Research Article, Vector Biol J Vol: 3 Issue: 1
Sublethal Effects of Ethanol Extract of Ocimum sanctum on Laboratory Bred Population of Dengue Mosquito Aedes aegypti L. (Diptera: Culicidae)
Mohd Shazad, Kamal Kumar Gupta*, Sunil Kayesth and Shailendra Kumar
Department of Zoology, Deshbandhu College, University of Delhi, Kalkaji, New Delhi
*Corresponding Author : Kamal Kumar Gupta
Insect Reproduction Laboratory, Department of Zoology, Deshbandhu College, University of Delhi, Kalkaji, New Delhi 110019, India
Tel: +91981182200
E-mail: kamal_suyash@yahoo.com
Received: April 20, 2018 Accepted: June 20, 2018 Published: July 05, 2018
Citation: Shazad M, Gupta KK, Kayesth S, Kumar S (2018) Sublethal Effects of Ethanol Extract of Ocimum sanctum on Laboratory Bred Population of Dengue Mosquito Aedes aegypti L. (Diptera: Culicidae). Vector Biol J 3:1. doi: 10.4172/2473-4810.1000128
Abstract
Objectives: Aedes aegypti is a vector of several diseases which cause serious discomfort and fatality to human being. Current investigations were carried out to assess sublethal effects of ethanol extract of the leaves of Ocimum sanctum against A. aegypti in order to find ecofriendly alternatives for integrated vector management�??.
Methods: Lethal and sublethal effects of ethanol extract of O. sanctum against fourth instar larvae were assessed by using WHO protocol. The studies were extended to evaluate the effects of the extract on growth and development of the treated larvae. The chemical components present in the extracts were analyzed by Gas chromatography and mass spectroscopy (GC-MS).
Results: Exposure of fourth instar larvae of A. aegypti revealed moderate toxicity. However, the treated larvae exhibited mortality during subsequent days of exposure. The treatment with the extract also has adverse impact on growth and development of the larvae. Consequently there was increase in the larval duration in the treated larvae. Larva-pupa intermediate and pupa-adult intermediate were the developmental anomalies observed in the treated fourth instar larvae. This suggested presence of juvenile hormone mimic or juvenile analogue activity in the ethanol extract of O. sanctum leaves. Presence of farnasene, a derivates of the intermediates in the juvenile hormone biosynthesis was reported in the GCMS analysis of the extract.
Conclusion: Our studied indicated moderate lethal and efficient sublethal effects of ethanol extract of O. sanctum against A.aegypti. The bioactive compounds present in the extract, individually or synergistically may be used in formulating a strategy for the mosquito management
Keywords: Aedes aegypti; Ocimum sanctum; Sublethal; Growth and development
Introduction
Aedes aegypti L. (Diptera: Culicidae) is the primary vector of diseases like dengue fever [1,2], yellow fever [3], chikungunya [4,5] and Zika virus [6]. The incidence of dengue fever has increased dramatically around the world, specifically in last decade. One recent estimate indicated 390 million dengue infections per year worldwide of which 96 million manifest clinically with severity of disease [7]. In fact the actual number of dengue cases is much more; many cases are misclassified and underreported [8]. In 2016, dengue outbreak occurred worldwide; the region of Americas reported more than 2.38 million cases [9]. Official records of Union health ministry of India reported a significant increase in the number of dengue cases in last few years [10]. It was estimated that that 3.9 billion people, in 128 countries, are at risk of infection with dengue viruses [11].
In the absence of effective vaccine against many viral disease transmitted by A. aegypti, control of the mosquito population is the only mean to prevent the spread of the diseases. Synthetic insecticides are widely used as larvicide in mosquito control programs worldwide. However, they are toxic to non-target organisms, have high mammalian toxicity and affect the environment by contaminating soil, water, and air [12]. In past few decades mosquito populations have developed resistance against a variety of synthetic organic chemicals. A. aegypti has developed resistant against deltamethrin [13], an extensively used insecticide in mosquito control programs [14].
Plant possesses a variety of phytochemicals. Many of these are secondary metabolites and are used for protection against herbivore insect predators. Therefore, they are natural candidates for the discovery of new compounds to combat insect menace [15]. Plant based chemicals have an advantage over the synthetic chemicals as they are target specific, biodegradable, environmental friendly and relatively nontoxic to human; also chances of development of resistance in insects against plant products are less [16,17].
Ocimum sanctum commonly called tulsi in India is “queen of the herbs” [18]. Essential oils present in the leaves of O. sanctum have been reported to possess wide range of activities in insects. Many species of Ocimum have larvicidal activity against A. aegypti and Culex quinquefasciatus [15,19,20]. Besides antifeedent activity [21] and repellent activity [22] of Ocimum has also been reported against Aedes and other insects. Some of the bioactive compounds present in Ocimum function as insect growth regulators and disrupt growth and development in insects [23].
Most of the studies on O. sanctum were focused on larvicidal activity and lethal effects on A. aegypti. Very few reports were available on delayed toxicity and sublethal effects of plant extracts on A. aegypti. Growth, development and reproduction are important components of insect life; aberration of these can hamper the insect population. Therefore, a schematic research on implication of ethanol extract of O. sanctum against fourth instar larvae of A. aegypti was initiated in search for effective affordable natural products which can be used in integrated vector management programme of A. aegypti.
Materials and Methods
Collection of plant material
The leaves of O. sanctum were collected from the botanical garden of Deshbandhu College, Kalkaji, New Delhi, India (28.5403° N, 77.2544° E), during April-May, 2017 and brought to the laboratory in polythene bags. The leaves were washed thoroughly with tap water in order to remove dust or any other particles stuck to them. These were kept in shade at temperature (30 ± 2°C) for about 30 days to dry completely.
Preparation of ethanol extract
Extract of O. sanctum was prepared by cold extraction method [24]. The dried leaves were mechanically grinded with the help of electric grinder to make a fine powder. The powder of the leaves was mixed with five times ethanol (weight to volume) and kept for 24h at a temperature 27°C. The extract was filtered by using Whatman filter paper No 1. The residue after first extraction was extracted again twice in the same manner. Subsequently, the extracts were pooled, and concentrated using a vacuum rotary evaporator (Buchi) at 45°C. After complete evaporation of the solvent, the concentrated extract was collected and stored as 10% stock soulution in a refrigerator at 4°C for further use.
Rearing of mosquitoes
The present investigations were employed on the dengue fever mosquito, A. aegypti. The stock culture was obtained from International Center for Genetic Engineering and Biotechnology (ICGEB), New Delhi, India. The colony was maintained in an insectary at temperature 28 ± 1°C, 80 ± 5% relative humidity and 14 L: 10 D photoperiod [25]. Adults were provided with freshly soaked deseeded raisins. Periodic blood meals were provided to female mosquitoes for egg maturation. The eggs were collected in a bowl lined with Whatman filter paper and were allowed to hatch in enamel trays filled with dechlorinated water. Larvae were provided with a mixture of yeast powder and grinded dog biscuits in a ratio on 1:3. The pupae were collected in enamel bowl filled with dechlorinated water and transferred to the cloth bag cages for adult emergence.
Larval toxicity bioassay
The protocol prescribed by WHO [8] was adopted to conduct bioassay of O. sanctum leaf ethanol extract against fourth instar larvae of A. aegypti. Laboratory reared A. aegypti larvae were used for the larvicidal bioassay. Twenty-five fourth instar larvae were kept in an enamel bowl (capacity 500mL) containing 249 mL of dechlorinated water and 1mL of ethanol leaf extract of O. sanctum. Concentrations of 400 ppm, 200 ppm, 100 ppm and 50 ppm of the extract were used for the bioassay. In control, 1 mL of ethanol was mixed to 249 mL of dechlorinated water. Normal food was provided to the experimental larvae. Mortality in the control and experimental set ups was recorded after 24h of exposure. The experiments were continued to observe day wise mortality, larval duration, and developmental abnormalities in the larvae and the pupae. The various developmental abnormalities were photographed using Nikon SMZ745T stereo zoom microscope The control mortality, if any, was corrected by using Abbott‟s formula [26]. All the experiments, unless specified, were replicated five times.
Statistical analysis
SPSS version 19 software was used for analysis of data. Anova: one factor was used to test the significant difference between groups. Tukey post hoc test was used to identify the significant difference within the group. Results with p<0.05 were considered to be statistically significant [27].
GC-MS Analysis of the Ethanol Leaf Extract of Ocimum sanctum
The phytochemicals present in the ethanol extract of O. sanctum were analyzed through gas chromatography and mass spectroscopy (GC-MS) [28]. The concentrated extract of O. sanctum was dissolved in ethanol and injected into the Gas chromatography unit (Shimadzu GC-MS QP2010). The injector temperature was maintained at 250 ºC. The detector used was flame ionization detector which was maintained at 280 ºC. The pressure of the carrier gas, nitrogen was kept at 10 psi. The oven temperature was set at 60 ºC to 280 ºC with a gradual increment of 10 ºC per min. The injected extracts were eluted in the DB-5 MS column of 30m long and 0.25 mm inner diameter and the eluted constituents were detected by flame ionization detector. The GC chromatogram was recorded. From the graph, the compounds were identified by comparing the data with the existing software libraries like WILEY08, NIST08 and NIST08s [29].
Result
The present study was carried out with an aim to develop a safe and environment-friendly strategy for mosquito management. Efficacy of ethanol extract of O. sanctum was assessed for survival and longevity of early fourth instar larvae of A. aegypti. The results of larvicidal bioassays showed moderate toxicity of the ethanol extract of O. sanctum against early fourth instars larvae of A. aegypti. In a population of the fourth larval instar, 80.00%, 24.00%, 4.80%, and 4.00% mortality was observed after 24h of the exposure to 400 ppm, 200 ppm, 100 ppm and 50ppm respectively (Table 1). Exposure to 400ppm of the ethanol extract of O. sanctum also caused mortality during subsequent days. It was recorded that at this concentration the mortality on second, third, fourth and fifth day was 5.6%, 0.8%, 1.6% and 0.8% respectively (Table 2).
| Concentration (ppm) | Percent mortality after 24 h of exposure* (Mean ± S.E.) | F value | P-value |
|---|---|---|---|
| 50 | 4a ± 2.53 | 82.368 | ≤ 0.001 |
| 100 | 4.8a ± 2.33 | ||
| 200 | 24b ± 6.32 | ||
| 400 | 80c ± 4.0 |
Table 1: Larvicidal activity of ethanol extracts of Ocimum sanctum against fourth instar larvae of Aedes aegypti.
| Concentration | Day wise mortality* (Mean ± S.E.) | |||||
|---|---|---|---|---|---|---|
| Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | |
| 50 ppm | 4 ± 0.06 | 0 | 0 | 0 | 0.8 ± 0.02 | 0.8 ± 0.02 |
| 100 ppm | 4.8 ± 0.05 | 0 | 0 | 0 | 0 | 0.8 ± 0.02 |
| 200 ppm | 24 ± 0.14 | 0.8 ± 0.02 | 0 | 0 | 0 | - |
| 400 ppm | 80 ± 0.09 | 5.6 ± 0.09 | 0.8 ± 0.02 | 1.6 ± 0.04 | 0.8 ± 0.02 | - |
Table 2: Day wise larvicidal activity of ethanol extract of Ocimum sanctum on fourth instar larvae of Aedes aegypti.
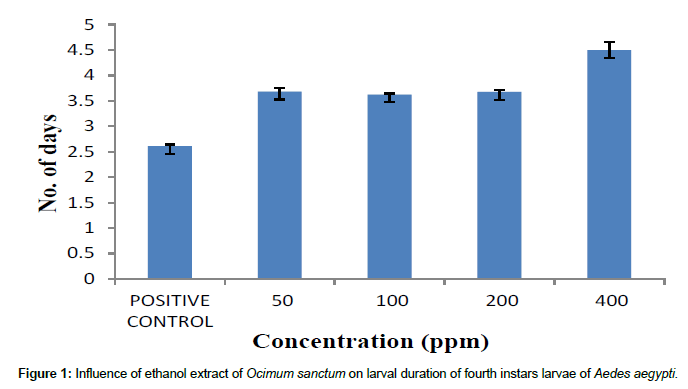
Ethanol extract of O. sanctum has an effect on the larval duration (Figure 1). Normally the fourth instars larvae developed into pupa after 2.61 days. However, the larval duration of the surviving fourth instar increased to 3.68, 3.62, 3.68 and 4.45 days after treatment with 50ppm, 100 ppm, 200 ppm and 400 ppm respectively. The results were statistically significant (P< 0.001).
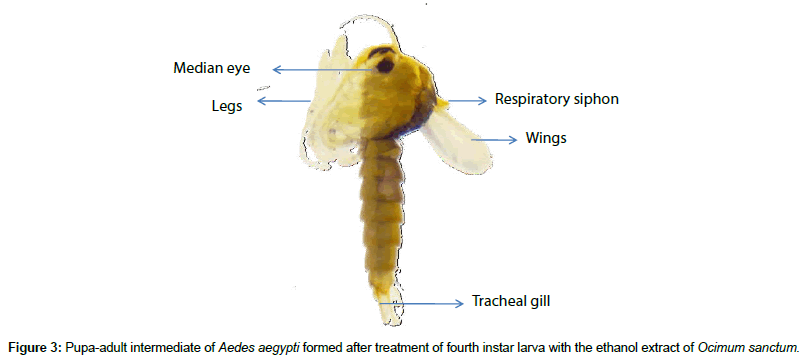
Treatment of the fourth instar larvae with the extract also resulted in formation of larva-pupa intermediate. The pupae formed from the treated larvae sometime moulted into pupa-adult intermediate. The body of the fourth instars larva of A. aegypti is differentiated into head, thorax and abdomen. A pair of eye spot is present in head region and respiratory siphon and tracheal gills are present on last abdominal segment. The body is covered with chitinous setae (Figure 2a). Body of pupa is divided into cephalothorax and abdomen. Cephalothorax region possesses single median eye, a pair of respiratory siphon and rudimentary appendages. The abdomen is attached to the cephalothorax at an angle which gives a characteristic comma shape to the pupa. Tracheal gills were present on last abdominal segment (Figure 2c). In case of the larva-pupa intermediates body divided into cephalothorax and abdomen. Like pupa a single median eye, a pair of respiratory siphon and rudimentary appendages were present in cephalothoracic region. The chitinous setae were absent. However, the abdomen was attached to the cephalothorax in a straight line. Consequently, characteristic comma shape of the pupa was absent in the larva-pupa intermediate. Like fourth instar larva, the respiratory siphon was also observed on the last abdominal segment of larva-pupa intermediate (Figure 2b). Pupaadult intermediates retained most of the pupal characters but showed presence of one pair of wings and three pairs of jointed legs in the cephalothoracic region (Figure 3). Formation of larvapupa intermediate was dose related. The results were statistically significant (P< 0.007) (Table 3). Pupa-adult intermediate was observed in the experimental set ups of treatment with 100ppm. GC-MS analysis of ethanol extract of O. sanctum revealed the presence of 51 compounds (Figure 4). Some of the compounds like eugenol, caryophyllene, alpha.-farnesene, farnesyl acetone and geranylgerniol have been described for their insecticidal, larvicidal, mosquitocidal, insect deterrent, and insect repellant activity (Table 4) [30-37].
| Concentration (ppm) | Percent larva pupa intermediate* (Mean ± S.E.) | F value | P-value |
|---|---|---|---|
| 50 | 0.0a ± 0.0 | 6.625 | 0.007 |
| 100 | 4.0bc ± 2.31 | ||
| 200 | 6.67c ± 1.33 | ||
| 400 | 0.0a ± 0.0 |
Table 3: Influence of ethanol extract of Ocimum sanctum on Larvae-pupa intermediate formation in the fourth instar larvae of Aedes aegypti.
| S. No | Name of the Compound | Molecular Formula | Compound Nature | Activity | References |
|---|---|---|---|---|---|
| 1 | Eugenol | C10H12O2 | - | Insecticide, larvicide, juvabional | [30]. |
| 2 | Caryophyllene | C15H24 | Sesquiterprnes | Antimicrobial, antioxidant, anti-tumor, antibacterial, anti-inflammatory, fungicide, insecticidal, antifeedant, insectifuge, larvicide, mosquitocide | [30-32] |
| 3 | Alpha-farnesene | C15H24 | Terpenoid | Insect deterrent, high mortality | [33] |
| 4 | Farnesyl acetone | C18H30O | - | Insect repellant | [34] |
| 5 | Geranyl geraniol | C20H34O | - | Bactericidal and precursors of Mevalonic acid pathway. | [31,35-37] |
Table 4: GC-MS analysis of ethanol extract of Ocimum sanctum.
Discussion
Mosquitoes affect the external and internal environment of human being by creating a biting nuisance that deter outdoor activities and by transmitting pathogens of deadly disease. Although, many mosquito species are not important in pathogen transmission, they take blood meal from a vertebrate host in order to complete the gonotropic cycle of ovaries and development of eggs [38]. Mosquito borne diseases can be controlled by the preventing mosquito bite to human beings; this can be achieved mainly by controlling the mosquito population. Since five decades synthetic chemicals have been used as a mean to control the mosquito population. Synthetic chemicals had larvicidal, adulticidal, ovicidal, oviposition-deterrent and repellent activities. Extensive use of synthetic organic chemicals resulted in environmental hazards and the development of resistance in mosquito species. This has necessitated the need for search and development of environmentally safe and low cost methods for mosquito control. Phytochemicals alter the different life stages of mosquito hence may be used as a potential weapon against the mosquito population.
Present study evaluated the potential role of ethanol extract of O. sanctum in the management of mosquito population by altering the growth and development of the fourth instar larvae. The study revealed that O. sanctum ethanol leaf extract had moderate larvicidal activity. These results are in agreement with that of [39] who reported the larvicidal activity in acetone, chloroform, ethyl acetate, hexane and methanol leaf and flower extract of Ocimum against A. aegypti and Culex quinquefasciatus. He reported moderate larvicidal activity in leaf extract of Ocimum [40] studied larvicidal effects of methanol extract of O. canum and acetone extract of O. sanctum against the larvae of A. aegypti (LC50=99.42 and 81.56 ppm) and against C. quinquefasciatus (LC50=44.54 and 38.30 ppm). Kelm and Nair [41] investigated that hexane extract of O. sanctum contained compounds like eugenol which was responsible for mortality in the fourth instar larvae of A. aegypti.
Sub-lethal effects of ethanol extract of O. sanctum were assessed by considering day wise mortality, larval duration and growth and development impairment. Although the extract could not inhibit the development completely, it mislead or the deranged the physiological cues which were required for the development of a normal adult. The ethanol extract of O. sanctum significantly prolonged the larval duration of A. aegypti.fourth instar larva. Nathan, et al. [42] reported similar activity in the leaf and seed extract of Melia azedarach against Anopheles stephensi. The inability of the fourth instar larva to molt in the pupa and formation of larva-pupa intermediate and moulting of pupa into pupa-adult intermediate indicated the presence of some JH mimic or JH analogue in the ethanol extract of O. sanctum. Parallel results of ethanol extract of O. sanctum leaf ethanol extracts were reported by Summarwar and Pandey [21] against the larvae of Spodoptera litura. Several developmental anomalies were reported in many insects as a sequel to plant extract treatment by many researchers. Kodandaram [43] reported the dose dependent effects of anonin, imidacloprid, karanjin, achook, and ecomeem against D. koenigii. Umamageswari [44] observed ecdysis inhibitor activity in the extracts of Acorus calamus, Ocimum sanctum, Neem, Parthenium and Eucalyptus against Dysdercus cingulatus.
GC-MS analysis of the Ocimum leaf ethanol extract revealed 51 peaks. Some of the important chemical compounds observed in the extract were eugenol, caryophyllene, geranylgerniol, hexadecanoic acid, etstigmasterol, alpha-farnesene, farnesyl acetone. Devendran and Balasubramanian [45] reported the presence of eugenol (43.88%), caryophyllene (26.53%), cyclopentane, cyclopropylidene-(1.02%), cyclohexane, 1,2,4-triethenyl (15.31%), octadecane, 1,1-dimethoxy-(2.04%) and benzene methanamine, N,N,a,4-tetramethyl-(2.04%) in hydroalcoholic extracts of O. sanctum. Methanol extract of O. sanctum showed the presence of three phytocomponents viz methyl-isoeugenol, caryophyllene and eugenol [46] reported the presence of terpenes, phenols, caryophyllene, eugenol, farnesene, geraniol, pthalic acid, terpieol and germacrene D in GC-MS analysis of essential oil of O. sanctum. Our study indicated that phytoconstituents present in the ethanol extract have chemical compounds responsible for insecticidal, growth and developmental disruptors against A. aegypti. eugenol has been reported to possess juvabional activity in a variety of insects [44]. Geranylgeraniol present in the extract acted as intermediate of mevalonic acid pathway of juvenile hormone biosynthesis [31]. These compounds individually or synergistically acted as a JH mimic or analogue and effect the growth and development.
Present investigation explored the prospective role of ethanol extract of O. sanctum as larvicide and growth disruptor against A. aegypti. The investigations suggested potential sublethal effects of the extract. The growth and development of A. aegypti population in natural environment can be impaired by using the extract. In this way the mosquito population can be hampered specially during early stages. The results however may vary depending on the weather conditions and environmental intricacies.
References
- Jansen CC, Beebe NW (2010) The dengue vector Aedes aegypti: what comes next. Microbes Infect 12: 272-279.
- Rosen L, Shroyer DA, Tesh RB, Freier JE, Lien JC (1983) Transovarial transmission of dengue viruses by mosquitoes: Aedes albopictus and Aedes aegypti. Am J Trop Med Hyg 32: 1108-1119.
- Aitken THG, Tesh RB, Beaty BJ, Rosen L (1979) Transovarial transmission of yellow fever virus by mosquitoes (Aedes aegypti). Am J Trop Med Hyg 28: 119-121.
- Paupy C, Ollomo B, Kamgang B, Moutailler S, Rousset D, et al. (2010) Comparative role of Aedes albopictus and Aedes aegypti in the emergence of dengue and chikungunya in central Africa. Vector Borne Zoonotic Dis 10: 259-266.
- Burt FJ, Rolph MS, Rulli NE, Mahalingam S, Heise MT (2012) Chikungunya: a re- emerging virus. Lancet 662-671.
- Thangamani S, Huang J, Hart CE, Guzman H, Tesh RB (2016) Vertical transmission of Zika virus in Aedes aegypti mosquitoes. Am J Trop Med Hyg 95: 1169-1173.
- Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, et al. (2013) The global distribution and burden of dengue. Nature 496: 504-507.
- World Health Organization (2005) Guidelines for Laboratory and Field Testing of Mosquito Larvicides. World Health Organization, Geneva, Switzerland.
- World Health Organization (2017) Monitoring Health for SDGs. World Health Organization, Geneva, Switzerland.
- National Vector Borne Disease Control Programme (NVBDCP) (2017) Dengue Cases and Deaths in the Country since 2010. Ministry of Health and Family Welfare, India.
- Brady OJ, Gething PW, Bhatt S, Messina JP, Brownstein JS, et al. (2012) Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl Trop Dis 6: 1760.
- Govindarajan M, Sivakumar R, Rajeswari M, Yogalakshmi K (2012) Chemical composition and larvicidal activity of essential oil from Mentha spicata (Linn.) against three mosquito species. Parasitol Res 110(5): 2023-2032.
- Gayathri V, Murthy PB (2006) Reduced susceptibility to deltamethrin and kdrmutation in Anopheles stephensi Liston, a malaria vector in India. J. Am. Mosq Control Assoc 22: 678-688.
- Rose RI (2001) Pesticides and public health: integrated methods of mosquito management. Emerg Infect Dis 7: 17.
- Cavalcanti ESB, Morais SM, Lima MAA, Santana EWP (2004) Larvicidal activity of essential oils from Brazilian plants against Aedes aegypti L. Mem Ins Oswaldo Cruz 99: 541-544.
- Isman MB (2000) Plant essential oils for pest and disease management. Crop Proc 19: 603-608
- Sharma P, Mohan L, Srivastava CN (2006) Phytoextract-induced developmental deformities in malaria vector. Bioresour Tech 97: 1599-1604.
- Makri O, Kintzios S (2008) Ocimum sp. (basil): Botany, cultivation, pharmaceutical properties, and biotechnology. J Herb Spi Medicinal Plants 13: 123-150.
- Murugan K, Murugan, P, Noortheen A (2007) Larvicidal and repellent potential of Albizzia amara Boivin and Ocimum basilicum Linn against dengue vector, Aedes aegypti (Insecta: Diptera: Culicidae). Bioresource Technol 98:32 198-201.
- Bagavan A, Rahuman AA, Kamaraj C, Geetha K (2008) Larvicidal activity of saponin from Achyranthes aspera against Aedes aegypti and Culex quinquefasciatus (Diptera: Culicidae). Parasitol Res 103: 223-229.
- Summarwar S, Pandey J (2015) Antifeedant activity of leaf extracts of Catharanthus roseus and Ocimum sanctum against fourth instar larvae of Spodoptera litura. Int J Pure App Zool 3: 259-262.
- Erlera F, Ulugb I, Yalcinkayac B (2006) Repellent activity of five essential oils against Culex pipiens. Fitoterapia 77: 491-494.
- Deshmukh PB, Renapurkar DM (1987) Insect growth regulatory activity of some indigenous plant extracts. Int J Tropic Insect Sci 8: 81-83.
- Mariajancyrani J, Chandramohan G, Brindha P, Saravanan P (2014) GC-MS analysis of terpenes from hexane extract of Lantana camara leaves. IJAPBC 3: 37-41.
- Kumar S, Warikoo R, Wahab N (2010) Larvicidal potential of ethanolic extracts of dried fruits of three species of peppercorns against different instars of an Indian strain of dengue fever mosquito, Aedes aegypti L. (Diptera: Culicidae). Parasitol Res 107: 901-907.
- Abbott WB (1925) A method for computing the effectiveness of an insecticide. J Econ Entomol 8: 265-267.
- Anshul N, Kalra A, Singh D (2014) Biological effect of sweet wormwood, Artemisia annua methanol extracts and essential oil against Helicoverpa armigera Hub. (Lepidoptera: Noctuidae). J Entomol Zoo Stud 2: 304-307.
- Bagavathi PE, Ramasamy N (2012) GC-MS analysis of phytocomponents in the ethanol extract of polygonum chinense L. Pharma Res 4: 11
- Kumar MS, Maneemegalai S (2008) Evaluation of larvicidal effect of Lantana camara Linn against mosquito species Aedes aegypti and Culex quinquefasciatus. Adv Biol Res 2: 39-43.
- Duke S (2015) Phytochemical and Ethnobotanical Databases. USA.
- Sharifi-Rad SJ, Hoseini-Alfatemi SM, Sharifi-Rad M, Sharifi-Rad M, Iriti M, et al. (2015) Phytochemical compositions and biological activities of essential oil from Xanthium strumarium L. Molecules 20: 7034-7047.
- Raman BV, Samuel LA, Pardha SM, Rao BN, Krishna ANV, et al. (2012) Antibacterial, antioxidant activity and GC-MS analysis of Eupatorium odoratum. Asian J Pharma Clin Res 5.
- Dancewicz K, Gliszczyńska A, Halarewicz A, Wawrzeńczyk C, Gabryś B (2010) Effect of farnesol and its synthetic derivatives on the settling behaviour of the peach potato aphid Myzus persicae (Sulz.). Pestycydy/Pesticides 1: 51-57.
- Innocent E, Joseph CC, Gikonyo NK, Nkunya MH, Hassanali A (2010) Constituents of the essential oil of Suregada zanzibariensis leaves are repellent to the mosquito Anopheles gambiae. J Insect Sci 10: 1-8.
- Perez CR, Nyati P, Noriega FG (2015) A corpora allata farnesyl diphosphate synthase in mosquitoes displaying a metal ion dependent substrate specificity. Ins Biochem Mol Biol 64: 44-50.
- Huang J, Marchal E, Hult EF, Tobe SS (2015) Characterization of the Juvenile Hormone Pathway in the Viviparous Cockroach, Diploptera punctata. PLoS One 10.
- Mayorala JG, Nouzovaa M, Navareb A, Noriegaa FG (2009) NADP-dependent farnesol dehydrogenase, a corpora allata enzyme involved in juvenile hormone synthesis. PNAS 106: 21091-21096.
- Knight RL, Walton WE, O‟Meara GF, Reisen WK, Wass R (2003) Strategies for effective mosquito control in constructed treatment wetlands. Ecological Engineering 21: 211-232.
- Anees AM (2008) Larvicidal activity of Ocimum sanctum Linn.(Labiatae) against Aedes aegypti (L.) and Culex quinquefasciatus (Say). Parasitol Res 103: 1451-1453.
- Kamaraj C, Rahuman AA, Bagavan A (2008) Antifeedant and larvicidal effects of plant extracts against Spodoptera litura (F.), Aedes aegypti L. and Culex quinquefasciatus Say. Parasitol Res 103: 325-331.
- Kelm MA, Nair MG (1998) Mosquitocidal compounds and a triglyceride, 1, 3- dilinoleneoyl-2-palmitin, from Ocimum sanctum. J Agri Food Chem 46: 3092-3094.
- Nathan SS, Savitha G, George DK, Narmadha A, Suganya L, et al. (2006) Efficacy of Melia azedarach L. extract on the malarial vector Anopheles stephensi Liston (Diptera: Culicidae). Bioresource Technol 97: 1316-1323.
- Kodandaram MH, Thakur NSA, Shylesha AN (2008) Toxicity and morphogenetic effects of different botanicals on red cotton bug Dysdercus koenigii Fab. (Hemiptera: Pyrrhocoridae) in North Eastern Hill (NEH) region of India. J Biopest 1: 187-189.
- Umamageswari P, Rajavel DS, Rani BU (2006) Studies on ecdysis inhibitory activity of certain botanicals on red cotton bug, Dysdercus cingulatus (Fab.). Hexapoda 13: 58-61.
- Devendran G, Balasubramanian U (2011) Qualitative phytochemical screening and GC- MS analysis of Ocimum sanctum L. leaves. Asian J Plant Sci Res 1.
- Khan A, Ahmad A, Akhtar F, Yousuf F, Xess I, et al. (2010) Ocimum sanctum essential oil and its active principles exert their antifungal activity by disrupting ergosterol biosynthesis and membrane integrity. Res Microbiol 161: 816-823.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi