Innovation and partnerships in deal making- Actual insights from pharmaceutical industry
Kuntal Chatterjee
Apotex Inc., Germany
: J Forensic Toxicol Pharmacol
Abstract
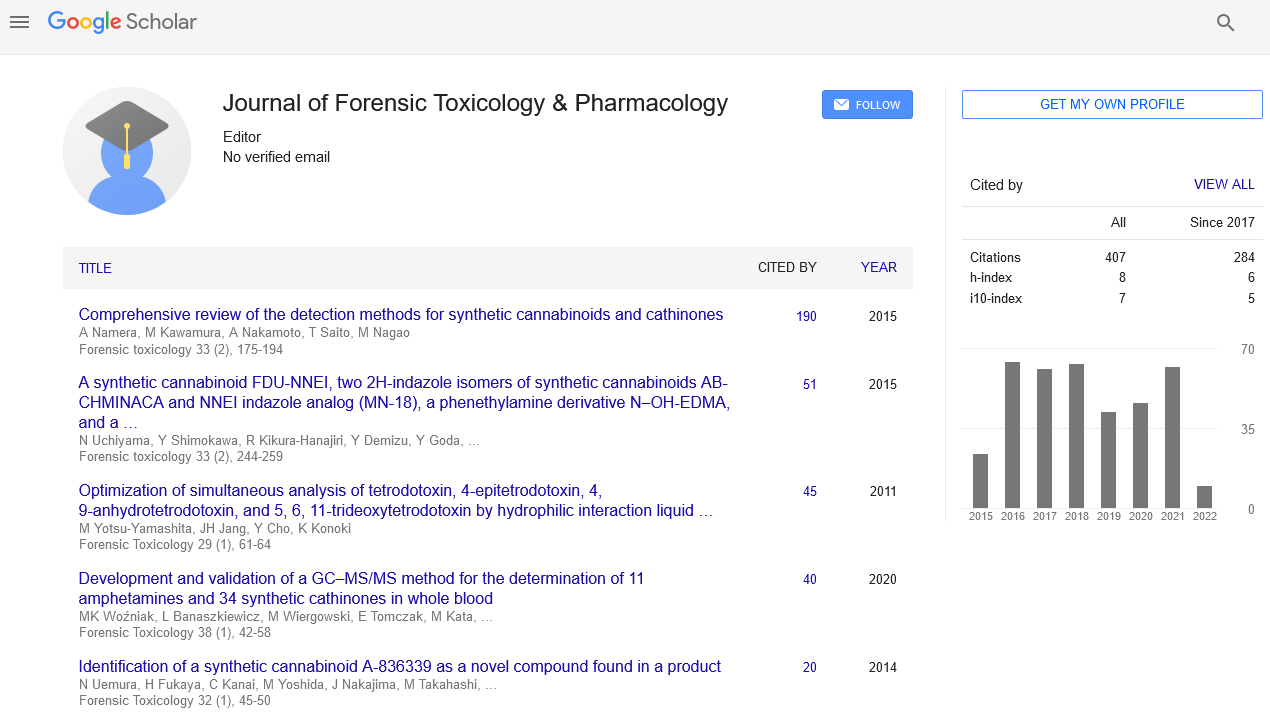
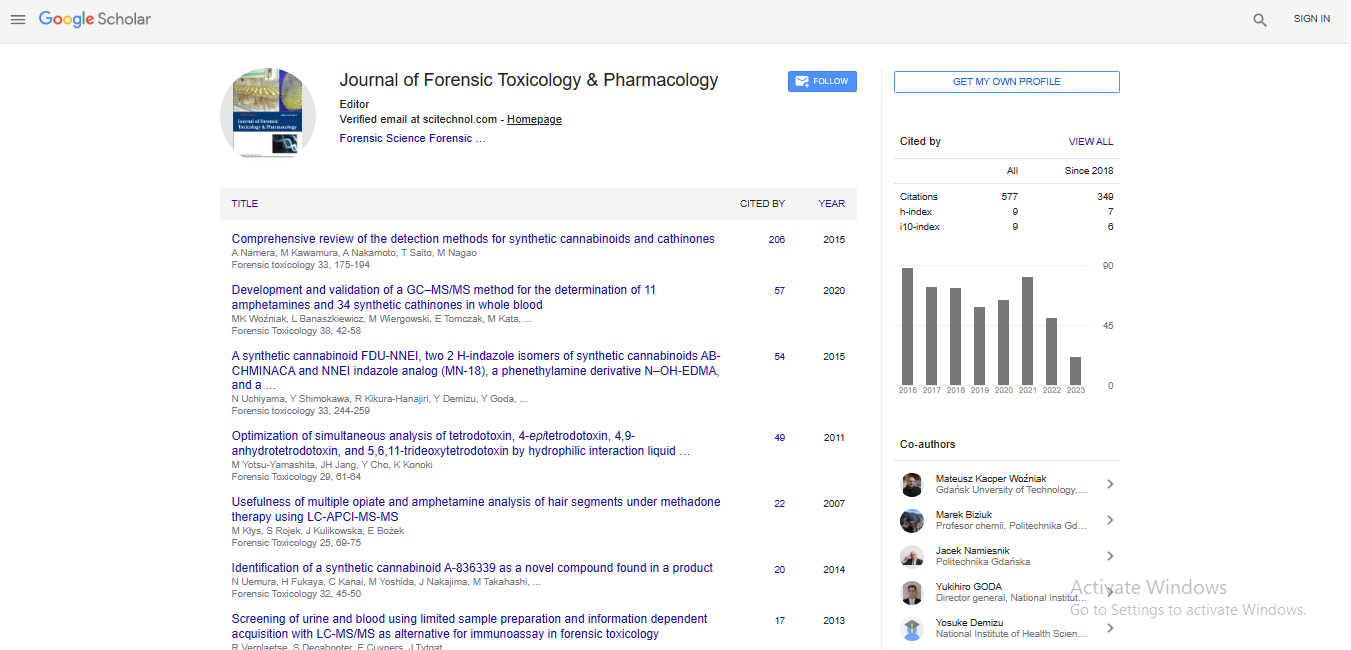
Deal Making is not an easy piece of cake. While it starts with active scouting, it is more relevant to follow the actual process flow and also active networking. The whole process starts with identifying gaps in product portfolio and then Sourcing and qualifying 3’rd party partners. In my 17 plus years of direct Pharma experience, I have found that Trust is the most important parameter in deal making and usually it grows with time. Also, to build trust and reach a preferred partner status, it is important to work together like a joint family concept. Another important aspect of Innovation and Partnerships lies with right communication. When we communicate well both internally and externally, we should clearly guide the reader or the team member with details. For example, in my MBA thesis, I was able to devise a ‘filing to market review (FTMR)” project wherein we mapped each process owner in terms of complexity and was able to reduce target launch dates by months in advance. Attached presentation is just a snapshot of what works well and what does not. Timing is also another important part of deal making. Even though we may have the best commercial case and molecule identified, if we are not respecting each other’s time and working towards a common goal, then we can easily fail. So, Innovation and partnerships in deal making is all about trust, timing and mutual growth.
Biography
Kuntal Chatterjee has 17 plus years of techno commercial experience. He has obtained his Master of Science in Microbiology from Government Institute of Science Aurangabad, India and also a MBA in Pharmaceutical management from NMIMS, Mumbai, India. At Dr Reddys he began his career in quality control and has been responsible for regulatory submissions, approvals –drug registration with USFDA directly. During his time with Dr Reddys, he implemented the FTMR process for EU –a process he developed as part of his MBA thesis project. This process aims to minimise gaps and maximise communication right from filing a dossier until launch in the specified territory. At Sandoz, he was responsible for Central and Eastern European markets. He was the lead portfolio strategist –undertook total gap analysis of the market, identified ‘market attractiveness’ and was involved with pipeline management for both Rx and OTC products. He looked at 20+ countries and obtained CEO excellence award from Jeff George (CEO Sandoz) for wiping out innovator from a local market (effective launch, pricing & KAM management). Currently at Apotex, he is mainly focusing largely on in-licensing aspects and also identifying CMOs/CDMOs to co-develop generic products and then enter into profit sharing deals with them. He also leads the due diligence team here for his BD projects and also manages contracts with global lawyers. He reports to the SVP and CBO role currently at Apotex.
E-mail: kchatter@apotex.com
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi