Short Communication, J Sleep Disor Treat Care Vol: 6 Issue: 3
How Sleep Bruxism and Tension Headaches Affect the Masseter Inhibitory Reflex
Luco K*
President at Luco Hybrid OSA Appliance Inc, University of Alberta, Kingston, Canada
*Corresponding Author : Ken Luco DDS
President at Luco Hybrid OSA Appliance Inc, 1419 Butternut Creek Road, Canada
Tel: 613 888 6019
E-mail: DrLuco@sympatico.ca
Received: March 14, 2017 Accepted: April 11, 2017 Published: April 18, 2017
Citation: Luco K (2017) How Sleep Bruxism and Tension Headaches Affect the Masseter Inhibitory Reflex. J Sleep Disor: Treat Care 6:3. doi: 10.4172/2325-9639.1000198
Abstract
Sleep bruxism (SB) is classified as a repetitive jaw-muscle activity characterized by clenching or grinding of the teeth and/or by bracing or thrusting of the mandible. In recent years, the inhibition of the masseter inhibitory reflex (MIR) has been shown to result in increased bite force during sleep bruxism events. Tension type headaches (TTH) are a hallmark symptom reported by sleep bruxism patients and affect the MIR very similarly to SB.
Keywords: Sleep Bruxism, Headaches, Masseter Inhibitory Reflex
Short Communication
Sleep bruxism (SB) is classified as a repetitive jaw-muscle activity characterized by clenching or grinding of the teeth and/or by bracing or thrusting of the mandible [1]. In recent years, the inhibition of the masseter inhibitory reflex (MIR) has been shown to result in increased bite force during sleep bruxism events. Tension type headaches (TTH) are a hallmark symptom reported by sleep bruxism patients and affect the MIR very similarly to SB [1-5].
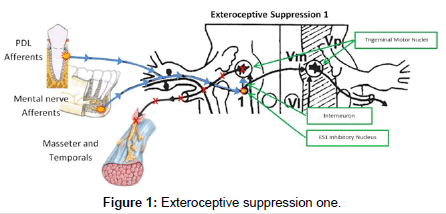
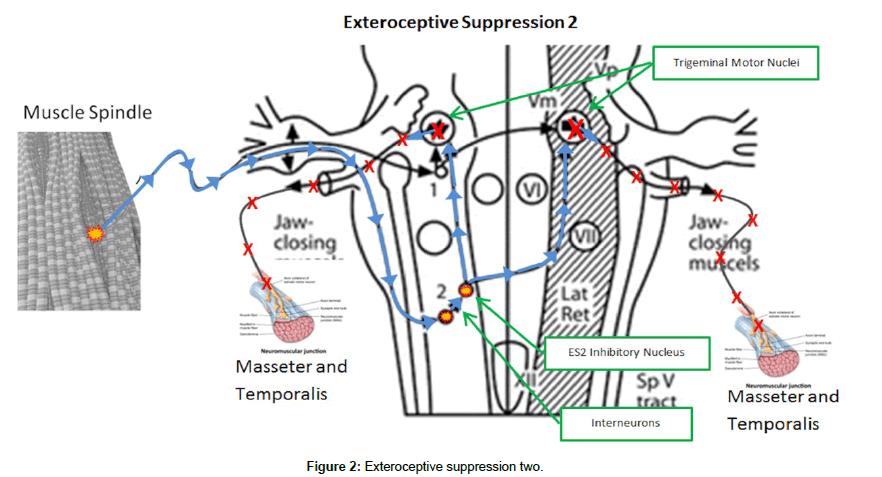
The masseter inhibitory reflex is a cranial reflex consisting of two distinct phases or silent periods termed exteroceptive suppressions (ES1 and ES2) [6]. This reflex is located in the trigeminal mesencephalic nucleus of the pons [7]. The first phase of the reflex, ES1, has its afferents located in the periodontal ligaments of the teeth as well as the mental nerve. The 1st inhibitory nucleus in the mesencephalic nucleus has only a single interneuron connecting it to the ipsilateral trigemenal motor nucleus (TMN), that controls the ipsilateral masseter and temporalis muscles. Normally, ES1 lasts 10-14ms (Figure 1). The 2nd phase, ES2, has its afferents located in the muscle spindle sensory organs located in the masseter and temporalis muscles. These afferents descend down the lateral trigemenal tract to the second inhibitory interneuron. There are multiple interneurons for ES2 connecting it balaterally to both TMN. Normally, ES2 lasts 40-50 ms6 (Figure 2). The result of the activation of the MIR is suppression of the TMN and inhibition of contraction of the masseter and temporalis muscles. This protective reflex is normally inactive and is activated when chewing softer foods and encountering a hard object. The reflex activates, suppressing further contraction of the masseter and temporalis, preventing damage to the oral structures [8-12].
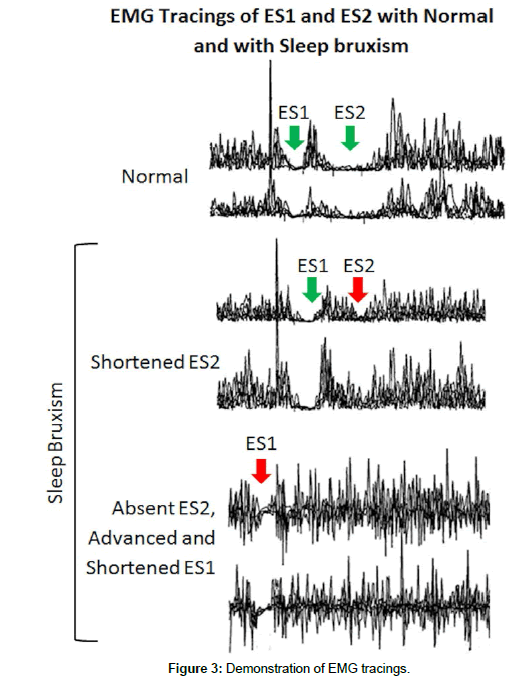
In sleep bruxism the ES2 phase and to a lesser extent, the ES1 phase are affected. The ES2 phase is shortened considerably or completely absent, whereas the ES1 phase is advanced slightly and shortened (Figure 2). There are reduced pain thresholds with TTH, relating to the decreased ES2 of the MIR. There is also excitability of the reticular nuclei which inhibit the medullary inhibitory internureons that mediate the ES2 phase of the MIR [2]. The EMG tracings in Figure 3 demostrate the significant reduction or complete abscense of ES2 in sleep bruxism [12].
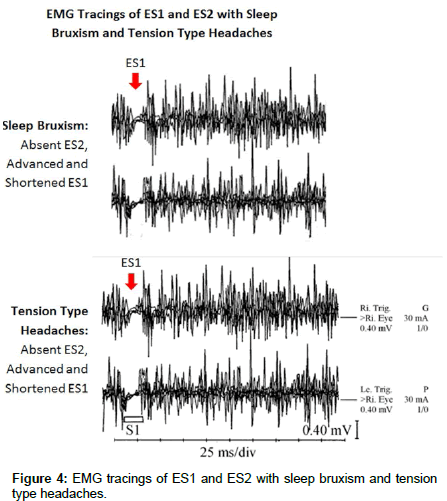
The EMG tracing in Figure 4 compares the ES2 suppression in SB bruxism with the ES2 suppression in TTH. The resulting suppression of the MIR (Figure 4) in TTH is remarkably similar to the suppresion of the MIR in SB, illuminating how SB and TTH are interrelated. This inhibition of the MIR in SB and TTH allows the masseter and temporolis muscles to contract with maxium uninhibited force resulting in the severe damage to teeth, muscles, TMJ and severe tension type headaches seen in SB and TTH [8,12-16].
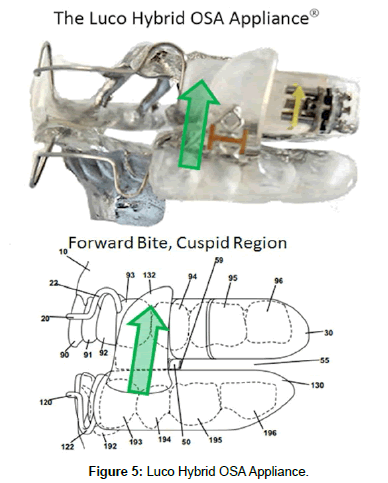
With oral appliance therapy for SB, the position of the bite is critical. If the bite on the device is placed in the molar region, the MIR will continue to be suppressed (with resulting excessive bite force) whereas if it is placed in the cuspid or incisal region, the MIR is reactivated and the bite force is limited to normal levels [17-20]. In the prototype stage of the Luco Hybrid OSA Appliance (Figure 5), these principles were implimented early on, achieving excellent results. The forward bite effectively reduced the symptoms of SB and TTH within 2 weeks in the majority of cases. This is attributed to the re-activation of the MIR with resulting control of bite force with the cuspid bite, confirming the previous research [10] and verified with EMG home sleep study recording.
This has significant implications in the treatment of obstructive sleep apnea (OSA) patients who also suffer from SB, as the two commonly occur together. The majority of OSA devices allow molar contact, which in SB maintains the suppression of the MIR. This often results is patient symptoms from SB and TTH. The FDA list the development of TMD as one of the known risks of mandibular advancement appliances in their guidance for these devices. The suppression of the MIR is likely the cause. The relationship between SB and TTH in supression of the MIR is strong. Research has shown that, even in young patients [21-33], this supression occurs, reinforcing the genetic cause [34].
Considering the MIR in the treatment of OSA and SB patients is critical. With a minimum of 8-10% of the population suffering from SB [1] and up to 95% suffering from primary headache [35] (which includes TTH) at some point in their life, this is a significant social problem. SB falls under dental treatment and the American Academy of Sleep Medicine classification of SB [1], states that a sleep study is not essential for a positive diagnosis of sleep bruxism. This puts this treatment in the realm of general dentistry. With the Luco Hybrid device (Figure 5) being the 1st treatment FDA cleared for the treatment of sleep bruxism/tension-migraine headaches (K160477), there is a safe and effective treatment available that any dentist can easily learn and impliment in their practices.
References
- American Academy of Sleep Medicine (2014) The International classification of sleep disorders 3rd edtn. Darien, USA.
- Mayer P, Heinzer R, Lavigne G (2016) Sleep Bruxism in Respiratory Medicine Practice. Chest 149: 262-271.
- Saito M, Yamaguchi T, Mikami S, Watanabe K, Gotouda A, et al. (2013) Temporal association between sleep apnea-hypopnea and sleep bruxism events. J Sleep Res 23: 96-203.
- Manfredini D, Winocur E, Guarda-Nardini L, Paesani D, Lobbezoo F (2013) Epidemiology of bruxism in adults: a systematic review of the literature. J Orofac Pain 27: 99-110.
- Calic A, Peterlin B (2015) Epigenetics and Bruxism: Possible Role of Epigenetics in the Etiology of Bruxism. Int J Prosthodont 28: 594-599.
- Inan R, Benbir G, Karadeniz D, Yavlal F, Kiziltan ME (2016) Brainstem reflexes in patients with sleep bruxism: Masseter inhibitory reflex responses and auditory startle reaction. Clin Neurophysiol 127.
- McMillan AS (1994) Motor-unit reflex inhibition in different regions of the human masseter muscle. Arch Oral Biol 39: 885-890.
- Tzvetanov P, Rousseff RT, Radionova Z (2009) Abnormalities of masseter inhibitory reflex in patients with episodic tension-type headache. J Zhejiang Univ Sci B 10: 52-56.
- Huang H, Song YH, Wang JJ, Guo Q, Liu WC (2014) Excitability of the central masticatory pathways in patients with sleep bruxism. Neurosci Lett 558: 82-86.
- Cruccu G, Cruccu G, Agostino R, Inghilleri M, Manfredi M, et al. (1989) The masseter inhibitory reflex is evoked by innocuous stumuli and mediated by A beta afferent fibers. Exp Brain Res 77: 447-450.
- Göbel H, Dworschak M (1996) [Exteroceptive suppression of activity of the temporal muscle. Principles and applications]. Nervenarzt 67: 846-859.
- Göbel H, Dworschak M, Wallasch TM (1993) Exteroceptive suppression of temporalis muscle activity: perspectives in headache and pain research. Cephalalgia 13: 15-19.
- Ä°nan R, Şenel GB, Yavlal F, Karadeniz D, Gündüz A, et al. (2016). Sleep bruxism is related to decreased inhibitory control of trigeminal motoneurons, but not with reticulobulbar system. Neurol Sci 38: 75-81.
- Neufeld JD, Holroyd KA, Lipchik GL (2000) Dynamic assessment of abnormalities in central pain transmission and modulation in tension-type headache sufferers. Headache 40: 142-151.
- Ongerboer de Visser BW, Cruccu G, Manfredi M, Koelman JH (1990) Effects of brainstem lesions on the masseter inhibitory reflex: functional mechanisms of reflex pathways. Brain 113: 781-792.
- Paulus W, Raubüchl O, Straube A, Schoenen J (1992) Exteroceptive suppression of temporalis muscle activity in various types of headache. Headache 32: 41-44.
- Schoenen JMD (1993) Exteroceptive suppression of temporalis muscle activity in patients with chronic headache and in normal volunteers: methodology, clinical and pathophysiological relevance. Headache 33: 3-17.
- Schoenen J, Bottin D, Sulon J, Gaspard U, Lambotte R (1991) Exteroceptive silent period of temporalis muscle in menstrual headaches. Cephalalgia 11: 87-91.
- Wallasch TM (1991) A study on the exteroceptive suppression of the masseter, temporalis and trapezius muscles produced by mental nerve stimulation in patients with chronic headaches. Cephalalgia 11: 162-163.
- Wallasch TM, Göbel H (1993) Exteroceptive suppression of temporalis muscle activity: findings in headache. Cephalalgia 13: 11-14.
- UginÄÂius P, Atiş ES, Türker KS (2014) Reflex responses of human masseter motor units to mechanical stimulation of the teeth. J Neurophysiol 111: 51-61.
- Wallasch TM, Reinecke M, Langohr HD (1991) EMG analysis of the late exteroceptive suppression period of temporal muscle activity in episodic and chronic tension-type headaches. Cephalalgia 11: 109-112.
- Wallasch TM, Niemann U, Kropp P, Weinschutz T (1993) Exteroceptive silent periods of temporalis muscle activity: correlation with neuropsychological findings. Headache 33: 121-124.
- Wang W, Schoenen J (1994) Reduction of temporalis exteroceptive suppression by peripheral electrical stimulation in migraine and tension-type headaches. Pain 59: 327-334.
- Wang W, De Pasqua V, Gerard P, Schoenen J (1995) Specificity and sensitivity of temporalis ES2 measurements in the diagnosis of chronic primary headaches. Headache 35: 85-88.
- Yu SK, Schmitt A, Sessle BJ (1973) Inhibitory effects on jaw muscle activity of innocuous and nocuous stimulation of facial and intraoral sites in man. Arch Oral Biol 18: 861-870.
- Zwart JA, Sand T (1995) Exteroceptive suppression of temporalis muscle activity: a blind study of tension-type headache, migraine, and cervicogenic headache. Headache 35: 338-343.
- Lipchik GL, Holroyd KA, O'Donnell FJ, Cordingley GE, Waller S, et al. (2000) Exteroceptive suppression periods and pericranial muscle tenderness in chronic tension headache: effects of psychopathology, chronicity and disability. Cephalalgia 20: 638-646.
- Headache Classification Committee of the International Headache Society (IHS) (2004) The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 33: 629-808.
- Schepelmann K, Dannhausen M, Kotter I, Schabet M, Dichgans J (1998) Exteroceptive suppression of temporalis muscle activity in patients with fibromyalgia, tension-type headache, and normal controls. Electroencephalogr Clin Neurophysiol 107: 196-199.
- Aktekin B, Yaltkaya K, Ozkaynak S, Oguz Y (2001) Recovery cycle of the blink reflex and exteroceptive suppression of temporalis muscle activity in migraine and tension-type headache. Headache 41: 142-149.
- Gastaldo E, Quatrale R, Graziani A, Eleopra R, Tugnoli V, et al. (2006) The excitability of the trigeminal motor system in sleep bruxism: a transcranial magnetic stimulation and brainstem reflex study. J Orofac Pain 20: 145-155.
- Ebinger F (2006) Exteroceptive suppression of masseter muscle activity in juvenile migraineurs. Cephalalgia 26: 722-730.
- Abe Y, Suganuma T, Ishii M, Yamamoto G, Gunji T, et al. (2012) Association of genetic, psychological and behavioral factors with sleep bruxism in a Japanese population. J Sleep Res 21: 289-296.
- Donnet A (2008) [Primary headaches]. Rev Prat 58: 608-615, 642.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi