Research Article, J Hydrogeol Hydrol Eng Vol: 0 Issue: 0
Assessment of hydrographic variables of Salt-pans water of Mumbai, West coast of India
Dr. Anis A. B. Choudhery Miss. Simeen Rumani
Department of Zoology, G.M. Momin Womenâ??s College, Bhiwandi, (Affiliated to University of Mumbai)
Department of Oceanography, Sindhu Swadhyay Sanstha (Integrated school of aquatic education), The University of Mumbai. Maharashtra, India.
*Corresponding author: S Rumani Department of Oceanography, Sindhu Swadhyay Sanstha (Integrated school of aquatic education), The University of Mumbai. Maharashtra, India.; email: rumanisimeen@gmail.com.
Citation: Rumani S. Anis , Choudhery, et al. (2021) Assessment of hydrographic variables of Salt-pans water of Mumbai, West coast of India. J Hydrogeol Hydrol Eng
Abstract
The monthly variation of the hydrographic variables of Salt-pans water of, Mumbai, west coast of India was investigated during a complete salt-production period. Ecologically significant parameters like water temperature, Salinity, pH value, dissolved oxygen (DO), carbon dioxide (CO2), Biochemical oxygen demand (BOD), phosphate, nitrate, nitrite, and silicate were analysed. Samples were collected bimonthly from November 2013- to June 2014 from the selected site of different types of pans (regions) of the Salt production area. The results indicate that hydrological parameters of Salt-pans are very dynamic in nature. It is a great journey of moderate saline, nutrient-enriched brackish water to transform into one of the utmost hypersaline, dead zone aquatic ecosystems. Dissolved oxygen found to be circulated in the aquatic system of different pans of salterns with Standard Deviation of mean value (SD) 1.7188 which is very high, similarly nutrients like Nitrate with SD 0.840, Nitrite SD 7.48, Phosphate SD 3.3278, Silicate SD 1.66. Maximum deviation observed in two factors, temperature ranges from 26C0 to 53 C0 (SD 10.237) and salinity ranges from 22 ppt to 535 ppt. (SD 139.124).
Keywords: Hypersaline, Salt production, hydrographic variables , correlation
Introduction
Salt exists in all animal and plant lives. Salt is the mineral of universal distribution. Nature seems to have been generous in the supply of this unique mineral. It is available in plenty in the seas, in spring, in varied other parts of the world. Human cultivation of salt is one of the ancient activities, and the initial known salt harvesting is believed to have occurred at Lake Yuncheng, in the Chinese province of Shanxi around 6000 BC.
Even though salt has been used in a range of ways, one of the most acknowledged, salting fish to preserve it appears in the proceedings of the Xia Dynasty by about 2000 B.C. (The Indian Economist, November 2020). There are a number of ways by which salt can be produced, but in Eastern North America Indians obtained most of their salt from salines located far from the coast. A few groups, such as the Chitimacha Indians of Louisiana, did indeed boil seawater, but this practice was rare in Eastern North America as a whole. Written records illustrate the manufacture and trade of sea salt in China, as well, and date to 1800 BC. The Chinese process involved “putting ocean water in clay vessels and boiling it until reduced to pots of salt crystals.” By 450 BC, innovator Yi Dun was boiling brine (salty water) in iron pans to distill salt. (Mark Kurlansky 2003 ).
The salt springs of western Arkansas and northwestern Louisiana were well-known to the eighteenth-century Indians, and many groups frequented these areas to produce and trade salt. This mineral was extremely important in the trade between the French and the various Caddoan groups (Clarence H. 1978). The Egyptians began trading salt (in the form of salted fish) to Middle Eastern societies like the Phoenicians circa 2800 BC. The Phoenicians, in turn, traded with everyone else around the Mediterranean. Other Lower Mississippi Valley groups heavily involved with the salt trade were the Quapaw, Korea, Taensa, and Tunica. (Department of the Arts of Africa 2000).
The salt is made along by a river, which, when the water goes down, leaves it upon the sand. As they cannot gather the salt without a large mixture of sand, it is thrown together into certain baskets they have for the purpose, made large at the mouth and small at the bottom. These are set in the air on a ridge-pole; and water being thrown on, vessels are placed under them wherein it may fall; then, being strained and placed on the fire, it is boiled away, leaving salt at the bottom (Elvas 1904).
The manufacture and trade of sea salt in China, as well, and date to 1800 BC. When the Romans moved their salt-works away, they also built their first road, the Via Salaria (or Salt Road).
India is the third largest Salt producing Country in the World after China and USA with Global annual production being about 230 million tonnes. The production of salt during 1947 was 1.9 million tones which have increased tenfold to record 30 million tones during 2018-19 as per Annual Report of Salt-Department GOI (2018-19).
Salt industry occupies a prominent role in the rural economy. Around 1.28 lakh persons are employed directly by the salt industry functioning in our country. Salt plays an important role in the human diet and is also widely used for preserving food. It makes possible the manufacture of glass, soap, plastic, medicine, etc., and others in the petrochemical industry.
In India salt manufacturing are carried out in the coastal states of Gujarat, Tamil Nadu, Andhra Pradesh, Maharashtra, Karnataka, Orissa, West Bengal Goa, and hinterland State of Rajasthan. The Private Sector plays a dominant role contributing over 95% of the salt production, while the public sector contributes about 2-3%. The co- operative sector contributes about 8% whereas the small-scale sector (less than 10 acres) accounts for nearly 40% of the total salt production in the country as described in Economy watch on June 29, 2019.
SALT PANS:
318,718 acres of coastal land in India are used for salt-production. Out of which 275,244 acres are recognized recognised by Salt Commissioners Organisation (SCO) and 43,474 acres of land are belongs to un-recognized units of the SCO. The procedure of issuing licenses was stopped in 1996 and units that came into existence thereafter are not registered and are considered as un-recognized units by SCO.
A salt pan is created when pools of seawater evaporate at a rate faster than it is replenished by rainfall. As the water evaporates, it leaves behind the minerals precipitated from the salt ions dissolved in the water.
These coastal water forms the most productive zone and is heavily exploited for renewable as well as non-renewable resources. Saltpans are extensive, man-made hyper saline habitats that are of great importance for migratory shorebirds owing to the high productivity and predictability in time and space, as well as their shallow depth.
THE PRESENT WORK:
Traditionally salt farming in Mumbai has been practiced for the past 1,500 years. In Mumbai saltpans occupies 5378 acres at 13 different pockets across the eastern suburbs of Ghatkopar, Chembur, Wadala, Kanjurmarg, Bhandup, Mandale, Turbhe, Nahur, and Mulund, and the western suburbs of Dahisar, Mira Road, Bhayander, Malvani, and Vihar. Close to 400 acres of salt pans may have been lost due to encroachments about four decades ago when sprawling residential colonies were built mainly in the eastern suburbs. These include residential enclaves such as Garodia Nagar in Ghatkopar, Chheda Nagar in Chembur, and Bangur Nagar in Goregaon and Star Builder in Bhandup. A further 44 acres are encroached upon by slums, while 359 acres are either lost in litigation or have ownership disputes. Another 483 acres were acquired by the state government to build the Eastern Express Highway, a sewage plant, and a cemetery; and 187 acres were given to the central government to build staff quarters as per times of India (2016).
The structure of salt pans consists of a series of rectangular beds, each bed bounded on all four sides and joined to the next one through an opening in the common bund. There are five different regions or zone or types of pans in salt-production areas.
Region1 or R1: -Main reservoirs: It is the main storage of the creek water it is large and vast area generally occupying 36.0 acres but varies from place to place. The size of Jenkin salt works, which is about 450 x 80 meters. The water depth is 31 to 41 (90 x 120 cms). The creek water enters this region and stockpile at least for 15 days for till Beaume numbers (0Be1) increase 00 to 30 (duration may vary according to climate condition) then it pours to the next block.
Region 2 or R2: - Sub reservoir:
These are numbers of smaller reservoirs having sizes about 110 x 30 meters and water height about 2’ to 2.5’ (60 to 75 cm). It accepts water from main reservoirs in these regain beaume number (0Be1) increases up to 60 resulting 50% decreases in the volume of brine water.
Region 3 or R3: Condenser:
These are a parallel arranged number of blocks holding brine for condensation. The sizes are 8 x 2 meters. The depth of water varies 0’ to 1’ (30 cm), density increases very rapidly in this area reaches up to 140 Be and while the volume of water reduces up to 10%.
Region 4 or R4: Feeding Channel: It is a narrow 2 to 3 feet broad and 100-meter-long channel having a depth of about 2 feet (60 cm). Here density increases up to 300 Be, with very heavy water passing in the final zone i.e. Crystallizers.
Region 5 or R5: Crystallizers: It is water tide landmasses having sizes of about 12 x 15 feet (3.6 x 4.5 meters) and depth is about 4 inches (10 cms). Numbers and sizes vary one salt works to other salt works. Beaume number fluctuates between 250 Be. to 300 Be. The crystallized NaCl (salt) is racked to the platform of the pan and is allowed to drain if beaume number exceeds 300 then such brine is discarded since it may include crystallization of other salts such as magnesium chloride magnesium sulfate, calcium sulfate, etc. Fresh brine is received to the crystallizer and the process is continued from December to early June.
In principle, the operation is not basically different from the one described previously. Differences could be seen only in optimization, brine transfer, and collection of salt due to some technological progress. It is because the whole process relies on natural energy, solar energy. Salt curing in saltpans commences from November and ends in June.
Ecologically the salt-pans are very active. It has far different hydrological features compared with another aquatic ecosystem. It is known for the most hypersaline ecosystem, extremely variable salinity, temperature, and nutrient richness. The natural aquatic system has very different equations and correlations
The purpose of the study was to study the hydrological features of salt- pan and to calculate the correlation between different hydrological factors
Material and Methods
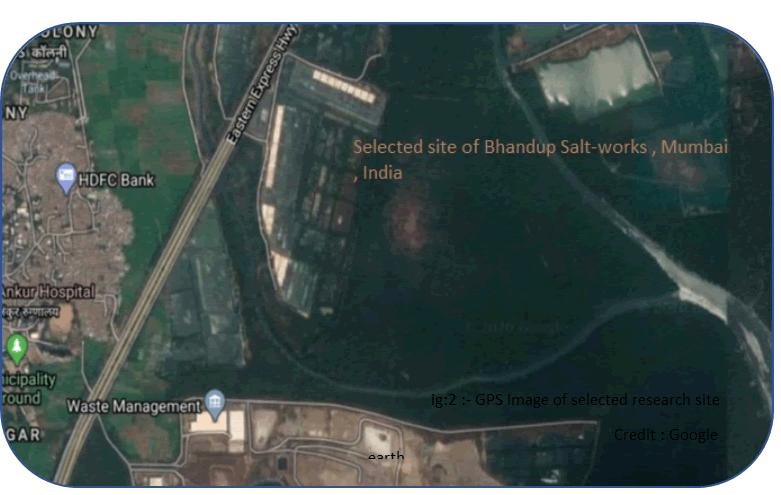
The present study was conducted in Jenkin salt works which lay both sides of the eastern highway near Thane creek and lie extending between that Latitude and longitude coordinates of selected are 19.1488° N, 72.9626° E. It is a part of Bhandup Salt-work, located at Bhandup, a suburban township of Greater Mumbai (figure-1 & 2). It is a perennial Salt-pans, surrounded by a huge lush green mangrove population. The physicochemical status of salt-pans was studied monthly for one salt-production period from November 2013 to June 2014. Water samples were collected bimonthly from five different regions. Water temperature was recorded by a standard centigrade thermometer on site. For the estimation of dissolved oxygen and other hydrological analysis were collected separately in 300 ml BOD bottles and oxygen was fixed at the time of sampling on the field.
The analyses were carried out for temp, pH, salinity, dissolved oxygen, free dissolve CO2. BOD, phosphate, nitrite, nitrate, and silicate. The parameters of physicochemical are determined by standard methods prescribed by American Public Health Association (APHA) (1999), Trivedy and Goal (1986), Kodarkar (1992) and ASTM (2003).
pH of the surface water in the selected sampling station was measured during high tide condition with a portable pH meter (sensitivity = ± 0.02).
Statistical analysis: The results were calculated as the mean value of the triplicate tests with the standard deviation. One Way analysis of variance (ANOVA) was applied to test for significant differences at p<0.05. The correlation matrix (Pearson’s correlation coefficients) was carried out by SPSS statistical software (Version 16.0 for Windows). The standard deviation, mean and correlation coefficient are estimated as by Ratner.B. (2009).
Result and discussion
The trophic status of the salt pan is influenced by the variation of the hydrological factors. There is a constant interaction between the biotic and abiotic components in the saltpans. In the context of the present study; the baseline information of the Physico-chemical characteristics of water would form a useful means for further ecological evaluation and monitoring of these saltpans.
Temperature:
A large number of ecological processes in the freshwater bodies is influenced by temperature. Graham et al., (1982) had shown the water temperature to regulate photosynthesis in the aquatic ecosystems.
Water temperature has a direct effect on most ecological processes in rivers, lakes, and wetlands, including photosynthesis, respiration, rates of growth and development, and ecological interactions. (B.T. De 2009) Water temperature had been regarded as a limiting factor in the development of zooplankton (Moore, 1978) and plays an important role in controlling the occurrence and abundance of algal diversity (Nazneen, 1980). Hoff and Ibara, (1977) reported a close correlation between water temperature and fish abundance and diversity in the Slocum River Estuary, South Eastern, New England.
In an established system, the water temperature controls the rate of all chemical reactions and affects fish growth, reproduction, and immunity. Drastic temperature changes can be fatal to fish. The lowering of temperature during monsoon was probably due to high precipitation, decreased solar radiation, presence of cloud, and low atmospheric temperature (Gopinath, 1985). It is important in the aquatic system because it can cause mortality and it can influence the solubility of D.O. and CO2.
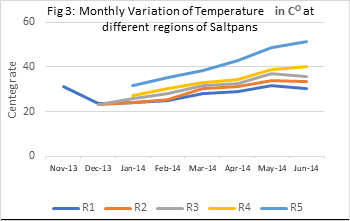
The pattern of seasonal variation in water temperature recorded in different regions of saltpans chosen for the present study resembles that of atmospheric temperature. In the present study, the minimum average monthly temperature was recorded 23.2 ºC at R3 areas in the month of December 2013 and the highest monthly temperature recorded 51.35 ºC at R5 in the month of June 2014 with SD 10.237. All the surface water temperature recorded between 10 am to 11 a.m.
Table 1: : Monthly Variation of Temperature in CO at different regions of Saltpans
| Months | R1 | R2 | R3 | R4 | R5 |
|---|---|---|---|---|---|
| Nov-13 | 31.25 | ||||
| Dec-13 | 23.55 | 23.3 | 23.2 | ||
| Jan-14 | 23.85 | 24.15 | 26 | 27.3 | 31.4 |
| Feb-14 | 24.75 | 25.55 | 28.25 | 30.2 | 35.1 |
| Mar-14 | 28.2 | 30.3 | 31.55 | 33.05 | 38.35 |
| Apr-14 | 29 | 31 | 32.45 | 34.4 | 42.7 |
| May-14 | 31.4 | 34 | 37 | 39 | 48.55 |
| Jun-14 | 30.45 | 33.5 | 35.55 | 40.1 | 51.35 |
The coefficient correlation study reveals that water temperature plays a very important role in the distribution of other hydrological factors. It was found that it showed a significant positive correlation with salinity (0.784), and for the rest of all the factors, it showed significant negative correlations. Among most significant negative correlations with temperatures were found Silicate (-0.536), CO2 (-0.455), and BOD (-0.405).
pH:
pH is an important hydrological parameter, it regulates the life processes, nutrient availability, and biological productivity. pH levels can decline in estuarine systems to levels that can adversely affect biological responses. The potential impacts on biological resources of even moderate decreases in pH, particularly in systems that naturally tend to have lower pH conditions, maybe more important than previously realized (Harris et.al 1999)
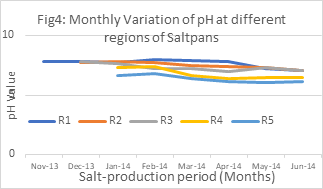
In the present investigation, pH is varied between 6.1-7.9 range in all the 5 regions for an entire salt-production period of 8 months. The pattern of seasonal variation in pH recorded from R1 to R5 fluctuated between 6.1 at R5 (June 2014) to 7.9 at R1 (March 2014) with mean SD 0.49295.
Table 2:Monthly Variation of pH at different regions of Saltpans
| Months | R1 | R2 | R3 | R4 | R5 |
|---|---|---|---|---|---|
| Nov-13 | 7.82 | ||||
| Dec-13 | 7.8 | 7.75 | 7.77 | ||
| Jan-14 | 7.75 | 7.8 | 7.65 | 7.3 | 6.6 |
| Feb-14 | 8 | 7.7 | 7.2 | 7.4 | 6.8 |
| Mar-14 | 7.9 | 7.5 | 7.25 | 6.65 | 6.4 |
| Apr-14 | 7.85 | 7.4 | 7 | 6.4 | 6.15 |
| May-14 | 7.25 | 7.3 | 7.3 | 6.5 | 6.05 |
| Jun-14 | 7.05 | 7.05 | 7.05 | 6.45 | 6.1 |
showed a significant positive correlation with DO (0.994), CO2 (0.986) and 0. 973, and negative significant correlation with temperature (-0.309), and salinity (-0.82).
Salinity:
Salinity is the saltiness or amount of salt dissolved in a body of water. It is the measure of all dissolved salts, It has major influences on species richness and diversity of the salt pan ecosystem. In the reclaimed saltpans at the Berg River estuary, South Africa, Velasquez, (1992) observed the quality of such artificial wetlands as foraging sites for shorebirds and wading birds depend largely on the abundance of benthic macrofauna which in turn was determined by salinity. Salinity governs physical characteristics like the density and heat capacity of the water. The high value of salinity in water and soil may cause native vegetation to become unhealthy or die and lead to a decline in biodiversity through the dominance of salt-resistant species, potentially altering ecosystem structures.
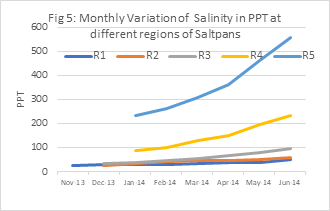
Substantial variation in the value of salinity recorded in saltpans with very high mean SD i.e.139.124. In the present research, the minimum average monthly salinity 24.47 ppt recorded at R1(November 2013) and the highest value observed 556.22 ppt at R5 (June 2014).
Table 3: Monthly Variation of Salinity in PPT at different regions of Saltpans
| Months | R1 | R2 | R3 | R4 | R5 |
|---|---|---|---|---|---|
| Nov-13 | 24.47 | ||||
| Dec-13 | 27.85 | 27.7 | 32.35 | p-value | p-value |
| Jan-14 | 28.44 | 34.43 | 39.73 | 88.95 | 232.49 |
| Feb-14 | 31.95 | 40.35 | 48.15 | 101.46 | 260.5 |
| Mar-14 | 33.75 | 45.58 | 54.9 | 128.7 | 309.05 |
| Apr-14 | 36.55 | 47.85 | 69.1 | 149.67 | 360.35 |
| May-14 | 39.27 | 49.99 | 78.01 | 194.5 | 460.26 |
| Jun-14 | 50.63 | 57.65 | 95.01 | 234 | 556.22 |
Salinity showed a significant positive correlation with temperature (0.784), and a significant negative correlation with all the other recorded hydrographic parameters. It showed a negative significant correlation with silicate (-0.8930) , CO2 (-0.889), and BOD (-0.863).
Dissolved Oxygen
According to Wetzel and Likens, (1979), among all the abiotic factors, dissolved oxygen is the most important factor in aquatic life as it provided valuable information about the biological and biochemical reactions going on in waters. According to Wetzel, (1975) dissolved oxygen is very essential for the metabolism of all aquatic organisms that process aerobic respiratory biochemistry.
During all the months of the study, it was observed that the dissolved oxygen of salt pans was always higher during the month of November as compared to in the month of June. The result establishes that dissolved oxygen had a definite relationship with the salinity. As the salinity increased, the dissolved oxygen content of the water decreased. This falls in line with the studies carried out by Javor (1989).
Oxygen molecules dissolved in water is the major indicator of water quality. Oxygen levels that remain below 1-2 mg/l for a few hours can result in large fish kills. The concentration of DO in water is affected by the physical and chemical properties of the water. Two of the key factors that influence DO concentration are temperature and salinity, which influence the solubility of oxygen in the water. The dissolved oxygen content of water is largely controlled by the balance between input and consumptive metabolism (of oxidizable matter received). Anthropogenic influences (industrial-including deforestation, agricultural and municipal wastes) tend to load the latter scale by the addition of organic effluents.
Its correlation with water body gives direct and indirect information e.g. bacterial activity, photosynthesis, availability of nutrients, stratification etc. (Premlata Vikal, 2009). During summer, the long days and intense sunlight seem to accelerate photosynthesis by phytoplankton, utilizing CO2 and giving off oxygen. This possibly accounts for the greater qualities of O2 recorded during summer (Krishnamurthy R, 1990).
The results of present investigation showed minimum average monthly DO of 0.15 mg/l at R5 (June 2014) and the highest average monthly DO 4.62 mg/l at R1(November 2013) with SD 1.7188.
It showed positive significant correlation with Nitrate (.998), CO2 (.990) and pH (0.994), and significant negative correlation with salinity (-0.856) and temperature (-0.386).
Free CO2: Carbon dioxide is the end product of the organic carbon degradation in almost all aquatic environments and its variation is often a measure of net ecosystem metabolism (Smith 1997,1993, Hopkinson 1985). Consequently, in aquatic biogeochemical studies, it is advantageous to determine parameters that characterize the carbon dioxide. CO2 is also the most important greenhouse gas on Earth. Its fluxes across the air-water or sediment- water interface is among the most important concerns in global change studies and are often a measure of the net ecosystem production/metabolism of the aquatic system. CO2 is also the most important greenhouse gas on Earth. Its fluxes across the air-water or sediment-water interface are among the most important concerns in global change studies and are often a measure of the net ecosystem production/metabolism of the aquatic system.
The monthly variation in CO2 recorded in different regions of saltpans varied with SD of 1.213746. The present investigation showed minimum CO2 1.1 mg/l at R5 (June 2014) and the highest CO2 observed 4.75 mg/l at R1 (February 2014).
It showed significant positive correlation with DO (0.99), Nitrate (.971), and PO4 (0.986) and significant negative correlation with Salinity and temperature, -0.856 and – 0.386 respectively.
BOD: It is a measure of organic material contamination in water, specified in mg/L. BOD is the amount of dissolved oxygen required for the biochemical decomposition of organic compounds and the oxidation of certain inorganic materials (e.g., iron, sulphites). Biochemical Oxygen Demand is an important water quality parameter because it provides an index to assess the effect discharged wastewater will have on the receiving environment. The higher the BOD value, is the greater the amount of organic matter or “food” available for oxygen consuming bacteria. Biochemical oxygen demand (BOD) forms the key indicator of organic load in any wastewater system.
The pattern of monthly variation in BOD recorded, it was found the minimum monthly average 0.02 mg/l in R5 (June 2014) and the highest 6.15 mg/l in R3 (February 2014) with mean SD value 2.673655.
BOD showed significant positive correlation with DO (0.927), silicate (0.927) , CO2 (0.918) , and significant negative correlation with Salinity (-0.863) and Temperature ( -0.425).
Phosphate:
The distribution and behavior of nutrients in the coastal water is influenced by local conditions such as rainfall, the quantity of freshwater inflow, tidal incursion and biological activities such as phytoplankton uptake and regeneration (SATPATHY et al., 2010) and also by human activities (CHANG et al., 2009).In the present
study, coastal waters of all the four stations studied showed a greater load of nutrients. Similar observations have also been made by KAMALKANTH et al. (2012) on the East coast of India. Phosphate is the most important inorganic nutrient that can limit the phytoplankton production in coastal ecosystems. In this study, it showed a high value during the post-monsoon and a low one during monsoon periods. The variation in phosphate content depends upon its concentration in the freshwater that mixes with the seawater, phytoplankton uptake, addition through localized upwelling, adsorption and desorption of phosphate, buffering action of sediment under varying environmental conditions and replenishment as a result of microbial decomposition of organic matter (POMEROY et al., 1965; SATPATHY et al., 2010). In biological systems, phosphorus is found as a free phosphate ion in solution and is called inorganic phosphate, to distinguish it from phosphates bound in various phosphate esters. Phosphates are the naturally occurring form of the element phosphorus, found in many phosphate minerals. In mineralogy and geology, phosphate refers to a rock or ore containing phosphate ions. Inorganic phosphates are mined to acquire phosphorus for use in agriculture and industry. In ecological terms, because of its important role in biological systems, phosphate is a highly sought-after resource. Once used, it is often a limiting nutrient in environments, and its availability may govern the rate of growth of organisms. This is generally true of freshwater environments, whereas nitrogen is more often the limiting nutrient in marine (seawater) environments. In the context of pollution, phosphates are one component of total dissolved solids, a major indicator of water quality, but not all phosphorus is in a molecular form that algae can break down and consume. Phosphate deposits can contain significant amounts of naturally occurring heavy metals. Mining operations processing phosphate rock can leave tailings piles containing elevated levels of cadmium, lead, nickel, copper, chromium, and uranium. Unless carefully managed, these waste products can leach heavy metals into groundwater or nearby estuaries. Uptake of these substances by plants and marine life can lead to the concentration of toxic heavy metals in food products. Phosphorus in the form of phosphate (PO4 -p) is an essential plant nutrient and is a major component of most fertilizers. Erosion and runoff can result in large amounts in bodies of water and lead to eutrophication, the depletion of oxygen from excessive aquatic growth. Animal die-off, toxins, and foul water can also result. These are some of the primary reasons that the monitoring of phosphate levels in water is extremely important. Anjane yalu and Vasanthi, (1989) observed the phytoplankton abundance in the Penguin fish farm in the periphery of the lake Kolleru, Andhra Pradesh, India to be correlated with nitrites, nitrates and phosphates. Phosphorus had been regarded as the major limiting nutrient of the water body by Heilman, (1968); Klopaters, (1978) and Brown, (1981). Further phosphate was considered to be the important nutrient limiting the growth of phytoplankton (Welch et al., 1978). A positive correlation between phosphates and algae had been reported by Kramer et al., (1979) and Sarkar et al., (1985). Murphy et al., (1984) had also found the nitrates and phosphates to influence the waterfowl populations. The current hydrological analysis showed minimum PO4 -p value of
0.15 ppm in R5 ( June 2014) and the highest PO4 -p value 7.55 ppm at R1 (December 2013).
It was found to have a significant positive correlation with nitrite (0.999), Nitrate (0.993), DO (0.985), and a significant negative correlation with salinity (-0.807) and Temperature (-0.365).
Nitrates
Variations in nitrate and its reduced inorganic compounds are predominantly the results of biologically activated reactions (KATHIRAVAN etal., 2013). The high concentration of nitrate was recorded during the pre-monsoon period which could be attributed to higher the activity of the oxidation of ammonia from nitrogen to nitrite and subsequently to nitrate during the summer
season and the enrichment of terrigenous deposit with a lot of nutrients as the terrestrial runoff during the monsoon and subsequent post-monsoon seasons (KALAIARASI et al., 2012). Usually, nitrates are built up during the dry season and high levels of nitrates are observed only during early rainy seasons (WOLFHARD; REINHARD, 1998) In an aquatic environment, nitrogen is present in combined forms of ammonia, nitrite, nitrate, urea, and dissolved organic compound; Nitrate is the oxidized form of nitrogen and usually is the most abundant, found in the combined form of inorganic nitrogen in any water body. It is an important nutritional factor and it’s quality in any water resource indicates the productivity of the system. In natural waters, the nitrate originates mainly from the oxidation of NH3 by nitrifying bacteria. According to Jhingram (1991) the values of dissolved organic nitrogen below 0.1 mg/L may be considered as indicative of poor productivity, those in the range 0.1 to 0.2 mg /l of average productivity and those above 0.2 mg/l as favorable for productive ponds.
Biological oxidation of organic nitrogenous substances present in domestic and industrial sewage and nitrifying bacteria and nitrates to the water body. Utilization by green plants and denitrifying bacteria help in the reduction of nitrates. Ganapati (1960), pointed out that tropical water particularly non-polluted are deficient in nitrate content.
The pattern of monthly variation in NO3-N recorded with SD 0.840833. In the present study, the minimum NO3-N recorded 0.15 mg/l at R5 (June 2014) and the highest NO3-N , observed 3.6 mg/l at R1 (November 2013)
It showed a significant positive correlation with DO and Silicate (0.988) and a significant negative correlation with Salinity (-0861) and Temperature (-0.309).
Nitrite:
Nitrogen is one of the important constituents of cells, responsible for the growth and development of phytoplankton and ultimately of primary production. Nitrite is an intermediate oxidative state of nitrogen, during the process of oxidative ammonia to nitrate and in the reduction of nitrate. Reduction of nitrate by facultative and anaerobic denitrifying bacteria such as Pseudomonas and oxidation of ammonia compounds by aerobic nitrifying bacteria like Nitromonas are chiefly involved in the formation of nitrite in the water body. Nitrite is unstable as an intermediate stage in the nitrogen cycle. Nitrite-Nitrogen is unstable and easily oxidized to the nitrate form. The nitrite concentration is much lower in natural wastes as compared to nitrates. It is essential to assess the nutrient load and their respective concentration in aquaculture practices.
The monthly variation recorded in NO2-N with SD 7.479765. In the present investigation, the minimum NO2-N recorded 0.15 mg/l at R5 (June 2014) and the highest NO2-N observed 3.58 mg/l in R1 (December 2013).
It showed a significant positive correlation with PO4 (0.999), DO (0.986) CO2 (0.965) and significant negative correlation with Salinity (-0.813) and Temperature (-0.371).
Silicates:
The Spatio-temporal variation of silicate in coastal water is influenced by several factors, most importantly the proportional physical mixing of seawater with freshwater, adsorption of reactive silicate into suspended sedimentary particles and biological removal by phytoplankton, especially by diatoms and silicoflagellates (ASTON, 1980). The higher value of silicate observed during the monsoon period in this study maybe due to the discharge of freshwater rich with dissolved silicate into the coastal water (PURUSHOTHAMAN; VENUGOPALAN, 1972). The lower values of silicate observed during the post- monsoon and pre-monsoon periods could be due to the uptake of silicates by phytoplankton for their biological activity (LISS; SPENCER, 1970; ASTON, 1980), which is confirmed by the negative correlation of silicate with phytoplankton community. The silicate content observed was higher when the salinity was low and this maybe due to heavy the influx of freshwater derived from the surrounding drains carrying silicate leached out of rocks. A similar result was also reported by KAMALKANTH et al. (2012) and this is evident from the significant negative correlation of silicate content with salinity.
Silicates are the most widespread of the minerals. They are made up of oxygen and silicon, the number one and number two most abundant elements in the earth’s crust. By themselves, they make up over 90% of the weight of the earth’s crust. Most rocks are composed mainly of this class of minerals. Silicates are well characterized as solids but are less commonly observed in solution. The anion SiO4−4 is the conjugate base of silicic acid, Si(OH)4, and both are elusive as are all of the intermediate species. Silicon is known to be present in all living organisms. This element occurs in the form of hydrated amorphous silica, referred to as opal, and is required for the production of structural materials in single-celled organisms through to higher plants and animals. For many life forms, silicon can even be an essential element. Water moving over and through natural deposits will dissolve a small amount of various silicate minerals, making silicates a common contaminant of most waters. Natural physical and chemical weathering processes also produce many extremely small particles or colloids of silicate minerals. Thus, silicates are commonly found suspended in most water sources, usually at low concentrations.
Increased silicate concentrations will affect the water ecosystem, but the effects and their intensity depend on the local situation, e.g., season, phosphorus, nitrogen and silicon concentration of the receiving water, and composition of the effluent (silicate or silicate in combination with phosphorus and nitrogen). Changes in ecosystems are generally regarded as undesirable. However, silicate will not only affect the ecosystem processes, but also the possibilities for the use of the water system by humans. Depending on the use functions of the water system, these effects can be positive as well as negative.
The monthly variation of silicate with SD 1.660723 recorded from different regions of saltpans. The minimum value of silicate recorded 0.02 mg/l R5 (June 2014) and highest silicate 6.15mg/l R3 (February 2014.
Silicate showed significant positive correlation with NO3 (0.988), NO2 (0.967) PO4 (0.967), and significant negative correlation with Salinity -0.893 and Temperature (-0.536).
Conclusion:
Low dept of water, excessive exposures to sunlight, and high atmospheric gaseous interventions provide great variations to the aquatic ecosystem of saltpans. Sampling areas belonged to different pans and have shown high monthly variations, which do not occur in natural climate. In the span of 8 months salinity increased 20 times and Do reduces from 4.5 mg to a negligible amount of 0.15 mg/l. It was also observed that similar types of pans do not show great variations and parameters are found with little variation.
They depend on several factors such as local climatic conditions, days of sampling, time of sampling etc. we can observe very consistent persistence of nutrients and other factors in the first three types of Saltpans i.e. R1, R2, and R3, and it could better place economic activities related to aquaculture.
Coefficient correlation analysis is very significant and we have found that salinity plays a most significant role in a dominant hydrological factor, controlling all other parameters except Temperatures. All other parameters are significantly correlated with each other.
In the present study, lower salinity value, and temperature recorded in R1, R2 and R3 areas, which showed a very good amount of nutrients and other elements. High temperature and salinity in R4 and R5 areas were found due to high evaporation rate and very high atmospheric temperature in the months of May and early June.
Dissolved oxygen (DO) is essential to most aquatic organisms and is greatly affected by their metabolism (TOULIABAH et al., 2002). Seasonal variation in dissolved oxygen is due to freshwater flow and the terrigenous impact of sediments (KUMAR et al., 2009). But in saltpans both conditions do not exist, DO depends on the primary preproduction and atmospheric mixing of oxygen, still, a good amount of DO was found in the first three reasons of saltpans. In the present study, dissolved oxygen showed an inverse trend to temperature and salinity. Several studies have reported that the greater solubility of oxygen might be due to low temperature and low salinity values (ROCHFORD, 1951; SATPATHY, 1996; PUTHIYA et al., 2009). This
is well explained by the significant negative correlation of dissolved oxygen with salinity and temperature, similar observation has been made by the investigator.
The distribution and behavior of nutrients in the coastal water is influenced by local conditions such as rainfall, the quantity of freshwater inflow, tidal incursion, and biological activities such as phytoplankton uptake and regeneration (SATPATHY et al., 2010) and also by human activities (CHANG et al., 2009). In the present study,
R1, R2 and R3 showed a greater load of nutrients. The high concentration of nutrients in R1 and R2 areas due to runoff containing the pollutants and fertilizers from the nearby dumping ground and another surrounding area paddy fields, sewage, and anthropogenic wastes.
The increase in temperature and rapid mixing of sub-surface and surface water during the pre-monsoon season might have favored the nitrate replenishment mechanism (RAINA et al., 1984). The same phenomenon has been observed in saltpans. Phosphate is the most important inorganic nutrient that can limit phytoplankton production in coastal ecosystems. In this study, it showed a high value in R1, R2, and R3 areas, during the month of December, January, and February. The Spatio-temporal variation of silicate in coastal water is influenced by several factors.
The higher value of silicate observed in R1 and R2 regions. It may be due to the discharge of freshwater rich with dissolved silicate into the coastal water (PURUSHOTHAMAN; VENUGOPALAN, 1972). The silicate content observed was higher when the salinity was low and this may be due to the heavy influx of freshwater derived from the surrounding drains carrying silicate leached out of rocks. A similar result was also reported by KAMALKANTH et al. (2012) and this is evident from the significant negative correlation of silicate content with salinity. The present investigation of the hydrographic variables of saltpans will provide useful information for further ecological assessment for economic development and blue economy perspective which is the utmost requirement for self-reliant India.
References
- Anjaneyalu, S.R., and Vasanthi, R. (1989): Ecological studies in Penguin fish farm located in the periphery of lake Kolleru (Andhra Pradesh, India). Pages. 42. In Proc, of seminar on wetland ecology and management, Bom. Nat. Hist. Soc., Keoladeo National park, Bharatpur, 23 â?? 25. February 1990. 154pp
- Annual Report 2018-19 Salt-Department Government of India .
- American Public Health Association (APHA-2012) Standard Methods for the Examination of Water and Waste
- ASTM International, (2003): Annual Book of ASTM Standards, Water and Environmental Technology (West Conshohocken, Pennsylvania, 01, 6-7).
- Aston, (1980) Nutrients dissolved gasses and general biochemistry in estuaries. Chemistry and Biogeochemistry of Estuaries. Wiley, New York,.233-262 p.
- T. De Stasio, T. Golemgeski, D.M. Livingstone (2009) Temperature as a Driving Factor in Aquatic Ecosystems Encyclopedia of Inland Waters, Pages 690-698
- Brown, L. (1981): Comparison of the structure, primary productivity and transpiration of Cyperus ecosystem in Florida. Ecol. Monogr. 51: 403-427.
- Chang, K. H.; Amano, A.; Miller, W.; Isobe, T.; Maneja, R.; Siringan, f. p.; Iimai, h.; Akano, (2009) S. Pollution study in Manila Bay: eutrophication and its impact on plankton community. Interdisciplinary Studies on environmental Chemistry â?? Environmental Research in Asia. Terra pub,.261-267 p.
- Department of the Arts of Africa, Oceania, and the Americas. (2000) ,â??The Trans-Saharan Gold Trade.â? http://www.metmuseum.org/toah/hd/gold/hd_htm (October 2000).
- Economywatch June 29, 2019
- Elvas in Bourne, (1904): Bourne, Edward G. (editor) (1904) Narratives of the career of Hernando de Soto, 2 vols. A.S. Barnes & Co. New
- Ganapathi V. (1960): Ecology of tropical waters. Proc. Symp. Algology. ICAR, New Delhi, pp: 204-218.
- Gopinathan, P., Rodrigo, 1. X., Mohamed Kasim, H. and Rajagopalan, S. (1994): Phytoplankton pigments in relation to primary production and nutrients in the inshore waters of Tuticorin, southeast coast of India. Indian J. Mar. Sci., 23 : 209-212.
- Graham, J.M., Auer, T., Canale, R.P., and Hoffman. J.P. (1982): Ecological studies and mathematical modelling of Cladophora in lake Huron; 4. Photosynthesis and respiration as functions of light and temperature. J. Great Lakes Research. 8: 100 â?? 111.
- Harris, j. o., g. b. Maguire, s. j. Edwards, and M. Hindrum. (1999). Effect of pH on growth rate, oxygen consumption rate, and histopathology of gill and kidney tissue for juvenile greenlip abalone, Haliotis laevigata Dononvan and blacklip abalone, Haliotis rubra Leach. Journal of Shellfish Research 18:611â??619.
- Heilman, D.E. (1968): Relationship of availability of phosphorus and cations to forest succession and log formation in interior Alaska. Ecology. 49: 331 â??
- Hoff, G., and Ibara, R.M. (1977): Factors affecting the seasonal abundance, composition and diversity of fishes in a south-eastern New England estuary. Est. Coast. Mar. Sci. 5: 665 â?? 671.
- Hopkinson, C.S, (1985): Shallow-water and pelagic metabolism: Evidence of heterotrophy in the near-shore Georgia Bight, Marine Biology, 87, pp
- Javor, B.J. (1989): Growth potential of halophilic bacteria isolated from solar salt environments: Carbon sources and salt requirements. Applied and Environmental Microbiology, 48 (2), 352-360.
- Jhingran, G. (1991): In: Fish and Fisheries of India, 3rd Ed. Hindustan Publishing Corporation, Delhi, India, 727 pp.
- Kalaiarasi, M.; Paul, ; Lathasumathi, C.; Stella, C. (2012) Seasonal variations in the physico-chemical characteristics of the two coastal waters of Palk-Strait in Tamil Nadu, India. Global J. Environ. Res., v. 6, p. 66-74.
- Kamalkanth, S.; Muniyan, M.; Christyponni (2012) A. Seasonal variations in physico-chemical parameters at Tranquebar Coastal Nagapattinam, India.Int. J. Environ. Biol., v. 2, p. 203-207,
- Kathiravan, ; Sundaramanickam, A.; Varadharajan,.D.; Balasubramanian, (2013) Seasonal variations in physico-chemical parameters of Parangipettai coast, South east coast of India. Int. J. Pharm. Biol. Arch., v. 4, p. 240-249.
- Klopaters, M. (1978): Nutrient dynamics of fresh water riverine marshes and the role of emergent macrophytes, pp. 195 â?? 216. In Freshwater wetlands: ecological processes and management potential. (eds) Wood, K.E. Whigham, F. and Simpson, R.L. Academic press, New York.
- Kodarkar, M. S. (1992): Methodology for water analysis, (physico-chemical, Biological and Microbiological Indian Association of Aquatic Biologists Hyderabad, Pub. 2, 50).
- Kramer, J.R., Herbes, S.E., and Allen, H.E. (1979): Phosphorus: analysis of biomass and sediment pp 582 â?? 1000. In Nutrients in Natural waters (eds) Allen, H.E. and Krams, J.R. Wiley Inter Science Publication, New York
- Krishnamurthy, R., (1990): Hydro-biological studies of Wohar reservoir Aurangabad (Maharashtra State) India, Journal of Environmental Biology, 11(3), 335-34
- Kumar, J. I.; George, B.; Kumar, R. N.; Sajish, R.; Viyol, S. (2009) Assessment of spatial and temporal fluctuations in water quality of a tropical permanent estuarine system â?? Tapi, West coast of India. Appl. Ecol. Environ. Res., v. 7, p. 267-276.
- Liss, S.; Spencer, C. P.A (1970). Biological process in the removal of silicate from seawater. Geochim. Cosmochim. Ac., v. 34, p. 1073-1088.
- Moore, W.J. (1978): Distribution and abundance of phytoplankton cycles in 153 lakes, rivers and pools in the Northwest Can. J. Biol. 56: 765 1173
- Murphy, S,M., Kessel, B., and Vining, L.J. (1984): Waterfowl population and limnologic characteristics of Taiga J.Wildl. Manage. 48(4): 1156 â??1163
- Nazneen S., (1980): Influence of Hydrological Factors on the Seasonal Abundance of Phytoplankton in Keenjhar Lake. Int. Ges.Hydrobiol, 65(20): 269-282.
- Pomeroy, C. R.; Smith, E. E.; Grant, C. M.(1965).The exchange of phosphate between estuarine water and sediments. Limnol. Oceanogr.,v. 10, p. 167-172,
- Premlata, (2009), Multivariant analysis of drinking water quality parameters of lake Pichhola in Udaipur, India. Biological Forum, Biological Forum- An International Journal, 1(2),pp 97-102.
- P.A.; Venugopalan, V. K. (1972).Distribution of dissolved silicon in the Vellar estuary. Indian J. Mar. Sci., v. 1, p. 103-105,
- Puthiya C.; Poongothai, S.; Neelakantan, M. A. (2009) Impact of industrial pollution on the physico-chemical characteristics of sea water in Thoothukudi coastal area. Rasayan J. Chem., v. 2, p. 912-919,.
- Raina, U.; Shah, A. R.; Ahamad, S. R. (1984) Pollution studies on river Jhelum: an assessment of water Indian J. Environ. Health, v. 26, v.107-120.
- Ratner, (2009). The correlation coefficient: Its values range between +1/â??1, or do they?. J Target Meas Anal Mark 17, 139â??142 https://doi.org/10.1057/ 2009.5.
- Rochford, D. J. (1951). Studies in Australian estuarine hydrology, Introductory and comparative features. Aust. J. Mar. Fresh. Res., 2, p. 1-116,
- Mark Kurlansky (2003) Salt: A World History ), pp 18-, 38 .
- Sarkar, S.K., Singh, B.N., and Choudhary, A. (1985): Composition and variation in the abundance of zooplankton in the Hoogly estuary, West Bengal. Proc, Indian Acad. Sci. 95: 125 â??
- Satpathy, K.; Mohanty, A. K.; Natesan, U.;Prasad, M. V. R.; Sarkar,
- K. (2010) Seasonal variation in physico-chemical properties of coastal waters of Kalpakkam, East coast of India with special emphasis on nutrients. Environ. Monit. Assess., v. 164, p. 153-171.
- Satpathy, K. K. (1996) Seasonal distribution of nutrients in the coastal water of Kalpakkam, East coast of India.Indian J. Mar. Sci., 25, p. 221-224,.
- Smith, V. and Hollibaugh, J. T, (1993): Coastal metabolism andthe oceanic organic carbon balance, Reviews of Geophysics, 31, pp 75-76
- Smith, V. and Hollibaugh, J. T, (1997): Annual cycle and interannual variability of ecosystem metabolism in a temperate climate embayment, Ecology/Ecological Monographs, 67, 509
- The Caddo Indians of Louisiana. Clarence H. Webb, Hiram Gregory. (1978).
- The Indian Economist, 2, November The Times of India, 16 November 2016,
- Touliabah, ; Safik, H. M.; Gaballah, M. M.; Taylor, W. D. (2002.) Phytoplankton and some abiotic features of El-Bardawil lake, Sinai, Egypt. Afr. J. Aquat. Sci., v. 27, p. 97-105,
- Trivedy, K. and Goel, P.K., (1986): Chemical and Biological method for Water pollution studies, Environmental Publications, Karad, India.
- Velasquez, C.R. (1992): Managing artificial saltpans as a waterbirds habitat: Species responses to water level manipulation. Colonial Waterbirds. 15: 43
- Welch, E.B., Strutevant, , and Perkins M.A. (1978): Dominance of phosphorus over nitrogen as the limits of phytoplankton growth rate. Hydrobiologia. 57(3): 209 â?? 215
- Wetzel R.N. (1975): Limnology W.B. Sunders Co.Philaded phia.
- Wetzel, RG and Likens, GE (1979): Limnological Analysis WB Saunder Co Philadelphia,
- Wolfar S.; Reinhard, B. (1998) The heterogeneity of runoff and its significance for water quality problems. Hydrol. Sci. J., 43, p. 103-113,
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi