Research Article, J Forensic Toxicol Pharmaco Vol: 11 Issue: 1
Aluminum Phosphide (ALP) Poisoning a Challenge in Developing Countries. Symptom, Diagnosis and Treatment Strategies
Saeed Aghebat Bekhier*
Department of Toxicology, Kerman university of medical science, Kerman, Iran
*Corresponding author:
Saeed Aghebat Bekhier, Department of Toxicology, Kerman university of medical science, Kerman, Iran
E mail: saeedaghebat@gmail.com
Received date: September 07, 2021; Accepted date: September 21, 2021; Published date: September 28, 2021
Citation: Saeed Aghebat Bekhier (2021) Aluminum Phosphide (ALP) Poisoning a Challenge in Developing Countries. Symptom, Diagnosis and Treatment Strategies. J Forensic Toxicol Pharmaco 10:6.
Abstract
Accidental or intentional poisoning by insecticides and rodenticide is a serious challenge. One of the most important of these toxins is aluminum phosphide(ALP), which is of great concern in developing countries. The main mechanism of toxicity of this substance is expressed in the form of inhibition of the electron transfer chain in mitochondria, which increases lipid peroxidation, decreases GSH and thus reduces ATP production. A range of symptoms for mild to severe AlP poisoning have been reported, with the leading cause of death in patients with Cardiovascular and respiratory disorders. The diagnostic aspect of poisoning is mostly based on the patient's history, self-expression, and clinical symptoms.
Keywords: Aluminum phosphide; Poisoning, Treatment; Rice tablet; Phosphine; ALP
Introduction
Pesticides have a broader meaning and include herbicides, rodenticides, nematocides, algaecides and fungicides. Pesticide poisoning is a worldwide health problem that can occur intentionally for the purposes of suicide, murder, and unintentional accident. In addition, self-poisoning accounts for about one-third of suicides worldwide. Poisoning is one of the leading causes of disability and death worldwide. Poisoning is reported to account for 1.0-2.0 and 1.3% of deaths respectively in developed and developing countries [1]. Aluminum phosphide(AlP) poisoning is one of the most common causes of poisoning in developing countries including India and Iran. Cases of poisoning with it have also been reported in developed countries. The lethal dose of this compound is about 0.5 grams, and acute poisoning with these compounds may be caused directly by ingestion of its salts or indirectly by accidental inhalation of phosphine gas released in contact with moisture during use. Due to the lack of specific antibodies, this pesticide is a serious challenge for the health of poisoned people. The mortality rate with it is very high and in the range of 45 to 100%. The usual lethal dose of AlP in an adult weighing an average of 70 kg is estimated at about 500 mg. Phosphine gas released by the reaction of AlP with moisture inhibits mitochondrial cytochrome oxidase and inhibits oxidative respiration. In AlP poisoning, superoxide and peroxide radicals are formed, leading to cell membrane damage due to lipid peroxidation. Target organs for AlP poisoning include lung, heart, brain, gastrointestinal tract, kidney, and liver. This compound damages several organs through cell membrane denaturation and eventually causes death due to cardiovascular collapse. Due to the fact that there is no specific treatment for poisoning with AlP tablets. The most important treatment strategy is supportive therapies and reduction of symptomatic complications and reducing the risk of further absorption of phosphine gas.
Mechanism of action ALP
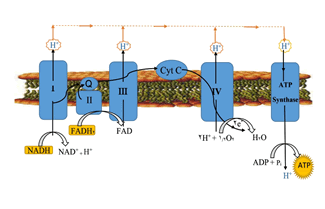
Despite much research on the mechanism of toxicity of aluminum phosphide(Alp), the exact mechanism of action of Alp is still unknown and mysterious. However, some early studies in animal studies Phosphine gas has been shown to bind mainly to cytochrome oxidase and alter the capacity of hemoglobin components (Figure 1).
It also creates oxidative stress and releases oxygen free radicals out of the mitochondria, leading to lipid peroxidation and denaturation of cell membrane proteins various organs. Oxygen free radicals are responsible for the hypoxia of the organs, which is the main feature of phosphine poisoning. Other mechanisms of phosphine-induced damage include impaired enzyme and protein synthesis in myocardial and lung cells. Phosphine denatures oxyhemoglobin and the enzyme is inhibited by reacting with phosphine. Thus it has a destructive effect on the membrane. They believe that oxidative stress is one of the main ones Mechanisms of action of Alp toxicity, which is somewhat similar to organophosphate (OP) compounds. In addition, AlP reduces glutathione, which is one of the main antioxidant defenses. In fact, AlP and OP are produced like toxic stress, which is associated with changes in glucose metabolism [2].
Showed that exposure to phosphine in vitro reduces human serum cholinesterase activity, depending on the duration and concentration of phosphine. Experimental and observational studies have shown that inhibition of cytochrome c oxidase and other enzymes leads to the production of superoxide radicals and cellular peroxides. Cell damage occurs by lipid peroxidation and other oxidant mechanisms. Metal phosphides have a wide range of effects on the body, including cellular toxicity, oxidative stress, inhibition of cholinesterase, circulatory failure, cardiac toxicity, gastrointestinal toxicity, liver damage, neurotoxicity, electrolyte imbalance, and metabolic disorders. A study to evaluate the effects of oxidative stress on pH3 poisoning using pathological and immunohistochemistry methods showed that severe PH3-induced damage in many systems, especially the nervous system, including nerve and axonal damage with increased expression of 4-hydroxy-2-trans-nonenal (4HNE), 8-hydroxy-2'-deoxyguanosine (8-OH-dG), and 3-nitrotyrosine (3-NT) in the brain showed that oxidative stress is an important mechanism for neuronal death Was PH3 in toxicity. The toxicity of AlP appears to be of greater concern at the cellular level, resulting in widespread damage leading to multiorgan dysfunction (MOD) and death. In vitro, phosphine has been shown to inhibit cytochrome c oxidase and impedes cellular oxygen consumption. However, inhibition of cytochrome oxidase C is not the main cause of poisoning. Mitochondria can be the potential site of maximum damage in ALP poisoning, result of mitochondrial damage, ATP production is reduced, which in turn disrupts cellular metabolic activity and disrupts MOD. It can also cause free radicals and damage and regenerate MOD due to electron disruption [3].
Physical and chemical properties and applications
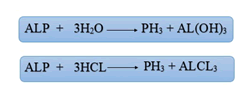
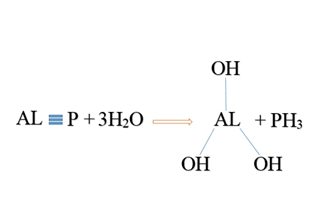
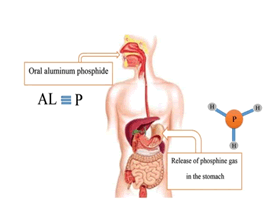
Aluminum phosphide(AlP) is a dark gray or yellow crystalline solid. AlP has a molecular weight of 57/9553 g / mol. This combination is known by different names, the most important of which are: Aluminium phosphide (AlP) , Phostoxin, Bhostoxin, Celphide, Quickphos, Gastion, AL-Phos and Etc. The compound reacts with moisture to release phosphine gas, a flammable and toxic gas and A non-toxic compound in the form of aluminum hydroxide. Aluminum phosphide(AlP) is used as an insecticide and rodenticide compound for grain storage. Phosphine gas in pure form , is colorless and odorless, but after AlP reacts with moisture and stomach acid and is released due to structural and chemical changes, it smells bad like rotten fish or garlic (Figure 2).
In recent years, many cases of poisoning with high mortality rates due to contact with aluminum, zinc and calcium phosphides have been reported, except in rare cases, which were accidental, most of them due to suicide (Figure 3).
Acute and lethal poisonings due to metal phosphides have been reported more than developing countries such as India, Sri Lanka, Iran and Jordan, and such poisonings are rare in other countries, especially European countries and the United States. The high rate of poisoning with these agents in these countries can be due to their cheapness and availability (Figure 4).
Epidemiology
Every year, a large number of deaths due to pesticide poisoning are reported worldwide.
Phosphide poisoning accounts for about 68% of the reported causes of poisoning in the Indian subcontinent. However, most reported cases of phosphide poisoning in India are mainly related to agricultural workers. AlP poisoning is a major concern in Iranian society. In previous studies in India and Iran, the ratio of men to women in poisoning with AlP tablets was two to one (Figure 5).
Exposure to aluminum phosphide
It can be said that ALP is the most common cause of poisoning in rural and urban areas of some developing countries such as India, which is usually swallowed for suicide. This substance is also used as a cause of suicide in Iran. There have been reports of poisoning with this substance in other countries due to occupational exposure. Therefore, the two main pathways of exposure to ALP include intentional exposure to suicide and accidental occupational exposure [3].
The amount of toxicity
The results of various studies performed to determine the toxicity of aluminum phosphide such as fatal dose(FT), Permissible exposure limit (PEL), short term exposure limit (STEL) and (LD50)are given in the table below (Table 1).
| Index | Dose | Description |
|---|---|---|
| FD | 150- 500 mg | for a 70 kg adult |
| PEL | 0.3 ppm | over an 8 h shift (for factory stuffs) |
| STEL | 1 ppm | - |
| LD | 400-600 ppm | Lethal in 30 min |
| LD50 in rate | 1.47 g/m3 | during 35–50 min of exposure (inhalation of fumes) |
| LD50 in mice | 0.68 g/m3 | during 65–75 min of exposure (inhalation of fumes) |
| LD50 for cats | 25 ppm | (2–4 h daily during 3 days) (inhalation of fumes) |
Table 1: Toxicity indexes of phosphide aluminum.
Processes of absorption , distribution, metabolism and elimination of ALP
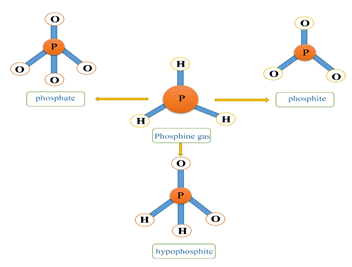
In oral administration of ALP, due to the contact of the tablet with the moisture in the oral cavity or due to contact with gastric acid, due to the hydrolysis of aluminum phosphide, phosphine gas is released which is rapidly absorbed through the gastrointestinal mucosa.In inhalation, phosphine gas is rapidly absorbed through the lungs. Dermal absorption of phosphine gas and aluminum phosphide is negligible. Following absorption, phosphine gas is widely distributed throughout the body. Phosphine irreversibly reacts with hemoglobin in red blood cells to produce methemoglobin.Phosphine gas is converted to inactive metabolites by metabolism. Its most important metabolites are phosphite, phosphate and hypophosphite. Most phosphine metabolites are excreted in the urine. Some phosphine gas is excreted unchanged through the lungs and through the exhaled air [4] (Figure 6).
The signs and symptoms of intoxication are nonspecific and vary according to the duration of exposure and the route of entry into the body of exposure and the amount of phosphine released, and initially include inflammation of the airways and shortness of breath (Figure 7).
Clinical Manifestations
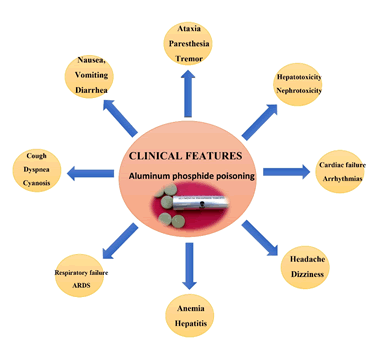
Other features may include dizziness, extreme tiredness, tightness in the chest, headache, nausea, vomiting, diarrhea, ataxia, numbness, paresthesia, tremors, muscle weakness, diplopia, and jaundice. Heart failure, cardiac arrhythmias, seizures and coma, and late manifestations of hepatotoxicity and nephrotoxicity may also occur in some cases. Clinical manifestations of intoxication usually occur within minutes after taking ALP. Mild intoxication usually begins with gastrointestinal symptoms such as nausea, vomiting, diarrhea, headache, abdominal pain, and cardiovascular symptoms such as tachycardia. And these patients usually recover. In moderate to severe poisoning, signs and symptoms in addition to gastrointestinal symptoms affect the cardiovascular, respiratory and nervous systems. Hepatic and renal failure may also occur over time. AlP toxicity specifically affects cardiovascular tissues, including: profound and refractory hypotension, congestive heart failure, electrocardiographic abnormalities (ECG), myocarditis, sinus tachycardia, and peritoneal arrhythmias. Respiratory symptoms may include cough, dyspnoea, cyanosis, pulmonary edema, respiratory failure, and ARDS. Metabolic acidosis may be due to the accumulation of lactic acid. Other unusual findings in ALP poisoning have been reported, include intravascular hemolysis, acute adrenal insufficiency, hepatitis, acute tubular necrosis, pancreatitis, hypoglycemia or hyperglycemia, methemoglobinemia, hemolytic anemia, and disseminated intravascular coagulation [5] (Figure 8).
Laboratory findings
Laboratory evaluation is performed to obtain a prognosis. In cases of severe AlP intoxication, leukopenia is observed. Elevated hepatic enzymes such as serum glutamic-oxaloacetic transaminase (SGOT) or serum glutamic-pyruvic transaminase (SGPT) have also been observed, as well as metabolic acidosis due to moderate to severe AlP intake. Plasma magnesium levels are decreased in AlP poisoning and in some cases a decrease or increase in serum potassium levels has been reported. Laboratory results show an increase in BUN and creatinine levels. Hemolysis is a rare manifestation of this poisoning. One study reported hemolysis due to G6PD deficiency after ALP. Plasma renin levels are important in toxicity because their levels are directly related to mortality and are proportional to the dose of AlP.It has also been observed that serum cortisol levels are usually reduced in severe AlP intoxication. There have been reports of hypomagnesemia, a hypermagnesemia that is associated with cardiac toxicity and extensive focal myocardial destruction. In one study, a positive association was reported between mortality and low serum magnesium levels and red blood cells [5].
Diagnosis and methods of identification and quantification
Diagnosis of AlP intoxication usually depends on clinical symptoms or history (self-reported or by attendants). For accurate management of patients with AlP poisoning, in case of doubt about the clinical signs of AlP poisoning, differential diagnosis can be made using by simple silver nitrate-impregnated paper test on the patient's stomach contents or respiration. The sensitivity of this test is high and it detects about 0.05 ppm. Unlike other reagents, the presence of ammonia and arsenic does not interfere with the results. Chemical analysis of a patient's blood or urine sample is useless to find phosphine because phosphine is rapidly oxidized to phosphite and hypophosphite. However, headspace gas chromatography coupled with mass spectrometry (HS-GC-MS) is the most specific and sensitive method that can be used to analyze airtight samples (viscera and stomach contents) collected in During the autopsy process. Another way to detect phosphine gas is to use a potentiometric method, a semi-quantitative test to determine the amount of phosphine gas. Another test that can be done to diagnose AlP poisoning on stomach contents is the ammonium molybdate test [2].
Treatment of acute poisoning Alp
One of the main treatment strategies is to inhibit the release of more phosphine gas. The acidic contents of the stomach convert AlP into toxic phosphine gas, so diluting the stomach contents with sodium bicarbonate reduces phosphine production. Reducing gastric acid secretion with proton pump inhibitor drugs such as omeprazole can help reduce the effects of AlP poisoning. Oxidation of phosphine to phosphate in the stomach using potassium permanganate is one of the recommended treatment strategies during the first 60 minutes of intoxication. Activated charcoal in the first minutes of poisoning reduces the absorption of phosphide particles, which can delay the onset of toxic effects. In many clinical and laboratory articles, the use of oral coconut oil has been recommended along with common treatments. Due to the fact that coconut oil contains saturated fatty acids, it seems to prevent the release of phosphine gas from AlP due to the physical and chemical properties of AlP. In some scientific sources, gastric lavage has been recommended as an efficient method to prevent the release and further absorption of phosphine [4].
Antidote therapy
so far Aluminum phosphide(AlP) poisoning has no specific antidote. The most important treatment strategy is to use supportive therapies and reduce the toxic effects of higher phosphine uptake. Numerous studies have shown that magnesium sulfate treatment reduces mortality by up to 50%. Research has shown that magnesium levels are reduced in AlP poisoning, so administering magnesium reinforces the hypothesis that improving magnesium levels can be helpful in the treatment of AlP poisoning. It has been suggested that hypomagnesemia may be the main cause of high mortality in patients with AlP poisoning and its correction.It will save the patients. A study examining the effect of magnesium on the recovery of patients with AlP poisoning has shown that there is a significant improvement in patients receiving magnesium. In AlP poisoning, an increase in lipid peroxidation and a decrease in glutathione levels have been observed. Since GSH activity is dependent on magnesium, magnesium deficiency reduces glutathione levels and increases lipid peroxidation. Studies have shown that one of the compounds that has been shown to have beneficial effects in relieving AlP poisoning is melatonin. Melatonin may specifically improve AlP -induced cardiac toxicity. Melatonin is also known as a powerful antioxidant. In addition, melatonin not only suppresses respiratory chain complexes but also increases ATP production. Therefore, melatonin use was suggested as a potentially beneficial approach against AlP -induced cardiotoxicity. Studies have shown that digoxin treatment increases myocardial contraction and blood pressure and improves cardiovascular function. The results of a study suggest that the administration of coenzyme Q10 as an antioxidant can be used as another supportive treatment. They claim that, based on previous studies in patients with heart failure, coenzyme Q10 may also increase systolic heart function. Studies on the positive effect of N-acetyl cysteine (NAC) on the survival rate of AlP poisoned rats(10, 20,40 mg/kg, IP).Administration of N-acetyl cysteine has been shown to reduce Alp-induced myocardial damage and increase survival time(95-98). Antioxidants such as (1000 mg every 12 h via slow IV infusion), vitamin E (400 Units, IM) and NAC (140 mg/kg oral as a loading dose followed by 70 mg/kg oral every 4 h, for up to 17 doses) have been considered as beneficial treatments in patients with AlP poisoning. The results of an animal study in rats showed that fresh RBC transfusion could improve metabolic acidosis due to AlP poisoning and increase survival in rats. It is suggested that an increase in fresh RBC may moderate the acid-base balance or prevent phosphine and hemoglobin chelation.
Conclusion
Metal phosphide is a rodenticide and is used to protect grain in warehouses and fields. Aluminum phosphide is widely used due to its unique properties such as strength, high efficiency, low cost and easy availability. Poisoning with this compound occurs accidentally and intentionally .AlP poisoning is one of the major concerns in Asia. Many studies are underway to find a definitive treatment for poisoning. There are two main obstacles to rescuing the poisoned, one is the delay between being exposure to the toxin (mainly through ingestion) to treatment and the other is the lack of specific antidotes that reduce the chances of rescuing patients. Since no specific antidote has been discovered so far, supportive therapies continue to be emphasized. Preventing poisoning is a fundamental and inexpensive solution.
Suggestions
- Restrict sales to young people and Offering licensed and banned tablet in public stores
- Changes in the shape and structure of the tablet packaging
- Close monitoring of production routes, distribution and consumption of this product and giving sufficient information to families
- Replacing AlP and Phostoxin with other less dangerous pesticides
- Educate farmers on how to use rice tablets and how to maintain them to prevent occupational poisoning.
Conflict of Interest
The authors declare no conflict of interest
Acknowledgement
Author is thankful to the Department of Toxicology and Pharmacology of Kerman University of Medical Sciences, Kerman, Iran.
References
- Kanchan T, Menezes RG, Kumar TM, Bakkannavar SM, Bukelo MJ, et al.(2010) Toxicoepidemiology of fatal poisonings in Southern India. J Forensic Leg Med 17: 344-347.
- Mohammadi AB, Nahandi MZ (2011) An Epidemiological Study of Aluminum Phosphide Poisoning in Patients Admitted in a Specialized Poisoning Referral Center in Northern Iran. Depiction of Health 2: 7-12.
- Chaudhary S, Momin SG, Vora DH, Modi P, Chauhan V, et al. (2013) An epidemiological study of fatal aluminium phosphide poisoning at Rajkot. IOSR J pharm 3: 17-23.
- Nosrati A, Karami M, Esmaeilnia M (2013) Aluminum phosphide poisoning: A case series in north Iran. Asia Pac J Med Toxicol 2: 111-113.
- Siwach SB, Yadav DR, Arora B, Dalal S (1988) Acute aluminum phosphide poisoning-an epidemiological, clinical and histo-pathological study. J Assoc Physicians India 36: 594-696.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi