Research Article, J Sleep Disor Treat Care Vol: 6 Issue: 4
Dispositional Experiential Emotion Regulation and the effects of PreSleep Negative Mood on Sleep
Vandekerckhove M1*, Houthuys J1, Kestemont J1, Verhaert V3, De Valck E4, Verbraecken J5, Weiss R2 and Cluydts R1
1Department of Clinical and Life Span Psychology, Experimental and Applied Psychology, Belgium
2Department of Biological Psychology, University of Cologne, Germany
3Biomechanics and Engineering Design (BMGO), KULeuven, Heverlee, Belgium
4Measure, model and manage bio-responses (M3-BIORES), KULeuven, Heverlee, Belgium
5Sleep disorders center, UZA, Edegem, Belgium
*Corresponding Author : Vandekerckhove M
Faculty of Psychology and Educational Sciences, Department of Clinical and Life Span Psychology and Experimental and Applied Psychology, Pleinlaan 2 | B - 1050 Brussel, Belgium
Tel: +02-6291594/04-96212066
E-mail: Marie.Vandekerckhove@vub.ac.be
Received: June 05, 2017 Accepted: June 16, 2017 Published: June 23, 2017
Citation: Vandekerckhove M, Houthuys J, Kestemont J, Weiss R, Verhaert V, et al. (2017) Dispositional Experiential Emotion Regulation and the effects of Pre-Sleep Negative Mood on Sleep. J Sleep Disor Treat Care 6:4. doi: 10.4172/2325-9639.1000201
Abstract
The present study investigates the impact of pre-sleep negative stress on sleep and unrecognized effects of dispositional emotion regulation. It was examined whether the induction of negative pre-sleep emotion had a negative impact on sleep physiology and second, whether a dispositional High ‘Emotional Approach’ (HEA) compared to a ‘Low Emotional Approach’ (LEA) defined by high versus low efforts to identify, process, and express emotions, assessed by the ‘Emotional Approach’ scale EAC, Stanton et al., facilitates the recovery from a pre-sleep failure-experience and its effects on sleep physiology. The induction of a personal failure experience resulted in a worsening of mood, a significant increase of ruminative thoughts about one’s own performance and impaired subsequent sleep physiology. The negative impact on sleep physiology was observed in decreases in sleep efficiency, (SE), total sleep time (TST), an increase in total time awake (TTA), and latency to the third phase sleep (N3) or slow wave sleep (SWS) and % REM-sleep. Our main objective of exploring emotion regulation as a personality trait in this study revealed an impact of dispositional differences in a high versus low emotional approach on the recovery of painful emotions within subjects and not between subjects. This result can be explained by the small group of participants- a characteristic of much effortful and exploratory sleep research. However and this is highly interesting: within the group with a LEA (8), more sleep parameters were affected in the night after the failure induction; a longer sleep onset latency, an enhanced latency to slow wave sleep, less % REM-sleep, more arousals, time awake, less total sleep time and sleep efficiency during the failure night relative to the neutral night was observed. Within the group of a HEA (7), only REM-% was decreased in the failure induction compared to the neutral condition. These preliminary results suggest an adaptive regulatory effect of a HEA in the recovery with a painful experience, an effect extending into sleep.
Keywords: Emotion; Emotion regulation; Sleep; Coping
Introduction
Daytime emotional stressful events, affect sleep quality resulting in longer sleep onset latency, elevated awakenings, arousal in sleep, slow wave sleep- and REM-sleep alterations [1-7]. Not only daytime events determines the affective state of an individual, also the individual's approach of emotion regulation of these events in order to regain emotional balance and wellbeing is crucial here [8,9]. Emotion regulation refers to methods of change related to the experience and expression of emotions, as well as the times in which emotions occur [10]. Although effective emotion regulation is widely seen as vital for mental wellbeing and research on emotion regulation is receiving more and more attention, the adaptive value of different emotion regulation modes for recovering from an emotionally painful event is still an open question. Some studies emphasize the adaptiveness of a more cognitive approach such as cognitive control or reappraisal [8], while others a more affective experiential approach [4,9,11]. It is surprising to note that despite a long history of interest in links between emotion regulation and psychopathology or physical disease [12], the link between specific emotion regulation strategies and sleep is largely a new field with various conflicting findings. A study on the role of coping in the effects of acute stress (e.g., giving a speech on the next morning) on sleep physiology [13], found significantly lower average REM-counts in the last REM-period without any moderating effects of coping styles (such as avoidance, problem-focused coping, seeking social support, self-blame, or wishful thinking). Another study on the relationship between emotion regulation, low and high periods of stress and sleep, found that a high score for ‘emotion focused coping’ as assessed by the ‘Ways of Coping’ questionnaire of Lazarus & Folkman [14], increased alertness and thus physiological arousal, which predicted a reduction in sleep time from the low to the high-stress period [15]. Morin and coworkers [16] also found that, in comparison with controls, insomnia patients relied more on emotion-focused coping strategies [14] and perceived their lives as more stressful. However, in order to recover from painful emotions, ‘facing’ them directly and not avoiding them as implied in the definition of ‘emotion focused’ by Lazarus & Folkman [14] by wishful thinking, distancing, minimization or avoidance of emotions) might be more adaptive. In other words, as applied in experiential client-centered therapies, an ‘openness to one’s own experiential processes’ may help the individual to recover from painful emotion [15,17-19]. In contrast, an emotion avoidant approach is considered a maladaptive way of coping with emotions and is associated with sustained negative mood states, cognitive biases, increased vulnerability to depression, and impaired problem solving as a consequence [20].
In order to investigate which emotion regulation strategy is effective in response to stressors -- also on the level of sleep physiology -- the present study explored the effectiveness of experiential dispositional emotion regulation after a personal failure experience on sleep. In this study two modes of emotion regulation are distinguished: (1) a ‘High Emotional Approach’ (HEA) and (2) a ‘Low Emotional Approach’ (LEA) defined by a high or low tendency to acknowledge, understand, identify and express actual emotional experiences and affective feelings about a situation and to focus on concrete moment-by-moment details of how emotional experience unfolds [1]. We hypothesize that a ‘HEA’ compared to a ‘LEA’ is more adaptive in successful emotional processing and recovery from painful events with differential effects on mood, rumination and sleep physiology. Examining these differences in healthy people may provide further insights into the specific adaptive value of emotion regulation strategies and their role in moderating numerous effects of a painful event on sleep fragmentation, the development and maintenance of sleep disturbances in people experiencing transient stressful life events, ontological discomfort, depression and posttraumatic stress disorder.
Methods
Participants
Participants were recruited for a study on cognition and sleep. They volunteered via different sources, such as mailing lists of the electronic communication platform of the ‘Vrije Universiteit Brussel’ and via acquaintances and colleagues of the researchers. Healthy Dutch speaking participants have been included in this study with an age range of 19–56 yrs (M=32.38, SD=12.23), with a good baseline sleep quality and no sleeping disturbances (including apnoea, insomnia), a mean between 6 and 9 hours of sleep per night and no psychiatric disorder or medication intake or medication influencing sleep and no psychotherapeutic treatment, pregnancy, shift working, or intake of more than 3 beverages containing caffeine or alcohol per day. Of the 43 people who responded, 21 subjects were screened with the semistructured interview M.I.N.I. [21,22] as well as with the short version of the multidimensional Symptom Check List 90 [23], to exclude psychiatric and psychological problems. To exclude people with sleeping problems, subjects filled out the Pittsburgh Sleep Quality Index (PSQI) [24]. In order to assess the participants’ emotion regulation styles the Emotional Approach Questionnaire EAQ; Stanton et al. [1] was completed. This questionnaire assesses two distinct ‘emotional approach’ coping factors: ‘Emotional processing’, which refers to actively approaching and understanding one’s emotions, and ‘Emotional expression’. The scale has a good internal item consistency (α=.80 to .90) and a high test-retest reliability for both men and women (rtt=.72 to .82). High and low emotional approach groups were selected from the top and bottom 50% [1]. Only moderate to good sleepers, with reported average SOL below 20 min participated, except for two subjects with an average SOL of 29.5 and 32.5 min. There were no habitual short or long sleepers. Participants gave their written informed consent and also, prior to the study, they were asked (1) to keep a sleep diary for 2 weeks, so as to check for irregular sleep-wake schedules, and (2) to maintain their normal sleep-wake patterns two weeks before the experiment. Finally, 15 participants completed the study: 7 with a LEA (Male=4; Female=3) and 8 with a HEA (Male=3; Female=5). High scores on the Emotional Approach Questionnaire ranged between 43 and 53 (M =46.63, SD =3.54) and low scores ranged between 26 and 36 (M =33.43, SD= 3.69). One person spent an extra night and two people spent two extra paid nights in the laboratory. Participants were paid 120 euros for the entire study, which was approved by the Ethics Committee at the ‘Vrije Universiteit Brussel’.
Procedure
Participants were scheduled for four nights with eight hours’ time in bed at the sleep laboratory of the ‘Vrije Universiteit Brussel’. The first night included a diagnostic sleep study to rule out any potential sleep disorder and to adapt the participants to the laboratory procedures. The second night served as the baseline night for sleep parameters. Following an adaptation and a baseline night, subjects underwent two experimental nights: one night with the experimental condition, and one night with a neutral or control condition. In order to avoid order effects, the two experimental nights were counterbalanced.
Adaptation and baseline night
Experimental night: Failure induction
The failure induction consisted of a failure feedback in response to different cognitive tasks which we introduced as an intelligence test [4]. In several prior pilot tests, we adapted the level of difficulty until all participants had the minimum score between 0 and 3 whereby the maximum score of the test was 9-11. After this phase, we validated the test on 12 persons. The tasks and failure feedback were presented on a computer in order to standardize to a maximum degree and to be able to individualize the scores based on their real performances.
On the experimental night participants arrived at the lab at 20.00h. After attaching electrodes, participants had to watch an emotionally neutral film (on nature) of 5 minutes’ duration which served as a baseline condition [26]. After rating their emotional experience with the PANAS, the experimenter entered the room again to introduce the procedure of the intelligence test which the participants had to fill out. The experimenter informed the participant that he or she would be participating in a new cognitive test reflecting their level of intelligence and predicting future professional achievement. They were told: “It is very important that you concentrate and do this intelligence test to the best of your possibilities. The test is being developed for international use and reflects your potential regarding general intelligence and professional achievements”. The test contained five subtests: (1) the spatial ability tasks, (2) logical steps tasks (figure completion) and mathematical problem solving tasks, (for instance; number completion: 100, 95, ?, 79, 68, ?, 40, 23), (3) a numeric and visual memory test and (4) a semantic reasoning task (5) The final task involved an impossible semantic test by presenting three unrelated words 10 times whereby the participants had to find the related word (blood, music, cheese) for a more detailed description; see Vandekerckhove et al. [4]. Between the different tasks, the experimenter entered the room several times giving several comments in an annoyed and irritated way and voice. The first time, the following feedback was given: “You are moving around too much, causing physiological artifacts making the data useless and full of artifacts. Please sit still.” After five minutes the experimenter entered the room again, sighing and clearly annoyed, checking the electrodes and then leaving the room without talking to the participants. After each subtest and at the final completion of the test the score obtained was presented on the computer screen accompanied by a failure feedback saying that the person’s test achievements were weak and clearly below the mean. At the end of the testing, the experimenter informed the participants that the physiological measurements so far were useless and that they would have to return and perform the task the next morning. In reality, no physiological data were obtained and the test was too difficult. Before the participants went to bed at 23.00 h they completed the PANAS to assess their emotional experience and recovery of negative affect and the level of rumination by the Impact of Event Scale [27,28].
After their morning awakening at 07.00 h, mood and intrusions were assessed again. After the electrodes were removed and before leaving the lab, a funnelled debriefing procedure was used. Depending on whether the night of the failure induction came before the night of the neutral condition (with the relatively easy tasks), they were told: “The task you performed yesterday is, in general, still far too difficult to fulfil in the given time. Everyone is obtaining a score between 0 and 3, which means that your score was not bad at all and that we definitively have to adapt our tests to a more normal level of difficulty.” It was emphasized that the tasks were obviously far too difficult which made their performance unrelated to intelligence and either academic or career success. The debriefing was done in a way that participants really understood the validating nature of the study and they felt well again.
For the setting in which the failure night came after the neutral condition, the same debriefing was given except that participants were explicitly asked whether they had any idea about the aim of the study. The debriefing was done in a way that once again participants really understood the true nature of the study and the reason of the manipulations and they felt well again.
Experimental night - Neutral condition
For the control condition, the same procedure was followed as in the night of the failure induction with identical but less difficult tasks to fulfil as in the experimental condition. Prior pilot testing adjusted the tasks to a medium-range level of difficulty and calibrated about the same time for solving the tasks so that participants could perform 60% and got neutral feedback. Although the experimenter entered also the room, no irritated comments of moving too much at the participants were given. At the end of the testing, the experimenter informed the participants that the physiological measurements were okay. In the debriefing phase, the experimenter said “The task you performed yesterday was okay and that their collaboration meant a step further in the validation of the study”.
Measurements
Polysomnography
Polysomnographic recordings were performed at the experimental sleep laboratory at the ‘Vrije Universiteit Brussel’. The recording consisted of six EEG electrodes referenced to a single mastoid (F3, C3, O1, F4, C4 and O2), two EOG electrodes referenced to a single mastoid (LOC-ROC), a bipolar submentalis EMG, tibialis EMG, and EKG. A 32 channel Embla N7000 recording system was used (Medcare) with a DC offset of 500 mV max and a fixed DC low cut filter at 0.3 Hz. The EEG and EOG signals were high pass filtered at 0.5 Hz and low pass filtered at 40 Hz, EMG channels were high pass filtered at 5 Hz and low pass filtered at 70 Hz. Absolute power (mV2) in the delta (1-3.5 Hz), theta (4–7.5 Hz), alpha (8–12 Hz), sigma (12– 14 Hz), beta-1 (12–20 Hz), beta-2 (20–35 Hz) and high beta (25.5-30 Hz) bandwidths was computed for both EEG sites. The data were scored blind by two trained specialists reaching an inter-reliability of 90%. Sleep stages were scored based on the new guidelines of the American Academy of Sleep Medicine [29] with the following sleep variables: ‘total sleep time’ (TST), ‘sleep onset latency’ (SOL) (from the moment the lights go out until the first minute of stage 1 sleep, N1 of the sleep period time, (SPT), ‘wake after sleep onset’ (WASO), ‘sleep efficiency’ (SE), % ‘slow wave sleep’ (SWS) of the sleep period time (SPT), latency to SWS, latency to REM, % REM-sleep of the SPT, number of REM periods, % Stage 1 sleep (N1) of the SPT, and % Stage 2 sleep (N2) of the SPT. Also the numbers of awakenings, number of awakenings from REM-sleep, as well as the number of arousals were scored. Movement artefacts were excluded from analysis. Spectral analysis was performed on C4-A1 to evaluate the sleep onset period (SOP) and NREM and REM-sleep. The SOP was defined as lights out to the first 5 minutes of stage 2 sleep [30]. The SOP has been divided into four quartiles to evaluate the EEG-dynamics. All epochs containing movements or EMG-artefacts were excluded from analysis.
Wake-EEG
A 5 minute measurement with eyes open and eyes closed was performed. An EOG and EMG submentalis were done to exclude artifacts of eye movements or muscle activity. Sampling rate was 256 Hz and impedances were kept below 10 Kohm. The recording equipment was identical to that of the polysomnography. Absolute and relative values were calculated. A Fast Fourier Analysis was performed on the wake EEG on a minimum of 90 seconds artifact-free data using Neuroguide software (Applied Neuroscience, Inc.).
Results
Negative emotion experience and rumination
A General Linear Model repeated measures ANOVA (SPSS, version 19) were used. In order to control for baseline emotional differences between participants, the relatively ‘neutral’ film was showed. In accordance, as expected no difference in negative affect was observed before the failure induction’ between the failure- and the neutral night (F (1, 13)=0.00, ns, η2 p=.0). ‘After the tasks’ (neutral: M =1.28, SD= 0.21 - failure: M =2.13, SD = 0.73) and ’the following morning’ (neutral: M = 1.22, SD = 0.24 – failure: M =1.55,SD =0.54), significant differences between both experimental (failure and control) nights were found (respectively (F (1, 14)=18.81, p =.001, η2 p)*** and (F (1, 14)=4.01, p<.10,=.22)*). Also rumination, measured with the IES, differed significantly between both nights and this was the case at the different moments (times) ‘presleep’ (neutral: M =3.27, SD =2.71 - failure: M =10.53, SD =6.77) and ‘postsleep’ (neutral: M = 2.33, SD =2.23 – failure: M =7.60, SD =6.30) (F (1, 14)=20.58, p=.000, η2 p=.59)*** and (F (1, 14)=11.14, p=.005, η2 p=.44)***.
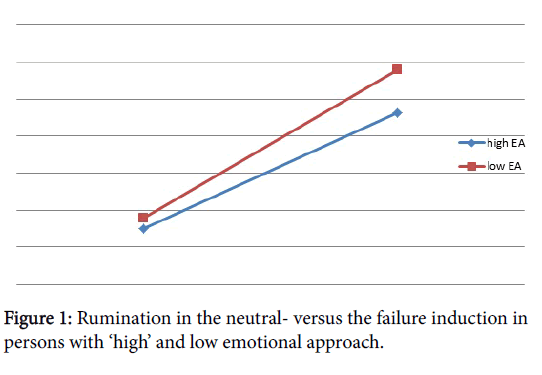
Within the failure night itself, (F (1, 13)=0.22, ns), the increase of negative experience in LEA, ‘after the tasks’ (M =2.23, SD =0.85) in comparison with the group with a HEA (M =2.05, SD = 0.66) was not significant. The same tendency has been found for rumination. The group with a HEA (M =9.62, SD =6.65) reported less rumination ‘presleep’ compared to the group with a LEA (M =11.57, SD =7.28) (F (1, 13)=0.29, ns) (Figure 1).
Within the failure night itself, although not significant (F (1, 13)=0.22, ns), the average negative experience in the group with a LEA was higher just ‘after the tasks’ (M =2.23, SD =0.85) in comparison with the group with a HEA (M =2.05, SD =0.66). This was not the case ‘the following morning’ where we found, although this was not significant (F (1, 13)=0.80, ns), that persons with a LEA reported less negative affect (M =1.41, SD =0.47) relative to persons with a HEA (M =1.66, SD= 0.60). The same tendency, but not significant, can be found for rumination. The group with a HEA (M =9.62, SD =6.65) reported less rumination ‘presleep’ compared to the participants with a LEA (M =11.57, SD =7.28) (F (1, 13)=0.29, ns. Only ‘Postsleep’ with a LEA (M =7.00, SD =4.24) reported less rumination relative to HEA (M =8.12, SD =7.95) (F (1, 13)=0.11, ns) (see also Table 1).
| Neutral night | Failure night | |||||||
|---|---|---|---|---|---|---|---|---|
| High EA | Low EA | Entire group | High EA | Low EA | Entire group | |||
| Emotional experience | ||||||||
| NA before the tasks (PANAS) | 1.17 (± 0.18) | 1.08 (± 0.13) | 1.12 (± 0.16) | 1.14 (± 0.15) | 1.10 (± 0.18) | 1.12 (± 0.17) | ||
| NA after the tasks (PANAS) | 1.31 (± 0.20) | 1.23 (± 0.24) | 1.28 (± 0.22) * | 2.05 (± 0.66) | 2.23 (± 0.85) | 2.13 (± 0.73)*** | ||
| NA following morning (PANAS) | 1.23 (± 0.27) | 1.21 (± 0.22) | 1.22 (± 0.24) | 1.66 (± 0.60) | 1.41 (± 0.47) | 1.55 (± 0.54) | ||
| Rumination presleep (IES) | 3.00 (± 2.93) | 3.57 (± 2.64) | 3.27 (± 2.71) | 9.26 (± 6.65) | 11.57 (± 7.28) | 10.53 (± 6.77)*** | ||
| Rumination postsleep (IES) | 1.87 (± 1.46) | 2.86 (± 2.91) | 2.33 (± 2.23)*** | 8.12 (± 7.95) | 7.00 (± 4.24) | 7.60 (± 6.30)*** | ||
| Sleep parameters | ||||||||
| SE(%) | 93.87 (± 10.98) | 93.24 (± 5.63)*a | 93.58 (± 8.60)**g | 89.95 (± 17.24) | 88.04 (± 8.80)*a | 89.06 (± 13.52)**g | ||
| WASO (min) | 12.71 (± 24.43) | 30.07 (± 48.18) | 21.39 (± 37.79) | 45.36 (± 81.25) | 43.00 (± 45.16) | 44.18 (± 63.16) | ||
| Total time awake (%) | 6.12 (± 10.98) | 6.76 (± 5.63)*a | 6.42 (± 8.60)*g | 10.74 (± 19.13) | 11.96 (± 8.80)*a | 11.31 (± 14.72)*g | ||
| Number of awakenings | 6.50 (± 6.87) | 12.57 (± 9.03) | 9.33 (± 8.27) | 8.37 (± 8.12) | 15.71 (± 9.50) | 11.80 (± 9.27) | ||
| TST (min) | 440.31 (± 53.46) | 451.93 (± 32.46)**a | 445.73 (± 43.78)*g | 432.25 (± 94.08) | 411.64 (± 56.36)**a | 422.63 (± 76.81)*g | ||
| SOL | 6.75 (± 3.76) | 9.29 (± 7.12)*a | 7.93 (± 5.53) | 12.37 (± 18.51) | 13.21 (± 8.12)*a | 12.77 (± 14.13) | ||
| % REM | 23.52 (± 6.23)*e | 20.79 (± 6.37)*a | 22.25 (± 6.23)**g | 17.47 (± 8.74)*e | 16.71 (± 4.38)*a | 17.12 (± 6.82)**g | ||
| Awakenings from REM | 1.00 (± 1.60) | 1.43 (± 1.51) | 1.20 (± 1.52) | 0.87 (± 0.99) | 2.00 (± 2.00) | 1.40 (± 1.59) | ||
| Number of REMs | 5.50 (± 2.20) | 4.86 (± 1.68) | 5.20 (± 1.93) | 4.12 (± 1.73) | 4.86 (± 1.07) | 4.47 (± 1.46) | ||
| Latency to REM (min) | 96.44 (± 43.54) | 121.93 (± 52.65) | 108.33 (± 48.05) | 145.31 (± 121.35) | 96.36 (± 17.53) | 122.47 (± 90.19) | ||
| Latency to SWS (min) | 23.94 (± 9.53) | 20.75 (± 8.30)***a | 22.37 (± 8.83)**g | 38.50 (± 33.32) | 28.43 (± 5.33)***a | 33.80 (± 24.38)**g | ||
| % N1 | 6.84 (± 2.61) | 15.40 (/) | 8.27 (± 4.20) | 10.70 (± 6.53) | 16.50 (/) | 11.67 (± 6.31) | ||
| % N2 | 38.14 (± 5.01) | 32.20 (/) | 37.15 (± 5.10) | 36.18 (± 15.84) | 20.80 (/) | 33.62 (± 15.50) | ||
| % N3 | 26.36 (± 6.02) | 25.70 (± 12.47) | 26.05 (± 9.22) | 26.49 (± 6.69) | 25.83 (± 8.89) | 26.18 (± 7.51) | ||
a= low emotional approach
e= high emotional approach
g= entire group
Table 1: Means and standard deviations for emotional experience, rumination, and sleep parameters for the entire group and in persons with ‘high’ and ‘low emotional approach’ at the neutral and the failure night.
Footnote
*Significant at the p<.10 level, **Significant at the p<.05 level, ***Significant at the p<.01 level
Effects of failure induction on polysomnographic variables
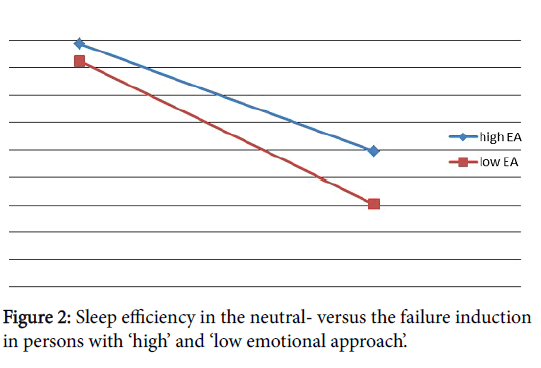
A statistically significant effect of the failure experience on SE was found (F (1, 14)= 7.63, p<.05, η2 p=.35)** indicating that SE in the night with the failure experience was significantly worse than in the neutral night. After the failure induction, compared to the neutral condition, TTA (F (1, 14)=6.76, p<.05, η2 p=.33)** and Latency to SWS (F (1, 14)=5.20, p<.05, η2 p=.27)** increased significantly. As a consequence, in the failure night relative to the neutral night, TST was enhanced (F (1, 14)=3.86, p<.10, η2 p=.22)*. Finally, % REM-sleep (F (1, 14)=8.46, p<.05, η2 p=.38)** was decreased in the failure night compared to the neutral night (see also Table 1). These effects were likely not directly due to other factors as effort or physiological arousal associated with the performance of the cognitive tasks. Possible confounding variables were controlled for by comparing the neutral condition including similar cognitive tasks but without negative feedback, with the baseline night for emotional experience and sleep physiology which did not reveal significant results with all F’ s smaller than 1.5. Also the significant increase in negative emotional experience in the failure night compared to the neutral night makes it probable that the results in this study reflect the emotional experience due to the failure experience more than the level of effort related with the fulfilment of the cognitive tasks (Figure 2).
Emotion regulation
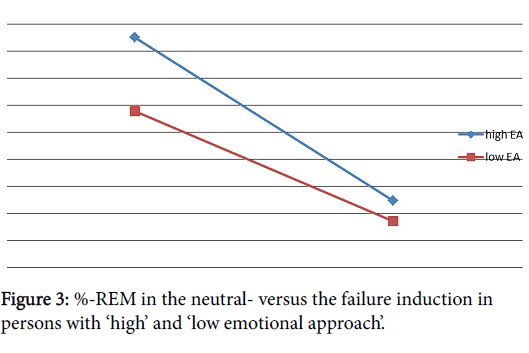
Implications of HEA and LEA were only observed ‘within subjects’. Within the group of HEA, only a slight decrease in % REM-sleep (t (7)=2.04, p<.10)* caused by the failure induction has been observed. When comparing the effects with the baseline night, HEA showed a decrease in REM-percentage (t (6)=2.22, p<.10)* and number of REMs (t (6)=2.12, p<.10)* in the failure night (Table 1). Within the group with a LEA more sleep variables were affected after the failure induction. They showed a decreased Sleep Efficiency in the failure night relative to the neutral night (t (6)=2.14, p <.10)*, and also more Time Awake during the failure night in comparison with their neutral night (t (6)=2.15, p<.10 )*. As a consequence, TST decreased during the failure night relative to the neutral one (t (6)=2.99, p<.05)**. Latency to SWSsleep was increased in the failure night compared to the neutral night (t (6)=-3.94, p<.01)*** as well as SOL (t (6)=-2.27, p<.10)* and decreases in % REM-sleep (t (6)=2.19, p<.10)* and number of arousals (t (6)=1.97, p<.10)* have been observed during the failure night. In comparison to the baseline night, participants with a LEA showed a lower % REM-sleep (t (6)=2.60, p<.05)** and less arousals (t (6)=2.52, p<.05)**. In comparison to the baseline night, the group of LEA showed significant increases on the failure night w ere found for number of awakenings (t (6)=-2.52, p<.05)** as well as for SOL (t (6)=-3.43, p <.05)** (Figure 3).
Gender effects
No differences were found when dividing participants into groups based on gender.
Discussion
Emotional stress has been considered as the main etiological factor of primary psychophysiological sleep disorders and depression. An increased incidence of emotionally stressful life events in association with individual vulnerability has been found to be of major influence in the precipitation and triggering of insomnia [31]. However, pathways determining the effects of emotional stress on sleep and sleep disorders are not well understood. Therefore, the current study examined whether dispositional emotion regulation affects the recovery of a negative emotional event on mood, rumination and subsequent sleep physiology. The induction of a personal failure experience resulted in a worsening of mood, a significant increase of ruminative thoughts about own performance and impaired subsequent sleep physiology. The negative impact on sleep physiology was observed in decreases in Sleep Efficiency, (SE), Total Sleep Time (TST), an increases in Total Time Awake (TTA), and latency to the third phase sleep (N3) or Slow Wave Sleep (SWS) and % REM-sleep. Regarding the global effects of the failure experience on sleep confirms a preceding study of our lab on ‘pre-sleep emotion and sleep’ [4] and also correspond with other studies on pre-sleep arousal and sleep, posttraumatic stress disorder and sleep that showed sleep disturbances on where affected sleep variables has been observed such as increased number of awakenings, decreased slow wave sleep, longer REM-sleeplatency and decreased sleep efficiency [32,33]. Reductions in total sleep time on the night before an exam [34,35] reduced slow wave sleep or on-call nights [36] and decrease in sleep efficiency and a longer slow wave sleep latency [37] are similar findings reacted to pre-sleep emotional stress.
Emotion regulation and sleep
Our main objective of exploring emotion regulation as a personality trait in this study revealed some preliminary findings. The results suggest an adaptive impact of dispositional differences in a high versus low emotional approach on the recovery of painful emotions only within subjects. These results correspond with a proceeding of our lab where both emotion regulation strategies were induced by instruction [4]. However, the present study could not confirm a similar effect of the induction of an experiential versus analytical approach between subjects, probably due to the small group - a characteristic of much effortful exploratory sleep research. Nonetheless, at the dispositional level, within the group with a low emotional approach, we observed a longer sleep onset latency, an enhanced latency to slow wave sleep, less % REM-sleep, more arousals, time awake, less total sleep time and sleep efficiency during the failure night relative to the neutral night. Within the group with a high dispositional emotional approach, only one variable was affected with a decrease of REM-% in the failure induction compared to the neutral condition. These exploratory results provisionally suggest that individuals with a dispositional high emotional approach recover better than individuals with a low emotional approach who remained more negatively affected by the failure induction, especially at the level of objective implicit physiological sleep patterns. As a consequence, in the case of a selfrelevant painful experience, such as a personal failure experience, it should be useful to apply a high emotional approach and acknowledge, identify, process, and express affective experiences associated with the stressor.
In sum, these preliminary findings confirm the need for further exploration and validation of the impact of applying an emotional approach in emotion recovery and sleep physiology in larger groups as well as in clinical populations too. Moreover, in future studies, it should be also important to broaden the palette of adaptive experiential strategies to regulate emotions approaches where increased openness, acceptance and recognition of mental events such as thoughts and affective feelings in the present is important [38,39] compared with more cognitive approaches such as cognitive reappraisal where the individual has to reinterpret a stimulus’ meaning, in a way that changes, among other things, the trajectory of the emotional response [8]. Despite the limitations of this study, the present findings challenge the still present-day emphasis within psychology and psychotherapy on cognitive strategies in approaching emotional and personally relevant stressors, and the neglect of more functional ‘experiential’ approaches applied in experiential, emotion and client-centered therapies with high emphasis on actual awareness of bodily felt affective experience intensifying experience of negative emotion at first, but decreasing negative emotion in the longer run [18,40]. In conclusion, this study is a preliminary step in highlighting the role of an individual tendency to apply an experiential emotion regulation strategy in resilience to emotional distress and sleep disruption.
References
- Stanton AL, Danoff-Burg S, Sworowski LA, Collins CA, Branstetter AD, et al. (2002) Randomized, Controlled Trial of Written Emotional Expression and Benefit Finding in Breast Cancer Patients. J Clin Oncol 20: 4160-4168.
- Cartwright R, Luten A, Young M, Mercer P, Bears M (1998) Role of REM-sleep and dream affect in overnight mood regulation: a study of normal volunteers. Psychiatry Res 81: 1-8.
- De Valck E, Cluydts R, Pirrera S(2004) Effect of cognitive arousal on sleep latency, somatic and cortical arousal following partial sleep deprivation. J Sleep Res 4: 295-304.
- Vandekerckhove M, Cluydts R (2010) The emotional brain and sleep: an intimate relationship. Sleep Med Rev 14: 219-226.
- Harvey AG (2005) Unwanted intrusive thoughts in insomnia. In: Clark DA, Intrusive thoughts in clinical disorders: Theory, research, and treatment, Guilford Press, New York, USA.
- Hobson JA, Stickgold R, Pace-Schott EF (1998) The neuropsychology of REM sleep dreaming. Neuroreport 9: R1-14.
- Cui R, Li B, Suemaru K, Araki H (2007) Psychological stress-induced changes in sleep patterns and its generation mechanism. Yakugaku Zasshi 128: 405-411.
- Gross JJ (1998) The emerging field of emotion regulation: An integrative review. Rev Gen Psychol 2: 271-299.
- Watkins E, Moulds M (2005) Distinct models of ruminative self-focus: Impact of abstract versus concrete rumination on problem solving in depression. Emotion 5: 319-328
- Gross JJ (1999) Emotion regulation: Past, present, future. Cogn Emot 13: 551-573.
- Stanton AL, Kirk SB, Cameron CL, Danoff-Burg S (2000) Coping through emotional approach: Scale construction and validation. J Pers Soc Psychol 78: 1150-1169.
- Hayes SC, Strosahl K, Wilson KG (1999) Acceptance and Commitment Therapy: An experiential approach to behavior change. Guilford Press, New York, USA.
- Germain A, Buysse DJ, Nofzinger E (2007) Sleep-specific mechanisms underlying posttraumatic stress disorder: Integrative review and neurobiological hypotheses. Sleep Med Rev 12: 185-195.
- Folkman S (1984) Stress: Appraisal and Coping. Encyclopedia of Behavioral Medicine, Springer Publishing Company, USA.
- Watkins E, Teasdale JD (2004) Adaptive and maladaptive self-focus in depression. J Affect Disord 82: 1-8.
- Morin CM, Rodrigue S, Ivers H (2003) Role of stress, arousal, and coping skills in primary insomnia. Psychosom Med 65: 259-267.
- Hunt MG (1998) The only way out is through: emotional processing and recovery after a depressing life event. Behav Res Ther 36: 361-384.
- Greenberg L, Vandekerckhove M (2008) Emotional Expression and Regulation in Psychotherapeutic Processes In: Vandekerckhove MMP, von Scheve C, Ismer S, Kronast S, Regulating Emotions: Social Necessity and Biological Inheritance. Blackwell Press, USA.
- Watkins E, Teasdale JD (2001) Rumination and overgeneral memory in depression: effects of self-focus and analytic thinking. J Abnorm Psychol 110: 353-357.
- Wyer RS, Martin LL, Tesser A (1986) Ruminative Thoughts: Advances in Social Cognition. Lawrence Erlbauw.
- Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, et al. (1998) The mini-international neuropsychiatric interview (MINI): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 59: 22-33.
- Van Vliet IM, De Beurs E (2007) Het Mini Internationaal Neuropsychiatrisch Interview (MINI): Een kort gestructureerd diagnostisch psychiatrisch interview voor DSM-IV en ICD-10 stoornissen. Tijdschrift of Psychiatry 49: 393-397.
- Arrindell WA, Ettema JHM (2003) SCL-90. Handleiding bij een multidimensionele psychopathologie-indicator, Swets Test Publishers, Tilburg, Netherlands.
- Buysse DJ, Reynolds III CF, Monk TH, Berman SR, Kupfer DJ (1989) The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res 28: 193-213.
- Watson D, Clark LA, Tellegen A (1988) Development and validation of brief measures of positive and negative affect: The PANAS scales. J Pers Soc Psychol 54: 1063-1070.
- Mauss IB, Cook CL, Cheng JYJ, Gross JJ (2007) Individual differences in cognitive reappraisal: Experiential and physiological responses to an anger provocation. Int J Psychophysiol 66: 1-8.
- Horowitz M, Wilner N, Alvarez W (1979) Impact of events scale: A measure of subjective stress. Psychosom Med 41: 209-218.
- Brom D (1985) Schok verwerkings lijst, Universiteit Utrecht, vakgroep Klinische Psychologie, Netherlands.
- Iber C, Ancoli-Israel S, Chesson A, Quan S (2007) The AASM manual for the scoring of sleep and associated events: rules, terminology, and technical specifications, USA.
- Lamarche CH, Ogilvie RD (1997) Electrophysiological changes during the sleep onset period of psychophysiological insomniacs, psychiatric insomniacs, and normal sleepers. Sleep 20: 724-733.
- Healey ES, Kales A, Monroe LJ, Bixler EO, Chamberlin K, et al. (1981) Onset of insomnia: role of life-stress events. Psychosom Med 43: 439-451.
- Edéll-Gustafsson UM (2002) Insufficient sleep, cognitive anxiety and health transition in men with coronary artery disease: a self-report and polysomnographic study. J Adv Nurs 37: 414-422.
- Germain A, Buysse DJ, Ombao H, Kupfer DJ, Hall M (2003). Psychophysiological reactivity and coping styles influence the effects of acute stress exposure on rapid eye movement sleep. Psychosom Med 65: 857-864.
- Holdstock TL, Verschoor GJ (1974) Student sleep patterns before, during and after an examination period, J Psychol 4: 16-24.
- Lester BK, Burch NR, Dossett RC (1967) Nocturnal EEG-GSR profiles: the influence of presleep states. Psychophysiology 3: 238-248.
- Torsvall L, Akerstedt T (1988) Disturbed sleep while being on-call: an EEG study of ships' engineers. Sleep 11: 35-38.
- Akerstedt T, Kecklund G, Axelsson J (2007) Impaired sleep after bedtime stress and worries. Biol Psychol 76: 170-173.
- Kabat-Zinn J (1994) Wherever you go there you are: Mindfulness meditations in everyday life, Hachette Books, New York, USA.
- Fletcher LB, Schoendorff B, Hayes St (2010) Searching for Mindfulness in the Brain: A Process-Oriented Approach to Examining the Neural Correlates of Mindfulness. Mindfulness 1: 41-63.
- Stiles WB, Barkham M, Mellor-Clark J, Connell J (2008) Effectiveness of cognitive-behavioural, person-centred, and psychodynamic therapies in UK primary-care routine practice: replication in a larger sample. Psychol Med 38: 677-688.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi