Research Article, J Hydrogeol Hydrol Eng Vol: 8 Issue: 6
Hydrochemical Assessment and Seasonal Variations in Surface Water Quality Parameters of Ved pond in Ratanpur in Bilaspur, Chhattisgarh
Renu Nayar*
Department of Chemistry, DP Vipra College, Billaspur, Chhattisgarh, India
*Corresponding Author : Dr Renu Nayar
Department of Chemistry, DP Vipra College, Billaspur, Chhattisgarh, India
Tel: +91 9977413760
E-mail: nayar.renu@yahoo.co.in
Received: August 02, 2019 Accepted: August 16, 2019 Published: August 26, 2019
Citation: Nayar R (2019) Hydrochemical Assessment and Seasonal Variations in Surface Water Quality Parameters of Ved pond in Ratanpur in Bilaspur, Chhattisgarh. J Hydrogeol Hydrol Eng 8:2.
Abstract
In the present work Ved pond which are situated around the remote area in Ratanpur. People residing near these sites use water for bathing, washing clothes and for their other daily activities. These samples were analysed for 13 Physico-chemical parameters to evaluate their suitability for domestic application. The various parameters that affects the water quality such as Temperature, pH, Electrical Conductivity, Total dissolved solid, Dissolved oxygen, Biological oxygen demand, Transparency, Alkalinity, Nitrate, Phosphate, Calcium, Magnesium ,Total suspended solid, were investigated. In the present work, we selected 08 sampling points on peripheral sites in the zone i.e. 02 Sites in North direction, and 02 sites each in South West and East and eight sampling sites on inner sites (centre) in the Ved pond. Water samples were collected from various sampling sites from four directions North South East and West. Sampling sites were selected on the basis of margin length of water reservoirs at interval of 100 meter and 100 meter inside to the ponds. Considering the depth, the samples were collected from water surface and bottom of the ponds. A total of 08 representative water samples in peripheral sites and 08 samples in inner sites were collected from August 2018 to May19. This research is aimed to investigate the quality of surface water for rural people. The maximum transparency value of 37.0 ± 0.40 was recorded at S-1in the East marginal sites and 30 ± 0.22 at the East inner sites in the rainy season. The maximum mean value of pH was found to be 9.9 ± 0.41at S-1(north peripheral) in the summer season. The alkaline nature of the pond water values may be due to sewage discharged by surrounding villages and agricultural fields. The total load of salts in a water body is directly related to its conductivity. The EC values of the study area had a maximum mean of 1044 ± 44.1 (north peripheral) at S-2 in the summer season and 982 ± 29.2 (West inner) at S-b in the rainy season, its indicates the load of salts in a water body is directly related to its conductivity. DO and BOD values were always above the WHO permissible limit of at all sampling stations. Maximum mean BOD 39 ± 2.1was found at S-2 (West peripheral) at in the summer season .The mean value of BOD increases in the summer season indicating increase in organic load in water .Ca2+and Mg2+ ions ranges from 27.2 ± 14.8 (S-2, South Marginal in the rainy season) to 133.9 ± 31.0 (S-2, East Marginal in the summer season).and for inner sites it varied between 9.6 ± 4.2 (S-1 South marginal in the rainy season) and 31.7 ± 4.1(S-1 west marginal in the summer season). During the study the highest mean concentration of total nitrogen during summer season was observed as 67.5 ± 18.2 at S-2 North peripheral and a maximum of 58.7 ± 12.7 at S-s south inner in the summer season. The tabulated data shows this seasonal variation. Since there is variation in monthly climate over different seasons, therefore the average values of the parameters have been also been tabulated. The study shows that water of Ved pond is found to be polluted as this pond is surrounded on all sides by residential localities and sewerage from residences is continuously flowing into the pond. A number of instances of skin diseases in the people who were in contact with the pond water living nearby has been reported. Thus measures have to be adopted for the treatment of the Ved pond water.
Keywords: Global water; Hydrosphere; Inorganic chemicals; Physico-chemical
Introduction
Life was first originated in water. The global water scarcity is increasing and as such it has become necessary to take new water sources that we earlier may have thought of as inaccessible in to consideration. Water is one of the most important constituents of the living organism in the world [1]. Water is universal and excellent solvent as it dissolves wide range of substances then other common solvent [2]. So no other compound can be compared to water as solvent. Water has many unique and global characteristics that make it suitable for living being to survive in different conditions for life. Water quality status in a single term that is helpful for the selection of appropriate treatment technique to meet the concerned issues. Hydrosphere covers more than 75% of earth surface as fresh water. Sea water, rivers, oceans, lakes and pond water form a hydrosphere, which contains more than 35 ppt of dissolved solids. Natural water always contains dissolved and suspended substances of organic and inorganic mineral. Mostly ground water contains dissolved substances while surface water is rich in suspended matter [3].
Ratanpur is situated about 25 km from Bilaspur and Korbamain road. It is the famous holy and mythological place of Adishakti Mahamaya Devi. Ratanpur is a Nagar Palika in Bilaspur district in the Indian state of Chhattisgarh, situated at 22.3°N and 82.17°E. The town Ratanpur is attractive as a religious canter and more than 150 Ponds situated during ancient period as cited in history. That is why; Ratanpur Nagar is famous known as “Temple City” as well as “City of Ponds”.
The Physico-chemical analysis of pond water in rural areas of India are still certain areas which should be targeted for determination of surface water quality where varied human activities are taking place [4]. Due to improper drainage system, septic tanks and solid waste disposal, contamination of pond water by organic, inorganic chemicals and micro-organisms is likely to occur. A survey of literature shows that there is no systematic study of quality of surface water in Ratanpur and its surrounding rural areas. Hence it is proposed to investigate the quality of water in Ratanpur and its surrounding areas where some major ponds are situated and used by people in the form of different activities [5]. To locate the sampling sites during present investigation, the entire area of Ratanpur Nagarpalika has been divided into four zones (A, B, C and D). Zone-B consists of a two large pond named as Ved and Ratneshwer. Zone B consists of two large ponds named as Ved and Ratneshwer. However for the present investigation we have chosen the largest pond Ved in zone B. Where the background of intense anthropological activities at the borders of the Ved pond. Due to improper drainage system, septic tanks and solid waste disposal, contamination of ground water by organic chemicals and microorganisms is likely to occur. Similarly contamination due to inorganic chemicals from seepage of effluents from industries is also possible. The pond is anthropogenic and its water used for domestic purpose and irrigation. The pond is surrounded by semi urban and semi agriculture area (Figure 1).
Objectives of the Present Work
There is much available work on the Physico-chemical quality of pond water in rural areas [6]. but there are still few areas which should be targeted to determine the surface water quality where different human activities are taking place. Due to improper drainage system, septic tanks and solid waste disposal, contamination of pond water by organic, inorganic chemicals and micro-organisms is likely to occur. A survey of literature shows that there is no systematic study of quality of surface water in Ratanpur and its surrounding rural areas. Hence it is proposed to investigate the quality of water in Ratanpur in Chhattisgarh and its surrounding areas where some major ponds are situated and used by people in the form of different activities such as washing clothes and dishes, bathing animals etc. A regular monitoring of pond will help in taking long-term precautions, which will be helpful in protecting public health in the study area. In the present study the Physico-chemical analysis of Ved pond water samples was done in rainy winter and summer season and mean values of the different measured samples were compared with the World Health Organization (WHO) Guideline Values (GVs) for drinking water quality [7].
Materials and Methods
Samples sites and water samples collection in four directions
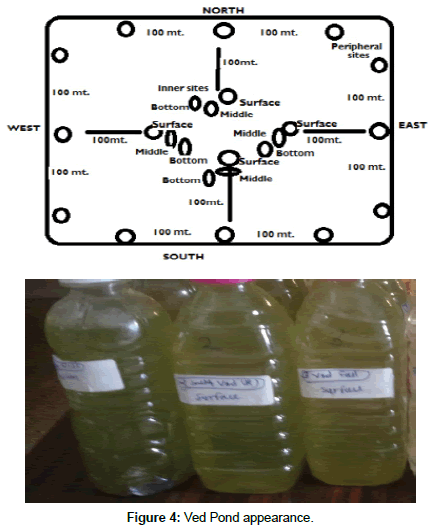
8 peripheral Sites and 8 inner sites covering all the directions were chosen for the Physico-chemical analysis of water of this pond. In the north of the pond over a spread of 430 m, and 380 m spread in the south direction. 08 sampling sites were chosen, each 100 m apart, these sites are termed as peripheral/Marginal sites in all direction. Nearly 100 m from the shore, 08 sites were chosen in the middle/inner of the pond (Table 1).
| Pond water | Peripheral length | Sampling sites | |||
|---|---|---|---|---|---|
| Direction | Length (approx. in meter) | Marginal sites | Inside | ||
| Depth (In feet) |
Inner sites | ||||
| Ved | NORTH | 430 | Vn-1, Vn-2, | 05 | Vn-S, Vn-B |
| EAST | 370 | Ve-1, Ve-2 | 06 | Ve-S, Ve-B | |
| SOUTH | 380 | Vs-1, Vs-2 | 05 | Vs-S, Vs-B | |
| WEST | 270 | Vw-1, Vw-2 | 05 | Vw-S, Vw-B | |
Table 1: Sampling sites determined as per water area of Ved Pond System.
To assess the quality of water in Ved pond, the Physico-chemical analysis of water samples were performed at seasonal interval. Samples were collected from different sites following the standard sampling methods at regular monthly intervals throughout the seasons (Rainy, winter and summer). Physical parameters of water samples were measured on the spot at the time of sample collection employing digital pH meter and thermometer (Table 2). For other parameters of chemical analysis, water samples were carried out to laboratory in icebox an insulated container and further analyses were preceded within 24 hours. The samples were kept in refrigerator maintaining below 5°C temperature that was used for further analysis. Sampling sites and the picture of four directions in Ved pond are shown in Figures 2-4. Various sampling stations (Table 1) around this pond have been selected which are having pond water. Water samples were collected every month for a period of one year August 2018 to June 2019.
| S.NO | Parameters | Abbreviations | Units | Method used |
|---|---|---|---|---|
| 1 | Temperature | Temp. | °C | Thermometer |
| 2 | pH | Water analyzer kit (systronic) model no 371 | ||
| 3 | Electrical conductivity | EC | mhos/cm | Water analyzer kit (systronic) model no 371 |
| 4 | Total dissolved solid | TDS | mg/l | Water analyzer kit (systronic) model no 371 |
| 5 | Dissolved oxygen | DO | mg/l | Water analyzer kit (systronic) model no 371 |
| 6 | Biological oxygen demand | BOD | mg/l | 5 days incubation at 20°C and titration of initial and final DO |
| 7 | Transparency | Trans. | cm | By Secchi disk |
| 8 | Alkalinity | -------- | mg/l | Titrimetric method (WITH .01 N H2SO4) |
| 9 | Nitrate | NO3- | mg/l | Spectrophotometer |
| 10 | Phosphate | P04--- | mg/l | Spectrophotometer |
| 11 | Calcium ions | Ca++ | mg/l | EDTA (.05N) Titrimetric method |
| 12 | Magnesium ions | Mg++ | mg/l | EDTA (.05N) Titrimetric method |
| 13 | Total suspended solid | TSS | mg/l | Gravimetric method |
Table 2: Methods used for determination of physicochemical parameters.
Physico-chemical and water quality analysis of Ved pond
People residing near the Ved pond use water for bathing, washing clothes and for their other daily activities. There were analysed for 13 Physico-chemical parameters to evaluate their suitability for domestic application. The various parameters that affects the water quality such as Temperature, pH, Total dissolved solid, Electrical Conductivity Dissolved oxygen, Biological oxygen demand, Alkalinity, Nitrate, Phosphate, Calcium, Magnesium, Total suspended solid, were investigated. Parameters such as pH, Temperature, EC, TDS, DO were measured at the sample collection sites using Water Quality Analyser Kit (Systronic model no 371). Spectrophotometric work was determined by using Carl-Zeiss Spekol-10 spectrophotometer for analysis of nitrate, and phosphate [8,9]. Determination of other parameters were carried out by standard methods as prescribed by APHA, Manivaskamand NEERI manual on water and waste water analysis. The samples were collected from each site at peripheral (about 100 m and inner (10 m from the shore) locations. Seasonal variation (Rainy, winter and summer) plays an important role in determining the Physico-chemical as well as microbial characteristics of water. The tabulated data shows this seasonal variation. Since there is variation in monthly climate over different seasons, therefore the average values of the parameters have been also been tabulated. Water was sampled in to 1 L polyethylene containers. Samples of surface water were collected for chemical analysis from peripheral areas, inner surface and bottom of the pond once a month. A total of 8 representative water samples in peripheral sites and 8 samples in inner sites were collected from August 2018 to June 2019. All water samplings were completed from 8 to 10 o’clock in the morning. The methods adopted for analysing the various Physico-chemical parameters are listed in table 2.
Results and Discussion
Water appearance of Ved pond
Water of Ved pond was appearing more turbid and greenish in colour. The colour of water is yellow-green in all four directions such as north east west and south and it has a smell of rotten eggs, in the North and south direction probably. A green bloom was floating on the surface in stagnant and shallow areas but bloom was invisible under the depth. Dense populations of small and big fish known to be present in and around the pond.
Water quality of ved pond
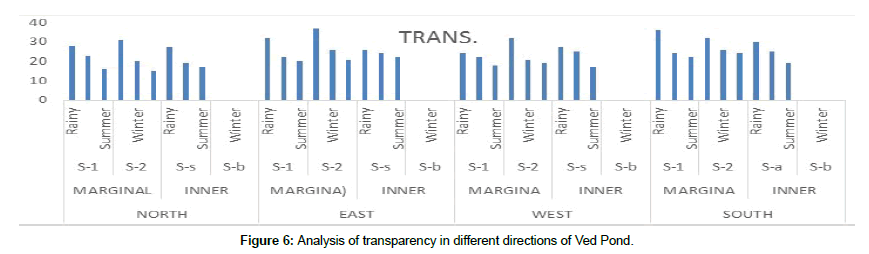
Transparency of natural waters is an indicator of productivity. The transparency of the fresh water is 35 to 45 cm which is suitable for the aquatic environment [10]. Suspended particles in our water behave similarly to dust in the atmosphere. They reduce the depth to which light can penetrate. Sunlight provides the energy for photosynthesis. The penetration depth of light in water determines the depth to which aquatic plants can grow. Transparency decreases with the presence of molecules and particles that can absorb or scatter light. Transparency measurement done was measuring the depth of light penetration into the body of water using the Secchi disc. The low-cost disc was self-fabricated and designed (Figure 5) as its price quoted by manufacturers was high. A higher transparency value was observed during summer due to absence of rain, runoff and flood water as well as gradual setting of suspended particles [11].
Observed data about Physico-chemical analysis of water samples collected during three seasons-Rainy (Up to October) Winter (Up to February) and summer (up to June) have been computed (Mean ± SD) as mentioned in Annexure 1. The maximum transparency value of 37.0 ± 0.40 was recorded at S-1in the East marginal sites and 30 ± 0.22 at the East inner sites in the rainy season. The transparency of water body is affected by the factors like plank tonic growth, rainfall, Suns position in the sky, angle of incidence of rays, cloudiness, visibility and turbidity due to suspended inert particulate matter. Our results revealed that the lowest water transparency value was recorded 16.0 ± 0.41 at S-1 in the North marginal sites during summer season (Figure 6). The temperature ranged between 31.7 ± 1.8 at (S-1) (south peripheral) in rainy season and 24.2 ± 1.6 (S-2) (north peripheral) in winter season whereas for the inner sites it varied between 38.7 ± 2.6 (west inner) in the Summer season to 23.2 ± 1.4 (north inner) in the winter Water temperature is generally low, ranging from 5°C-36°C. This is standard for fish and other aquatic organisms. The range of water temperature was found within the standard range (17.2°C-33.4°C) and higher in rainy season due to moistures in atmosphere and high air temperature. During this study we observed the difference of 2°C to 3°C between water surface and inner site.
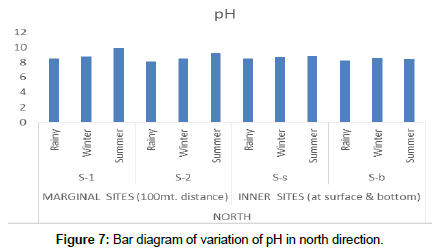
pH of an aquatic system is an important indicator of water quality and the extent of pollution in watershed areas. During the assessment period the maximum mean value of pH was found to be 9.9 ± 0.41 at S-1(north peripheral) in the summer season, and at the inner sites a maximum of 9.1 ± 0.47 at S-s (South inner) surface in the rainy season. The alkaline nature of the pond water values may be due to sewage discharged by surrounding villages and agricultural fields (Figure 7). Sewage and agricultural discharges are generally a complex combination of natural organic and inorganic materials and man-made compounds. It usually contains many fertilizers, metals, sediments, pesticides, nutrients, salt, sodium, calcium, potassium, chlorine, phosphate, bicarbonate etc. [12].
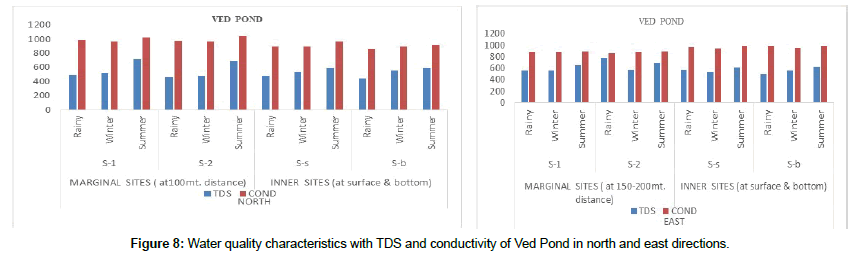
In our study area, the Total Dissolve Solids (TDS) was found maximum in the rainy season. TDS has a positive correlation with water temperature not exceeding the standard level. The TDS value of Ved pond is not harmful for aquatic life. Maximum TDS of 771 ± 16.8 was observed at S-2 (East Peripheral) in the rainy season (Figure 8). Total suspended solids include clay, sand, various silicates, and microorganisms etc. [13]. The suspended TSS matter is determined by filtering the sample, drying the residue and determining its weight by difference. Total suspended solids mostly occur in wastes and sewage material. The TSS values were always above the WHO permissible limit of 50 mg/l. Total suspended solids include the coarser as well as, the reparable particulates. Suspended solids containing much of organic matter may cause putrefaction and consequently result in depletion of dissolved oxygen [14]. Mineral matter in suspended form may lead to silting, thereby resulting in damages to aquatic plant and animal life. Presence of suspended solids also increases the turbidity, making the water unsuitable for domestic use too. In the present investigation, the maximum total suspended solids was 127.3 ± 11.4 at S-2 (North peripheral) in the summer season and 86.5 ± 9.7 (East inner) at S-b in the winter season.
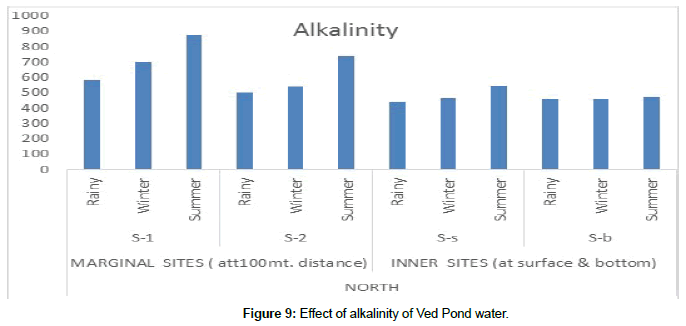
Conductivity is a measure of the ability of water to conduct electricity. It depends on the ionic concentration and water temperature. The total load of salts in a water body is directly related to its conductivity. The EC values of the study area had a maximum mean of 1044 ± 44.1 (north peripheral) at S-2 in the summer season and 982 ± 29.2 (West inner) at S-b in the rainy season (Figure 9) Alkalinity of natural waters is normally due to the presence of bicarbonate, carbonate and hydroxide compounds of calcium, magnesium, sodium and potassium. Maximum alkalinity 873.7 ± 47.7 was observed at S-1(North peripheral) in the summer season and 750.66 ± 40.6 at S-b (west inner sites) in the rainy season [15].
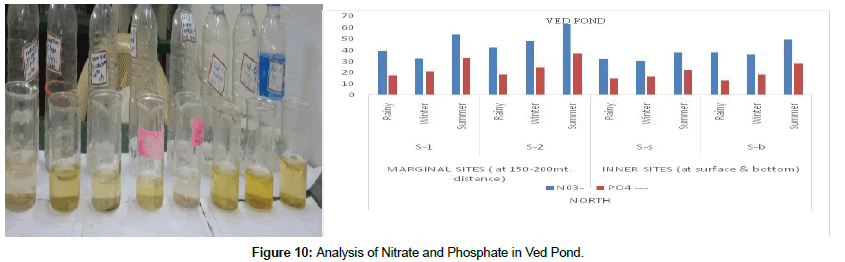
Dissolved oxygen values were always above the WHO permissible limit of 4 mg/l at all sampling stations. It is a physical phenomenon and depends upon the solubility of oxygen, which in turn is influenced by water temperature. Maximum value of DO 4.8 ± 0.43 was observed at S-2 (West Peripheral) in the winter season and 4.6 ± 0.43 at S-b (North Inner) in the winter season. This means DO decreases with high humidity and a rise in water temperature. Maximum mean BOD 39 ± 2.1 was found at S-2 (West peripheral) at in the summer season. The mean value of BOD increases in the summer season indicating increase in organic load in water. Calcium is the major component of natural waters. It is associated with rocks, sediments and also with natural activities. Rocks in the surrounding, catchments area and in contact with water are its major source. Domestic effluents also contribute towards calcium contents of any water body. During the study, the mean concentration of actions such as Ca2+ and Mg2+ ions ranges from 27.2 ± 14.8 (S-2, South Marginal in the rainy season) to 133.9 ± 31.0 (S-2, East Marginal in the summer season) and for inner sites it varied between 9.6 ± 4.2 (S-1 South marginal in the rainy season) and 31.7 ± 4.1 (S-1 west marginal in the summer season). Magnesium is essential for normal plant growth and is desirable in water for irrigation. The main sources of magnesium in water are natural rocks, agricultural practices and industrial effluents etc. During the study the highest mean concentration of total nitrogen during summer season was observed as 67.5 ± 18.2 at S-2 north peripheral and a maximum of 58.7 ± 12.7 at S-s south inner in the summer season (Figure 10). Nitrogen fertilizers are applied in very large amounts in field crops. Since the plants cannot utilize all the nitrogen applied to the fields, some is left in the soil and it can leach into ground water. In addition, not all the applied nitrogen gets into the soil and some are washed off the fields in the form of runoff and it flows into surface waters. Increased level of nitrate-nitrogen in water bodies causes nutrient enrichment, which results in enormous growth of aquatic filamentous green algae, leading to eutrophication. This huge algal growth creates anaesthetic conditions in the water body. Some of the blue green algae species are reported to be toxic. Anaerobic conditions are developed in the water body due to the oxygen uptake during night time, which in turn, is not compensated by photosynthesis. Phosphorus occurs in natural waters and waste waters in the form of various phosphates. Surface waters seldom contain high concentration of phosphates, since they are utilized by plants whereas ground waters usually contain appreciable amounts of phosphate.
Conclusion
The study shows that water of Ved pond is found to be polluted as this pond is surrounded on all sides by residential localities and sewerage from residences is continuously flowing into the pond. A number of instances of skin diseases in the people who were in contact with the pond water living nearby have been reported. Thus measures have to be adopted for the treatment of the pod water.
Suggestions
With sensible policies, water sources can be protected from pollution, the drainage canals and septic tanks must be constructed far away from the wells. Generally, water pipelines and septic pipe lines of the town run parallel to each other thereby giving chances for contamination of drinking water. Enough care should be taken by municipal authorities to separate these lines to prevent faecal contamination. Faecal contamination may takes place because of open defecation practices, running of water pipes close to sewage lines. Open air defecation along the canal bunds should be banned. Good sanitation practices should be implemented near open water sources. Regularly biochemical and microbiological tests should be carried out so as to protect the public from waterborne disease outbreaks. Ozonisation in place of chlorination is recommended. Activities including dredging of sediments, construction of new sewerage treatment plants, outlets of sewerage water should be diverted.
Acknowledgement
The author acknowledges the support from State planning commission Raipur (Chhattisgarh) for the financial assistance in form of Major Research Project and expresses gratitude to the Vice-Chancellor Dr G.D.Sharma of the Atal Bihari Vajpayee University, Bilaspur Chhattisgarh for his expert guidance, excellent supervision and constant encouragement given to me throughout the period of the work. I express my deep gratitude to Dr DK Shrivastav, Professor and Head of the department of Microbiology, Govt. Science College, and Bilaspur for his keen interest shown in the progress of this work and for constantly motivation. I am thankful to NEERI, Nagpur and Orissa University of Agriculture and Technology, Bhubneshswer for providing library and laboratory facilities.
References
- Padmavathy S, Amali J, Raja RE, Nagarajan P (2003) A study of fluoride level in potable water of Salem district and an attempt for de fluoridation with lignite. Indian Journal of Environmental Protection 23: 1244-1247
- Abdul S, Mallikarjun ND, Vijay Kumar K (2012) Correlation-regression model for physicochemical quality of groundwater in the south India city of Gulbarg. African J of Environmental Science and Technology 6: 353-364
- Upadhyay K, Mishra P, Gupta AK (2010) Studies on the physico-chemical status of two ponds at Varanasi and Bhadohi under biotic stress. Plant Archives 10: 691-693.
- Sayadi MH, Sayeed MRG, Suyash K (2010) Short-term accumulative signatures of heavy metals in river bed sediments in the Industrial area, Tehran, Iran. Environ Monit Assess 162:465-473.
- Devendra Singh P, Seema G (2017) Assessment of distribution patterns of wetland birds between unpolluted and polluted ponds at Ratanpur District Bilaspur Chhattisgarh, India. Indian J Sci Res 12: 204-215.
- Pankaj N, Dhan Raj (2018) Water quality assessment using physico-chemical parameters of palasani pond, Jodhpur District, Rajasthan, India. IJRAR
- APHA (2005) Standard Methods for the examination of water and waste water. American Public Health Association
- Manivaskam N (2000) Physico chemical examination of water, sewage and industrial effluents.
- NEERI (1988) Manual on water and waste water analysis, National Environmental Engineering Research Institute. Nagpur, India.
- Hossain KM, Hassan M, Parvin N, Hasan S, Islam S, et al. (2012) Antimicrobial, cytotoxic and thrombolytic activity ofCassia sennaLeaves (Family: Fabaceae). J Applied Pharm Sci 2: 186-190.
- Khan MAG, Chowdhury SH (1994) Physical & chemical Limnology of lake Kaptai, Bangladesh Trop Ecol 35: 35-51.
- Vaghasiya Y, Dave R, Chanda S (2011)Phytochemical analysis of some medicinal plants from Western region of India. Res J Med Plant 5: 567-576.
- Medudhula T, Samatha C, Sammaiah CH (2012) Analysis of water quality using physico-chemical parameters in lower manair reservoir of Karimnagar district, Andhra Pradesh. Inter J of Environ Sci 3: 172-177
- Trivedi RK, Goel PK (1986) Chemical and biological method for water pollution studies.
- Venkatesharaju K, Ravi Kumar P, Somashekar RK, Prakash KL (2010) Physico-chemical and Bacteriological Investigation on the river Cauvery of Kollegal Stretch in Karnataka. Int J Innov Res Sci Eng Technol 6: 50-59.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi