Case Report, Clin Oncol Case Rep Vol: 3 Issue: 4
Response to PARP Inhibitor in PALB2-Mutated Metastatic Pancreatic Adenocarcinoma: A Case Report
Michael J Allen1*, Amy Zhang2, Samuel D Saibil1, Sarah Picardo1, Shari Moura1, Grainne M O’Kane1 and Jennifer J Knox1*
1Department of Medical Oncology, Wallace McCain Centre for Pancreatic Cancer, Princess Margaret Cancer Centre, University Health Network, Toronto, Canada
2PanCuRx Translational Research Initiative, Ontario Institute for Cancer Research, Toronto, Canada
*Corresponding Author : Michael J Allen
Department of Medical Oncology, Princess Margaret Cancer Centre, 700 University Avenue, Toronto, ON M5G 1X6, Canada
E-mail: michael.allen@uhn.ca
Received: July 18, 2020 Accepted: September 01, 2020 Published: September 11, 2020
Citation: Allen MJ, Zhang A, Saibil SD, Picardo S, Moura S, et al. (2020) Response to PARP Inhibitor in PALB2-Mutated Metastatic Pancreatic Adenocarcinoma: A Case Report. Clin Oncol Case Rep 3:4.
Abstract
Abstract
PALB2 (Partner and Localizer of Breast Cancer Type 2 (BRCA2)) pathogenic variants increase sensitivity to poly (ADP-ribose) Polymerase Inhibitors (PARPi) in a variety of malignancies, yet no documented objective response to treatment in metastatic pancreatic cancer has been reported. We describe a case of a PALB2-mutated metastatic pancreatic cancer patient who had achieved a partial response to platinum-based chemotherapy, and was then observed to achieve an ongoing partial response with a further 42% reduction in tumor size over 6-months of treatment to date whilst on the PARPi olaparib.
Keywords: Pancreas adenocarcinoma; PALB2; PARP inhibitor; Olaparib
Introduction
Pancreatic ductal adenocarcinoma is the 7th leading cause of global cancer-related deaths [1]. Chemotherapy remains the mainstay of treatment with either combination fluorouracil, irinotecan and oxaliplatin (mFOLFIRINOX) or gemcitabine+nab-paclitaxel the preferred options. Outcomes remain poor with overall survival 11.1 m and 8.5 m respectively [2,3].
PALB2 (Partner and Localizer of Breast Cancer Type 2 (BRCA2)) plays a pivotal role in DNA damage repair [4]. Germline PALB2 mutations have been observed in breast and gynaecological cancer patients and has an approximate prevalence of 4% in familial pancreatic cancers [4,5]. The prevalence of PALB2 mutations in nonfamilial pancreatic cancers is unknown. It has been reported that PALB2 pathogenic variants increase sensitivity to poly (ADP-ribose) Polymerase Inhibitors (PARPi), similar to that observed in BRCA1 and BRCA2 mutated disease [6].
This case describes a partial response to olaparib in PALB2- mutated metastatic pancreatic adenocarcinoma following treatment with mFOLFIRINOX. Consent was obtained from the patient for publication of this article.
Clinical Investigation
A 60 year old male patient was diagnosed with metastatic pancreatic adenocarcinoma in December 2018 after presenting with a 6-month history of lethargy, vague upper abdominal pain, 20 kilograms weight loss, intermittent nausea and vomiting. Initial radiological investigations with Computed Tomography (CT) identified a 4.8 cm × 4.2 cm × 4.0 cm mass in the tail of pancreas that was inseparable from the left adrenal gland. The liver was enlarged measuring 20 cm with bilobar intrahepatic metastases. A heterogenous necrotic metastasis measuring 4.5 cm × 4.3 cm was present in segment 4a/8 with surrounding satellite lesions. A further dominant metastasis was present in segment 7 measuring 4.3 cm × 3.7 cm. Enlarged peripancreatic lymph nodes were observed.
A liver biopsy confirmed the diagnosis, with immunohistochemistry positive for CK7, CK19 and negative for CK20, TTF-1, Napsin and CDX2. A research liver biopsy was performed as part of the prospective COMPASS trial (Canadian Comprehensive Molecular Characterization of Advanced Pancreatic Ductal Adenocarcinoma for Better Treatment Selection) [7]. Whole genomic sequencing was performed on the Illumina HiSeq 2000/2500 platform as described by Connor et al. [8] identifying a germline stopgain PALB2 (c.T2192G:p.L731X) mutation and loss of the wild type allele (Figure 1). The tumour contained 3 of the 4 main driver genes for pancreatic ductal adenocarcinoma. KRAS (Kirsten rat sarcoma 2 viral oncogene homolog) was activated by a pathogenic G12D (c.G35A:p.G12D) mutation with loss of the wildtype allele by somatic Loss of Heterogenicity (LOH), while TP53 was biallelically inactivated by somatic frameshift with LOH, and CDKN2A lost by homozygous deletion. Wildtype KRAS has an inhibitory effect on oncogenic KRAS signalling and loss of the wildtype allele may promote a more aggressive tumour phenotype [9]. The tumour also had a one copy loss of SMAD4 without a somatic variant. The tumour mutational burden was 3.4 mutations per mega-base.
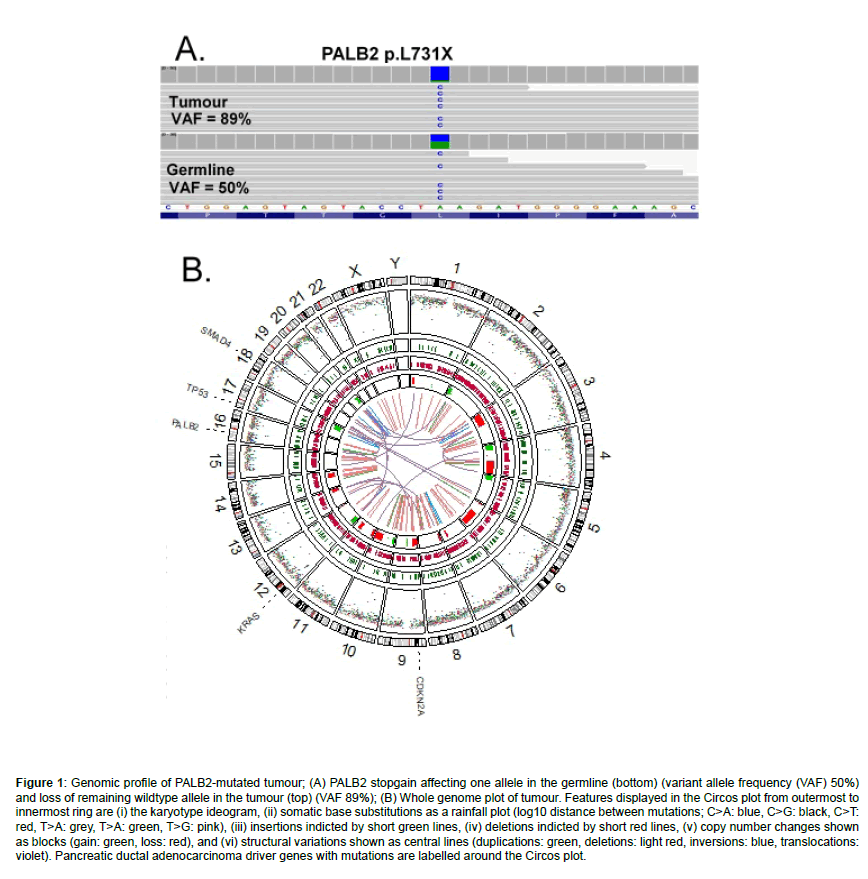
Figure 1: Genomic profile of PALB2-mutated tumour; (A) PALB2 stopgain affecting one allele in the germline (bottom) (variant allele frequency (VAF) 50%) and loss of remaining wildtype allele in the tumour (top) (VAF 89%); (B) Whole genome plot of tumour. Features displayed in the Circos plot from outermost to innermost ring are (i) the karyotype ideogram, (ii) somatic base substitutions as a rainfall plot (log10 distance between mutations; C>A: blue, C>G: black, C>T: red, T>A: grey, T>A: green, T>G: pink), (iii) insertions indicted by short green lines, (iv) deletions indicted by short red lines, (v) copy number changes shown as blocks (gain: green, loss: red), and (vi) structural variations shown as central lines (duplications: green, deletions: light red, inversions: blue, translocations: violet). Pancreatic ductal adenocarcinoma driver genes with mutations are labelled around the Circos plot.
The patients past medical history was notable for a diagnosis of non-insulin dependent diabetes mellitus 6-months prior to his cancer diagnosis. He had previously been diagnosed with hypertension and had undergone a gastric bypass 10 years prior. Medications at diagnosis included Metformin 500 mg bd, Amlodipine, Olmesartan/ Hydrochlorothiazide, Bisoprolol, Hydromorphone Contin and Hydromorphone immediate-release prn. He had a 30 pack year smoking history, quitting 2 months prior to his diagnosis. He did not drink alcohol. Family history included a sister diagnosed with tonsillar carcinoma (53 y) and a maternal aunt with pancreatic adenocarcinoma (80 y).
The patient was initially treated with mFOLFIRINOX (oxaliplatin 85 mg/m2, irinotecan 144 mg/m2 and infusional fluorouracil 2400 mg/ m2 over 46 hrs) administered every 2 weeks. He received 11 cycles, requiring a 20% dose reduction in oxaliplatin and 40% dose reduction in irinotecan due to toxicity (reduced performance status, nausea, vomiting) after cycle 1. He developed grade 2 peripheral neuropathy in his hands and lethargy after 10 cycles. His European Clinical Oncology Group (ECOG) performance status was 1. He had a marked improvement in pain symptoms. After 11 cycles he was switched to ‘maintenance’ olaparib 300 mg twice daily. He has been on olaparib for 6 months without complication with his current performance status ECOG 0, and is living an active life with work and travel.
Radiological assessment was conducted as per RECISTv1.1. Three target lesions were identified: A pancreatic tail mass, segment 4a/8 liver lesion, and segment 7 liver lesion. The sum measurement at baseline was 151 mm. After 6 months of mFOLFIRINOX the sum measurement of the target lesions had reduced to 78 mm with no new lesions identified (48% from baseline; cPR), at which point olaparib was commenced. Three subsequent CT scans have been performed all indicating a further reduction in the size of target lesions, initially 64 mm, 59 mm, and 45 mm (70% reduction from baseline; 42% reduction from commencement of olaparib) (Table 1 and Figures 2 and 3).
↓: Reduction in percentage
Table 1: Response to olaparib as per RECISTv1.1 observed in PALB2-mutated metastatic pancreatic adenocarcinoma.
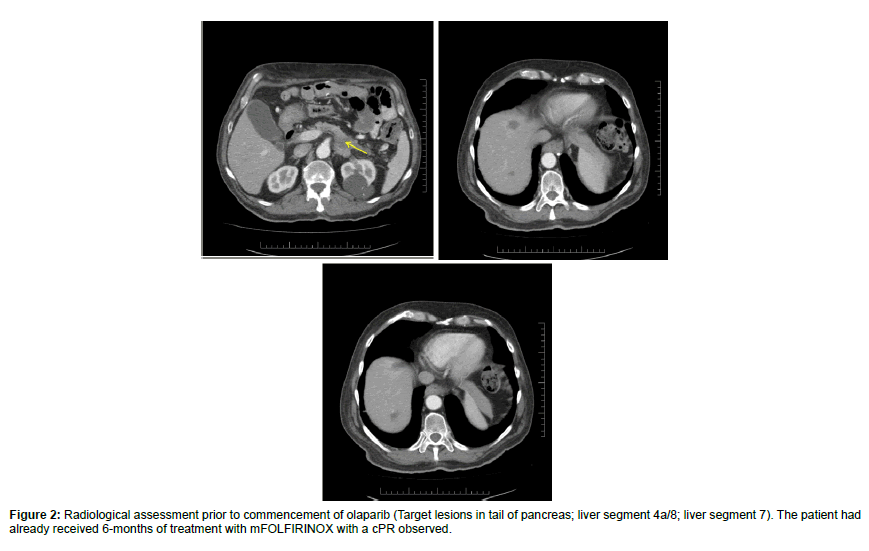
Figure 2: Radiological assessment prior to commencement of olaparib (Target lesions in tail of pancreas; liver segment 4a/8; liver segment 7). The patient had already received 6-months of treatment with mFOLFIRINOX with a cPR observed.
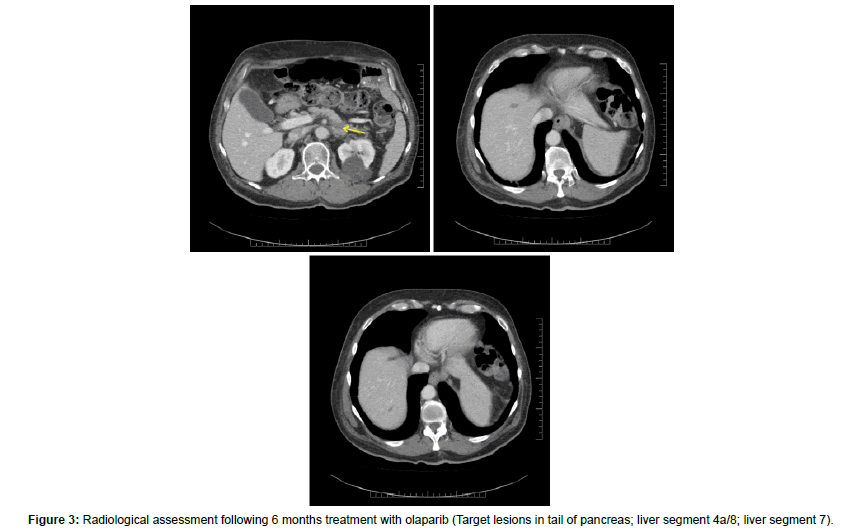
Figure 3: Radiological assessment following 6 months treatment with olaparib (Target lesions in tail of pancreas; liver segment 4a/8; liver segment 7).
Carbohydrate antigen 19.9 (Ca19.9) was 576 kU/L (normal<34) at baseline, reducing to 14 kU/L with chemotherapy. On olaparib a nadir of 6 kU/L was observed and it currently remains at 7 kU/L.
Discussion
Homologous Recombinant Repair Deficiency (HRD), and the subsequent accumulation of DNA double-stranded breaks, as occurs with the silencing of BRCA1/2, also occurs with the genetic inactivation of other genes, including PALB2 [10,11]. PALB2 is essential for the anchoring of BRCA2 to nuclear structures and recruitment of doublestranded DNA breaks, acting as a link between BRCA1 and BRCA2, with PALB2-mutant cancers also susceptible to PARPi [12,13]. In part due to its rarity and lack of testing, identification of PALB2-mutated tumours is rare, and subsequent treatment with PARPi is scarcely reported in the literature, specifically in pancreatic cancers.
This case demonstrates both the clinical and radiological benefit that can be achieved with targeted treatment of a PALB2-mutant pancreatic cancer who had previously achieved a partial response to standard first-line therapy with mFOLFIRINOX. A deeper partial response was observed, with a further 42% reduction in target tumour size over 6 months while on olaparib (Table 1). Similar outcomes were observed in the POLO (Pancreas Cancer Olaparib Ongoing) trial, which examined the benefit of olaparib as maintenance therapy following initial treatment with mFOLFIRINOX in germline BRCA-mutated metastatic pancreatic cancer. Whilst there was no overall statistically significant improvement in response rate, 23% (n=18) achieved a response including 2 patients with a complete response [14]. Maintenance olaparib resulted in an improved median progression-free survival. While some ongoing response on olaparib could be attributed to the prior chemotherapy it is highly unlikely this efficacy would persist for 6 months or more. A further publication has suggested that olaparib may have a clinical benefit without evidence of radiological response in the treatment of PALB2-mutated prostate cancer [15-17].
Of note, there was no observed response to single agent veliparib, another PARPi in HRD metastatic pancreatic cancer suggesting possible heterogenicity in response to different PARPi. Additionally, veliparib was not found to be of any further benefit when combined with platinum-based chemotherapy (cisplatin/gemcitabine) in BRCA/PALB2 mutated pancreatic cancer. This single study argues for sequential dosing of platinum-based chemotherapy followed by a PARPi rather than concurrent.
Conclusion
With improved and more accessible germline and somatic tumour genomic analysis, PALB2 mutations will be diagnosed more often, expanding the cohort of HRD tumours in pancreas cancer where PARPi may be therapeutic. This case report adds to the rationale supporting genomic analysis in all patients with metastatic pancreatic adenocarcinoma eligible for systemic treatments.
Acknowledgements
The authors would like to acknowledge the contribution of all clinical and non-clinical members of the COMPASS research team. Particularly, Professor Steven Gallinger, Stephanie Ramotar and Shawn Hutchinson for their input and advice regarding this manuscript.
References
- Rawla P, Sunkara T, Gaduputi V (2017) Epidemiology of pancreatic cancer: Global trends, etiology and risk factors. World J Oncol 10: 10-27.
- Conroy T, Desseigne F, Ychou M, Bouche O, Guimbaud R, et al. (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364: 1817-1825.
- Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, et al. (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369: 1691-1703.
- Nepomuceno TC, De Gregoriis G, de Oliveira FMB, Suarez-Kurtz G, Monteiro AN, et al. (2017) The role of PALB2 in the DNA damage response and cancer predisposition. Int J Mol Sci 18: 1886.
- Jones S, Hruban RH, Kamiyama M, Borges M, Zhang X, et al. (2009) Exomic sequencing identifies PALB2 as a pancreatic cancer susceptibility gene. Science 324: 217.
- Spugnesi L, Gabriele M, Scarpitta R, Tancredi M, Maresca L, et al. (2016) Germline mutations in DNA repair genes may predict neoadjuvant therapy response in triple negative breast patients. Gen Chromos Can 55: 915-924.
- Aung KL, Fischer SE, Denroche RE, Jang GH, Dodd A, et al. (2018) Genomics-driven precision medicine for advanced pancreatic cancer: Early results from the compass trial. Clin Cancer Res 24: 1344-1354.
- Connor AA, Denroche RE, Jang GH, Lemire M, Zhang A, et al. (2019) Integration of genomic and transcriptional features in pancreatic cancer reveals increased cell cycle progression in metastases. Cancer Cell 35: 267-282.
- Mueller S, Engleitner T, Maresch R, Zukowska M, Lange S, et al. (2018) Evolutionary routes and KRAS dosage define pancreatic cancer phenotypes. Nature 554: 62-68.
- Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, et al. (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly (ADP-ribose) polymerase. Nature 434: 913-917.
- Castroviejo-Bermejo M, Cruz C, Llop-Guevara A, Gutierrez-Enriquez S, Ducy M, et al. (2018) A RAD51 assay feasible in routine tumor samples calls PARP inhibitor response beyond BRCA mutation. EMBO Mol Med 10: e9172.
- Buisson R, Masson JY (2012) PALB2 self-interaction controls homologous recombination. Nucleic Acids Res 40: 10312-10323.
- Xia B, Sheng Q, Nakanishi K, Ohashi A, Wu J, et al. Control of BRCA2 cellular and clinical functions by a nuclear partner, PALB2. Mol Cell 22: 719-729.
- Golan T, Hammel P, Reni M, Van Cutsem E, Macarulla T, et al. (2019) Maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. N Eng J Med 381: 317-327.
- Horak P, Weischenfeldt J, von Amsberg G, Beyer B, Schutte A, et al. (2019) Response to olaparib in a PALB2 germline mutated prostate cancer and genetic events associated with resistance. Cold Spring Harb Mol Case Stud 5: a003657.
- Lowery MA, Kelsen DP, Capanu M, Smith SC, Lee JW, et al. (2018) Phase II trial of veliparib in patients with previously treated BRCA-mutated pancreas ductal adenocarcinoma. Eur J Cancer 89: 19-26.
- O'Reilly EM, Lee JW, Zalupski M, Capanu M, Park J, et al. (2020) Randomized, multicenter, phase II trial of gemcitabine and cisplatin with or without veliparib in patients with pancreas adenocarcinoma and a germline brca/palb2 mutation. J Clin Oncol 38: 1378-1388.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 