Research Article, J Biochem Physiol Vol: 8 Issue: 1
The Role of Euphol, a Tetracyclic Triterpene Possible Potential as Anti-Cancer with Computational Evidence (In-silico Insight)
Muhammad Muzammil Nazir1, Shabana Naz1 , Saima Muzammil2, Iqra Farzeen1 and Shahla Faisal3 and Asma Ashraf1*
1Department of Zoology, Government College University, Faisalabad, Pakistan
2Department of Microbiology, Government College University, Faisalabad, Pakistan
3Department of statistics, Government College University, Faisalabad, Pakistan
*Corresponding Author: Asma Ashraf,
Department of Zoology, Government College University, Faisalabad, Pakistan
E-mail: asmabinm@gmail.com
Received date: 30 August, 2024, Manuscript No. JBPY-24-146857;
Editor Assigned date: 02 September, 2024, PreQC No. JBPY-24-146857 (PQ);
Reviewed date: 17 September, 2024, QC No. JBPY-24-146857
Revised date: 12 February, 2025, Manuscript No. JBPY-24-146857 (R);
Published date: 20 February, 2025, DOI: 10.4172/jbpy.1000184.
Citation: Nazir MM, Naz S, Muzammil S, Farzeen I, Faisal S, et al. (2025) The Role of Euphol, a Tetracyclic Triterpene Possible Potential as Anti-Cancer with Computational Evidence (In-silico Insight). J Biochem Physiol 8:1.
Abstract
Cancer-related death and morbidity are largely attributable to its widespread proliferation and invasiveness, which disrupts the normal functions of affected organs. Natural therapies such as plants have been used to treat cancer. Cancer can develop resistance to anticancer drugs over time, leading to treatment failure and tumor recurrence. A tetracyclic triterpene alcohol, euphol from Euphorbia. Tirucalli sap, contain anti-inflammatory, antiviral, and analgesic properties. This review article mainly accentuated the different anticancer perspectives of euphol like breast cancer, pancreatic cancer, glioma cell line, and gastric cancer. In this review, we described on the possible effect of euphol in cancer prevention in in vitro assays and techniques such as cell viability/proliferation, apoptosis, cellular senescence, invasion and migration, oxidative stress and antioxidant effects, gene and protein expression, and angiogenesis in cancer drug discovery. Furthermore, role of bioactive compound (euphol) against cancer analyzed through network pharmacology (ADME, Networking and Docking). Kegg analysis showed euphol is effective through different cancer pathways. Docking analysis performed by receptor protein 7aei and 7aem that have best fit with the compound: With docking scores of −8.8 and -8.6 respectively. In conclusion, euphol can be characterized as novel compound in drug development. The current review summarizes the most recent investigation of euphol.
Keywords: Euphol, Cancer, Biological effect, Cell lines, Breast cancer, Pancreatic cancer, Glioma cell line, Gastric cancer
Introduction
In the past decade, extensive research has recognized millions of phyto-compounds from herbal medicines that have anti-cancerous activities [1]. The phytochemicals in question represent a great source of cancer cells as they are or after different physio-chemical changes, which can be used effectively in different therapeutic methods [2]. Natural compounds are used to develop new treatments because of their great diversity, which comprehended by the fact that plants are unique and there is a need to isolate and purify the bioactive compounds [3]. The discovery of new cancer drugs is very important because of high prevalence, which is one of the important sources of worldwide deaths. It is widely accepted that microbes, plants, and marine organisms are useful sources for the discovery of anti-cancer drug [4]. Natural products and their derivatives have proven useful for chemotherapy development throughout history, due to their diverse structures, molecular properties, and pharmacological properties [5].
In traditional medicinal system, plants have been used to treat cancer. Alkaloids, flavonoids, terpenes, and phenols are among the classes of natural compounds that have shown efficacy in clinical trials [6]. The early drugs that were isolated from Vinca rosea L. (Apocynaceae) and entered in clinical trials are vincristine, vinblastine, vindesine, and vinorelbine [7]. There are numbers of tumors like leukemia, lung cancer, breast cancer, and Hodgkin’s lymphoma has been treated with vinca alkolides by Food and Drug Administration (FDA) [8]. In 1961 and 1963, Vinblastine and vincristine respectively FDA approved and are currently in clinical use. In 1994, the FDA approved vinesine and vinorelbine as active ingredients, and in 2012 the European Medicines Agency (EMA) approved vinflunine to treat metastatic cancer in the urothelial surface [9]. The World Health Organization has also listed vinblastine, vincristine, and vinorelbine as essential medicines for the treatment of follicular lymphoma, acute non-small cell lung cancer, metastatic breast cancer Kaposi's sarcoma, testicular germ cell tumors, Hodgkin's lymphoma, ovarian germ cell tumors, Ewing's sarcoma, lymphoblastic leukemia, retinoblastoma, and rhabdomyosarcoma [10].
Phyto-compound characteristics
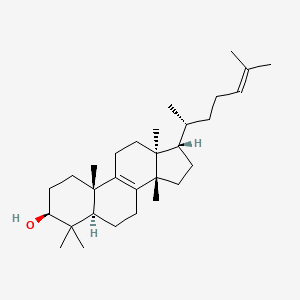
E tirucalli, a species of the genus Euphorbia, is commonly used in folk medicine [11]. The main components, tetracyclic triterpene alcohol of E. tirucalli sap is euphol, contain anti-inflammatory, antiviral, and analgesic properties [12]. Euphorbia tirucalli can be used to treat various cancers as shown in Figure 1. The mechanism of the anticancer effect of euphol has not been determined. In mouse models of acute colitis and arthritis, euphol showed anti-inflammatory effects in the case of human colon cancer cells [13]. Euphol specifically advances apoptosis through mitochondria-subordinate caspase-3 enactment and development capture through p27kip1 acceptance and cyclin B1 hindrance in human CS12 gastric malignant growth cells. It likewise showed cytotoxicity and solid intensity against different gatherings of human disease cell lines like glioblastoma (a typical and aggressive kind of mind tumor). It has also been reported that euphol exhibits anti-nociceptive effects in neuropathic pain models of rats and mice [14]. Also, euphol has been shown to hinder switch transcriptase in human immunodeficiency infection type 1. It has been proposed against malignant growth impacts. In vitro examinations on stomach and bosom cancer cells have detailed that Euphol diminishes cell suitability [15]. There is evidence in an in vivo study of Ehrlich's ascites tumor model, E. Hydroalcoholic Thirukalli Extract (ETHE) that showed best results in animal survival [16]. From these studies, an increased interest in the therapeutic compounds from E. tirucalli, the major is euphol in oncology as shown in Figure 1.
Figure 1: Chemical structure of Euphol.
Materials and Methods
The previous components have been obtained and assembled, and pertinent research and review papers were taken into account. A thorough literature search was carried out utilizing specialized and dedicated search platforms and websites such as Google Scholar, PubMed, ScienceDirect, EBSCO, Sci-Hub, etc. to investigate the cancerous pharmacological actions of euphol. The focus was mostly on the literature including keywords related to anticancer properties.
Compound feature mapping
The physicochemical characteristics of euphol are directly related to its pharmacological activities, and Lipinski's rule of five physicochemical parameters, such as molecular weight, lip hydro partition coefficient, hydrogen donor and acceptor, and rotatable bonds, have been used to assess a compound's potential as a drug. Estimating the pertinent physicochemical features of active substances can reveal information about how much they similar with drugs. Prior to target prediction, the compounds were screened using the Absorption, Distribution, Metabolism, and Excretion (ADME) method [17].
Target fishing and classification
The potential targets of euphol were obtained by the use of the Swiss target prediction online programme, which employed the reverse pharmacophore matching approach to determine targets based on the SMILES number of each ingredient. The next preliminary stage to investigate the molecular mechanism of medicinal herbs to cure various diseases and disorders is the prediction of genes connected to disease.
Three databases such as GeneCards database (https:// www.genecards.org/), the Online Mendelian Inheritance in Man (OMIM) database (http://omim.org/) and DisGeNet (https:// www.disgenet.org/search) were used to find the targeted genes associated with cancer. Bioinformatics and evolutionary genomics tools were used to determine the intersection from the common genes list of euphol targets and targeted genes associated diseases (https:// bioinformatics.psb.ugent.be/webtools/Venn/), Swiss Target Prediction (swisstargetprediction.ch/), PubChem (pubchem.ncbi.nlm.nih.gov/), and Drug Bank [18]. Putative target of euphol were retrieved by Swiss target prediction. Cytoscape 3.10.1 software applied for network construction following the collection and screening.
Euphol targets prediction and network construction
A network diagram of the "active component-biological target" was made after possible protein targets evaluation using Cytoscape 3.10.1 software [19]. Nodes in the network reflect biological targets and active elements. Active components and biological targets were joined via edges.
Construction and intersection of targets between euphol and cancer Protein-Protein Interaction (PPI)
The integrative efficacy of the euphol was examined through Swiss target prediction database. The String database constructed network analysis using the interactions of targets with high scores [20]. The specie selection was limited to “Homo sapiens”. The PPI network discovered in the String database was then visualized and subjected to additional analysis using the Cytoscape software 3.10.1, a publically accessible interface for importing, visualising and investigating and analyzing molecular interaction. The network was analyzed further using the cytoHubba plugin App, and the top 10 genes were chosen based on their degree value. The Database of Annotation, visualization and Integrated Discovery (DAVID, https://david.ncifcrf.gov/) was used to assess the signaling pathways based on the Kyoto Encyclopaedia of Genes and Genomes (KEGG) databases.
Molecular docking
The molecular docking technique was used to validate important targets. The X-ray crystal structures of prospective targets were obtained using the RCSB Protein Data Bank. PDB is a single global repository for data about the three-dimensional structures of nucleic acids and proteins. Additionally, PyRx software was used to execute target docking between core targets and active compounds, and Bio Discovery Studio was used to complete the structure's refinement. For additional examination, the best-docked positions with the lowest binding energy and Root Mean Square Deviation (RMSD) were chosen. One of the main evaluation criteria used to weed out possible constituents and their probable targets was the docking scores between the core target and the compounds.
Results and Discussion
Role in cancer therapy
Cancer is a serious disease resulting from the disruption of the proliferation and homeostasis cell circuit. Decades of intensive research into the molecular biology of cancer have produced knowledge about the pathways that are commonly regulated in cancer. Although it is not far enough, this point has led to the creation of a new treatment method for the treatment of cancer, and the hospitals has confirmed that it is now in clinical trials. Still, despite these advances, cancers pose a major threat to public health and remain an important reason for worldwide death. Cancer-related death and morbidity are largely attributable to its widespread proliferation and invasiveness, which disrupts the normal functions of affected organs. To date, pharmacotherapy has still been considered one of the treatment options for various types of cancers. However, some cancer drugs are associated with serious side effects, influencing the personal satisfaction of disease patients. The possible result for different assays which were performed with euphol is shown in Table 1. In addition, cancer can develop resistance to anticancer drugs over time, leading to treatment failure and tumor recurrence. Overall, there is still a need to develop new and effective cancer prevention mediators.
In Human cancer cell lines, euphol promotes cytotoxicity. The antitumor impact of euphol in vitro was assessed utilizing the MTS test on 73 human disease lines from 15 strong cancer models (colon, bladder, pancreas, prostate, breast, placental choriocarcinoma lung, squamous cell carcinoma, oesophagus, cervix, medulloblastoma, head and neck, glioblastoma, ovarian carcinoma, and melanoma) They developed a complete portion reaction bend and IC50 values for this euphol treatment. Different cell lines of each tumor type display different euphol response patterns.
| Activity | Test | Target | Result |
| Cancer therapy | MTS test | 15 strong cancer models (cell carcinoma, oesophagus, cervix, medulloblastoma, head and neck, glioblastoma, ovarian carcinoma and melanoma) | Anti-tumor impact |
| Pancreatic cancer | Cell viability assay | Panc-1 and Mia-Dad Ca-2) | Inhibition in terms of cell viability |
| Breast cancer | Complement system assay, chemotaxis assay, MTT assay, Cytotoxic assay | Different cell lines | Apoptosis |
| Cell propagation | MTT assay | T47D cells | decrease in cell viability |
| Cell cycle arrest | stream cytometry | T47D | G0/G1 phase arrrest |
| Transcriptional level arrest of cell cycle | qPCR analysis | CDK1, CDK2 | Decrease in CDK1 and CDK2 mRNA |
| Articulation of cell cycle | Western blot | Cyclins A, B1, D1, and E, CDK2, p21, p27, Rb, and p-Rb level balanced | cyclins A, B1, D1 level increases, p21, p27 level decreases |
| Cell cycle protein separation | Western blot | Cyclins A, B1, D1 CDK2, p21, p27, Rb, and p-Rb level | cyclin D1 and p-Rb levels |
| Glioblastoma (GBM) cancer | MTS assay | Twelve glioma cell lines | High cytotoxicity index |
| Glioma cell cancer | CAM assay | GAMG cells | Decrease in tumor perimeter |
| Gastric cancer | WST-1 assay | CS12 and AGS cell | Apopotosis induction |
Table 1: Euphol role in treatment of different types of cancer.
Euphol biological effect on pancreatic cancer
It examined whether cytotoxic or cytostatic effects of euphol on pancreatic malignant growth cell lines, the most delicate kind of cancer. The efficacy of euphol on the propagation of pancreatic cell lines was also evaluated by incorporating BrdU to determine its cytotoxic or cytostatic effects. The expansion of pancreatic disease cell lines (Panc-1 and Mia-Dad Ca-2) was dose-dependent but varied between cell lines. In two cancer cell lines, small doses of euphol reduced proliferation, showing a dose-dependent effect on cell viability. At 17.51 μM, it could almost prevent 50% of the expansion, however, the most grounded hindrance was seen at 35.1 μM following 72 hours with 39.4% for Mia-Dad Ca-2 cells and 51% for Panc-1 cells. Moreover, this same concentration resulted in 22.4% inhibitory action against Panc-1 cells and 10.7% for Mia-Pa-Ca-2 cells in terms of cell viability. Thus, comparing its effect on both cell life and concentration, euphol appears to be more cytotoxic when it comes to growth inhibition.
Biological effect on breast cancer
In the world, there are more than 1,300,000 cases of breast cancer and 450,000 deaths each year as a result of this disease. The number of breast cancer deaths is decreasing, but breast cancer stays the subsequent driving reason for malignant growth passing in women.
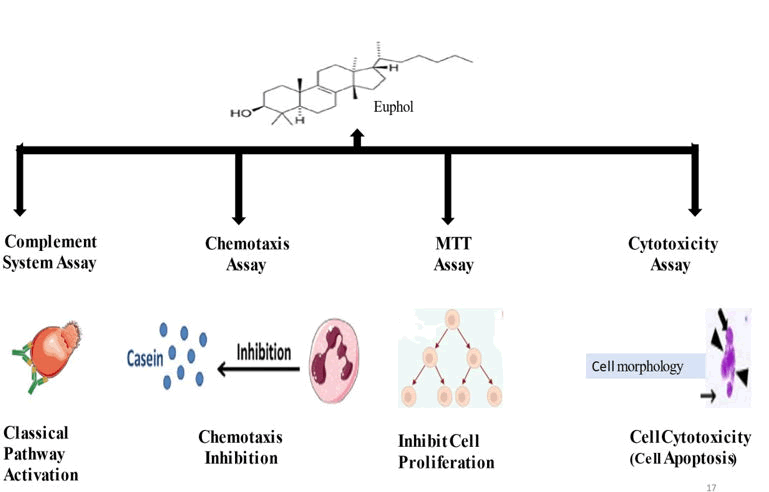
The primary tumor is resected surgically, but the recurrence can occur within months to 40 years of the beginning of side effects, and 15% of patients ultimately foster some type of disease. There is no cure for this disease. This makes it necessary for treatments that can cure the disease. Different treatments were shown in Figure 2.
Figure 2: Different anticancer assays result with euphol.
Conventional cytotoxic treatment kills neoplastic cells through various instruments, but it also affects normal healthy cells. There is no description of these agents causing negative effects, such as shortterm (myelosuppression) or long-term (cardiomyopathy and acute leukaemia) toxicities. Identifying new targeted therapies and developing targeted therapies, also known as targeted therapies, has resulted in a better understanding of the genetic mechanisms involved in cancer. For cancer, phenotype molecules are involved in molecular events that are highly specific to targeted therapies. A major part of human breast cancer and other types of cancer are increasing, and impaired cell cycle function leads enhance cell growth.
Cell propagation inhibition: The effects of euphol on cell proliferation in breast cancer, MTT assays were carried out on T47D cells, a particularly aggressive triple-negative type. The experiments tested the impact of 0.01, 0.03, 0.1, and 0.3 mM concentrations of the drug for a period of 24, 48, and 72 hours. There was a significant decrease in viable cells when treated with increasing doses of euphol most especially with 0.03 mM which revealed IC50 values to be 0.26, 0.22, and 0.13 mM when treated for 24, 48, and 72 hours respectively. Ultimately indicating that euphol can restrain cancerous cell growth in a demonstrable manner.
Euphol induces cell cycle arrest: The exposure of euphol to T47D cells showed how euphol affects cell growth by regulating the cell cycle. Those cells exposed to euphol were distinguished using PI and surveyed utilizing stream cytometry. After 24 hours of treatment with 0.3 mM euphol, there was stability in the transition of the cell cycle, but a marginal rise in the G0/G1 phase and a dive in the G2/M phase and S phase. Inclination toward euphol (0.3 mM) outcomes in a major increase from 59.63 to 71.41% for the G0/G1 zone, as well as a sharp drop from 26.39 to 18.76% for the G2/M zone. After 72 hours of 0.3 mM euphol exposure, there was a huge expansion in the G0/G1 extent, while the S and G2/M subpopulations fell (Control, 58.91%; 0.3 mM d Euphol, 71.44%; G2/M: control, 31.08%, 0.3 mm euphol, 21 .90%; S: Control, 6.81%. 0.3 mM euphol, 2.57%;). No changes were observed in sub-G1 levels at different concentrations and time interval (24h: 3.21%, 48h: 4.68%, 72h: 3.24%). It seems that euphol hinders cell proliferation by arresting cells in the G0/G1 phase before entrance to S phase.
Euphol directs transcription levels of cell cycle-related gens: After treatment with euphol, genes related to cell cycle at mRNA levels were observed to further validate euphol role in stimulating genes involved in cell cycle progression. The parameters selected for investigation were cyclin A2, B1, D1, and E, cyclin subordinate kinase 1 (CDK1), CDK2, cell division cycle 25, and p21 and p27 (CKI). According to the qPCR analysis, exposure to euphol (0.3 mM) for 24 hours, 48 hours, and 72 hours significantly reduced cyclin A2 and B1 mRNA levels. As a result of euphol (0.03 mM) treatment for 48 hours, CDK1 and CDK2 mRNA levels decreased, while p21 mRNA levels increased significantly.
Euphol controls the articulation of cell cycle-related proteins: The effect of euphol on cell cycle reprogramming proteins during cell cycle arrest was reported. It was reported that after 48-72 hours of euphol treatment, the western blots results revealed the levels of cyclins A, B1, D1, and E, CDK2, p21, p27, Rb, and p-Rb. The exposure of euphol (0.03 mM) for 48 -72 hours, caused a decrease in cyclin A, B1, and D expression, but increased p21 and p27 expression. CDK2 expression was diminished following 48 hours treatments. Moreover, p- Rb levels were significantly diminished in response to euphol (0.03 mM). However, there was no difference in the Rb expression within control and euphol-treated cells. These findings imply that the replenishment of some cyclins' translation, along with the alteration of p21 and p27 were enhanced by euphol, and this could inhibit the cell cycle-related protein and eventually stunt growth. Euphol regulates proteins connected with the cell cycle.
To distinguish the proteins engaged with euphol-incited cell cycle arrest: During cell cycle distribution, euphol affected the levels of proteins involved. The result of western blot analysis on whole cell lysates after euphol treatment for 48-72 hours to determine cyclins A, B1, D1 and E expression levels, as well as CDK2, p21, p27, Rb, and p- Rb levels. Within 48-72 hours of euphol (0.03 mM) treatment caused a reduction in the expression level of cyclin A, B1, and D; while it amplified p21 and p27 expression. Treatment of T47D cells with euphol (0.03 mM) for 48 hours reduced CDK2 expression and the level of p-Rb compared to control cells, without changing Rb expression. It indicates that the upregulation of certain cyclins, together with changes to p21 and p27 expression caused by euphol, could result in the arresting of the cell cycle and cessation of growth. Indeed, euphol produced an anti-proliferative effect causing G0/G1 arrest. When analyzing cell cycle distribution and variations in cell cycle regulatory molecules, a lower concentration of cyclin D1 might explain the G0/ G1 arrest induced by euphol in addition to the hang-up of CDK2. While cyclin E does not appear to be affected, the decrease in CDK2 likely indicates a diminished activity of both cyclin E-CDK2. This is reflected by the reduction in cyclin D1 and p-Rb levels that eupol caused, which may be attributed to its CKI-mediated enhancement. It appears that these decreases are all related to the influence of eupol on CKI.
For DNA synthesis in mammalian cells, cyclin A is required and it limits the rate of the G1/S transition. Cyclin B is a mitotic cyclin that collects at the G2/M progress and cooperates with cyclin A during early mitosis. Related to lower cell expansion in S and G2/M stages, cyclin A and B1 mRNA and protein levels were diminished by euphol treatment. The decreased expression in cyclin A and B is related to a decline in cells in S and G2, and an expansion in cells in the G1 stage, in non-little cell tumors. The zenith of G1 seen by FACS assessment is a direct result of the shortfall of DNA in the apoptotic cells. The trait of sub-G1 can be utilized as a marker of apoptotic cells in cell cycle examination, albeit the shortfall of changes in the G1 stage can likewise be because of the deficiency of harmed cells during washing management. In that review, the sub-G1 subpopulation stayed at a low level, showing practically no apoptosis in T47D cells treated with euphol. This is reliable with the exploration that showed that T47D cells among 8 human bosom cancer cell lines from the Public Malignant Growth Foundation Malignant Growth Medication Screening are impervious to apoptosis and that there is no apoptosis in 7-hydrxystaurosporine (UCN-01) and less apoptosis is. It is related to camptothecin. This protection from apoptosis is seen as the MDM-2/p53 assurance is reliant upon p53. The less euphol apoptosis recognized in T47D cells can be made sense of by this system. Moreover, the degree to which MDM2/p53 adds to protection from apoptosis requires further explanation.
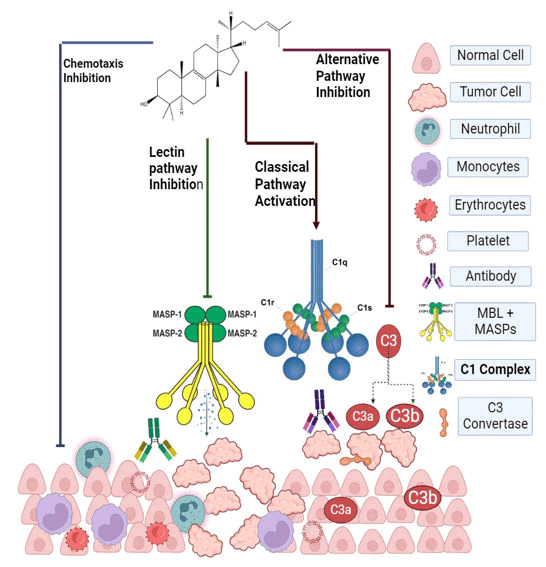
Euphol has biological effect in cancer therapy by different pathways such as chemotaxis inhibition, Lectin pathway inhibition, classical pathway and alternative pathway inhibition. In chemotaxis inhibition, cytokines and immunomodulatory cells do not migrate towards tumor cells. Hence, migration of neutrophils, monocytes and blood cells inhibited. By ELISA test, it was performed that Lectin pathway inhibited by euphol following C4 33. Classical pathway activation is an important application in cancer in hemolytic assay, euphol cause inhibition of alternative pathway. Inhibition of alternative pathway is an efficient strategy for cancer therapy. It inhibits conversion C3 molecules into C3A and C3b by C3 Convertase. Main biological activities by euphol are summarized in Figure 3.
Figure 3: Possible biological effect of Euphol on cancer therapy.
Biological effect on Glioblastoma (GBM) cancer
Evaluation of the anticancer outcome of euphol was conducted with an MTS assay on twelve glioma cell lines derived from viable cultures of adult and child patients, as well as an immortalized astrocyte cell line. This active extract induced varied response profiles in these cells, each with its own IC50 value ranging from 5.98-31.05 μM, with a mean of 19.38 μM. To identify effective treatment options for GBM, new agents urgently need to be explored.
Pediatric glioma cell lines displayed the most sensitive response towards euphol, with a mean IC50 of 13.6 μM, associated with the main culture and mature glioma cell lines with an IC50 of 15.3 and 24.1 μM respectively. For more precise results, the authors adopted a 15 μM dose-limiting (GI) criterion since it was in close alignment with the IC50 values of all cell lines in the initial test. The outcome showed that 7 out of 14 cell lines were resistant, 4 were sensitive and 3 derived differently. Furthermore, euphol has a higher selective cytotoxicity index (0.64 to 3.36) than TMZ (0.11 to 1.13). When tested together, euphol and TMZ demonstrated synergistic effects (combination index CI and lt; 1) for 8 out of 12 commercial glioma cell lines studied at an average CI range of 0.48 to 0.96.
Euphol biological effect on glioma cell lines
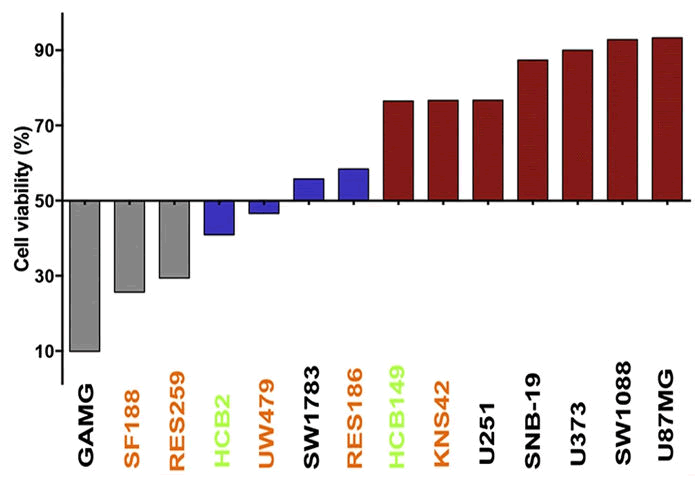
The report of Silva et al. explored the cytotoxic or cytostatic effect of euphol on glioma cells. Cell lines are exposed to different doses of euphol for 72 hours, and the level of proliferation was measured by BrdU incorporation. They observed a dose-dependent increase in its cytotoxicity effects. For instance, at 15 μM, euphol restrained 35.44% expansion in GAMG cells and 28.71% in U373 cells. The strongest inhibition resulted in an 88.86% reduction in cell viability for GAMG cells and 13.9% for U373 cells. These data demonstrate that euphol has a noteworthy cytotoxic effect on the harbor subordinate multiplication of hateful glioma lines. Furthermore, its effect on cell migration was investigated; 8 μM-IC50 value showed no significant influence in GAMG cells, yet 30 μM-IC50 value significantly reduced U373 cell migration at 48 and 72 hours when compared to untreated samples. Interestingly, GAMG cells required an extra 72 hours for the wound to close completely within the untreated group, proposing lower migratory capacity which may explain why euphol affects GAMG cells more than in U373 (Figure 4).
Figure 4: Cytotoxicity profile of 14 human glioma cell lines, exposed to euphol compound. Bars represent the cell viability at 15 μM of euphol. Colors represent the GI score classification and cancer cell lines subgroups. Gray (HS=Highly Sensitive); Blue (MS=Moderate Sensitive); Red (R=Resistant); Black (Adult glioma cell line); Orange (Pediatric glioma cell line) and Green (glioma primary culture).
Euphol has ability to repress cell invasion and separate growth was also gauged. Not only did it fail to impede cell invasion or diminish the number or size of glioma cells, but also these cells didn't halt proliferation or apoptosis. To better comprehend euphol and its effects on the cell cycle and programmed cell death, they scrutinized alterations in signaling proteins utilizing a proteome network involving 61 proteins pertinent to programmed cell death, the cell cycle, and anxiety. Results from this cell-stress network assay demonstrated that GAMG (drug-responsive) cells exposed to 6 h of euphol caused reductions of many proteins; the sole exception being an upsurge of SOD2. As for resistant U373 cells, their investigation showed a decrease in BCl-2 and NF-κB1 signaling, with little deviation in other proteins including HSP60 and HIF1. Euphol had a major impact on several proteins. In GAMG cells, it enhanced P53 (S15) activation and reduced P53 (S46) activation, while Trail R1/DR5, BCL-X, XIAP, Trail R1/DR4, and BAX expressions were decreased. However, an increase was observed in SOD2, thioredoxin-1, P21 CIP1, HSP60 and HSP70 activity levels. U373-resistant cell protein expression levels also changed due to euphol in particular after 24 hours. The results of this study suggest that euphol alters protein profiles in glioma cell lines differently. Upon treating the cells with euphol (8 and 30 μM, 72 h) FACS analysis showed no significant effect on the G1, G2/M, or S phases of the cell cycle for GAMG and U373.
Annexin V-FITC/PI assays further confirmed that euphol had no impact on apoptosis in the glioma cells studied here. An examination of LC3-II expression indicated that euphol exposure (8 and 30 μM, 2 to 6 hours) provoked a substantial rise in GAMG cells compared to those untreated. Furthermore, when combined with Baf, this increase was also observed in U373 cells. To further research autophagy, the cells were stained using acridine orange, a color that identifies the lysosomotropic changes linked to Acidic Vesicular Organelles (AVO). This led to a remarkable rise in acridine orange fluorescence from 15.5-43.8% when compared to the control, suggesting partial or total acidity among AVOs. Subsequent tests such as Baf representative ATPase inhibitor connected to the synthesis of autophagosomes and lysosomes) were carried out to further examine euphol-induced AVOs.
The researchers noted that euphol either alone or in combination with Baf (10 nM GAMG and 20 nM U373) caused an increase in AVO. Compared to Baf alone, euphol treatment increased AVO formation in U373 cells by 25.9-66.8%. This suggests that euphol may be related to autophagy induction, so they tested the impacts of inhibiting autophagy on euphol cytotoxicity. U373 and GAMG cells were treated with different concentration of euphol for 72 hours, with Baf added 3 hours after the onset of treatment. Euphol at 350 nM caused a reduction in the proliferation of GAMG cells by 100-45%when treated with Baf; whereas CElls in U373 were reduced by 100-30% when treated with 5.85 μM euphol. The cytotoxicity effect of euphol on different cell lines is shown in Figure 3.
The induction of apoptosis by Baf was found to be ineffective in treating euphol cells. Researchers discovered that euphol can activate the PKC signaling pathway in glioma cells, with an anti-inflammatory effect observed in mice skin as a direct result of its inhibition of PKCα. Additionally, they evaluated PKCs (cPKCs: PKCα, p-PKCα/βII, pPKCpanβII; nPKCs: PKCδ, p-PKCδ, pPKCδ/θ, p-PKCθ; atypical- PKCs): PKCζ/λ, PKCζ and PKD1/PKCμ) through immunoblotting. U373 cells displayed decreased activation of these isotypes after being treated with euphol on Ser744 and particularly Ser916 residues. Whereas the drug-resistant U373 cells displayed no change in the situation with generally complete or phosphorylated PKC types upon treatment with euphol.
In vivo euphol Effect by CAM assay: In an in vivo study, tumor growth and angiogenesis impact of euphol was assessed. The CAM study showed that there were no differences between the relative sizes of tumors formed in DMSO (1500.5 ± 265.3 μm) and euphol (1600 ± 50.4 μm) cells in U373. However, GAMG cells treated with euphol cause decrease in mean tumor perimeter from 2000 μm ± 15.1 μm to 1550 ± 30.3 μm compared to control cells (n=20). A significant difference in the number of vessels conformed GAMG growths and euphol-treated cells while contrasting control cancers (40 ± 5) with euphol (29 ± 4), suggesting that this treatment affected angiogenesis. No variations were observed in the U373 cell line.
Euphol biological effect on pancreatic cancer
On pancreatic cancer cell lines, the researchers study the biological effects of euphol. Panc-1 Mia-Pa-Ca-2 cell lines showed a weak migratory behavior, with wound closure taking over 72 hours. After being exposed to 8.46 and 21.47 μM euphol, the Mia-Pa-Ca-2 cells decreased motility as associated with the control cells; however, the effect of euphol on Panc-1 was not detectable due to its pre-existing low mobility phenotype. The results showed that euphol can be cytotoxic but does not affect the relocation of this particular cell line. Besides cell proliferation, migration, and colony formation, the metastatic capability of malignant growth cells can be evaluated in vitro. The impact of euphol on the autonomous development of Panc-1 malignant growth cells was studied. The results showed a significant reduction in the number and size of colonies formed 20 days subsequently of incubation; with the euphol reducing them by up to 90% as compared to the control. In addition, when looking at the Mia- Pa-Ca-2 cell line, no colonies were observed even in controls, recommending a low tumorigenic effect on pancreatic cell line.
Human gastric cancer
Euphol had a repressive effect on the proliferation of CS12 colon cancer cells, as well as CSN, CS12, AGS, and MKN45 cells, at concentrations of 2, 5, 10, 20, 40, and 60 μg/mL). The WST-1 assay revealed that it caused a dose-dependent reduction in growth for both CS12 and AGS cell lines in addition to commercially available MKN45 cells. Annexin V/PI staining showed that upon exposure to euphol (20 μg/mL), the apoptosis rate was greater for colon cancer cells compared to normal ones. Moreover, from the same test results, it could be seen that euphol induced cell death significantly in CS12, AGS, and MKN45 gastric cancer cell lines but not in CSN cells. Finally, IC50 values were determined as 49.6 (for CSN cells), 12.8 (CS12), 14.7 (AGS), and 14.4 (MKN45).
Initiation of ERK1/2 phosphorylation in CS12 cells in Euphol: ERK1/2 MAPK role in euphol-induced cell apoptosis, AGS, MKN45, CS12, and CSN cells using 20 μg/mL euphol observed. Results showed that ERK1/2 enactment was seen in a period subordinate way in CS12 cells. Similarly, there was a substantial increase in phosphorylated ERK1/2 after 72 h of treatment in AGS and MKN45 gastric cancer cells. However, no correlation between ERK1/2 activity and euphol treatment was found in CSN cells under the same conditions. In addition, to check the role of ERK1/2 MAPK pathway in suppression by euphol, CS12 cells were exposed to the inhibitor PD98059. Results revealed that PD98059 had a weak repressive effect on apoptosis induced by euphol in this cell line, indicating that the ERK1/2 MAPK pathway may contribute to CS12 apoptotic cell death mediated by euphol.
Role of ERK1/2 in the euphol-induced mitochondrialdependent programmed cell death pathway: Western blot analysis was employed to assess the role of ERK1/2 in euphol-induced apoptosis of CS12 cells and to a degree Bcl-2 and BAX expression. The results showed that euphol treatment of cells for 72 hours caused a significant upregulation of BAX in a dose-dependent manner, whilst decreasing Bcl-2 protein expression. Interestingly, PD98059 was able to reverse the effects of euphol on BAX levels. Mitochondrial membrane potential can be disrupted by pro-apoptotic BAX translocation, thus inducing caspase-mediated apoptosis. Flow cytometry further revealed that treatment with 20 μg/mL euphol led to a decrease in Mito Tracker fluorescent curves relative to vehicletreated control; however, pre-incubation with 13.4 ug/mL PD98059 resulted in an opposing shift from left to right, indicating dependency on ERK1/2 regulation of the mitochondrial programmed cell death pathway. The role of the possible biological effect of euphol in cancer therapy is shown in Figure 4.
In slico study
Euphol is a potent secondary metabolite. Table 2 shows the in silico studies with ADME predictions of euphol using the Swiss ADME online tool. Physiochemical properties of plant significantly affect metabolic state of body. Table 3 displays that euphol molecular weight fulfill the Lipkin rule criteria. It has less than five hydrogen bond donors, less than ten rotatable bonds, and an octanol-water partition coefficient (ClogP) of less than five and meet the requirements for oral bioavailability.
| Physiochemical properties of euphol | ||||||||||
| Compound | Formula | MW | Fraction Csp3 | RB | HBA | HBD | MR | TPSA | Log p | ESOL |
| Euphol | C30H50O | 426.72 g/mol | 0.87 | 4 | 1 | 1 | 137.04 | 20.23 A² | -7.09 | Poorly soluble |
| Pharmacokinetics properties | ||||||||||
| Compound | GIA | BBB | P-gpS | CYP1A2 inhibitor | Inhibitor CYP2C19 | Inhibitor CYP2C9 | Inhibitor CYP2D6 | Inhibitor CYP3A4 | Log Kp | |
| Euphol | Low | No | No | No | No | No | No | No | -2.58 cm/s | |
| Lipophilicity and drug-likeness | ||||||||||
| Compound | Lipinski | Ghose | Veber | Egen | Muegge | BAS | PAINS | Lead likeness | SA | |
| Euphol | Yes; 1 violation: | No | Yes | Yes | No; 1 violation | 0.55 | 0 alert | No | 6.07 | |
Table 2: Properties of Euphol.
|
Compound |
Pubchem ID |
2D structure |
Radar |
Canonical smiles |
|
Euphol |
441678 |
|
|
CC(=CCCC(C1CCC2(C1(C)CCC1=C2CCC2C1(C)CCC(C2(C)C)O)C)C)C |
Table 3: Characterisitcs of Euphol.
Topological polar surface area is a crucial component linked to medication bioavailability (TPSA). Compounds with a highly oral bioavailable and passively absorbed have a TPSA less than 140 Å2. As shown in Table 4, euphol have polar TPSA values, which range from 52.58 Ų. Low solubility restricts medication absorption in the gastrointestinal system, whereas high solubility facilitates maximal absorption in drug development plans focused on oral delivery. Table 4 indicated euphol is soluble with log p value between -7.09.
Pharmacokinetics properties: The Blood-Brain Barrier (BBB) penetration indicates the medication's relative affinity for either brain tissue or blood. Phenyl ring phosphospholipidosis require two substances that are not substrates of P-gp are needed: phytol and bis(2- ethylhexyl) phthalate. The cytochrome P450 (CYP) superfamily plays a crucial role in drug elimination through metabolic biological transformation). The molecule is less skin permeant if the lesser the log Kp (cm/s) value is. Euphol examined have log Kp readings -2.58 cm/s as displayed in Table 4.
| Rank | PubChem ID | Common name | Score |
| 1 | 9606.ENSP00000356438 | PTGS2 | 30 |
| 2 | 9606.ENSP00000263025 | MAPK3 | 28 |
| 3 | 9606.ENSP00000287936 | HMGCR | 26 |
| 4 | 9606.ENSP00000405330 | ESR1 | 26 |
| 4 | 9606.ENSP00000379683 | CYP19A1 | 21 |
| 6 | 9606.ENSP00000437955 | HIF1A | 21 |
| 6 | 9606.ENSP00000231509 | NR3C1 | 17 |
| 8 | 9606.ENSP00000229795 | MAPK14 | 17 |
| 9 | 9606.ENSP00000310928 | PPARD | 17 |
| 9 | 9606.ENSP00000354859 | DRD2 | 15 |
Table 4: Top 10 in STRING network ranked by degree method.
Lipophilicity and drug-likeness: As it was discovered to be within the range of -0.7 to +5.0, excellent permeability and oral absorption are anticipated. According to Meeran, et al., drug-likeness is a qualitative test that determines a compound's potential as an oral medication candidate based on its bioavailability
Lipinski's rule-of-five: If it fails to meet even one of the following four requirements, Lipinski's rule-of-five indicates that it is unlikely to be developed as a potential oral medication. This indicates the series' potential importance for the development of drug-like molecules.
Medicinal chemistry: Substances known as PAINS have substructures that, regardless of the protein receptor, cause a false response with a physiologically potent output. Table 2 shows that euphol did not restored PAINS recovery. Euphol had high synthesis feasibility because their SA scores were determined to be less than six. The standardized range of the synthetic accessibility (SA) score is 1 (easy synthesis) to 10 (hardly synthesis). Euphol candidates in the library had SA scores of less than four, indicating a high likelihood of synthesis.
To begin lead optimization, a medicinal chemist can specify the necessary chemical by using the lead-like rule-based technique. In Table 2, there is not a single violation related to lead likeness. It demonstrates that every substance is appropriate for biological optimization.
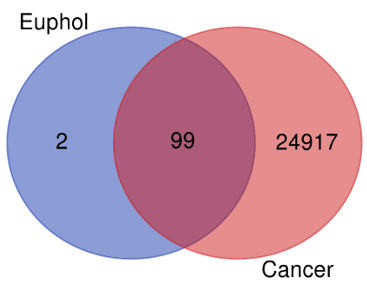
Protein interaction network construction: Euphol target gene were retrieved from swiss target Prediction. Following the discovery of a promising euphol target, target gene affiliated with Cancer were retrieved from OMIM, GeneCards and DisGeNet. The overlap genes between the targeted genes of tested. Euphol target genes and diseaserelated targets were uploaded to the Venn diagram. Thus, for further investigation, the shared targets of active ingredients and illness were identified (Figure 5). A total of 99 overlapping genes were observed.
Figure 5: Venn diagram of the common targets of euphol and cancer.
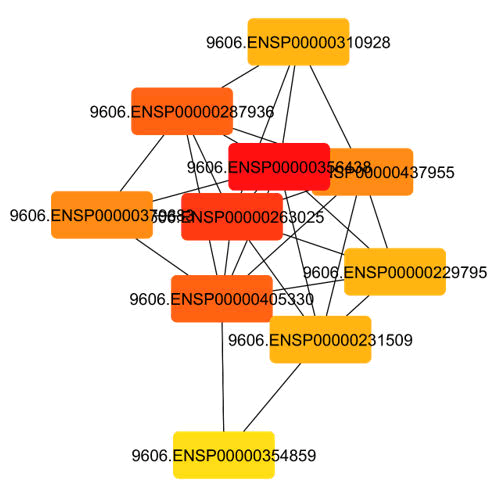
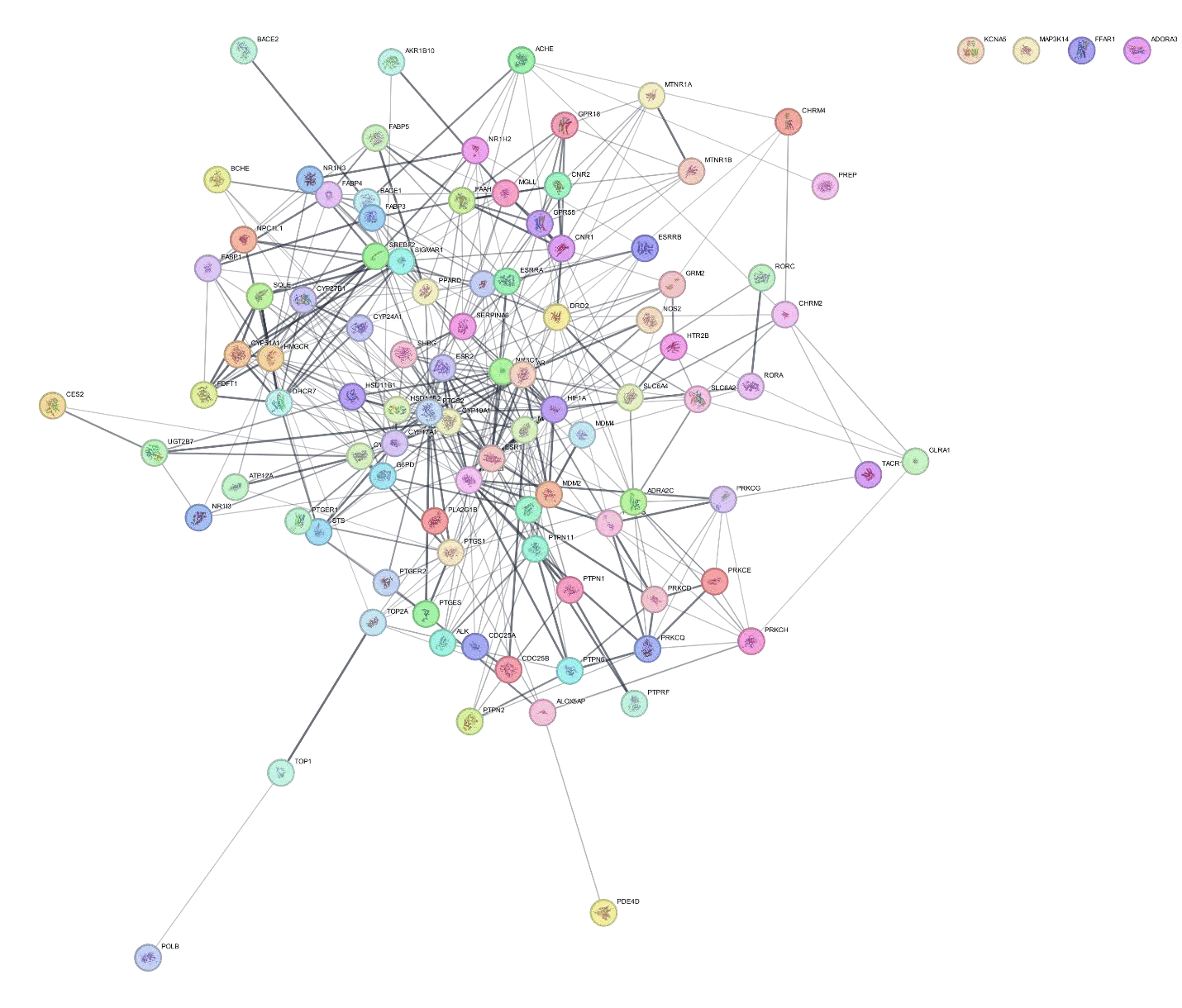
Using the String database, 99 genes that overlapped and the pathway they were related with that had the greatest number of genes were selected to form a PPI network. A PPI network illustrates the interplay between various targets as a disease develops (Figure 7). The nodes and their relationships demonstrate how these compounds cooperate. This suggests that all of the genes with the highest degree are highly correlated with one another and could therefore be hub targets Figure 6.
Figure 6: Top hub genes from cytohubba plugin.
Figure 7: PPI network of overlapping genes.
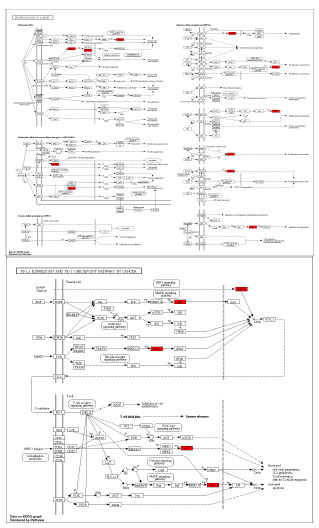
KEGG analysis
KEGG pathway carried out to identify interacting pathways between Euphol and Cancer. Notably, the majority of the genes were engaged in the pathways that follow cancer (Figure 8).
Figure 8: Representation of KEGG pathway analysis.
Molecular docking
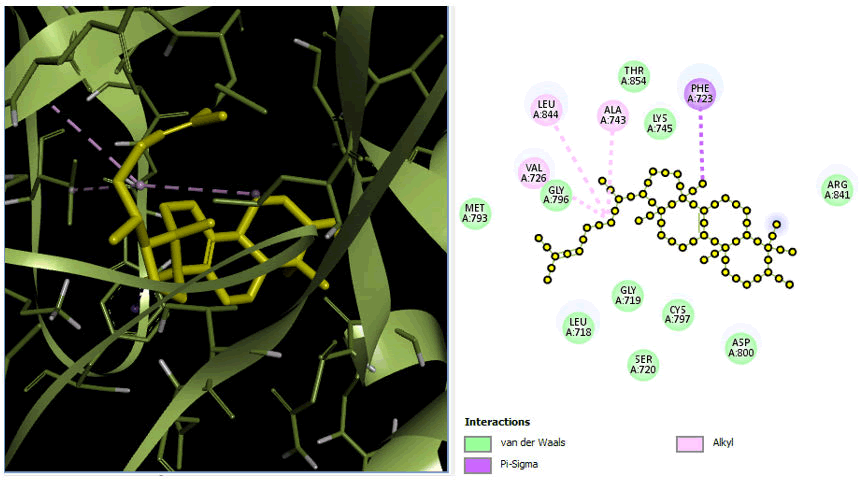
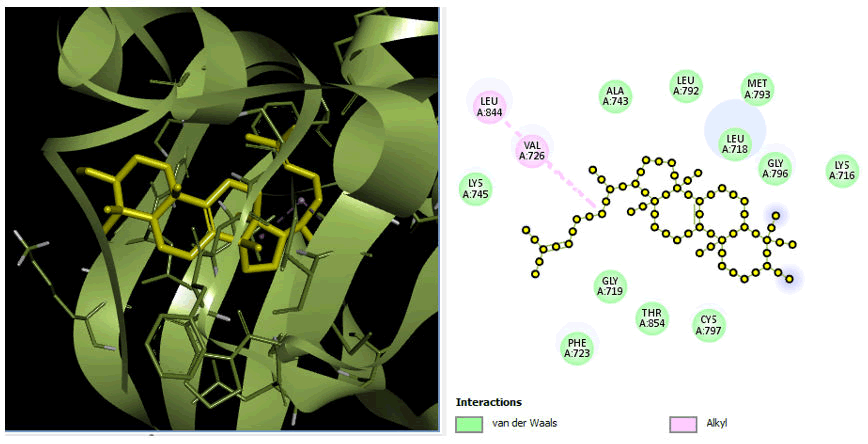
Minimize the energy of ligands via Pyrex docking. The docking analysis investigated the molecular interactions between receptor protein PDB ID: 7aei and 7aem with Euphol to determine the receptors' effectiveness. The coordinate structure of these receptors was retrieved from the PDB and progressively imported into Pyrex to finish docking.
The chemical that best interacts with the receptor was found by the Pyrex software. The best drug was selected on the base of docking score and binding fitness of the ligand that had the highest binding affinity to the receptor molecule. The findings were evaluated based on binding compatibility.
We fulfilled the criteria and performed docking with the receptor protein 7aei and 7aem have best fit with the compound: Euphol having docking scores of -8.8 and -8.6. The discovery studio's best poses, in 2D, with their protein-ligand interactions. The dock scoring prediction shows that the compound has the best ratio of all factors, allowing them to be considered as having an effective chemotherapeutic potential. The discovery studio's greatest poses were shown in 2D with their protein-ligand interactions presented in Figures 9 and 10.
Figure 9: 3D and 2D view of protein 7aei.
Figure 10: D and 2D view of protein 7aem.
Conclusion
For many years, medicinal plants have been significantly studied for their phyto-medicinal properties which bring known medicinal virtues. Phytochemical screening of Euphol reveals it, as a valuable medicine as anti-cancer. Several studies described that Euphol targeted the mitochondria of cells in cancer. Euphol exerts anticancer effects on cancer cell lines and tumors by modulating different pathways. Euphol promotes loss of cell viability, apoptosis, and autophagy. As the literature suggests, euphol has a wide range of therapeutic benefits with no adverse effects. It is predicted that euphol medications would control a variety of malignant conditions without causing any negative side effects. Furthermore, euphol anticancer activity confirmed through network pharmacology (ADME, Networking and Docking). Kegg analysis and molecular docking showed euphol is a promising approach as anticancer drug. Docking analysis performed by receptor protein 7aei and 7aem that have best fit with the compound: A significant number of compound, Euphol shown potential to anticancer activities against the different hallmarks of cancer, such as cell growth inhibition, antimitotic activity (anti-tubulin effects), apoptosis and/or autophagy induction, and migration, invasion, or metastasis inhibition. Although, at this moment there is no clinical evidence that euphol can prevent or treat cancer in humans based studies discussed here. Furthermore, several drugs have a synergetic influence so typical research and evolving work should be carried out for the conservation of euphol for their better therapeutic and commercial utilization.
Funding
This research received no external funding.
Informed Consent Statement
Not applicable
Acknowledgments
Not applicable
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ahmad A (2019a) Breast cancer statistics: Recent trends. Adv Exp Med Biol 152: 1-7.
[Crossref] [Google Scholar] [PubMed]
- Ahmad A (2019b) Breast cancer statistics: recent trends. Breast cancer metastasis and drug resistance: challenges and progress, 1-7.
- Andleeb R, Ijaz MU, Rafique A, Ashraf A, Bano N, et al. (2021) Biological activities of methanolic extract of Aegle marmelos against HN protein of newcastle disease virus. Agronomy 11: 1784.
- Assenov Y, Ramirez F, Schelhorn SE, Lengauer T, Albrecht M (2008) Computing topological parameters of biological networks. Bioinfo 24: 282-284.
[Crossref] [Google Scholar] [PubMed]
- Bailon-Moscoso N, Cevallos-Solorzano G, Carlos Romero-Benavides J, Isabel Ramirez Orellana M (2017) Natural compounds as modulators of cell cycle arrest: Application for anticancer chemotherapies. Curr Genomics 18:106-131.
[Crossref] [Google Scholar] [PubMed]
- Bates D, Eastman A (2017) Microtubule destabilising agents: Far more than just antimitotic anticancer drugs. Br J Clin Pharmacol 83: 255-268.
[Crossref] [Google Scholar] [PubMed]
- Bennouna J, Delord JP, Campone M, Nguyen L (2008) Vinflunine: A new microtubule inhibitor agent. Clin Cancer Res 14: 1625-1632.
[Crossref] [Google Scholar] [PubMed]
- Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, et al. (2000) The protein data bank. Nucleic Acids Res 28: 235-242.
[Crossref] [Google Scholar] [PubMed]
- Bhavana V, Sudharshan SJS, Madhu D (2017a) Natural anticancer compounds and their derivatives in clinical trials. Antican Plan Clinic Trials Nanotech 3: 51-104.
- Bhavana V, Sudharshan SJS, Madhu D (2017b) Natural anticancer compounds and their derivatives in clinical trials. In Anticancer plants: clinical trials and nanotechnology. Springer 51-104.
- Bonofiglio D, Giordano C, De Amicis F, Lanzino M (2016) Natural Products as Promising Antitumoral Agents in Breast Cancer: Mechanisms of Action and Molecular Targets. Mini Rev Med Chem 16: 596-604.
[Crossref] [Google Scholar] [PubMed]
- Daina A, Michielin O, Zoete V (2019) SwissTargetPrediction: Updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res 47: W357-W364.
[Crossref] [Google Scholar] [PubMed]
- Dallakyan S, Olson AJ (2015) Small-molecule library screening by docking with PyRx. Methods Mol Biol 1263: 243-250.
[Crossref] [Google Scholar] [PubMed]
- de Bono JS, Tolcher AW, Rowinsky EK (2003) The future of cytotoxic therapy: Selective cytotoxicity based on biology is the key. Breast Cancer Res 5: 1-6.
[Crossref] [Google Scholar] [PubMed]
- de Oliveira TL, Bavia L, Fontana PD, Cruz LS, Paludo KS, et al. (2021) Immunomodulatory and cytotoxic activities of euphol. Life Sci 280: 119700.
[Crossref] [Google Scholar] [PubMed]
- El-Hawary SS, Lithy NM, Amin E, AbouZid SF, Mohammed R (2021) Anti-trypanosomal activity and DNA fingerprinting of fifteen Euphorbia species using ISSR and SCoT markers. Beni-Suef Univ J Basic Appl Sci 10: 1-8.
- Fernandes LS, Costa YFGd, Bessa MED, Ferreira ALP, Correa JOdA, et al. (2021) Metabolic profiling and antibacterial activity of Eryngium pristis Cham and Schltdl-prospecting for its use in the treatment of bacterial infections. Arch Pharm Pharma Sci 5: 020-028.
- Gerullis H, Wawroschek F, Kohne CH, Ecke TH (2017) Vinflunine in the treatment of advanced urothelial cancer: clinical evidence and experience. Ther Adv Urol 9: 28-35.
[Crossref] [Google Scholar] [PubMed]
- Gu J, Gui Y, Chen L, Yuan G, Lu HZ, et al. (2013) Use of natural products as chemical library for drug discovery and network pharmacology. PloS One 8: e62839.
[Crossref] [Google Scholar] [PubMed]
- Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4: 44-57.
[Crossref] [Google Scholar] [PubMed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi