Research Article, J Med Toxicol Res Vol: 1 Issue: 1
Methadone Extraction by Modified QuEChERS Method in Post-mortem Samples by Gas Chromatography-Mass Spectrometry
Seyed Majid Salami Asl1*, Mohammad Javad Khodayar2, Zahra Mousavi3 and Maryam Akgagri4
1Department of Toxicology, Pharmaceutical Sciences Branch, Islamic Azad University, Tehran, Iran
2Department of Toxicology, School of Pharmacy, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
3Department of Pharmacology -Toxicology, Faculty of Pharmacy, Pharmaceutical Sciences Branch, Islamic Azad University, Tehran, Iran (IAUPS)
4Departments of Forensic Toxicology, Legal Medicine Research Center, Legal Medicine Organization, Tehran, Iran
*Corresponding Author : Seyed Majid Salami Asl
Department of Toxicology, Pharmaceutical Sciences Branch, Islamic Azad University, Tehran, Iran
E-mail: majid.salimi2012@yahoo.com
Received: October 31, 2017 Accepted: January 08, 2017 Published: January 15, 2018
Citation: Asl SMS, Khodayar MJ, Mousavi Z, Akgagri M (2018) Methadone Extraction by Modified QuEChERS Method in Post-mortem Samples by Gas Chromatography-Mass Spectrometry. J Med Toxicol Res 1:1.
Abstract
Background: Extraction in the first line toxicology laboratory is the stage of analysis and diagnosis, the diagnosis process of the drug is completely dependent on the extraction method. New high-efficiency extraction methods, replace past practices. QuEChERS extraction method has already been introduced in pesticide extraction. In forensic toxicology, autopsy samples from the bodies to investigate the cause of death after extraction and analysis are interpreted by different devices.
Objective: In this paper, extraction of methadone drug substance by QuEChERS method was investigated in autopsy samples.
Method: QuEChERS extraction method an easy and fast tube method using MgSO4 and NaCl salts and ethyl acetate solvent based on the diagnosis with Gas Chromatography-Mass Spectrometry.
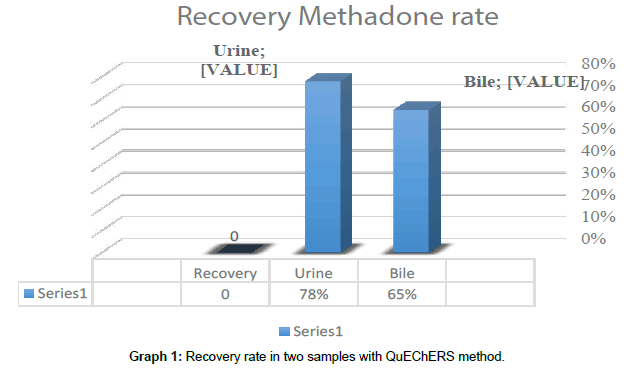
Results: Methadone recovery rates in urine specimens and contents of gallbladder were evaluated. The recovery rate of methadone analysis in the samples was found to be 65%-78% by the QuEChERS method (N = 30).
Conclusion: The modified QuEChERS extraction method, as a fast, effective, and green tube method, can replace alternate methadone detection methods in postmortem samples.
Keywords: Autopsy; Gallbladder; GCMS; Methadone; Urine
Introduction
QuEChERS abbreviation of Quick, Easy, Cheap, Effective, Rugged, and Safe. Introduction in Forensic Toxicology, Extraction is usually done by Liquid-Liquid Extraction (LLE) and Solid Phase extraction (SPE). The SPE method is expensive because of the cost of the cartridge and the cleaning of the homogenizer probe creates a risk of pollution and cross-reactivity, and therefore the SPE method is somewhat tedious and time-consuming and cannot be performed in many centers, but the QuEChERS method is a simple method. , Fast and reliable [1]. This method is widely used to determine pesticides and was first introduced in 2003[2]. Anastassiades and colleagues first introduced this method in 2003 to extract veterinary drugs from animal tissue. An important step in the analysis of extracted toxicological samples is that liquid-liquid extraction and solid phase extraction are used as methods for pre-toxic analysis [3,4]. Preparation of the sample is one of the critical steps in the proper diagnosis of a wide range of drugs and toxins by mass spectrometric chromatography (GC-MS) and liquid-mass spectrometry (LC-MS) chromatography, which extraction methods such as protein deposition, LLE and SPE has been designed and used [5,6]. Clinical toxicology and forensic medicine are heavily dependent on the science of decomposition chemistry. Blood, urine, and solid tissue as alternative liquids for oral, hair, and meconium are commonly used for toxicology [7,8]. The liquid-liquid extraction method as well as the extraction method by sedimentation of proteins, in spite of simplicity, inadequate sample extraction and contamination of the tool reduces the value of these methods. The SPE method has a lower selective performance than the protein precipitation extraction (PPE) or LLE interactions, but this method is relatively laborious and time-consuming. The QuEChERS method was proposed for the analysis of legal toxicology [9]. The QuEChERS approach is similar to PPE and LLE in terms of simplicity and is also similar to SPE with high selective performance [9,10]. At the present time, the Cochrane method is used in analyzing the pesticide residues of chemical fertilizers in food products [9,11]. There are two major problems in the extraction of drugs in chromatography techniques: one is the involvement of a matrix, and another is the suppression of ions [12]. Generally QuEChERS extraction is based on extracting with a solvent such as acetonitrile or ethyl acetate and dehydration in the presence of salts such as magnesium sulfate and sodium chloride. It is mainly administered in the treatment of heroin addiction and has a morphine-like function [13]. Some methadone is metabolized to the 2-ethyldiin-1,5-dimethyl-3,3-diphenyl-pyrrolidine (EDDP) by the enzyme cytochrome P450 hepatic (CYP3A4). The EDDP ratio to parametric methadone is used in the diagnosis of long-term and short-term treatment [14]. But EDDP as an artificial side effect is also the result of high temperature GC effects on methadone [15].
The main goal of this paper is to investigate the extraction of methadone with QuEChERS method in post-mortem samples by GC-MS. In forensic toxicology, autopsy samples from the bodies to investigate the cause of death after extraction and analysis are interpreted by different devices.
Materials and Methods
Methadone standard was used at concentration of 100 μg/ml (Sigma-Al-Drech, Germany), methanol, ammonia, hydrochloric acid, ethyl acetate, waterless magnesium sulfate, sodium chloride and deionized dispersed dispersant (Merck, Germany). Methadone standard 100 μg ml was prepared in methanol at a concentration of 2000 ng/ml as a working solution, then prepared from a standard dilution serial dilution of 2000, 1000, 500, 250, 125 and 62.25 ng/ml.
Sampling and extraction of samples by QuEChERS method
Urine samples, contents of the gallbladder bodies were dumped in accordance with legal requirements and according to their records at the site of forensic medicine. It was kept in order to avoid secondary variations until the time of doing it at a negative temperature of 20°C

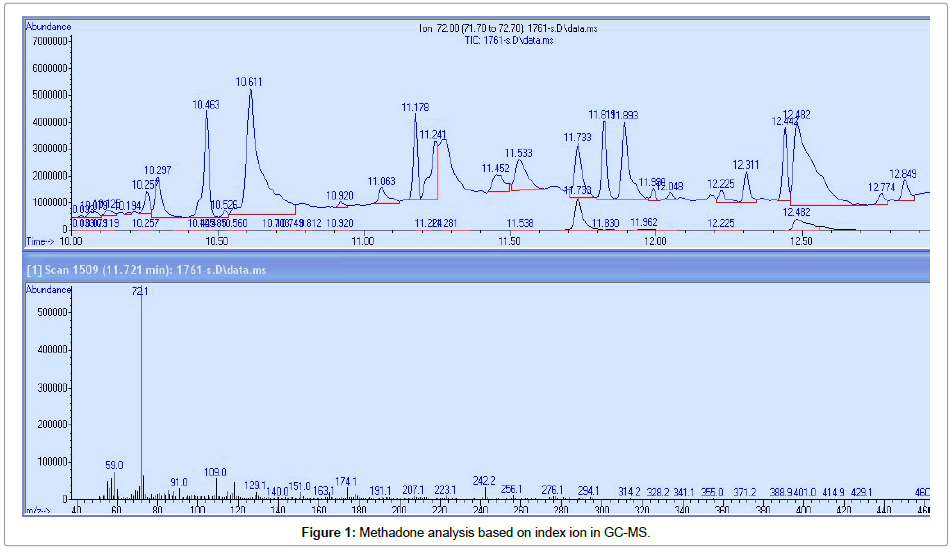
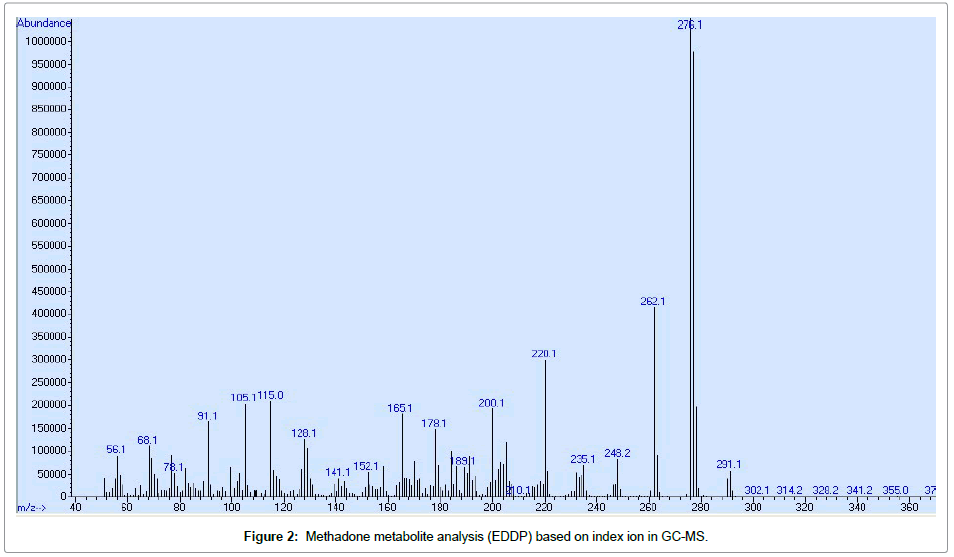
For analyzing the gas chromatography model 7890A, connected to the Agilent mass spectrometer Model 5975C equipped with a column of 30 m * 0.25 mm * 0.25 μm, which was used as a fixed phase with 5% phenyl methyl. The oven temperature increased from 60°C to 280°C at a temperature of 10°C and was kept constant at 280°C for 10 min. Helium gas was used with a purity of 99.999 at a flow rate of 1ml/ min and a mass energy of 70 ev. Split less injection mode and runtime length of 30min. Methadone identification was performed based on ion index and SIM method Table 1. The results were evaluated using the index ion and mass (m/z) with the Wiley7n.1 library software, GC-MS device (Figures 1 and 2).
| Analyte | Quantifier(m/z) | Qualifier(m/z) | RT |
|---|---|---|---|
| Methadone | 72 | 294.1223165 | 11.7 |
| EDDP | 276 | 262.12201165 | 11.1 |
Table 1: Specifications of analyte and index ions in GC-MS.
Validation method
Selectivity: Selectivity is the ability of an analytical method to detect and measure the analyte in the presence of other endogenous components within the sample (2).
Carry-over effect: During the validation process, the injection of a standard methadone sample containing 100ng/ml of a solvent of ethyl acetate or methanol-free methadone was injected, which revealed the results of the lack of recognition of methadone in the solvent.
Limit of Detection (LOD) and Limit of Quantitation (LOQ): Based on the signal to noise ratio, noise was measured by comparing the signals. A typical standard concentration sample was used to determine the minimum concentration level in which the analysis was performed. Signal-to-noise ratios of 3:1 and 1:10 were considered acceptable for LOD and LOQ, respectively. LOD and LOQ values were 29.1 ng/ml and 97.3 ng/ml, respectively [16]. The reason why the LOQ values obtained from legal data are higher than the published material is that most of the data are reported by MSL (LC-MS) chromatography with high pressure fluid (HPLC), which is used to analyze and Drug analysis is very sensitive [17]. Nevertheless, GC-MS is described as a useful method for analyzing legal and clinical cases due to its high repeatability in the diagnosis of opioids, Lerch et al. [2,18].
Linearity: The preparation of standard concentrations and the drawing of the standard curve of linearity were determined by drawing the standard curve and least squares regression. In order to prepare standard solutions, a stock solution was first provided at a concentration of 100 μg / ml, and then standard solutions were prepared by dilution to plot the standard curve and the regression equation. 3 concentrations of 62.5, 250 and 2000 ng/ml in methanol were prepared and then injected into GC-MS device in three replicates volume of 1 μl.
Precision: The precision of the analytical method describes the closeness of the individual measurements for an analyte. The exact daily data was measured by analyzing the area of the surface below the level of the analyzed analyte in the three levels of the following standard internal scale at three different concentrations (low of 62.5, average of 250 and above 2000 ng/ml). For the calculation of daily precision, the values of three concentrations injected for three consecutive days were used and CV% of the individual concentrations were obtained. The CV% represents the precision of the method, and the less the accuracy of the method is higher (Table 2).
| N=3 | Y=mx + b | Concentration range (ng/ ml) | LOD (ng/ml) | LOQ (ng/ml) | Quality Control Concentration (ng/ml) | Precision (n=3%) | |
|---|---|---|---|---|---|---|---|
| yaD=1 | y= 0.00006 + 0.0018 | 62.5-2000 | 62.5 | 13.5 | |||
| Methadone | Day=2 | y= 0.00004+ 0.0071 | 62.5-2000 | 29.1 | 97.3 | 250 | 8 |
| Day=3 | y= 0.00005+ 0.0038 | 62.5-2000 | 2000 | 11 |
Table 2: Methadone validation and its results.
Accuracy: Accuracy is an analytical method; the closeness of the determined value from the method to the true concentration of the analyte is described as 50%. The accuracy of the data is based on the calculation of the recovery rate obtained from different concentrations of the sample. The recovery rate set by the ICH for testing accuracy is 80 to 120 percent (International Conference on Harmonization, 1995). To perform the recovery test, three different concentrations (low 62.5, average 500 and above 1000 ng/ml) were added to the gallbladder three times a day from standard methadone solution to homogeneous urine specimen. After extraction by QuEChERS extraction and liquidliquid extraction, the samples were injected into the GC-MS machine and the percent recovery was calculated. Accuracy is through the addition of methadone to the body samples (urine, contents of the gallbladder and liver), which had already been negated by methadone.
The recovery rate of methadone analysis in different samples in the QuEChERS method was 65% -78% (Graph 1). Usually recovery methods should be between 80% - 120%. According to Anzillotti et al., For QuEChERS, recovery of 60-70% for intermediate and nonpolar compounds is acceptable [19].
Discussion
The most commonly used method of extraction is in most of the LLE forensic centers. Due to the large volume of required samples and the high amount of solvents that require a lot of time and money, along with other problems such as inadequate extraction of samples and contamination of tools and the environment, the value of this The method is reduced [9]. Potential properties of carcinogenicity and mutagenicity of compounds such as chloroform and ammonia are a threat to the health of staff and experts in forensic centers; in addition, the use of high volumes of solvents and environmental chemicals also threatens human society. As a result, finding an efficient way with lesser risks along with a greener and nonpolluting environment will force us to use new methods such as SPE and QuEChERS method. QuEChERS was approved as a sensitive, renewable and relatively simple method for qualitative and quantitative analysis of drugs and drugs. Since this method does not require specific equipment and requires less time, it shows that it has great potential for analyzing clinical and legal samples 2. In the case of samples such as the liver, kidney, lung, muscle tissue and adipose tissue, more needs to be done for the preparation process to increase the use of developed methods in clinical and legal toxicology [20,21]. The high LOQ values obtained from legal data in comparison with printed materials is that most of the data in the articles have been reported by LC-MS high pressure liquid chromatography (HPLC), which is used to analyze drugs It is very sensitive [17]. However, in a study by Lerch et al., GC-MS is a useful method for legal and clinical diagnosis of narcotics due to its repeatability [2,18]. Therefore, with these changes during these years, the Cochrane Extract Method has become capable of becoming an inclusive approach to the separation of many drugs and drugs.
Conclusion
Most articles and outcomes of the study are on non-corpuscular samples and for launching and extraction methods but bodies of bodies are not fully applicable to post-mortem samples due to phenomena such as post-mortem corruption, post-mortem distributions, post-mortem distribution, and so on. It also requires access to body samples that complicate the legal and legal barriers. In this study, there was no significant difference between the results of the detection of methadone found in body samples in terms of clinical, legal, and judicial analysis of cases; As a result, due to higher methadone recovery in the QuEChERS method, this method can be a better alternative than liquid-liquid extraction (LLE) in post-mortem samples.
Conflict of Interest
In the results of this thesis, there was no conflict with legal and legal issues.
Financing and Support
The cost of this thesis was completely personalized and with the scientific support of the Department of Toxicology, Faculty of Pharmacy, Jundishapur, Ahvaz, and the Legal Medicine Organization of Khuzestan-Ahvaz Province and the Toxicology Group of the Pharmaceutical Sciences University of Islamic Azad University, Tehran.
Acknowledgments
At the end of the family, Stahid and friends who helped me in this study, I have the great thanks.
References
- Yoshie H, Yui l, Tadashi H, Masaki H, Usui K et al. (2014) Application of modified QuEChERS method to liver samples for forensic toxicological analysis. Forensic Toxicol 32: 139-147.
- Amorim Alves E, Ana SA, Sara MC, Carlos MA, Annibal DPN et al. (2017) GC-MS Method for the Analysis of Thirteen Opioids, Cocaine and Cocaethylene in Whole Blood Based on a Modified Quechers Extraction. Curr Pharm Anal 13: 215-223.
- Bogusz MJ (2000) Liquid chromatography-mass spectrometry as a routine method in forensic sciences: a proof of maturity. J Chromatogr B Biomed Sci Appl 748: 3-19.
- Sauve E, Langødegård M, Ekeberg D, Øiestad AM (2012) Determination of benzodiazepines in ante-mortem and post-mortem whole blood by solid-supported liquid–liquid extraction and UPLC–MS/MS. J Chromatogr B 883:177-188.
- Maurer HH (2009) Mass spectrometric approaches in impaired driving toxicology. Anal Bioanal Chem 393: 97-107.
- Maurer HH (2013) What is the future of (ultra) high performance liquid chromatography coupled to low and high resolution mass spectrometry for toxicological drug screening? J Chromatogr A 1292: 19-24.
- Dinis-Oliveira R, Carvalho F, Duarte JA, Remião F, Marques A, et al. (2010) Collection of biological samples in forensic toxicology. Toxicol Mech Methods 20: 363-414.
- Dinis-Oliveira RJ, Magalhães T (2013) Forensic toxicology in drug-facilitated sexual assault. Toxicol Mech Methods 23: 471-478.
- Anastassiades M, Steven JL (2003) Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J AOAC Int 86: 412-431.
- Payá P, Anastassiades M, Mack D, Sigalova I, Tasdelen B, et al. (2007) Analysis of pesticide residues using the Quick Easy Cheap Effective Rugged and Safe (QuEChERS) pesticide multiresidue method in combination with gas and liquid chromatography and tandem mass spectrometric detection. Anal Bioanal Chem 389: 1697-1714.
- Lehotay SJ, Son KA, Kwon H, Koesukwiwat U, Fu W, et al. (2010) Comparison of QuEChERS sample preparation methods for the analysis of pesticide residues in fruits and vegetables. J Chromatogr A 1217: 2548-2560.
- Soltaninejad K, Mohammad K, Alireza N, Bahram D (2016) Simultaneous Determination of Six Benzodiazepines in Spiked Soft Drinks by High Performance Liquid Chromatography with Ultra Violet Detection (HPLC-UV). Iran J Pharm Res 15: 457- 463.
- Ferrari A, Coccia CP, Bertolini A, Sternieri E (2004) Methadone-metabolism, pharmacokinetics and interactions. Pharmacol Res 50: 551-559.
- George S, Braithwaite R (1999) A pilot study to determine the usefulness of the urinary excretion of methadone and its primary metabolite (EDDP) as potential markers of compliance in methadone detoxification programs. J Anal Toxicol 23: 81-85.
- Galloway FR, Bellet NF (1999) Methadone conversion to EDDP during GC-MS analysis of urine samples. J Anal Toxicol 23: 615-619.
- van Amsterdam P, Companjen A, Brudny-Kloeppel M, Golob M, Luedtke S, et al. (2013) The European Bioanalysis Forum community’s evaluation, interpretation and implementation of the European Medicines Agency guideline on Bioanalytical Method Validation. Bioanalysis 5: 645-659.
- Bjørk MK, Nielsen MK, Markussen LØ, Klinke HB, Linnet K, et al. (2010) Determination of 19 drugs of abuse and metabolites in whole blood by high-performance liquid chromatography-tandem mass spectrometry. Anal Bioanal Chem 396: 2393-2401.
- Lerch O, Temme O, Daldrup T (2014) Comprehensive automation of the solid phase extraction gas chromatographic mass spectrometric analysis (SPE-GC/MS) of opioids, cocaine, and metabolites from serum and other matrices. Anal Bioanal Chem 406: 4443-4451.
- Jones AW, Holmgren A, Kugelberg FC (2008) Driving under the influence of opiates: concentration relationships between morphine, codeine, 6-acetyl morphine, and ethyl morphine in blood. J Anal Toxicol 32: 265-272.
- Ferner R (2008) Post-mortem clinical pharmacology. Br J Clin Pharmacol 66: 430-443.
- Launiainen T, Ojanperä I (2014) Drug concentrations in post-mortem femoral blood compared with therapeutic concentrations in plasma. Drug Test Anal 6: 308-316.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi