Research Article, J Sleep Disor Treat Care Vol: 7 Issue: 3
A Study Comparing Primary and Secondary Sleep Bruxism and their Effect on the Trigeminal Cardiac Reflex
Ken Luco*
Luco Hybrid OSA Appliance Inc, University of Alberta, Kingston, Canada
*Corresponding Author : Ken Luco, DDS
President at Luco Hybrid OSA Appliance Inc, 1419 Butternut Creek Road, Canada
Tel: 613 888 6019
Fax: 613 544 0885
E-mail: DrLuco@sympatico.ca
Received: June 28, 2018 Accepted: July 06, 2018 Published: September 13, 2018
Citation: Luco K (2018) A Study Comparing Primary and Secondary Sleep Bruxism and their Effect on the Trigeminal Cardiac Reflex. J Sleep Disor: Treat Care 7:3. doi: 10.4172/2325-9639.1000218
Abstract
Sleep bruxism is classified as a repetitive jaw-muscle activity characterized by clenching or grinding and/or bracing or thrusting of the mandible during sleep. It has been proposed that sleep disordered breathing is causative regarding sleep bruxism, in a protective roll, assisting in stabilization of the airway during these abnormal breathing events. This paper will attempt to present evidence of two distinct forms of sleep bruxism: primary and secondary. It has been shown that sleep bruxism stimulates the trigeminal cardiac reflex affecting heart rate, respiration, blood pressure and cranial pressure. This paper compares the similarities and differences between primary sleep bruxism (occurring independently), secondary sleep bruxism occurring with sleep disordered breathing and secondary sleep bruxism resulting from administration of selective serotonin reuptake inhibitor type medications. It also examines how they all affect the trigeminal cardiac reflex similarly and meet the most recent classification of trigeminal cardiac stimulation.
Keywords: Sleep bruxism; Trigeminal cardiacreflex; Sleep disordered breathing; Desaturation; Polysomnography
Abbreviations
TCR: Trigeminal Cardiac Reflex; SSRI: Selective Serotonin Reuptake Inhibitors; SB: Sleep Bruxism; SB1: Primary Sleep Bruxism; SB2A: Secondary Sleep Bruxism due to sleep disordered breathing; SB2B: Sleep bruxism due to SSRI type medications; PSG: Polysomnography; ICDS3: International Classification of Sleep Disorders 3rd Edition; SBD: Sleep Disordered Breathing.
Introduction
Sleep bruxism (SB) is classified by the American Academy of Sleep Medicine (AASM) in the International Classification of Sleep Disorders (3rd Edition) (ICSD3) as a sleep related movement disorder characterized by clenching or grinding and/or bracing or thrusting of the mandible [1]. It is classified by the World Health Organization (WHO) under the code ICD-10-CM G47.63 in the International Classification of Diseases ICD-10 (and under the code 7A83 in draft of IDC-11) [2]. SB consists of tonic or sustained contractions lasting at least 2 s, phasic or rhythmic 1 Hz contractions lasting 0.25 to 2 s, or a mixed pattern with characteristics of both. Under ICD-11, there are three severity levels described, mild (XS5W), moderate (XSOT and severe (XS25). Mild is described as 5-15 SB events per hour, moderate as 15-25 per hour and severe as greater than 25 per hour. Sleep bruxism affects 8%-10% of the adult population, affecting men and women equally [1]. It usually occurs in shallower stages of sleep but has been shown to occur in all stages.
It has been proposed that sleep bruxism is a protective event during obstructive sleep disorders that helps stabilize the pharyngeal musculature assisting in respiration, however others propose that it is has a complex etiology and is variable between individuals [3,4]. SB also results in increased electroencephalographic (EEG) activity, seen as a sleep arousal and the extreme forces produced may result in dental damage, TMD, myofascial pain, and tension/migraine type headaches (migraines without auras) [1,5-7]. SB has been shown to result in activation of the trigemino-cardiac reflex (TCR), a powerful cranial reflex that alters heart rate, respiration, blood pressure, gastric motility and cranial pressure [8,9]. This stimulation of the TCR may be mechanical or chemical.
The TCR was first reported in 1999 and has been extensively reported in medical journals during maxillofacial and head and neck surgery. Inadvertent stimulation of the branches of the trigeminal nerve can result in rapid and significant changes [10-19], often seriously complicating the surgical procedure. The diver’s reflex is a classic example of the TCR stimulation resulting in rapid decrease of heart rate, blood pressure, respiration and cranial pressure. The TCR is considered oxygen conserving and may be stimulated along the course of all three branches of the trigeminal nerve as well as at the level of the brainstem (pons). It has been associated with arrhythmias, asystole, apnea, gastric mobility, sudden infant death syndrome (SIDS) [10], sleep apnea, and most recently, with sleep bruxism [8,9]. At most levels of the trigeminal nerve, stimulation results in bradycardia, hypotension, apnea, increased gastric motility and decreased intracranial pressure. Only at the level of the gasserian ganglion, do we see the paroxysmal effect of tachycardia, hypertension, hyperpnea, gastric hypomobility and increased cranial pressure. This has been described in a recent work by Meuwly et al. in their revised classification of the TCR [7]. Stimulation of the TCR may be chemical or mechanical. It is medically accepted that an increase/ decrease of 20% or more in heart rate is required before a diagnosis of TCR activation may be made [3]. It is also accepted that minor stimulations do not activate the TCR while major stimulations do [3].
What makes SB unique is that it affects the trigeminal nerve at the level of the Gasserion ganglion, neurochemically. The stimulation occurs through afferents from the periodontal ligaments of the teeth, the mental nerve of the mandible and muscle spindle organs located in the masseter and temporalis muscles [8]. At this level, both sympathetic and parasympathetic modulation may be observed as tachycardia or bradycardia, a decrease or increase in respiration or as an increase or decrease in blood pressure, gastric motility and cranial pressure. Regarding TCR stimulation by SB, the effects seen are a result of sympathetic hyper-stimulation of the TCR, seen as tachycardia, tachypnea, and inhibition of gastric motility, hypertension, and increased intracranial pressure.
Sleep bruxism has been shown to occur in the primary form [12], secondarily to sleep disordered breathing and secondarily to certain psychotropic drugs [18-22]. Genetic mutations associated with sleep bruxism include the HTR2A C Allele rs2770304 as a polymorphism affected the brain as well as the DRD3 rs6280, also as a polymorphism [23,24]. The precise mechanism for how this affects SB is currently not known but is under investigation.
Study Design
The purpose of the study was to examine three groups of subjects: those with primary sleep bruxism (SB1), those with sleep bruxism occurring concurrently with sleep disordered breathing (SB2A) and those taking selective serotonin reuptake inhibitor (SSRI) type medications (SB2B), and to assess if there is a commonality between them on the effect on the TCR and sleep bruxism. Polysomnographic recordings (PSG) were obtained using Medibyte Event type 3 sleep recorders with EMG Electrode Yokes (home sleep studies (HSS)). The EMG leads were attached to the masseter and temporalis muscles as well as the mastoid region (ground). Audio recordings of the SB events were completed in real time, to differentiate true from false recordings. Scoring was completed following the AASM guidelines on PPS scoring for sleep bruxism [25]. 74 subjects were underwent HSS recordings and divided into the three groups. In each group 15 representative subjects were chosen to represent the group.
The successful candidates consisted of 15 subjects with SB1, 15 with SB2A (with concurrent SDB) and 15 with SB2B due to SSRI medication, selected from a potential group of 74. To be representative of SB, equal numbers of male and female were selected. All subjects were 18 or older and provided informed consent.
For the SB1 group, all subjects were required to demonstrate SB events greater than 10 per hour in the absence of sleep disordered breathing (SDB). Subjects in the SB2A group were required to demonstrate both SBD and SB2A events simultaneously on a polysomnograph recording. The last group, the SB2B group, we required to test negative for SB prior to administration of the SSRI medications by history and polysomnographic recording. The SSRI medications were initiated and titrated to optimum levels over a six week period and a second HSS completed [22]. All subjects who did not meet these criteria were excluded from the study. All subjects completed the study. All sleep tracings were conducted following the AASM Manual for the Scoring of Sleep and Associated Events, Rules, Terminology, and Technology [26].
Statistical analysis
The one-way ANOVA followed by Dunnett’s multiple comparisons test was performed using GraphPad Prism version 7.00 for Windows, GraphPad Software, La Jolla California USA, www. graphpad.com determine significance.
Results
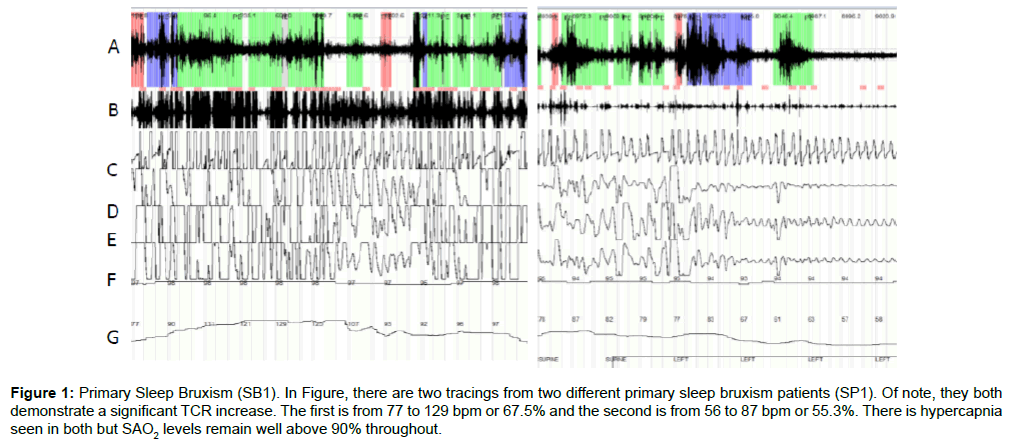
Table 1 lists the 15 subjects with SB1 (8 female, 7 male), mean age of 32 years 6 months and range of 19 years 9 months to 58 years 6 months. All subjects scored an apnea hypopnea index (AHI) of less than 5 (with a mean of 1.8 and range 0.1 to 4.1) suggesting the absence of SDB. All demonstrated an elevated bruxism index (hourly rate of bruxism events) greater than 10, with a mean of 30.6 and range 12.3 to 61.1. There was an associated increase in heart rate of 85.8% (with a range of +40.0% to +122.0%), indicating hyper-stimulation of the TCR. Figure 1 demonstrates 2 HSS tracings from 2 different subjects in the SB1 group. Both tracings demonstrate no oxygen desaturation with peripheral capillary oxygen saturation (SP02) levels remaining between 92% and 95% throughout the tracing. In both, significant SB events can be seen as well as the changes in heart rate.
| # | Gender | Age (years/months) | Bruxism Index | Mean HR | Max HR | % Increase | AHI |
|---|---|---|---|---|---|---|---|
| 1 | F | 26 y 0 m | 22.5 | 74.3 | 104 | 40 | 4.1 |
| 2 | F | 34 y 9 m | 19.8 | 66.2 | 133 | 100.9 | 1.2 |
| 3 | M | 24 y 6 m | 29.1 | 67.8 | 119 | 75.5 | 0.9 |
| 4 | M | 22 y 4 m | 33.6 | 71.4 | 114 | 59.7 | 2.3 |
| 5 | F | 42 y 9 m | 27.6 | 70.8 | 142 | 100.6 | 3.7 |
| 6 | M | 43 y 8 m | 18.4 | 59.8 | 129 | 115.7 | 2 |
| 7 | M | 20 y 3 m | 12.3 | 66.6 | 101 | 51.7 | 0.1 |
| 8 | M | 24 y 0 m | 41.2 | 62.1 | 133 | 114.2 | 4.1 |
| 9 | F | 33 y 1 m | 20 | 72.4 | 127 | 75.4 | 3.7 |
| 10 | F | 33 y 4 m | 33.3 | 68.3 | 137 | 100.6 | 2.2 |
| 11 | F | 21 y 8 m | 61.1 | 62.5 | 112 | 79.2 | 1.7 |
| 12 | M | 37 y 3 m | 49.7 | 59.9 | 133 | 122 | 0.5 |
| 13 | M | 58 y 6 m | 27.4 | 77.1 | 119 | 54.3 | 0.8 |
| 14 | F | 55 y 3 m | 29.5 | 69.9 | 141 | 101.7 | 0.1 |
| 15 | F | 19 y 9 m | 33.8 | 62.3 | 122 | 95.8 | 0.2 |
| Mean | 32 y 6 m | 30.6 | 67.4 | 124.4 | 85.8 | 1.8 |
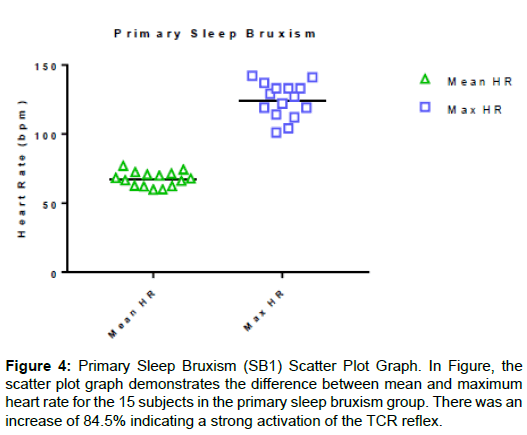
Table 1: Group 1 SB1 Primary Sleep Bruxism, Table 1 lists the 15 subjects studied who were diagnosed with primary sleep bruxism. Mean age was 32 y 6 m with a range of 19 y 9 m to 58 y 6 m. Mean AHI was 1.8 with a range of 0.1 to 4.1. Mean bruxism index was 30.6 with a range of 12.3 to 61.1 indicated severe sleep bruxism. The mean increase in TCR activation was 85.8%, well above the 20% threshold for a diagnosis of TCR activation.
Figure 1: Primary Sleep Bruxism (SB1). In Figure, there are two tracings from two different primary sleep bruxism patients (SP1). Of note, they both demonstrate a significant TCR increase. The first is from 77 to 129 bpm or 67.5% and the second is from 56 to 87 bpm or 55.3%. There is hypercapnia seen in both but SAO2 levels remain well above 90% throughout.
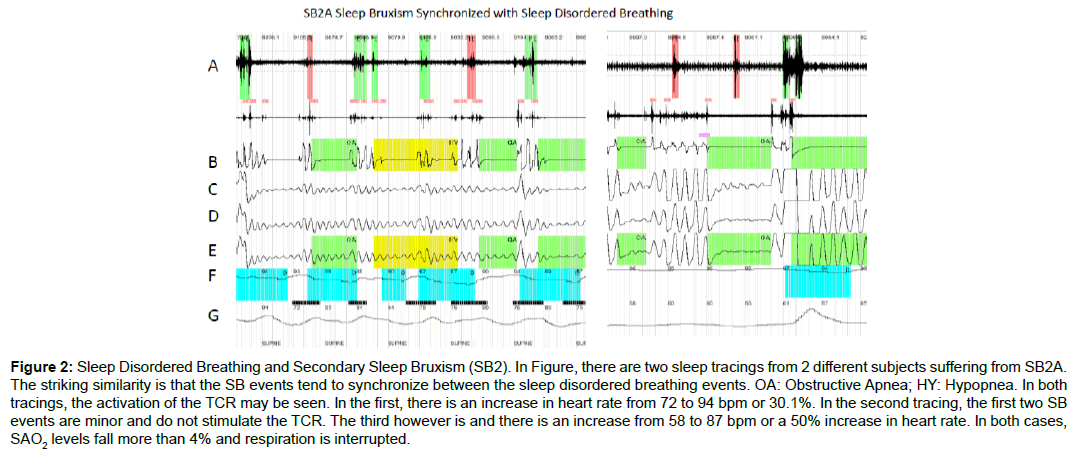
Table 2 lists the 15 subjects demonstrating SB2A concurrent with SBD (7 female, 8 male) with a mean age of 32 years 6 months and range of 19 years 7 months to 58 years 6 months. In these cases the AHI was well into the range of sleep apnea (mean 27.5, range 17.3 to 42.7). The bruxism index was elevated (mean 36.9, range 14.7 to 61.5), as well as heart rate (due to TCR stimulation) with a mean increase of 85.8%, range 74.4% to 116.5%. In Figure 2, tracings of SB2, one striking difference from Figures 1 and 2 is that in SB2A, the sleep bruxism events are synchronized at the beginning and end of each sleep apnea event. This is typical of SB when it occurs with SDB. The heart rate in Figure 2 is affected and oscillates with the SB events in a wave pattern due to repeated stimulation of the TCR. The second tracing demonstrates that minor SB events did not activate the TCR whereas a major event does (as is evidenced by the increase in heart rate).
| # | Gender | Age (years/months) | Bruxism Index | Mean HR | Max HR | % Increase | AHI |
|---|---|---|---|---|---|---|---|
| 1 | M | 49 y 7 m | 14.7 | 72.6 | 127 | 74.9 | 19.4 |
| 2 | F | 40 y 3 m | 28.3 | 74.1 | 131 | 76.8 | 26.8 |
| 3 | M | 32 y 1 m | 15.3 | 69.4 | 139 | 100.3 | 17.8 |
| 4 | F | 23 y 0 m | 42.7 | 64.9 | 116 | 78.7 | 33.3 |
| 5 | F | 28 y 6 m | 39.8 | 71.1 | 124 | 74.4 | 24.7 |
| 6 | M | 34 y 4 m | 33.1 | 72.8 | 131 | 79.9 | 28.2 |
| 7 | M | 46 y 3 m | 41.8 | 66.9 | 137 | 104.8 | 37.5 |
| 8 | F | 40 y 2 m | 55.7 | 63.7 | 129 | 102.5 | 31 |
| 9 | F | 55 y 1 m | 29.1 | 59.7 | 127 | 112.7 | 17.7 |
| 10 | F | 38 y 4 m | 36.6 | 58.1 | 114 | 96.2 | 26.4 |
| 11 | F | 22 y 7 m | 31.7 | 64.2 | 139 | 116.5 | 24.8 |
| 12 | M | 24 y 0 m | 49.8 | 72.3 | 141 | 95 | 29.7 |
| 13 | M | 31 y 6 m | 61.5 | 78.1 | 143 | 83.1 | 35.1 |
| 14 | M | 53 y 1 m | 49.4 | 72.6 | 135 | 86 | 42.7 |
| 15 | M | 41 y 7 m | 23.7 | 75.4 | 128 | 69.8 | 17.3 |
| Mean | 37 y 6 m | 36.9 | 69.1 | 130.7 | 90.1 | 27.5 |
Table 2: Group 2 SB2A Secondary Sleep Bruxism Sleep Disordered Breathing, Table 2 lists the 15 subjects studied who were diagnosed with secondary sleep bruxism relating to sleep disordered breathing. Mean age was 37 y 6 m with a range of 22 y 7 m to 55 y 1 m. Mean AHI was 27.5 with a range 17.3 to 37.7. Mean bruxism index was 36.9 with a range of 14.7 t0 61.5 indicating severe sleep bruxism. The mean % increase in heart rate was 90.1% with a range of 69.8% to 116.5%, well above the 20% threshold for a diagnosis of TCR activation.
Figure 2: Sleep Disordered Breathing and Secondary Sleep Bruxism (SB2). In Figure, there are two sleep tracings from 2 different subjects suffering from SB2A. The striking similarity is that the SB events tend to synchronize between the sleep disordered breathing events. OA: Obstructive Apnea; HY: Hypopnea. In both tracings, the activation of the TCR may be seen. In the first, there is an increase in heart rate from 72 to 94 bpm or 30.1%. In the second tracing, the first two SB events are minor and do not stimulate the TCR. The third however is and there is an increase from 58 to 87 bpm or a 50% increase in heart rate. In both cases, SAO2 levels fall more than 4% and respiration is interrupted.
Table 3 lists the SSRI group baseline, prior to administration of the SSRI medication (8 male, 7 female). Mean age was 32 years 3 months with a range of 23 years 6 months to 42 years 9 months. The mean AHI was 3.1 with a range of 0.8 to 4.9 indicating no sleep disordered breathing or excessive sleepiness. The mean bruxism index was 2.7 with a range of 0.7 to 4.8, indicating it was within the normal range. The mean increase in maximum heart rate was 6.7% with a range of 2.0% to 12%, well below the 20% threshold, indicating no stimulation of the TCR.
| # | Gender | Age (years/months) | BI | Mean HR | Max HR | % Increase | AHI |
|---|---|---|---|---|---|---|---|
| 1 | F | 28 y 1 m | 4.1 | 64.1 | 71.8 | 12 | 4 |
| 2 | F | 23 y 6 m | 0.7 | 67.4 | 74.8 | 11 | 2.6 |
| 3 | M | 33 y 5 m | 1.4 | 57.2 | 61.9 | 8.2 | 3.7 |
| 4 | F | 40 y 0 m | 2.2 | 69.9 | 73.4 | 5 | 3.2 |
| 5 | F | 28 y 7 m | 3.9 | 64.2 | 70.5 | 9.8 | 1.1 |
| 6 | M | 32 y 4 m | 3.7 | 76.8 | 78.3 | 2 | 1.2 |
| 7 | M | 37 y 6 m | 3.4 | 60.1 | 61.5 | 2.3 | 1.9 |
| 8 | F | 24 y 0 m | 1.1 | 63.4 | 68.3 | 7.7 | 2.5 |
| 9 | F | 24 y 3 m | 2.3 | 66.1 | 69.1 | 4.5 | 4.1 |
| 10 | F | 26 y 7 m | 0.9 | 68 | 72.1 | 6 | 4.8 |
| 11 | M | 49 y 11 m | 4.8 | 59.7 | 63.6 | 6.5 | 4.3 |
| 12 | M | 34 y 0 m | 4.6 | 74.1 | 75.8 | 2.3 | 3.6 |
| 13 | M | 22 y 7 m | 3.4 | 57.6 | 63.1 | 9.5 | 3.1 |
| 14 | M | 40 y 2 m | 2.9 | 62.2 | 66.6 | 7.1 | 3.1 |
| 15 | M | 42 y 10 m | 1.7 | 68.7 | 73.6 | 7.1 | 2 |
| Mean | 37 y 6 m | 2.7 | 4.3 | 69.6 | 6.7 | 3.1 |
Table 3: Group 3 SB2B before SSRI Administration, Table 3 lists the 15 subjects studied before use of SSRI type medications. Mean age was 37 y 6 m with a range of 22 y 3 m to 49 y 11 m. Mean AHI was 3.1 with a range of 1.1 to 4.8. Mean bruxism index was 2.7 with a range of 0.7 to 4.8 well within the normal range for sleep bruxism. Mean increase in TCR activation was 6.7% with a range of 2.0 to 12.0%, well below the 20% threshold for a diagnosis of TCR activation.
Table 4 lists the same group as Table 3, 6 weeks after administration and titration of the SSRI. Of significance, the bruxism index increased from 2.7 to 40.1 with a range of 18.9 to 71.6, indicating severe sleep bruxism was occurring. Heart rate increased from 6.7%, before the SSRI, to 92.9% with a range of 52.2% to 143.5% after, indicating the SSRI had significantly stimulating the TCR.
| # | Gender | Age (years/months) | Bruxism Index | Mean HR | Max HR | % Increase | AHI |
|---|---|---|---|---|---|---|---|
| 1 | F | 28 y 1 m | 36.3 | 65.2 | 71.8 | 82.5 | 4.2 |
| 2 | F | 23 y 6 m | 28.7 | 68.8 | 74.8 | 87.5 | 2.7 |
| 3 | M | 33 y 5 m | 33.1 | 57.9 | 61.9 | 143.5 | 3.3 |
| 4 | F | 40 y 0 m | 19.7 | 71.6 | 73.4 | 52.2 | 3.5 |
| 5 | F | 28 y 7 m | 66.6 | 66.2 | 70.5 | 67.7 | 0.8 |
| 6 | M | 32 y 4 m | 47.2 | 77.1 | 78.3 | 56.9 | 1.1 |
| 7 | M | 37 y 6 m | 29.8 | 59.9 | 61.5 | 112 | 2.1 |
| 8 | F | 24 y 0 m | 44.7 | 64.3 | 68.3 | 106.8 | 2.9 |
| 9 | F | 24 y 3 m | 37.2 | 66.2 | 69.1 | 94.9 | 3.8 |
| 10 | F | 26 y 7 m | 36.1 | 68.1 | 72.1 | 83.6 | 4.9 |
| 11 | M | 49 y 11 m | 58.7 | 61.8 | 63.6 | 116.8 | 4.7 |
| 12 | M | 34 y 0 m | 51.2 | 73.3 | 75.8 | 99.2 | 3.3 |
| 13 | M | 22 y 7 m | 71.6 | 58.6 | 63.1 | 104.8 | 3.8 |
| 14 | M | 40 y 2 m | 21 | 64.7 | 66.6 | 130.3 | 2.8 |
| 15 | M | 42 y 10 m | 18.9 | 69.3 | 73.6 | 54.4 | 1.9 |
| Mean | 37 y 6 m | 40.1 | 66.2 | 69.6 | 92.9 | 3.1 |
Table 4: Group 3 SB2B 6 Weeks After SSRI Administration, Table 4 lists the same 15 subjects as Table 3, 6 weeks after the administration of SSRI type medications. Mean AHI was 3.1 with a range of 0.8 to 4.9. Mean bruxism index was 40.1 with a range of 18.9 to 71.6 indicating severe sleep bruxism. The mean increase in TCR activation was 92.9%, well above the 20% threshold for a diagnosis of TCR activation.
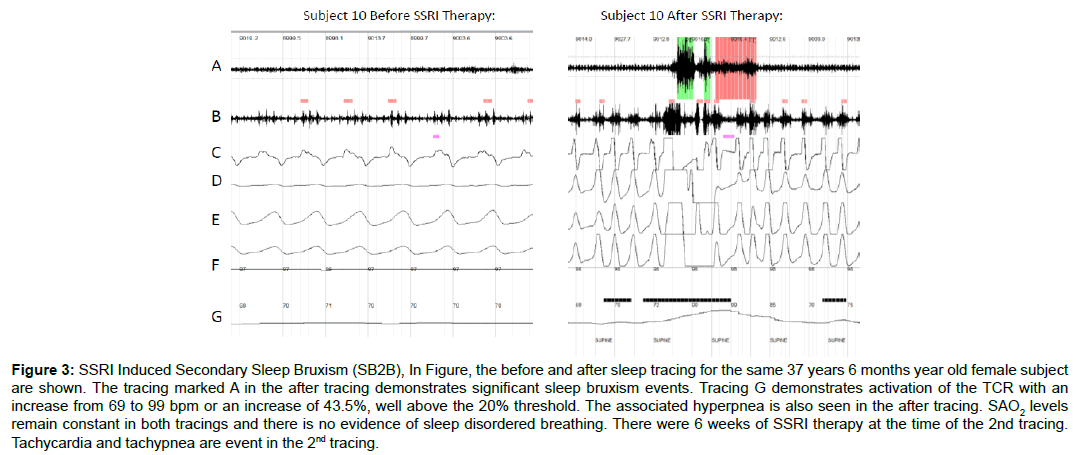
Figure 3 demonstrates two representative tracings: before and after SSRI administration. The changes in respiration, heart rate and jaw movement activity are readily apparent. Both tracings are of the same subject (subject 10, 26 years 7 months female). The significant effect on the sympathetic response demonstrates the SSRI effect on sleep bruxism. The other subjects in the SB2B group all showed similar effects in the second HSS. It was remarkable how the SSRI converted this group from the normal range into severe sleep bruxism with hyper-stimulation of the TCR.
Figure 3: SSRI Induced Secondary Sleep Bruxism (SB2B), In Figure, the before and after sleep tracing for the same 37 years 6 months year old female subject are shown. The tracing marked A in the after tracing demonstrates significant sleep bruxism events. Tracing G demonstrates activation of the TCR with an increase from 69 to 99 bpm or an increase of 43.5%, well above the 20% threshold. The associated hyperpnea is also seen in the after tracing. SAO2 levels remain constant in both tracings and there is no evidence of sleep disordered breathing. There were 6 weeks of SSRI therapy at the time of the 2nd tracing. Tachycardia and tachypnea are event in the 2nd tracing.
Discussion
The results strongly suggest three distinct forms of SB, however there were no statistically significant differences between the actual sleep bruxism intensity and frequency between the three groups. This implies that when SB occurs, regardless of the cause, the presentation is similar and the effect on the TCR is similar.
The scatter plot graphs in Figures 4-6 demonstrate the increase in heart rate (TCR stimulation). It clearly shows the increase seen for all subjects with tight groupings. Figure 7 is a scatter plot graph comparing the % increase in heart rate (TCR stimulation) for the three groups and demonstrates there was no significant difference between them. Figure 8 depicts the Bruxism Index (hourly rate) and, again, there is no statistically significant difference between them. In all subjects, the increase in heart rate significantly exceeded the 20% threshold implying SB has a very strong influence on the TCR. In comparison of the three presentations demonstrating sleep bruxism, there was no significant difference in the increase in heart rate and stimulation of the TCR (P=0.0523, between columns P=0.5855) suggesting that regardless of how sleep bruxism occurs, it results in the same pronounced hyper-stimulation of the TCR (Figure 7). There was also no significant difference found between the three groups regarding their bruxism indices (P=0.2001, between columns P=0.2001), suggesting the severity of sleep bruxism did not depend upon the group and cause (Figure 6).
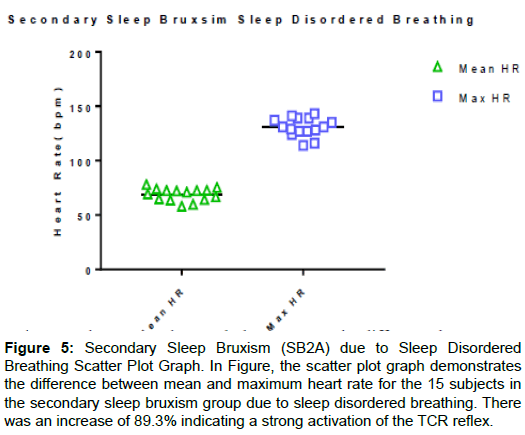
Figure 5: Secondary Sleep Bruxism (SB2A) due to Sleep Disordered Breathing Scatter Plot Graph. In Figure, the scatter plot graph demonstrates the difference between mean and maximum heart rate for the 15 subjects in the secondary sleep bruxism group due to sleep disordered breathing. There was an increase of 89.3% indicating a strong activation of the TCR reflex.
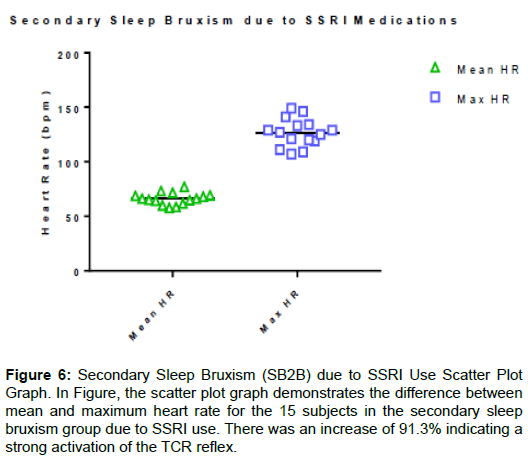
Figure 6: Secondary Sleep Bruxism (SB2B) due to SSRI Use Scatter Plot Graph. In Figure, the scatter plot graph demonstrates the difference between mean and maximum heart rate for the 15 subjects in the secondary sleep bruxism group due to SSRI use. There was an increase of 91.3% indicating a strong activation of the TCR reflex.
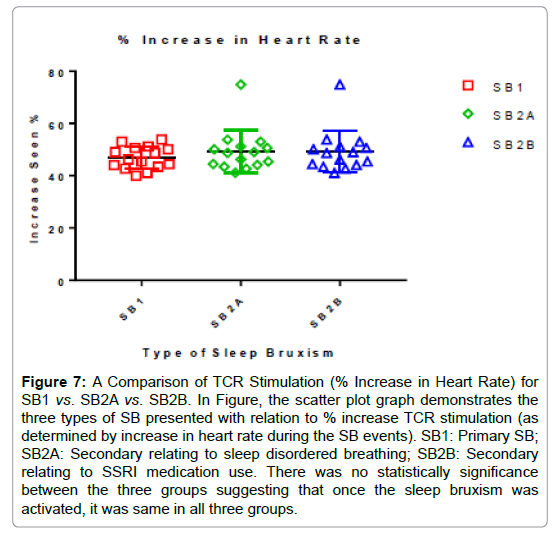
Figure 7: A Comparison of TCR Stimulation (% Increase in Heart Rate) for SB1 vs. SB2A vs. SB2B. In Figure, the scatter plot graph demonstrates the three types of SB presented with relation to % increase TCR stimulation (as determined by increase in heart rate during the SB events). SB1: Primary SB; SB2A: Secondary relating to sleep disordered breathing; SB2B: Secondary relating to SSRI medication use. There was no statistically significance between the three groups suggesting that once the sleep bruxism was activated, it was same in all three groups.
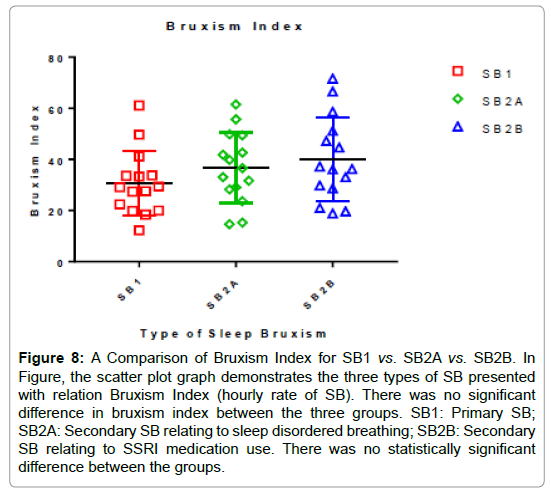
Figure 8: A Comparison of Bruxism Index for SB1 vs. SB2A vs. SB2B. In Figure, the scatter plot graph demonstrates the three types of SB presented with relation Bruxism Index (hourly rate of SB). There was no significant difference in bruxism index between the three groups. SB1: Primary SB; SB2A: Secondary SB relating to sleep disordered breathing; SB2B: Secondary SB relating to SSRI medication use. There was no statistically significant difference between the groups.
Conclusion
Further studies may elucidate even more supporting evidence. One significant finding however is that regardless of the initiating factor, the presentation of sleep bruxism appears consistent with regard to TCR stimulation and SB severity.
Caution should be exercised when prescribing SSRI type medications and a thorough familial history should be undertaken prior to prescribing these medications with particular attention to a history of SB or a history of SSRI use. With the HTR2A polymorphism known to underlie sleep bruxism, reducing re-uptake of serotonin has been shown in this study and others to initiate and/or intensify the effects of sleep bruxism in an otherwise asymptomatic patient. For patients suffering from sleep disordered breathing, depression is a common finding. Caution should be exercised before prescribing SSRI type medications to these patients, as SB2A may already be present but latent and the SSRI could initiate it or exacerbate it resulting in hyper-stimulation of the TCR. 92.9%, well above the 20% threshold for a diagnosis of TCR activation.
References
- Sateia MJ (2014) International classification of sleep disorders-third edition. Chest 146: 1387-1394.
- World Health Organization (1992) The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines. Geneva, Switzerland.
- Manfredini D, Guarda-Nardini L, Marchese-Ragona R, Lobbezoo F (2015) Theories on possible temporal relationships between sleep bruxism and obstructive sleep apnea events. Sleep Breath 19: 1459-1465.
- Jokubauskas L, Baltrušaitytė A (2017) Relationship between obstructive sleep apnoea syndrome and sleep bruxism: a systematic review. J Oral Rehabil 44: 144-153.
- Schmitter M, Kares-Vrincianu A, Kares H, Bermejo JL, Schindler HJ (2015) Sleep-associated aspects of myofascial pain in the orofacial area among Temporomandibular Disorder patients and controls. Sleep Med 16: 1056-1061.
- Fernandes G, Lucia AF, Goncalves DA, Speciali JG, Bigal ME, et al. (2013) Temporomandibular Disorders, Sleep Bruxism and Primary Headache are Mutually Associated. J Orofac Pain 27: 14-20.
- Meuwly C, Chowdhury T, Golanov E, Erne P, Schaller B (2015) Trigeminal Cardiac Reflex: New Thinking Model About the Definition Based on a Literature Review. Medicine 94: e484.
- Luco K (2018) The Relationship of the Trigemino-Cardiac Reflex to Sleep Bruxism. On J Neuro & Br Disord 1.
- Luco K (2018) Short-Term Effects of the Luco Hybrid Obstructive Sleep Apnea Appliance in Adults with Sleep Bruxism: A Quasi-Experimental Study. J Sleep Disor: Treat Care 7.
- Gyaninder PS, Chowdhury T, Bindu B, Schaller B (2016) Sudden infant death syndrome –Role of the trigeminal cardiac reflex. Front Neurol 7: 221.
- Mohan S, Flis DW, O'Leary MA (2014) A Case of Trigeminocardiac Reflex During Infrastructure Maxillectomy. JAMA Otolaryngol Head Neck Surg 140: 563-564.
- Singh GP, Chowdhury T (2017) Brain and heart connections: The trigeminocardiac reflex!. J Neuroanaesthesiol Crit Care 4: 71-77.
- Chowdhury T, Schaller B (2016) The Role of Acute Trigemino-Cardiac Reflex in Unusual, Non-Surgical Cases: A Review. Front Neurol 7: 186.
- Lapi D, Scuri R, Colantuoni A (2016) Trigeminal Cardiac Reflex and Cerebral Blood Flow Regulation. Front Neurosci 10: 470.
- Vasudev S, Reddy KS (2015) Trigemino-Cardiac Reflex During Orbital Floor Reconstruction: A Case Report and Review. J Maxillofac Oral Surg 14: 32-37.
- Cha ST, Eby JB, Katzen JT, Shahinian HK (2002) Trigenocardiac reflex: a unique case pf recurrent asystole during bilateral trigeminal sensory root rhizotomy. J Craniomaxillofac Surg 30: 108-111.
- Schaller B, Probst R, Strebel S, Gratzl O (1999) Trigeminocardiac reflex during surgery in the cerebellopontine angle. J Neurosurg 90: 215-220.
- Schaller B, Graf R (2004) Cerebral ischemia and reperfusion: the pathophysiologic concept as a basis for clinical therapy. J Cereb Blood Flow Metab 24: 351-371.
- Schaller B (2005) Trigemino-cardiac reflex during trans sphenoidal surgery for pituitary adenomas. Clin Neurol Neurosurg 107: 468-474.
- Schaller B (2005) Trigemino-cardiac reflex during microvascular trigeminal decompression in cases of trigeminal neuralgia. J Neurosurg Anesthesiol 17: 45-48.
- Molyneaux E, Telesia LA, Henshaw C, Boath E, Bradley E, et al. (2018) Antidepressants for preventing postnatal depression. Cochrane Database Syst Rev 4.
- Melo G, Dutra KL, Rodrigues FR, Ortega AOL, Porporatti AL, et al. (2018) Association between psychotropic medications and presence of sleep bruxism: a systematic review. J Oral Rehabil 45: 545-554.
- Oporto GH, Bornhardt T, Iturriaga V, Salazar LA (2016) Genetic polymorphisms in the serotonergic system are associated with circadian manifestations of bruxism. J Oral Rehab 43: 805-812.
- Lavigne G, Kato T, Kolta A, Sessle BJ (2003) Neurobiological Mechanisms Involved in Sleep Bruxism. Crit Rev Oral Biol Med 14: 30-46.
- Oksenberg A, Arons E (2002) Sleep bruxism related to obstructive sleep apnea: the effect of continuous positive airway pressure. Sleep Med 3: 513-515.
- Berry RB, Brooks R, Gamaldo CE, Harding SM, Marcus CL, et al. (2012) The AASM manual for the scoring of sleep and associated events. Rules, Terminology and Technical Specifications, American Academy of Sleep Medicine, Darien, Illinois, USA.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi