Case Report, Clin Oncol Case Rep Vol: 3 Issue: 5
Secondary Cutaneous Myeloid Sarcoma Treated with Nanoliposomal Encapsulation of Daunorubicin and Cytarabine
Andrew Gdowski1*, Lawrence Aung1, Tucker Pope1, Megan Kressin2 and Om Pandey1,3
1Department of Internal Medicine, The University of Texas at Austin Dell Medical School, Austin, TX
2Department of Pathology, The University of Texas at Austin Dell Medical School, Austin, TX
3Department of Oncology, The University of Texas at Austin Dell Medical School, Austin, TX
*Corresponding Author : Andrew Gdowski, Department of Internal Medicine
The University of Texas at Austin Dell Medical School, Austin, Texas
E-mail: andrewgdowski@gmail.com
Received: August 04, 2020 Accepted: September 16, 2020 Published: September 29, 2020
Citation: Gdowski A, Aung L, Pope T, Kressin M, Pandey O (2020) Secondary Cutaneous Myeloid Sarcoma Treated with Nanoliposomal Encapsulation of Daunorubicin and Cytarabine. Clin Oncol Case Rep 3:5. DOI: 10.37532/cocr.2020.3(5).144
Abstract
Cutaneous myeloid sarcoma is a rare malignancy and most often arises in the setting of acute myeloid leukemia. Here, we present the unique case of a patient who developed secondary cutaneous myeloid sarcoma after developing myelodysplastic syndrome from breast cancer treatment. We describe complete resolution of her lesions after treatment with a nanoliposomal dual encapsulation of daunorubicin and cytarabine and to our knowledge the first known treatment of a patient with cutaneous myeloid sarcoma with this regimen.
Keywords: Myeloid; Sarcoma; Breast cancer; Cytarabine; Nanoliposomal
Introduction
The diagnosis of AML and myeloid sarcoma are similar with one key exception: myeloid sarcoma consists of extramedullary myeloid blasts that develop into a mass by effacing normal tissue architecture. The WHO has set out guidelines for the diagnostic terminology and criteria of myeloid sarcoma and other names have also been used in the diagnosis: granulocytic sarcoma, extramedullary myeloid tumor, or chloroma. The most frequent sites of involvement are the skin, lymph nodes, gastrointestinal tract, bone, soft tissue, and testis. Other sites can also be involved.
Diagnosing myeloid sarcoma presents a challenge, particularly if the patient doesn’t present with a known hematological disorder. The clinical presentation, radiographic images, immunohistochemical results, and cytogenetic markers all should be taken into account during the diagnostic process. Morphologically myeloid sarcoma appearance is the same as AML with blast forms, high nuclear-tocytoplasmic ratios, fine chromatin and prominent nucleoli.
Case Presentation
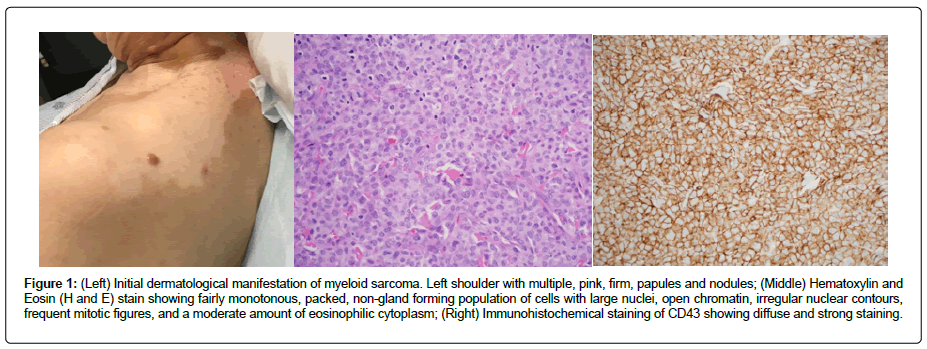
A 68-year old woman with a past medical history of vitiligo and lobular carcinoma of the breast treated with lumpectomy, adjuvant chemotherapy, radiation therapy, and tamoxifen for 5 years presented to the clinic with multiple, intermittently appearing, pink firm papules and nodules on multiple areas of the body including: right forehead, left neck, bilateral posterior shoulders, back and upper extremities (Figure 1). Skin biopsy of one of the nodules was consistent with the diagnosis of myeloid sarcoma (Figure 1). A bone marrow biopsy was performed and she was found to have myelodysplastic syndrome with multi-lineage dysplasia, however, there was no evidence of leukemia. Next generation sequencing panel was performed on the cutaneous lesion which showed mutations in NPM1, DNMT3A, as well as KRAS and FLT3 mutation negative.
Figure 1: (Left) Initial dermatological manifestation of myeloid sarcoma. Left shoulder with multiple, pink, firm, papules and nodules; (Middle) Hematoxylin and Eosin (H and E) stain showing fairly monotonous, packed, non-gland forming population of cells with large nuclei, open chromatin, irregular nuclear contours, frequent mitotic figures, and a moderate amount of eosinophilic cytoplasm; (Right) Immunohistochemical staining of CD43 showing diffuse and strong staining.
Thus it was concluded that she had a secondary entirely cutaneous myeloid sarcoma which developed after breast cancer therapy. This has never been reported before in the literature hence no standard treatment protocols exist. Treatment with a dual encapsulated daunorubicin and cytarabine nanoliposomal formulation was selected based on its approval in the setting of therapy related Acute Myeloid Leukemia (AML) in patients ≥ 60 years old as it has shown an improved median overall survival benefit compared to the standard combination of daunorubicin and cytarabine (7+3 regimen) [1]. Her treatment course thus far has included: induction chemotherapy with the same dosing used in the clinical trial (daunorubicin 44 mg/m2 and cytarabine 100 mg/m2 on day 1, 3 and 5). Following induction therapy, she developed neutropenic fever from which she recovered. After induction chemotherapy, the cutaneous nodules completely resolved. Two cycles of consolidation therapy were performed with two days of chemotherapy rather than three days. The consolidation therapy was well tolerated.
A bone marrow biopsy at four months, again showed no evidence for therapy-related myeloid leukemia. She is now more than a year after her initial diagnosis of cutaneous myeloid sarcoma and is without evidence of disease. One more additional bone marrow biopsy is planned.
Discussion
This case is unique as it is the first reported case of secondary cutaneous myeloid sarcoma arising in the setting of myelodysplasia due to breast cancer therapy. Additionally, experience with therapy for secondary cutaneous myeloid sarcoma in this setting is limited without prior reports.
The diagnosis of AML and myeloid sarcoma are similar with one key exception: myeloid sarcoma consists of extramedullary myeloid blasts that develop into a mass by effacing normal tissue architecture [2]. The WHO has set out guidelines for the diagnostic terminology and criteria of myeloid sarcoma and other names have also been used in the diagnosis: granulocytic sarcoma, extramedullary myeloid tumor, or chloroma [3]. The most frequent sites of involvement are the skin, lymph nodes, gastrointestinal tract, bone, soft tissue, and testis. Other sites can also be involved [4].
Cutaneous myeloid sarcoma can arise in the setting of AML, relapse of AML, through blast transformation in chronic myeloproliferative syndrome or myelodysplasia, or de novo. In one small case series, the diagnosis was most likely simultaneously seen with AML or AML developing after diagnosis (within 10.5 months on average) [3,5]. In the case of the patient presented here, it appears her cutaneous myeloid sarcoma arose is the setting of myelodysplasia. However, it is peculiar that repeat bone marrow biopsy did not show evidence of myelodysplasia or myeloid leukemia. Thus, it may be possible that the myelodysplasia initially identified in the bone marrow was transient.
Diagnosing myeloid sarcoma presents a challenge, particularly if the patient doesn’t present with a known hematological disorder. The clinical presentation, radiographic images, immunohistochemical results, and cytogenetic markers all should be taken into account during the diagnostic process [6]. Morphologically myeloid sarcoma appearance is the same as AML with blast forms, high nuclearto- cytoplasmic ratios, fine chromatin and prominent nucleoli. Immunohistochemical results of myeloid sarcoma are identical to AML, thus stains such as CD34 and CD117 are employed to identify immature cells and MPO, CD68/KP1, lysozyme, CD33, CD14, CD43, CD168 as well as others are used to determine if the cell type is myeloid or monocytic [7,8]. Some of the more common cytogenetic abnormalities seen in myeloid sarcoma are: monosomy 7 and 16, trisomy 8, trisomy 11, Mixed Lineage Leukemia (MLL) gene rearrangements, inversion of chromosome 16, as well as 16q, 5q, and 20q deletions [8]. Additionally, mutations in Nucleophosmin (NPM) 1 have been noted in cases of myeloid sarcoma [9]. This patient had a pathological mutation in NPM1 but not in FLT3 gene which is favorable in AML and may have improved outcomes with high-dose induction therapy [10-12]. She also had a mutation in DNT3A which is also associated with improved outcomes when treated with high dose daunorubicin therapy [11].
Treatment options in myeloid sarcoma involve chemotherapy, surgical resection, allogenic hematopoietic stem cell transplant, and radiation therapy with chemotherapy being the most common modality [6]. Due to her age and her funding status, she was not a candidate for allogenic stem cell transplant. Since no therapeutic guidelines exist for secondary myeloid sarcoma, it was hypothesized that since myeloid sarcoma shares many pathologic and genetic characteristics with AML, she could benefit from a regimen used for AML.
The decision was made to treat with the dual drug nanopliposomal encapsulation of daunorubicin and cytarabine. This formulation was recently approved following a phase 3 clinical trial showing improved overall survival (9.56 v 5.95 months) in patients 60-75 years old with secondary acute myeloid leukemia compared to standard of care cytarabine plus daunorubicin (7+3) chemotherapy. Similar adverse events were seen in both arms [1]. Current NCCN guidelines give this therapy a category 1 recommendation for induction therapy for therapy-related AML and category 2A recommendation for consolidation therapy. There have been no reported cases of secondary myeloid sarcoma treated with this therapy. However, it was extrapolated that the same properties which make this formulation successful at treating secondary AML should prove beneficial in secondary cutaneous myeloid sarcoma. The improved cytotoxicity demonstrated toward cancer cells in preclinical models stems from the ability of the dual-drug liposomal encapsulation of cytarabine and daunorubicin to deliver a 5:1 drug ratio to cancer cells. This fixed ratio has been reported to have synergistic activity in killing cancer cells [13].
The prognosis of myeloid sarcoma is difficult to reliably determine due to the limited number of cases published. From one study, there is no significant difference in prognosis whether the myeloid sarcoma is secondary or arises de-novo. Reported overall survival had a broad range from 2 months to 10 years [2].
Conclusion
In this case report we presented a rare case of secondary cutaneous myeloid sarcoma following breast cancer therapy. We also discussed the first reported use of a dual-drug nanopliposomal encapsulation of daunorubicin and cytarabine for the treatment of secondary cutaneous myeloid sarcoma with overall positive results.
References
- Lancet JE, Uy GL, Cortes JE, Newell LF, Lin TL, et al. (2018) CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J Clin Oncol 36: 2684-2692.
- Wang HQ, Li J (2016) Clinicopathological features of myeloid sarcoma: Report of 39 cases and literature review. Pathol Res Pract 212: 817-824.
- Kudva R, Monappa V, Solanke G, Valiathan m, Rao ACK, et al. (2017) Myeloid sarcoma: A clinicopathological study with emphasis on diagnostic difficulties. J Cancer Res Ther 13: 989-993.
- Yilmaz AF, Saydam G, Sahin F, Baran Y (2013) Granulocytic sarcoma: A systematic review. Am J Blood Res 3: 265-270.
- Neiman RS, Barcos M, Berard C, Bonner H, Mann R, et al. (1981) Granulocytic sarcoma: A clinicopathologic study of 61 biopsied cases. Cancer 48: 1426-1437.
- Huang XL, Tao J, Li JZ, Chen XL, Chen JN, et al. (2015) Gastric myeloid sarcoma without acute myeloblastic leukemia. World J Gastroenterol 21: 2242-2248.
- Aneja A (2018) Myeloid sarcoma pathology. Medscape Pathol.
- Pileri SA, Ascani S, Cox MC, Bacci F, Piccioloi M, et al. (2007) Myeloid sarcoma: clinico-pathologic, phenotypic and cytogenetic analysis of 92 adult patients. Leukemia 21: 340-350.
- Falini B, Lenze D, Hasserjian R, Couplant S, Jaehen D, et al. (2007) Cytoplasmic mutated nucleophosmin (NPM) defines the molecular status of a significant fraction of myeloid sarcomas. Leukemia 21: 1566-1570.
- Becker H, Marucci G, Maharry K, Radmacher MD, Mrozek K, et al. (2010) Favorable prognostic impact of NPM1 mutations in older patients with cytogenetically normal de novo acute myeloid leukemia and associated gene and microRNA-expression signatures: A cancer and leukemia group B study. J Clin Oncol 28: 596-604.
- Patel JP, Gonen M, Figueroa ME, Fernandez H, Sun Z, et al. (2012) Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. N Engl J Med 366: 1079-1089.
- Falini B, Nicoletti I, Martelli MF, Mecucci C (2007) Acute myeloid leukemia carrying cytoplasmic/mutated nucleophosmin (NPMc+AML): biologic and clinical features. Blood 109: 874-885.
- Mayer LD, Harasyn TO, Tardi PG, Harasym HL, Shew CR, et al. (2006) Ratiometric dosing of anticancer drug combinations: controlling drug ratios after systemic administration regulates therapeutic activity in tumor-bearing mice. Mol Cancer Ther 5: 1854-1863.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi