Case Report, Clin Oncol Case Rep Vol: 5 Issue: 1
Therapeutic Activity of a TIM-3 Blocking Antibody Against Recurrent High Grade Astrocytomas
Dimitrios Mathios1, Maria Adelita Vizcaino Villalobos2, Xiaobu Ye1, Patrick M Forde3, Jon Weingart1, Kaisorn Chaichana4, Matthias Holdhoff3, Drew M Pardoll5, Henry Brem1, and Michael Lim1,6*
1Department of Neurosurgery, Johns Hopkins University School of Medicine, Baltimore, USA
2Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, USA
3Department of Oncology, Johns Hopkins University School of Medicine, Baltimore, USA
4Department of Neurosurgery, Mayo Clinic, Jacksonville, USA
5Bloomberg-Kimmel Institute for Cancer Immunotherapy, Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, Baltimore, USA
6Department of Neurosurgery, Stanford University, Palo Alto, USA
*Corresponding Author:
Michael Lim
Department of Neurosurgery,
Johns Hopkins University School of Medicine,
Baltimore, USA
E-mail: mklim@stanford.edu
Received: November 30, 2021, Manuscript No: COCR-21-47756;
Editor assigned: November 21, 2021, PreQC No: P-47756;
Reviewed: December 18, 2021; QC No: Q-47756;
Revised: December 22, 2021, Manuscript No: R-477556;
Published: January 10, 2022, DOI: 10.4172/cocr.5(1).209
Citation: Mathios D, Villalobos MAV, Ye X, Forde PM, Weingart J, et al. (2022) Therapeutic Activity of a TIM-3 Blocking Antibody against Recurrent High Grade Astrocytomas. Clin Oncol Case Rep 5:1
Abstract
The antitumor activity of TIM-3 blocking antibodies has been investigated in preclinical models of cancer with promising results so far. Here we report in detail the first clinical experience with a human TIM-3 blocking antibody against high grade astrocytomas. Among a cohort of four brain tumor patients studied, three were treated with TIM-3 blocking antibody alone and one was treated with a combination of TIM-3 and PD-1 blocking antibodies. All patients had radiographic progression with progression free survival between 3-6.5 months. Three patients had transient grade 3 neurological toxicity. Three patients required dose reduction of TIM-3 due to symptomatic treatment-related edema. All patients had low percentage of TIM-3 positive immune cells and variable expression of PD-1, PD-L1, LAG-3 in the tumor microenvironment before treatment initiation. One of the four patients developed significant tumor edema on imaging during treatment that decreased significantly with administration of steroids. Histological examination of the tumor obtained before anti-TIM-3 initiation as well as after treatment showed significant tumor immune infiltration, compared to other tumors in this cohort. Overall, while there was no measurable clinical benefit among patients with progressive astrocytomas that were treated with TIM-3 blocking antibody in this pilot cohort, anti-TIM-3 may alter the tumor microenvironment.
Keywords: TIM-3; Immunotherapy; Brain tumors; PD-1
Introduction
High Grade Astrocytomas (HGA) constitutes a group of very aggressive adult brain tumors. Immunotherapy and more specifically immune checkpoint inhibition has shown promising results in other aggressive tumor types [1,2]; however, no significant anti-tumor activity is evident in recurrent glioblastoma [3]. Preclinical work from our group has shown that TIM-3 blockade in combination with PD-1 blockade and radiation could lead to 100% cure of mice bearing glioblastoma [4].
We here present a cohort of patients with HGA enrolled in the NCT02608268 Phase I-Ib/II Open-label Multi-center Study of the Safety and Efficacy of sabatolimab (anti-TIM-3) as Single Agent and in Combination with spartalizumab (anti-PD-1) in Adult Patients with Advanced Malignancies in our institution [5]. A total of four patients with HGA are included.
Materials and Methods
Study patient population
We analyzed the demographic, radiographic, laboratory, pathological, genetic, immunological and survival data of four patients with histologically confirmed HGA treated with anti-TIM-3 alone or with anti-PD-1 antibody on clinical trial NCT02608268 from September 2016 to September 2017 at our institution. The study was conducted under the approval of the Institutional Review Board of Johns Hopkins University, School of Medicine and after written informed consent from each patient. The primary outcome of this trial is safety and tolerability of treatment with these agents, treatment related adverse effects, overall response rate per Immune Related Response Evaluation Criteria in Solid Tumors (irRC). Secondary outcomes were progression free survival and overall survival.
Immunohistochemistry (IHC) in tumor samples and molecular analysis
Paraffin embedded tumor tissue was stained for IBA-1, CD4, CD8, CD20, PD-1, PDL1, TIM-3, LAG-3. See Supplementary Methods for more details. To evaluate the staining pattern for each antibody we performed a semiquantitative analysis and determined the mean percentage of positive cells.
IDH1 mutation status (Wild Type (WT) vs Mutant (MT)) and MGMT (O6-Methylguanine-DNA Methyltransferase) methylation status were determined by standard clinical molecular analyses performed in our institution as part of standard of care.
Case Description
Patient 1
The first patient was a Hispanic man in his late 70s with a history of recurrent left frontal IDH1 WT, MGMT unmethylated glioblastoma. After initial resection, he received standard radiation and chemotherapy (EORTC 26981–22981/NCIC CE3) [6]. Nine months after treatment completion, he underwent re-resection. Postoperatively, the patient enrolled on the NCT02608268 trial and was initiated on anti-TIM-3 therapy. The patient experienced worsening aphasia and right sided weakness after his first dose and his second dose was held. He was administered steroids and his symptoms improved. The patient was given a second reduced dose of anti-TIM-3. On a two month follow up MRI, the nodular enhancement appeared increased but with progressively decreased flair signal changes (Supplementary Figure 1). The patient stopped anti-TIM-3 treatment and was started on bevacizumab. The patient expired 5 months after entering the clinical trial.
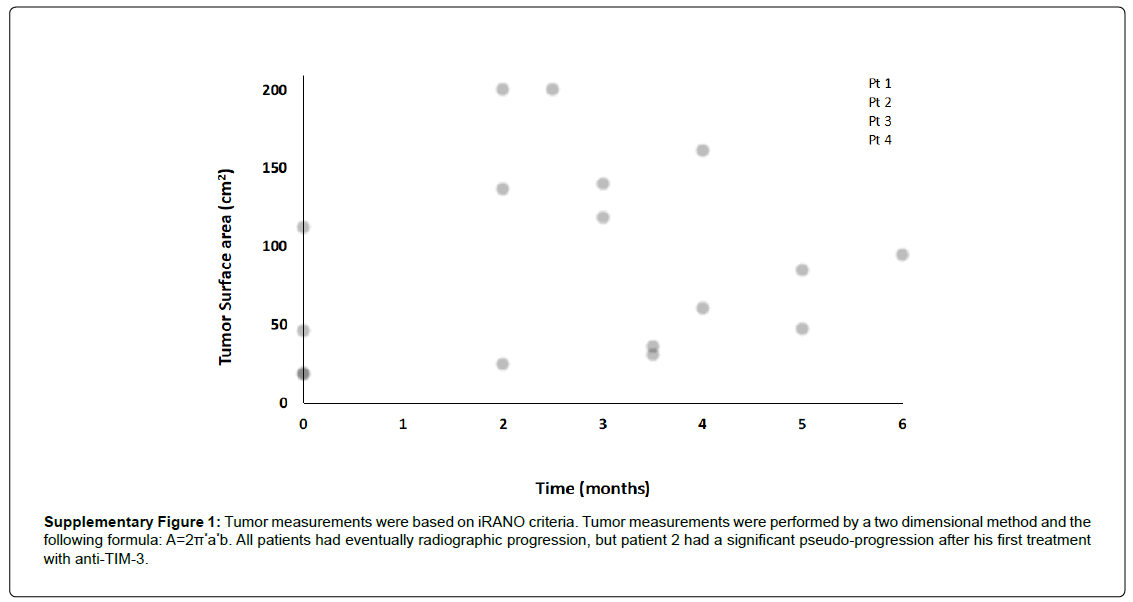
Supplementary Figure 1: Tumor measurements were based on iRANO criteria. Tumor measurements were performed by a two dimensional method and the following formula: A=2Ï?*a*b. All patients had eventually radiographic progression, but patient 2 had a significant pseudo-progression after his first treatment with anti-TIM-3.
Patient 2
The second patient was a Caucasian man at his early 60s with a history of left temporal glioblastoma (MGMT unmethylated, IDH1 WT) after initial resection, radiation and temozolomide and re-resection 9 months later. The patient enrolled in the NCT02608268 trial and was started on anti-TIM-3. The patient received 2 doses of anti-TIM-3 and 90 days after treatment initiation he developed increased somnolence and speech difficulty. The treatment was stopped due to symptomatic and radiographic progression. (Supplementary Figure 1). The patient was started on steroids with dramatic symptom improvement and significant decrease of tumor enhancement and T2 hyperintensity. The patient was restarted on a lower dose of anti-TIM-3, 1 month after the last dose of the drug. Repeat MRI 1 month later showed again interval increase of the nodular enhancement. The patient underwent biopsy of the enhancing lesion that confirmed active tumor. The patient withdrew from this trial and enrolled in another trial in our institution with no tumor response. He was then started on palliative temozolomide treatment that had to be stopped due to thrombocytopenia. The patient eventually expired due to disease progression 11 months after his enrolment on the anti-TIM-3 trial.
Patient 3
The third patient was a Caucasian man in his late 40s with history of a right temporal lobe grade II astrocytoma in 2011 surgically resected, with further progression in 2014 to grade III (IDH1MT, MGMT promoter methylated). He underwent radiation and temozolomide treatment in 2015. Subsequent MRI showed recurrence of his HGA. In 2016 he underwent biopsy of a newly appearing enhancing lesion, confirming the diagnosis of glioblastoma. The patient enrolled in the NCT02608268 trial, he received two doses of anti-TIM-3 and the day after his second infusion he experienced moderate headache and confusion. MRI brain revealed new leptomeningeal enhancement suggestive of venous congestion from cerebral edema. The patient was started on steroids and his symptoms immediately improved. The patient continued with treatment at a lower dose. MRI after the third dose suggested tumor progression. The patient was taken off the trial and continued on radiation only. The patient had progressive disease and he expired 9 months after enrolment in the trial.
Patient 4
The fourth patient was a Caucasian woman in her late 30s with history of right frontoparietal low grade astrocytoma that underwent initial resection in 2003 and subsequent second surgery as well as radiation in 2010 for tumor progression. A third surgery was performed that led to a diagnosis of IDH1MT, MGMT unmethylated anaplastic astrocytoma. The patient received postoperative radiation and temozolomide. Since 2016 the patient had progressive contrast enhancement around the resection cavity leading to an additional surgery. She was then treated with lomustine in 2017 for ongoing radiographic progression. Due to further progression the patient enrolled on the anti-PD-1/anti-TIM-3 arm of the NCT02608268 trial. She received 14 treatment doses of anti-PD-1/anti-TIM-3 with eventual radiographic tumor progression. The patient was taken off the trial and was started on palliative temozolomide and later on bevacizumab. The patient eventually worsened symptomatically and was transitioned to hospice. The patient was still alive at the last time of follow up with an overall survival of 19 months since her enrollment in the clinical trial.
Results
Patient characteristics
The four patients enrolled in this study had a pathologically confirmed diagnosis of glioblastoma (patients 1-3) or anaplastic astrocytoma (patient 4) that had all recurred after prior treatments. Patients 1-3 were initially treated with the anti-TIM-3 antibody at 800 mg/dose IV every 2 weeks. The dose of anti-TIM-3 in all three patients was decreased to 240 mg every 2 weeks due to symptomatic treatment-related edema. Patient 4 received 80 mg of anti-TIM-3 antibody IV every 2 weeks and were treated with concurrent anti-PD-1 antibody at a dose of 80 mg/dose IV every 2 weeks.
All patients had discontinued treatment at the last time of follow up (October 20, 2018) due to disease progression. All adverse events were grade ≤3 and only transient. See Table 1 for baseline patient characteristics, adverse events and overview of treatment outcomes.
The median number of anti-TIM-3 doses administered was 3 (2-14) before treatment discontinuation and the patients were treated on average for 4 months before treatment discontinuation (2.5-6.5 months).
Adverse Events (AE)
No grade 4 adverse events were observed in this cohort of patients. Three of four patients experienced transient grade 3 neurological AE. Patient 1 and 2 had a transient altered mental status during treatment related to increased peritumoral edema that improved with initiation of steroids, delay in treatment and decrease in the dose of the study drug. Patient 4 experienced left arm weakness associated with peritumoral edema that improved with increase in steroid dosing. No significant changes were observed in the number of circulating lymphocytes or platelets during the course of treatment (Supplementary Figure 2). All other adverse events were grade ≤2. See Table 1 for more details.
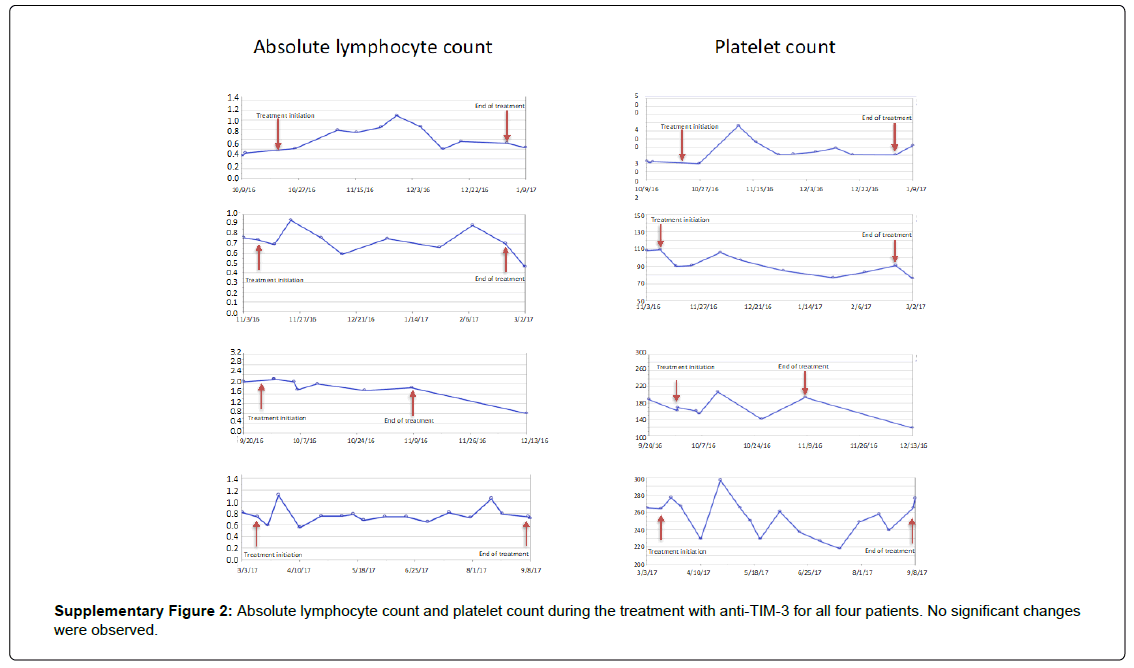
Figure 2: Absolute lymphocyte count and platelet count during the treatment with anti-TIM-3 for all four patients. No significant changes were observed.
Exploratory efficacy
All four patients had eventual disease progression. Patients 1 and 3 had clear progressive disease. Patient 2 developed a significant amount of edema and contrast enhancement when he was treated with anti-TIM-3 that subsequently decreased significantly with steroid treatment. This observation indicates development of a substantial peritumoral immune reaction that however did not lead to a sustained anti-tumor response (Supplementary Figure 1). Patient 4 showed signs of progression 6.5 months after treatment initiation. Table 1 shows the overall survival and progression free survival in this cohort that appears to be similar to historical data on recurrent HGA.
Immunophenotype of the tumor microenvironment
Two of the four patients (patient 2 and 3) exhibited a significant tumor immune infiltration with both perivascular and parenchymal immune cell infiltration. This mainly consisted of microglia/macrophages, CD4+ cells, followed by CD8+ cells and B cells (Supplementary Table 1). When comparing matched primary tumors with recurrent tumors in each patient, the recurrent tumors had an increase in immune infiltration compared to the primary tumors. The genetic profile of these two patients did not have any commonalities. Patient 2 had an IDH1 WT, MGMT promoter unmethylated tumor while patient 3 had an IDH1 MT, MGMT promoter methylated phenotype. All four patients had undergone the standard of treatment including radiation and temozolomide before undergoing treatment with anti-TIM-3. Patients 2 and 3 had a significantly higher percent of tumor infiltrating lymphocytes expressing PD-1, TIM-3 and LAG-3. Interestingly, patient 2 had a significant PD-1, TIM-3 and LAG-3 expression on the primary tumor as well as at the recurrent setting. However, patient 3 had low levels of PD-1, TIM-3 and LAG-3 expression at the primary resection with a significant increase in expression of all three molecules in the recurrent setting. Representative pictures of the IHC are shown in Supplementary Figure 3.
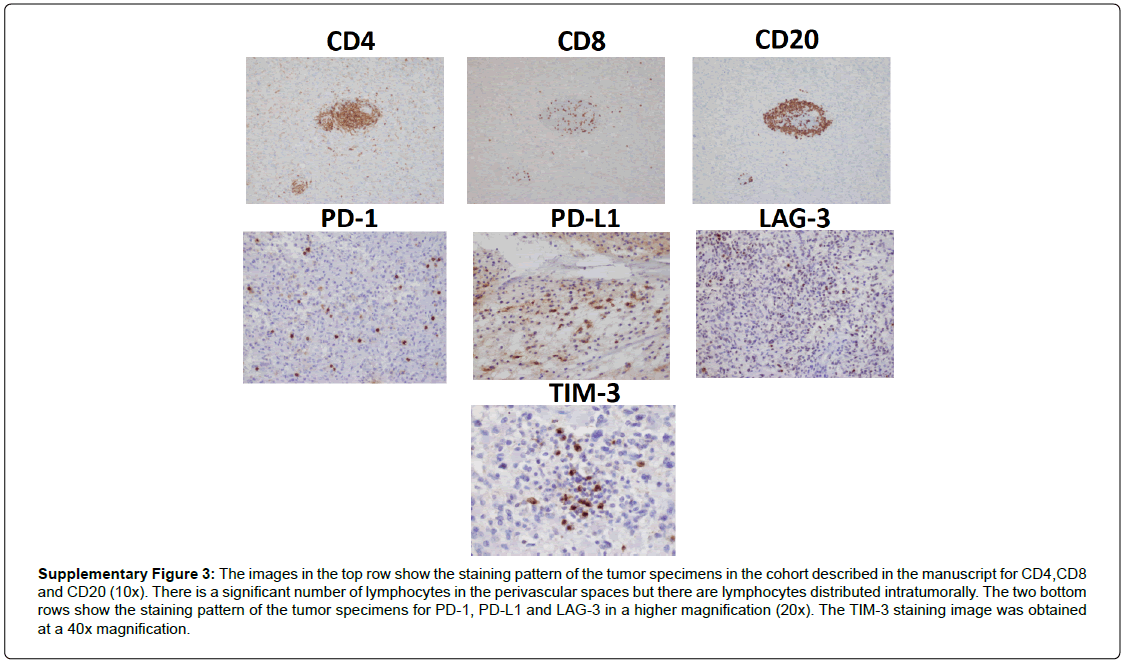
Supplementary Figure 3: The images in the top row show the staining pattern of the tumor specimens in the cohort described in the manuscript for CD4,CD8 and CD20 (10x). There is a significant number of lymphocytes in the perivascular spaces but there are lymphocytes distributed intratumorally. The two bottom rows show the staining pattern of the tumor specimens for PD-1, PD-L1 and LAG-3 in a higher magnification (20x). The TIM-3 staining image was obtained at a 40x magnification.
Discussion
TIM-3 inhibition has been evaluated in combination with other treatments in preclinical models of many cancer types with great results [4-7]. We here present detailed clinical, radiographic and immunological data on a first cohort of four HGA patients treated with anti-TIM-3 therapy as part of a larger trial [5]. Our data suggest that high doses of anti-TIM-3 may lead to significant increase in brain edema, however lower doses of anti-TIM-3 appear to be better tolerated. All patients with HGA had radiographic progression after TIM-3 inhibition with or without use of PD-1 inhibition with a median PFS of 2 months. Overall, there was weak expression of TIM-3 in all the samples analyzed with a trend for greater TIM-3 expression in recurrent tumors compared to primary tumors. This finding is in line with prior studies that indicate TIM-3 being a late marker of immune exhaustion [7]. Patients 2 and 3 exhibited the highest tumor immune infiltration with evidence of increased edema during the course of treatment. The increase in brain tumor edema implies enhancement of immune response, however, it did not affect tumor progression in a measurable way.
Interestingly, 3 out of 4 patients exhibited perivascular staining of B cells, and the increase in the inflammation in patients 2 and 3 was related with an increase in CD20 cells (B cells) as well. The significance of these findings is yet to be investigated in glioblastoma patients.
Conclusion
In conclusion, our study shows that while the use of anti-TIM-3 antibody in recurrent HGA is safe, and appears to be inducing a tumor related inflammatory response. Larger studies are needed to assess true efficacy.
Acknowledgements
We would like to thank the Tumor Microenvironment Core Facility of the Bloomberg Kimmel Institute for Cancer Immunotherapy and the Technology Development Center Manager Liz Engle for performing the immunohistochemistry staining of the tumor specimens.
Contributions
DM collected clinical data, genetic data, radiographic data and pathology data. MAVV interpreted the histology slides and the staining of the tumor samples. XY, PMF, KC, JW, MH, HB, DMP, ML participated in writing the manuscript. ML, DM designed the study.
Competing Interests
PMF receives research funding from AstraZeneca, BMS, Corvus, Novartis, Kyowa and is consultant for Amgen, AstraZeneca, BMS, Novartis, Janssen, Iteos, Sanofi. HB is a consultant for Perosphere, AsclepiX Therpeutics, StemGen, Accelerating Combination Therapies, Catalio Nexus Fund II. LLC, LikeMinds, Inc., Acuity Bio Corp, InSightec, Galen Robotics, Nurami Medicalâ?? , ML receives research funding from Arbor, Aegenus, Altor, BMS, Immunocellular, Celldex, Accuray, Novartis and is consultant for Aegenus, BMS and Stryker. All other authors have no competing interests.
Availability of data and material: All datasets created are presented in this manuscript.
Funding: Philanthropic grants from private families
References
- Forde PM, Chaft JE, Smith KN, Anagnostou V, Cottrell TR, et al. (2018) Neoadjuvant PD-1 blockade in resectable lung cancer. N Engl J Med. 21: 1976-86.
- Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ, Topalian SL, et al.(2012) Safety and activity of anti–PD-L1 antibody in patients with advanced cancer. N Engl J Med 26: 2455-2465.
- Omuro A, Vlahovic G, Lim M, Sahebjam S, Baehring J, et al.(2018) Nivolumab with or without ipilimumab in patients with recurrent glioblastoma: results from exploratory phase I cohorts of CheckMate 143. Neuro-oncol. 5: 674-686.
- Kim JE, Patel MA, Mangraviti A, Kim ES, Theodros D,et al. (2017) Combination therapy with anti-PD-1, anti-TIM-3, and focal radiation results in regression of murine gliomas. Clin Cancer Res 1: 124-136.
- Curigliano G, Gelderblom H, Mach N, Doi T, Tai WM, et al. (2021) Phase I/Ib clinical trial of Sabatolimab, an Anti-TIM-3 antibody, alone and in combination with spartalizumab, an anti-pd-1 antibody, in advanced solid tumors. Clin Cancer Res 27: 3620-3629.
- Stupp R, Hegi M, Weller M. (2010) Neuro-oncology, a decade of temozolomide and beyond. Expert rev. anticancer ther. 11: 1675-1677.
- Liu JF, Wu L, Yang LL, Deng WW, Mao L, et al. (2018) Blockade of TIM3 relieves immunosuppression through reducing regulatory T cells in head and neck cancer. J Exp Clin Cancer Res. 1: 1-8.
Google Scholar CrossRef
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 