Research Article, J Fashion Technol Textile Eng S Vol: 0 Issue: 5
Volatile Organic Compounds (VOCs) Treatment by a Functionalized Textile
Gidik H1*, Almuhamed S1, Dupont D1, Derue L2, Thielemans W3, Kignelman G3 and Lahem D2
1UCL, Hautes Etudes d’Ingénieur (HEI), GEMTEX, 13 rue de Toul, F-59000 Lille, France
2Materia Nova, 1 Avenue Nicolas Copernic, 7000 Mons, Belgium
3KU Leuven Campus (Kulak), Department of Chemical Engineering, Etienne Sabbelan 53, 8500 Kortrijk, Belgium
*Corresponding Author : Hayriye Gidik
Associate Professor, HEI, une école d’Yncréa Hauts-de-France, Lille, France
Tel: + 33 (0)3 28 38 48 58
E-mail: hayriye.gidik@yncrea.fr
Received: September 15, 2017 Accepted: September 27, 2018 Published: October 03, 2018
Citation: Gidik H, Almuhamed S, Dupont D, Derue L, Thielemans W, et al. (2018) Volatile Organic Compounds (VOCs) Treatment by a Functionalized Textile. J Fashion Technol Textile Eng S5:002. doi:10.4172/2329-9568.S5-002
Abstract
The past couple of years, buildings with low energy consumption and a higher thermal comfort have become more and more common but this has a negative impact on Indoor Air Quality (IAQ) [1,2]. The majority of pollutants present in the indoor air are Volatile Organic Compounds (VOCs) that originate from sources insidethese buildings. The ultimate aim of this study is to functionalize a textile substrate with a hybrid photo catalytic material based on titanium dioxide (TiO2) and cellulose nano crystals (CNC) in order to photo degrade VOCs under visible light.
Keywords: Indoor air quality; Photocatalysis; Textile; TiO2; Volatile organic compound (VOC)
Introduction
As the people in industrialized countries spend approximately 90% of their time indoors, the Indoor Air Quality (IAQ) has become an important health and safety concern [3]. However, the past couple of years, the buildings with low energy consumption and a higher thermal comfort, through for example tightening of building envelopes and decreased ventilation rates, have become more and more common which may have a negative impact on IAQ [1,2]. The majority of pollutants, which are present in the indoor air of buildings, are Volatile Organic Compounds (VOCs) that originate from different sources, i.e. building products and room furnishings, paints, glues and varnishes and occupant activities [4].
There are several methods including condensation, absorption, adsorption, incineration, bio-filtration and photocatalysis, available to remove these VOCs. Each method can be used for a particular domain in terms of pollutant concentration and air flow. The photocatalysis presents a real advantage for indoor air applications as it is able to treat low concentrations and flow [5]. The semiconductor photocatalysis, which is a green technology, has been widely applied in removing the organic pollutants from air. Especially, semiconductor compounds like ZnO and TiO2 exhibit good photocatalytic activity under UV light radiation [6].
As it was presented previously, TiO2 has a photocatalytic activity under UV light radiation. As it will be used for indoor applications, it is necessary to have photocatalytic activity under visible light. Thus, the first part of this study focuses on doping the TiO2 in order to extend its photocatalytic activity to the visible light domain. The second part presents its application on a textile substrate, i.e. polyester (PES) woven fabric, with coating technology. As the PES is a synthetic fiber, it has poor wetting properties which negatively affect its process ability. The surface of fibers should be modified in order to introduce new carboxyl and hydroxyl groups. In doing this, the binding efficiency of TiO2 can be enhanced [7]. In this study, alkaline hydrolysis was used for surface modification of PES and cellulose nanocrystals were preferred as incorporation vector between textile substrate and TiO2.
Materials and Methods
Materials
100% PES fabric with plain weave from Subrenat (France) was used as textile substrate. Triton® X-100, sodium carbonate, didecyldimethylammonium chloride and sodium carbonate from Fluka (Switzerland) were used for washing and hydrolysis. Ultra-high viscosity carboxymethyl cellulose (CMC) from C. E. Roeper GmbH (Germany) was preferred in order to prepare the coating paste. TiO2/ CNC suspension was prepared as explained below.
Methods
100% PES fabric with plain weave was washed with 2 g.L-1 Triton® X-100 and 2 g.L-1 sodium carbonate at 60°C during 1 hour. The samples were dried at 105°C during 5 minutes. Three different didecyldimethylammonium chloride concentrations, i.e. 0 g.L-1, 0.1 g.L-1 and 0.5 g.L-1, were combined with two different pH, i.e. 9 and 12, for hydrolysis in order to find out the optimal conditions. The samples were treated either 30 minutes or 90 minutes at 95°C with exhaustion. Then, they were dried at 105°C during 5 minutes.
The non-doped TiO2 was prepared by a low-temperature sol-gel route [8] and combined with CNC suspension resulting from cotton wool hydrolysis with sulfuric acid. This suspension was mixed with ultra-high viscosity CMC in order to prepare the coating paste. The mass ratio is CNC/TiO2/CMC ≈ 1/1/1. Prepared paste was applied on PES fabric using knife coating technology. The angle and the distance between the fabric and the knife were 90° and 0.01 mm, respectively.
Characterization
Five different methods were used for characterization of samples: Contact angle measurements, Attenuated Total Reflectance (ATR), Energy-Dispersive X-ray (EDX) spectroscopy, Scanning Electron Microscope (SEM) and Thermogravimetric Analysis (TGA).
Contact angle measurements
In order to compare different hydrolysis processes presented previously, the contact angle measurements were realized by using distilled water. An optical microscope (Zeiss, Germany) with SPOT Basic software was used to get images and they were treated by using Image J software.
Attenuated total reflectance (ATR)
ATR spectra of the non-hydrolyzed and hydrolyzed PES fabric (with and without CNC/TiO2/CMC treatment) and each product separately (TiO2, CNC, CMC) were characterized by using an IR spectroscopy (Varian, USA). The samples were analyzed in the transmittance mode in the range of 400 – 4000 cm-1. Each spectrum was collected by cumulating 64 scans at a resolution of 4 cm-1. The experimental data was collected and analyzed using the Varian Resolutions Pro software.
Scanning electron microscope and energy-dispersive X-ray (SEM/EDX)
A Scanning Electron Microscopy coupled with an Energy Dispersive X-ray spectroscopy (SEM/EDX) was used to analyze the morphology and chemical composition of CNC/TiO2/CMC samples. Characterizations were performed by a FE-SEM (Hitachi SU8020 Ultra-High Resolution Scanning Electron Microscope) equipped with an EDX detector (Htermo Scientific Ultradry Noran System 7).
Thermogravimetric analysis (TGA)
commercial TiO2 P25® (EVONIK, Germany) and cellulose nanocrystals samples were characterized separately by using a TGA 4000 thermogravimetric analyzer (Perkin Elmer, USA) in the temperature range of 30 – 900°C. PES samples with and without CNC/TiO2/CMC coating were characterized with same analyzer in the temperature range of 30 – 700°C. The heating rate was maintained at 10°C.min-1. The measurements were made under a constant air flow rate of 20 mL.min-1. The experimental data was collected and analyzed using the Perkin Elmer Pyris Manager software.
Results and Discussion
Choice of optimized hydrolysis procss
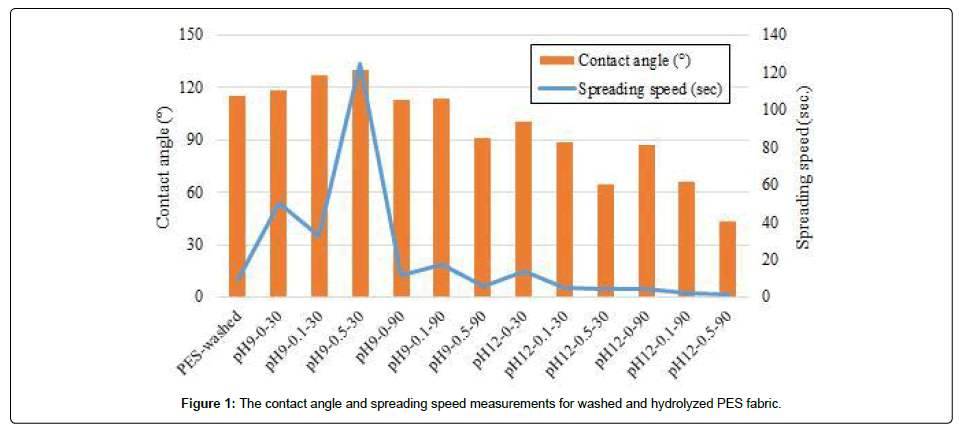
The Figure 1 presents measured contact angles and spreading speeds for different hydrolysis processes. As it was explained previously, three different didecyldimethylammonium chloride concentrations, i.e. 0 g.L-1, 0.1 g.L-1 and 0.5 g.L-1, two different pH, i. e. 9 and 12, and two different treatment times, i.e. 30 minutes and 90 minutes, were used for optimization.
As it is clear from Figure 1, the PES becomes much more hydrophilic with increasing pH, thus under alkaline conditions. Thus, pH 12 was chosen for future studies. The decreasing contact angle and spreading speed confirmed that the increasing didecyldimethylammonium chloride concentration and exhaustion time have a positive influence on hydrophilicity for pH 12. Thus, the fabric which was hydrolyzed with 0.5 g.L-1 didecyldimethylammonium chloride and pH 12 at 95°C for 30 minutes (pH12-0.5-30) was chosen for coating process.
ATR
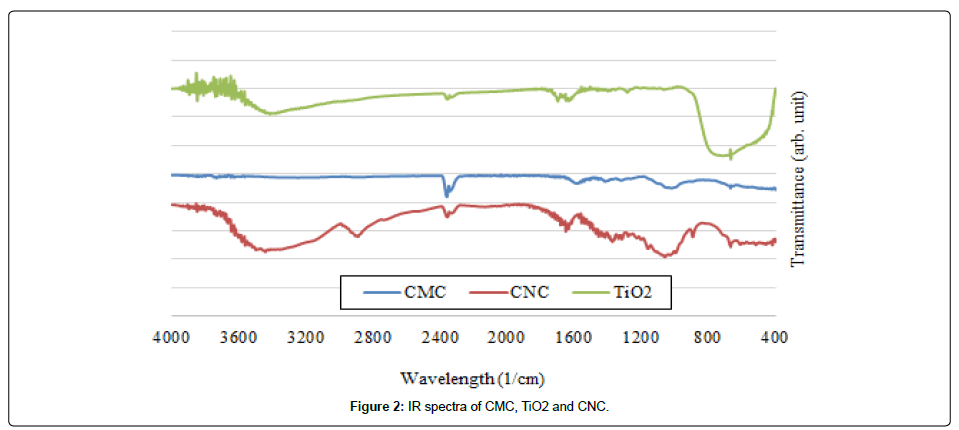
The Figure 2 presents the spectra of carboxymethyl cellulose (CMC), titanium dioxide (TiO2) and cellulose nanocrystals (CNC).
ATR spectra of the CNC shows the band at 3400 cm-1, 2897 cm-1 indicates the stretching vibration of aromatic OH and CH stretching vibration, respectively. The intense band at 1621 cm-1 indicates the presence of CO stretching vibration of cellulose rings as well as − OH bending vibration of absorbed water molecule and 1028 cm-1 band is related to the C−OH stretching vibration of secondary and primary alcohols of CMC and CNC. In addition, it is worthwhile to note peak at 614 cm-1 indicates C−OH out of plane bending vibration [9].
For CMC, the bands in the region 1354-1450 cm-1 are due to symmetrical deformation of CH2 and OH groups. The ether bonds in CMC are presented by the band between 1250 and 1050 cm-1 [10].
In case of TiO2, the broad vibration band between 3650 and 2600 cm-1 is associated with the OH stretching mode of water molecules (physisorbed molecular water), while that at 1620 cm-1 is assigned to their bending modes. The bands in the range 1600-1300 cm-1 are generally associated with carbonate groups; thus, the bands at around 1420 cm-1 are assigned to carbonate species formed by the adsorption of atmospheric CO2. The absorption in the range 1000-600 cm-1 is due to the surface vibrations of the Ti-O bonds [11].
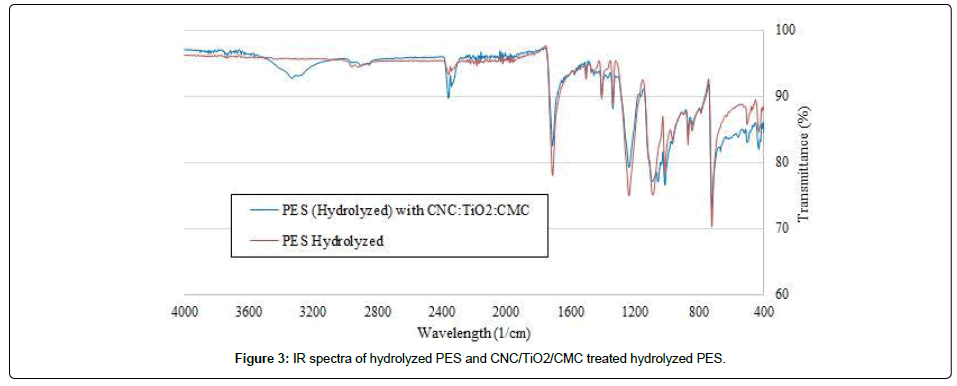
The Figure 3 presents the spectra of hydrolyzed PES fabric and CNC/TiO2/CMC treated hydrolyzed PES fabric.
The first results confirmed that hydrolysis increases the binding efficiency of TiO2 on PES. In addition to this, the spectrometric analysis showed the presence of CNC at around 3400 cm-1, CMC/ CNC at around 1028 cm-1 and TiO2 at around 600 cm-1 on the PES fabric.
SEM-EDX
The Figure 4 presents the SEM observation of CNC/TiO2/CMC treated hydrolyzed polyester fabric. The Figure 4 showed that the CNC/TiO2/CMC deposition was observed for certain zones of fabric. In order to deepen the research SEM analysis was completed with EDX composition analysis. Figure 5(b) and 5(c) present the EDX analysis of the areas shown on the SEM image of Figure 5(a) of CNC/ TiO2/CMC treated hydrolyzed polyester fabric.
The EDX and SEM analysis showed that the CNC/TiO2/CMC coating paste wasn’t applied on whole fabric surface homogenously. It can be seen the areas with TiO2 deposit (Zone 1) and others without TiO2 deposit (Zone 2).
The ATR, SEM and EDX analysis showed the presence of TiO2 particles on the fabric. In order to have quantitative results, the hydrolyzed PES fabric and CNC/TiO2/CMC treated hydrolyzed PES fabric were characterized by TGA.
TGA
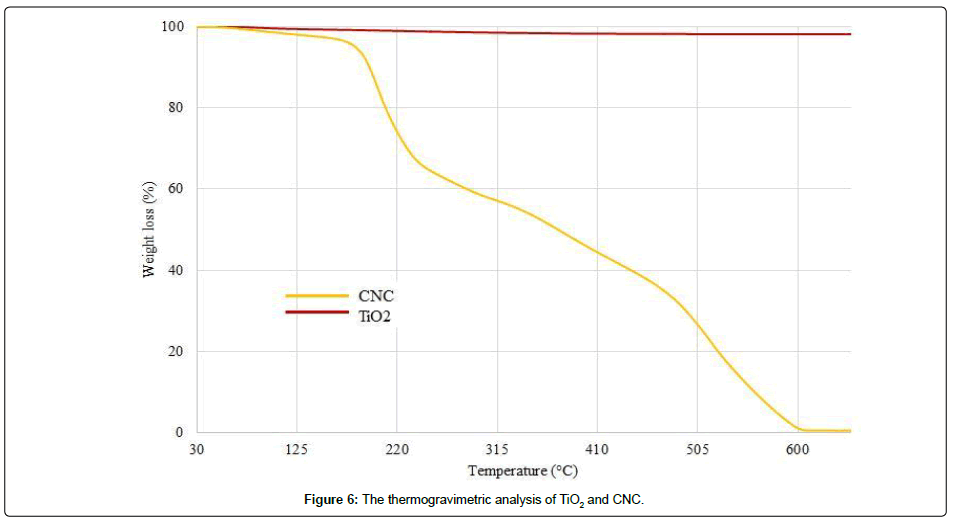
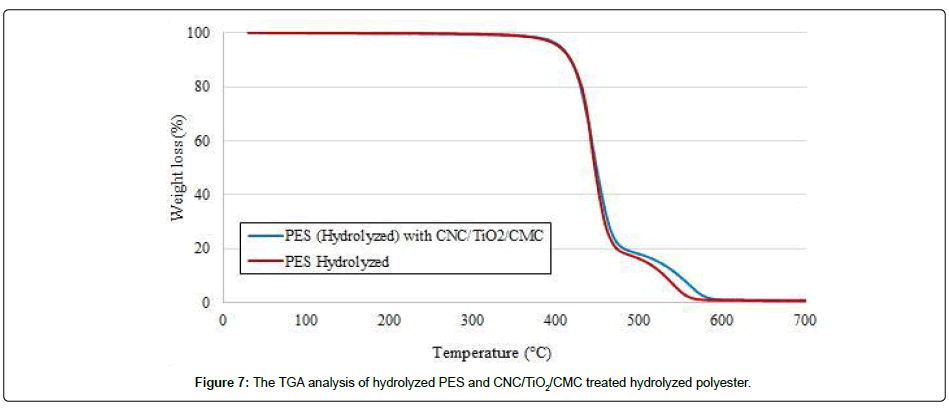
The Figure 6 presents the thermogravimetric analysis of titanium dioxide (TiO2) and cellulose nanocrystals (CNC), separately. As the TiO2 is a metal oxide, its degradation temperature is higher than 600°C. Thus, it didn’t show any change in weight till 900°C. However, CNC presented a weight loss from 160°C till 600°C and it was reached 0% at around 600°C. The Figure 7 presents the thermogravimetric analysis of hydrolyzed PES fabric and CNC/TiO2/CMC treated hydrolyzed PES fabric.
The thermogravimetric analysis showed that the degradation of fabrics was started at around 340°C and reached maximum rate around 600°C. From 580°C, the weight loss is 0.6% for hydrolyzed PES fabric. This percentage can be caused by the TiO2 which was used during spinning process as whitening agent. However, the weight of residue is 0.9% regarding to initial fabric weight for CNC/TiO2/ CMC treated hydrolyzed PES fabric from 600°C. As it was presented previously (Figure 6), the CNC was degraded at round 600°C and TiO2 stayed stable at this temperature. Thus, this 0.3% represents the quantity of TiO2 on fabric surface. The photocatalytic activity of the treated samples will be tested by using a measuring unit at laboratory scale. By doing this, it can be decided if this concentration is enough or not. If the concentration is insufficient, both faces of fabric can be coated, concentration of TiO2 in CNC/TiO2 suspension can be increased and the coating process can be optimized in order to have a homogenous deposition.
Conclusion
This preliminary study gave satisfactory results for application of non-doped TiO2 and CNC on a synthetic fiber based textile substrate. Future studies look at studying with doped-TiO2 which can be realized by three processes, i.e. sol-gel, physical vapor deposition (PVD) or ion implantation, in order to have a photocatalytic activity with visible light. The modification of fiber surface can be realized with plasma treatment instead of alkaline hydrolysis as a green technology. Another approach possible to functionalize the synthetic substrate with TiO2/CNC mixture is by grafting amine groups (NH2) on the surface of the polyester fibers in order to increase the polarity of the fibers surface. This will enhance the adhesion of TiO2/CNC on the substrate, consequently.
The photocatalytic activity of the treated samples will be tested by using a measuring unit at laboratory scale for IAQ. By doing this, the parameters like needed TiO2 amount on textile surface and textile treatment (plasma, alkaline …) can be defined in order to optimize the photocatalytic activity. In addition to these, the durability properties (washing, abrasion …) of developed depolluting textile will be evaluated.
Acknowledgment
This study is realized thanks to the Interreg V France-Wallonie-Vlaanderen program (TEXACOV Project of Portfolio GoToS3). Thus, the authors gratefully acknowledge the European Regional Development Fund (ERDF), the Walloon Region and West Vlaanderen Provincie for their financial support.
References
- Kaunelien V, Prasauskas T, Krugly E, Stasiulaitien I, Ciuzas D, et al. (2016) Indoor air quality in low energy residential buildings in Lithuania. Build Environ 108: 63–72.
- Langer S, Beko G, Bloom E, Widheden A, Ekberg L (2015) Indoor air quality in passive and conventional new houses in Sweden. Build Environ 93: 92–100.
- Brown T, Dassonville C, Derbez M, Ramalho O, Kirchner S, et al. (2015) Relationships between socioeconomic and lifestyle factors and indoor air quality in French dwellings. Environ Res 140: 385–396.
- Langer S, Ramalho O, Derbez M, Riberon J, Kirchner S, et al. (2016) Indoor environmental quality in French dwellings and building characteristics. Atmos Environ 128: 82–91.
- Le Cloirec P (1998) Les composés organiques volatiles dans l'environnemented Tec & Doc Lavoisier(Ecole des Mines de Nantes).
- Türkyılmaz Ş, Güy N, Özacar M (2017) Photocatalytic efficiencies of Ni, Mn, Fe and Ag doped ZnO nanostructures synthesized by hydrothermal method the synergistic/antagonistic effect between ZnO and metals. J Photochem. Photobiol 341: 39–50.
- Radetic M (2013) Functionalization of textile materials with TiO2 nanoparticles. J Photochem Photobiol 16: 62–76.
- Hu L,Wang J, Zhang J, Zhang Q, Liu Z (2014) An N-doped anatase/rutile TiO2 hybrid from low-Temperature direct nitridization enhanced photoactivity under UV-visible-light. RSC Advances 4: 420–427.
- Wanqing L, Changqing F, Xing Z, Qian Y, Shaofei P, et al. (2018) Cellulose nanocrystals obtained from office waste paper and their potential application in PET packing materials. Carbohydr Polym 181: 376-385.
- Ismail NM, Bono A, Valintinus ACR, Nilus S, Chng LM (2010) Optimization of reaction conditions for preparing carboxymethylcellulose. Appl Sci 10: 2530-2536.
- Ayoub H, Kassir M, Raad M, Bazzi H, Hijazi A (2017) Effect of dye structure on the photodegradation kinetic using TiO2 nanoparticles. Mater Sci Chem Eng 5: 31-45.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi