Research Article, J Sleep Disor Treat Care Vol: 4 Issue: 2
Association between Sleep Duration and Personality-Gene Variants: Sleep Duration is Longer in S/S Homozygotes of Serotonin Transporter than in L Allele Genotypes
| Akiko Koga1, Akiko Fukushima2, Keiko Sakuma2 and Yasuo Kagawa1* |
| 1Department of Medical Chemistry, Kagawa Nutrition University, Japan |
| 2Department of Molecular Nutrition, Kagawa Nutrition University, Japan |
| Corresponding author : Yasuo Kagawa, Ph.D Department of Medical Chemistry, Kagawa Nutrition University, 3-9-21 Chiyoda, Sakado City, Saitama, 350-0288, Japan Tel: +81 (49) 282 3618; Fax: +81 (49) 282 3618 E-mail: kagawa@eiyo.ac.jp |
| Received: February 10, 2015 Accepted: June 30, 2015 Published: July 03, 2015 |
| Citation: Koga A, Fukushima A, Sakuma K, Kagawa Y (2015) Association between Sleep Duration and Personality-Gene Variants: Sleep Duration is Longer in S/S Homozygotes of Serotonin Transporter than in L Allele Genotypes. J Sleep Disor: Treat Care 4:2. doi:10.4172/2325-9639.1000156 |
Abstract
Heavy Issue: Clarifying AHI Elevation after Contemporary Airway Surgery for OSA – The MACHO Graph
Objective: Personality genes and lifestyle and depression scores of young women were analyzed to reevaluate conflicting findings regarding the relationship between worry and sleep schedule (sleep duration, bedtime etc.).
Methods: Genes encoding serotonin transporter (5-HTT; S/S, S/L, L/L, and S/XL genotypes) and dopamine D4 receptor (DRD4; 2/4, 4/4, and other genotypes) and three clock genes were genotyped for 42 healthy female Japanese students (age range: 20-21). Their mood was assessed via the Center for Epidemiologic Studies Depression Scale (CES-D). Their personalities were assessed via the NEO Five-Factor Inventory (NEO-FFI). They maintained a diary and completed a questionnaire about sleep and dietary intake.
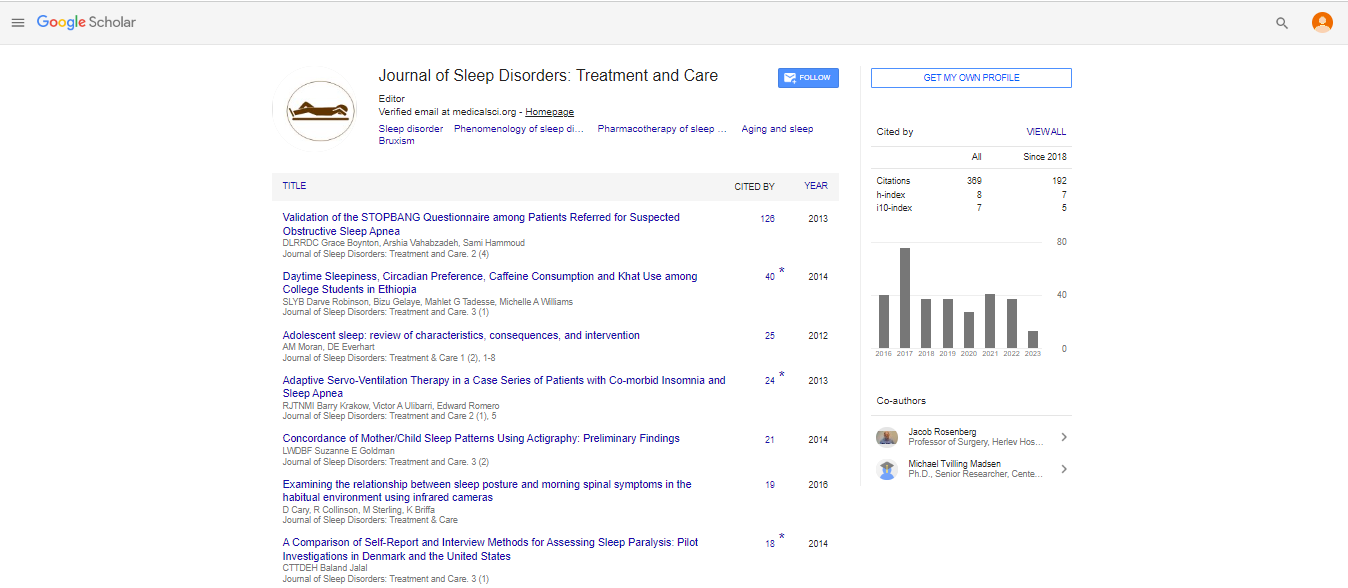
Results: The variant frequencies for S/S homozygote of 5-HTT and 4/4 homozygote of DRD4 were higher among this group than among Caucasians. Sleep duration was 40.8 min longer (7.13 ± .94 hrs) in 5-HTT S/S homozygotes than in others (6.45 ± .69
hrs) (p=0.018), and the bedtime was earlier (0:16 ± 1:05 h:min) for 5-HTT S/S homozygotes than for L allele genotypes (1:14 ± 0:41 h:min) (p=0.005). A “delayed bedtime phenotype” was evident in 55% of S/S homozygotes and 100% of L allele-bearing individuals. Sleep duration was also 42.0 min longer in 4/4 homozygotes (7.09 ± 0.94 hrs) of DRD4 than that in 4/2 heterozygotes (6.39 ± .77 hrs) (p=0.042). An inverse correlation (r=-.316, p=.043) between ppppsleep duration and CES-D score was detected. When groups with high and low CES-D scores were compared, “Delayed bedtime phenotypes”, Neuroticism, and Conscientiousness of NEO-FFI were 100%:59.3%, 34.0:25.5, and 24.6:28.2, respectively.
Conclusion: Both stress-susceptible genotypes, S/S homozygotes of 5-HTT and 4/4 homozygotes of DRD4, slept longer than genotypes with the L-allele of 5-HTT, and 4/2 heterozygotes of DRD4, respectively. The high CES-D group comprised short
sleepers, irrespective of genotype.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi