic improvement correlated with the PD-L1 expression was noted in the POPLAR trial, even patients with low or undetectable PD-L1 levels in the OAK trial showed improved survival with atezolizumab.
Similar to the PD-L1 expression, Microsatellite Instability (MSI) and the Tumor Mutation Burden (TMB) are considered new predictive biomarkers for identifying patients likely to benefit from immune checkpoint inhibitors [5-14]. However, the overlap between the TMB, MSI, and PD-L1 differs among cancer types, and only 0.6% of cases were positive for all three markers in NSCLC [15].
We herein report a remarkable response to atezolizumab in a patient with previously treated advanced NSCLC who had TMB-high and MSI-high status and negative PD-L1 expression.
Case Presentation
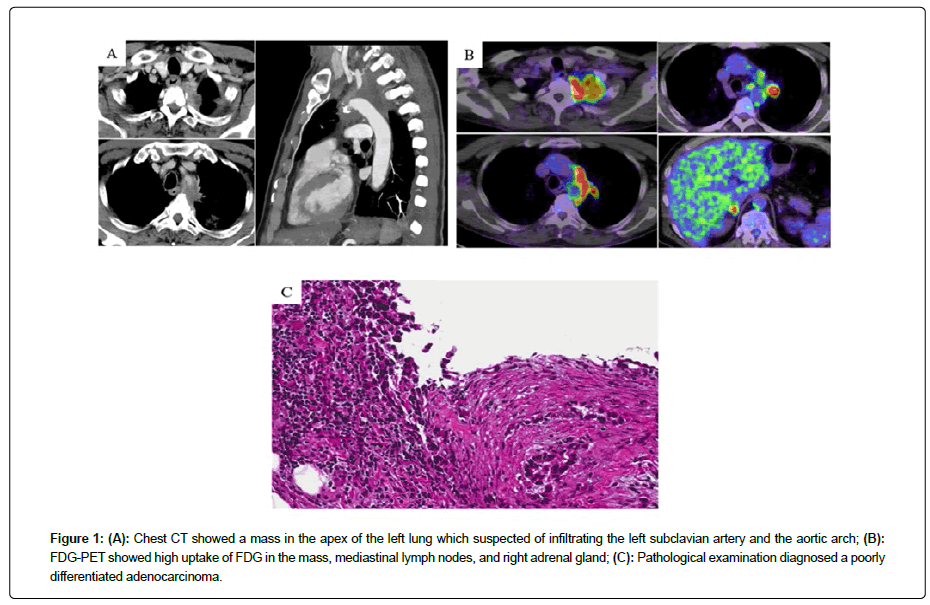
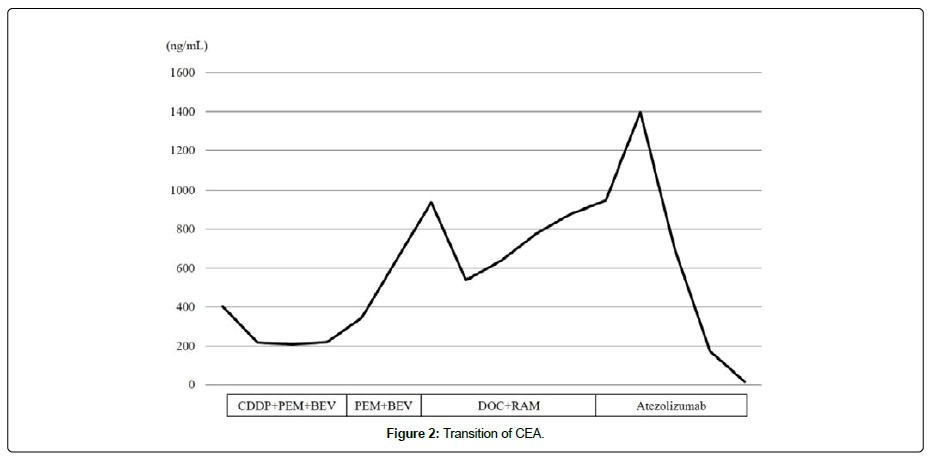
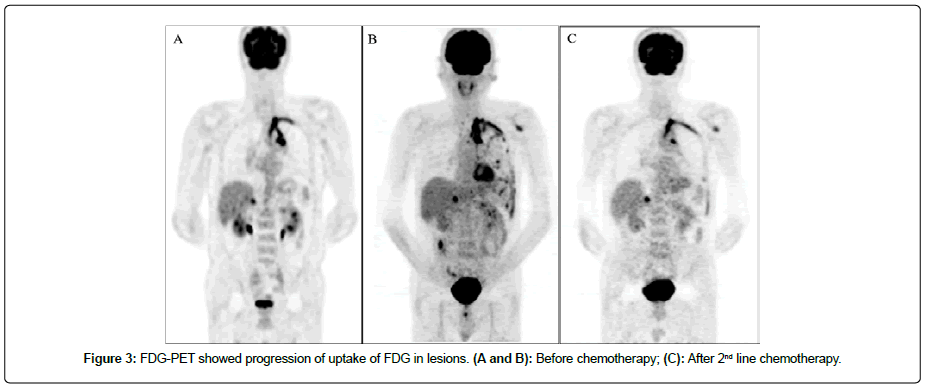
A 63-year-old man without a smoking history was aware of left shoulder pain. Chest Computed Tomography (CT) showed a mass in the apex of the left lung that was suspected of infiltrating being the left subclavian artery and aortic arch (Figure 1A). 18F-fluoro-2- deoxyglucose (18F-FDG) Positron Emission Tomography(PET) showed a high uptake of FDG in the mass, mediastinal lymph nodes, left 9th rib, and right adrenal gland (Figure 1B). The patients underwent a biopsy of pleural dissemination, and a pathological examination confirmed a poorly differentiated adenocarcinoma (Figure 1C). The pathological stage was pT4N2M1c, stage IVB, without Epidermal Growth Factor Receptor (EGFR) mutation or Anaplastic Lymphoma Kinase (ALK) translocation, rearrangement of ROS1, or expression of PD-L1. The patient received four Cycles of Chemotherapy with Cisplatin (CDDP), Pemetrexed (PEM), and Bevacizumab (BEV), followed by PEM and BEV maintenance therapy. However, Carcinoembryonic Antigen (CEA) was gradually elevated, and CT showed enlargement of the lesion at the left upper lung (Figure 2). Four cycles of Docetaxel and ramucirmab as second-line chemotherapy were administered. After that, CEA was further elevated (Figure 2), and PET showed the further uptake of FDG in the lesions (Figure 3A and 3B). The patient received atezolizumab as third-line chemotherapy. After eight cycles of atezolizumab, CEA was normalized (Figure 3C).
Figure 1: (A): Chest CT showed a mass in the apex of the left lung which suspected of infiltrating the left subclavian artery and the aortic arch; (B): FDG-PET showed high uptake of FDG in the mass, mediastinal lymph nodes, and right adrenal gland; (C): Pathological examination diagnosed a poorly differentiated adenocarcinoma.
After starting atezolizumab therapy, the TMB and MSI were analyzed in this patient’s pleural biopsy samples. The TMB was identified to be 16.1 mutations per megabase (mut/Mbp) by Next generation Sequencing (NGS). The MSI was analyzed by the Bethesda panel assay, three microsatellite loci (D2S123, D5S346, D17S250) were MSI positive. Therefore, this patient was defined as TMB-high and MSI-high (MSI-H) [16-18].
Discussion
Treatment of advanced NSCLC has evolved rapidly, and immunotherapy is a relatively new paradigm. The PD-1 receptors of activated T-cells are engaged by the tumor-expressed ligands PD-L1 and PD-L2 to reduce T-cell activation and facilitate tumor immune escape [5,18,19]. PD-1 and PD-L1 inhibitors for the treatment of advanced NSCLC are currently available and have demonstrated antitumor activity [3-6,20-22]. PD-1 and PD-L1 inhibitors significantly improved the Overall Survival (OS), Progression-Free Survival (PFS), and Objective Response Rate (ORR) in advanced NSCLC patients with PD-L1 expression. Furthermore, high PD-L1 expression was likely to be associated with increased benefits. However, PD-1 and PD-L1 inhibitors have also been reported to improve the OS in the population with PD-L1 <1% [22].
This discrepancy may be attributable to other biomarkers associated with the efficacy of PD-l and PD-L1 inhibitors. The TMB was recently confirmed to be a biomarker of the efficacy of PD-1 and PD-L1 inhibitors [8-11,23,24]. The TMB is defined as the total number of somatic mutations of the genomic coding area and associated with the emergence of neoantigens that trigger anti-tumor immunity [8]. The TMB is calculated based on the number of nonsynonymous somatic mutations identified by NGS. Although objective cut-off points for the TMB are not universally established, the cut-off points have been set at around 10 mut/Mbp in previous studies. Therefore, our case, which showed a TMB of 16.1 mut/Mbp, was defined as TMB-high [3,8,11].
MSI is also considered an independent predictive biomarker of immune checkpoint inhibitors [12-14,24]. MSI is the condition of genetic hypermutability and represents the phenotypic results of Mismatch Repair (MMR) deficiency. The five microsatellite loci (BAT-25, BAT-26, D2S123, D5S346, and D17S250) were amplified in a single multiplex Polymerase Chain Reaction (PCR). Cancers with instability at two or more of these loci are defined as MSI-H, while those with instability at a single locus are defined as MSI-low (MSI-L), and those with no instability at any of these loci are defined as Microsatellite Stable (MSS). In our case, three microsatellite loci (D2S123, D5S346, D17S250) were recognized, so the patient was defined as MSI-H.
In addition to the PD-L1 expression, TMB, and MSI are considered predictive biomarkers for selecting patients likely to benefit from immune checkpoint inhibitor.The overlap rates between PD-L1, TMB, and MSI are reportedly low in NSCLC [15]. TMB-high and MSI-H are found in 0.5%, TMB-high and PD-L1 positivity in 7.7%, MSI-H and PD-L1 positivity in 0.4%, and positivity in all 3 markers in 0.6% of NSCLC cases. Of note, most MSI-H patients had TMB-high (30/31) in a recent analysis of 5895 lung cancer tumors [25].
TMB-high and the neoantigen burden in tumors with MMR deficiency are associated with a favorable response to immune checkpoint inhibitors. A previous study reported an ORR of 53% and a 64% 2-year survival rate in cases of MMR deficiency or MSI-H tumors treated with pembrolizumab in a range of different cancer types, but this study did not include NSCLC patients.Although the efficacy of immune checkpoint inhibitors for NSCLC with TMB-high and neoantigen burden in tumors with MMR deficiency is uncertain, the present data showed that atezolizumab was markedly effective for NSCLC with TMB-high and MSI-H in the present case.
Conclusion
While PD-L1, TMB, and MSI can help identify patients who may benefit from immune checkpoint inhibitors, they are not complete predictors of favorable response in NSCLC. In the future, an analysis of the relationship among these biomarkers associated with the efficacy of immune checkpoint inhibitors is desired.
References
- Bray F, Ferlay J, Soerjomataram I, Siegel R, Torre LA, et al. (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68: 394-424.
- Postmus PE, Kerr KM, Oudkerk M, Senan S, Waller DA, et al. (2017) Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 28: 1-21.
- Fehrenbacher L, Spira A, Ballinger M, Kowanetz M, Vansteenkiste J, et al. (2016) Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): A multicentre, open-label, phase 2 randomised controlled trial. Lancet 387: 1837-1846.
- Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, et al. (2017) Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): A phase 3, open-label, multicentre randomized controlled trial. Lancet 389: 255-265.
- Taube JM, Klein A, Brahmer JR, Xu H, Pan X, et al. (2014) Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res 20: 5064-5074.
- Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, et al. (2015) Nivolumab versus docetaxel in advanced nonsquamous no-small-cell lung cancer. N Engl J Med 373: 1627-1639.
- Patel SP, Kurzrock R (2015) PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther 14: 847-856.
- Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, et al. (2015) Cancer immunology: Mutation landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348: 124-128.
- Hellmann MD, Ciuleanu TE, Pluzanski A, Lee JS, Otterson GA, et al. (2018) Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med 378: 2093-2104.
- Castro MP, Goldstein N (2015) Mismatch repair deficiency associated with complete remission to combination programmed cell death ligand immune therapy in a patient with sporadic urothelial carcinoma: Immunotheranostic considerations. J Immunother Cancer 3: 58.
- Bouffet E, Larouche V, Campbell BB, Merico D, Aronson M, et al. (2016) Immune checkpoint inhibition for hypermutant glioblastoma multiforme resulting from germline biallelic mismatch repair deficiency. J Clin Oncol 34: 2206-2211.
- Overman M, Kopetz S, McDermott R, Leach J, Lonardi S, et al. (2016) Nivolumab ± ipilimumab in treatment of patients with metastatic colorectal cancer with and without high microsatellite instability (MSI-H): CheckMate-142 interim results. J Clin Oncol 34: 3501.
- Vanderwalde A, Spetzler D, Xiao N, Gatalica Z, Marshall J (2018) Microsatellite instability status determined by next-generation sequencing and compared with PD-L1 and tumor mutational burden in 11,348 patients. Cancer Med 7: 746-756.
- Chapelle A, Hampel H (2010) Clinical relevance of microsatellite instability in colorectal cancer. J Clin Oncol 28: 3380-3387.
- Murphy KM, Zhang S, Geiger T, Hafez MJ, Bacher J, et al. (2006) Comparision of the microsatellite instability analysis system and Bethesda panel for determination of microsatellite instability in colorectal cancers. J Mol Diagn 8: 305-311.
- Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12: 252-264.
- Velcheti V, Schalper KA, Carvajal DE, Anagnostou VK, Syrigos KN, et al. (2014) Programmed death ligand-1 expression in non-small cell lung cancer. Lab Invest 94: 107-116.
- Melosky B, Chu Q, Juergens R, Leighl N, McLeon D, et al. (2016) Pointed progress in second-line advanced non-small-cell lung cancer: Rapidly evolving field of checkpoint inhibition. J Clin Oncol 34: 1676-1688.
- Passiglia F, Galvano A, Rizzo S, Incorvaia L, Listi A, et al. (2018) Looking for the best immune-checkpoint inhibitor in pre-treated NSCLC patients:An indirect comparison between nivolmab, pembrolizumab and atezolizumab. Int J Cancer 142: 1277-1284.
- Zhao Q, Xie R, Lin S, You X, Weng X (2018) Anti-PD-1/PD-L1 antibody therapy for pretreated advanced or metastatic nonsmall cell lung carcinomas and the correlation between PD-L1 expression and treatment effectiveness: An update meta-analysis of randomized clinical trials. Bio Med Res Int 3820956: 9.
- Hendricks LE, Rouleau E, Besse B (2018) Clinical utility of tumor mutational burden in patients with non-small cell lung cancer treated with immunotherapy. Transl Lung Cancer Res 7: 647-660.
- Prelaj A, Tay R, Ferrara R, Chaput N, Besse B, et al. (2019) Predictive biomarkers of response for immune checkpoint inhibitors in non-small-cell lung cancer. Eur J Cancer 106: 114-159.
- Schrock A, Sharma N, Peled N, Bufill J, Srkalovic G, et al. (2017) MA14.01 updated dataset assessing Tumor Mutation Burden (TMB) as a biomarker for response to PD-1/PD-L1 targeted therapies in lung cancer (LC). J Thorac Oncol 12: 422.
- Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, et al. (2015) PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med 372: 2509-2520.
- Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, et al. (2017) Mismatch repair deficiency predicts response of solid tumors to PD-1 blokade. Science 357: 409-413.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi