Case Report, J Sleep Disor Treat Care Vol: 6 Issue: 3
Obstructive Sleep Apnea in a Patient with Bilateral, Sequential Non-Arteritic Anterior Ischemic Optic Neuropathy
Mashaqi S1*, Lee M2 and McClelland C2
1Department of Sleep Medicine, Sanford Health, University of North Dakota School of Medicine, Grand Forks, North Dakota, USA
2Department of Ophthalmology and Visual Neurosciences, University of Minnesota, Minneapolis, Minnesota, USA
*Corresponding Author : Mashaqi S
Sanford Sleep Center– University of North Dakota, 2801 University Dr. S Fargo, ND 58104, USA
Tel: 3046900586
E-mail: saif.mashaqi@sanfordhealth.org
Received: June 19, 2017 Accepted: July 04, 2017 Published: July 11, 2017
Citation: Mashaqi S, Lee M, McClelland C (2017) Obstructive Sleep Apnea in a Patient with Bilateral, Sequential Non-Arteritic Anterior Ischemic Optic Neuropathy. J Sleep Disor: Treat Care 6:3. doi: 10.4172/2325-9639.1000199
Abstract
Obstructive sleep apnea (OSA) is a common sleep disorder which affects many organ systems. There are well-known associations between OSA and several ophthalmic disorders. This case reports the association between OSA and non-arteritic anterior ischemic optic neuropathy (NAION). The exact mechanism through which OSA may contribute to NAION is not fully understood. While the association between NAION and OSA is now well established, there is no strong evidence whether positive airway pressure (PAP) therapy can prevent the development of NAION. We report this case to encourage ophthalmologists, pulmonologist and primary care providers to consider screening for OSA in patients who are recently diagnosed with NAION.
Keywords: Non-arteritic anterior ischemic optic neuropathy; Obstructive sleep apnea; Optic nerve; Continuous positive airway pressure; Blindness
Abbreviations
NAION: Non-arteretic Anterior Ischemic Optic Neuropathy; OSA: Obstructive Sleep Apnea; RDI: Respiratory Disturbance Index; AHI: Apnea Hypopnea Index; APAP: Automated Positive Airway Pressure
Introduction
Obstructive sleep apnea (OSA) has been associated with several ophthalmic disorders including glaucoma, floppy eyelid syndrome, retinal vein occlusion, central serous chorioretinopathy, and nonarteritic anterior ischemic optic neuropathy (NAION) [1]. Thought to occur due to hypoperfusion and ischemia of a susceptible optic nerve head with an innate crowding, NAION usually presents with sudden, painless monocular visual loss often discovered upon awakening [2]. Classic associated exam features include an ipsilateral relative afferent pupillary defect, altitudinal visual field defects involving the upper or lower half of vision, and optic disc swelling with associated hemorrhages. NAION is more prevalent in elderly patients (above the age of fifty) with other cardiovascular risk factors (e.g. hypertension, diabetes mellitus, hyperlipidemia, hyperhomocystinemia and atherosclerosis) [3]. Studies show low risk of developing another episode of NAION in the same eye (3.6% in 3 year follow up) while the risk of developing “sequential” NAION in the contralateral eye is markedly higher (24% in 3 years) [4,5]. In theory, appropriate treatment of OSA may help reduce the risk of sequential NAION and avoid devastating blindness in those patients with OSA who have already lost vision in one eye.
Case Report
A 72-year-old Caucasian man with a past medical history of hypertension, hyperlipidemia, and episodic bradycardia requiring pacemaker placement developed acute onset painless vision loss in the right eye. Seven years prior, he suffered acute painless vision loss in the left eye with a severely swollen optic nerve. The diagnosis of NAION was established and optic nerve swelling transitioned to atrophy with no visual recovery. At that time, his right optic nerve had normal function with a full visual field and the appearance of a normal congenitally crowded optic nerve.
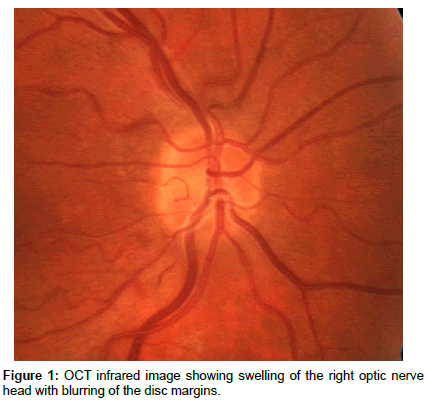
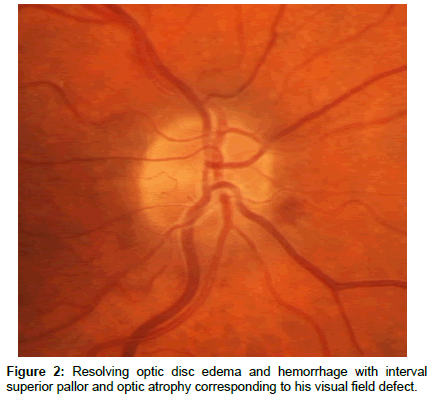
Upon presentation of vision loss in the right eye the patient had new interval development of decreased visual acuity (20/50), a right relative afferent pupillary defect, an inferior altitudinal visual field defect and optic disc swelling (Figure 1). A diagnosis of sequential NAION was made and over the course of three months his optic disc swelling transitioned to atrophy (Figure 2) while his vision remained unchanged.
The patient endorsed sleep related symptoms since 2009 when he was diagnosed with NAION in the left eye, which prompted referral to sleep clinic. He complained of excessive daytime sleepiness and daytime fatigue. The Epworth Sleepiness Scale was 13/24. He denied insomnia of sleep initiation or maintenance. He takes multiple naps (two to three during the daytime). He used to wake up with morning headache, dry mouth and sweating. He had witnessed snoring and apnea episodes. He felt sleepy driving especially for long distances and had to pull over and take a nap sometimes. He denied any symptoms suggestive of restless legs syndrome but did have witnessed leg movements during sleep. He denied any parasomnia of any kind. He denied any family history of sleep related breathing disorders. He used to drink 2-3 cups of coffee in the morning and two beers every night but no liquors. He is an ex-smoker. His past medical history includes hypertension and hyperlipidemia. Both are controlled using lisinopril, metoprolol and simvastatin. He did have repeated episodes of symptomatic bradycardia requiring pacemaker placement.
The physical exam showed a blood pressure of 144/72 mmHg and a body mass index of 27 kg/m2. There was no evidence of nasal septal deviation. Mallampati scale was IV. He underwent tonsillectomy and adenoidectomy. He had very mild retrognathia. Neck circumference was 17.25 inches. The rest of physical exam was unremarkable. He underwent overnight polysomnography (Table 1) which showed a mild degree of OSA that is exacerbated to the severe degree in supine position. Accordingly, he was started on Auto Positive airway pressure (APAP) device with settings of 5-15 cm H2O. In the follow up with sleep clinic, he reported significant improvement in hypersomnia, resolution of morning headache and better sleep quality, however there is no change in his visual symptoms. The download data showed 67% use => 4 hours with average use of 6 hours and 34 minutes. The 90th percentile pressure was 10 cmH2O. Average AHI was 12 events per hour. Ramp time was 30 minutes and ramp starting pressure was 5 cm H2O. Average large leak was 42 seconds.
| Sleep Architecture | |
|---|---|
| Total recording time | 551 minutes |
| Total sleep time | 406 minutes |
| Sleep efficiency | 74% |
| Sleep Latency | 34 minutes |
| REM latency | 142 minutes |
| N1 | 42% |
| N2 | 46% |
| 11N3 | 0% |
| REM | 12% |
| Supine time | 95 minutes |
| Spontaneous arousal index | 22 events per hour |
| Cardiorespiratory events | |
| Apnea Hypopnea index (AHI) | Obstructive (12.4 events per hour), Central (2.8 events per hour) |
| REM AHI | 27 events per hour |
| Supine AHI | 44 events per hour |
| Oxygen saturation (mean, nadir) | 93%, 83 % |
| Heart rate and Rhythm | 65 BPM. Sinus Rhythm with rare episodes of wide complex tachycardia resembling ventricular tachycardia |
| Limb movements | |
| Periodic limb movement index | 0 |
| Periodic limb movement arousal index | 0 |
Table 1: Overnight polysomnography.
Discussion
The association between OSA and NAION had been evaluated from multiple angles including the prevalence of OSA in NAION patients and vice versa. Mojon and colleagues showed a dramatically higher prevalence of OSA in NAION patients (71%) as compared to controls (18%) [5]. Palombi et al. conducted a prospective cohort study and showed an OSA prevalence of 89% in NAION patients [6]. Both studies used overnight polysomnography for the diagnosis of OSA with mean respiratory disturbance index (RDI) of 25 events per hour and apnea hypopnea index (AHI)>15 events per hour in the affected groups respectively. Stein et al. showed a higher prevalence of NAION in OSA patients. The incidence of NAION was stratified by OSA treatment where there was 16% hazard increase in the group who were not treated with CPAP [7]. On the other hand, the role of CPAP in preventing the development of NAION was debated in one study published by Behbehani et al. [8] where three patients developed NAION (one bilateral and two unilateral) despite being treated with CPAP for 4-6 months prior to the onset of NAION.
Many theories have been proposed to explain the pathophysiology of NAION. One leading theory is that nocturnal hypotension with subsequent vascular insufficiency and altered vascular auto regulation leads to hypoperfusion and ischemic infarction of the anterior portion of the optic nerve [9].
Some of the mechanisms suggested to link OSA and NAION include direct damage to the optic nerve head secondary to repetitive hypoxemia associated with obstructive events and direct compression of retinal ganglion cell axons secondary to increased intracranial pressure. The release of vasoactive substances (e.g. VEGF and endothelin-1) secondary to hypoxemia can lead to hypoperfusion and vascular dysregulation [10].
While the Ischemic Optic Neuropathy Decompression Trial (IONDT) McNab et al. [2] showed that 43% of NAION eyes with moderate to severe vision loss demonstrated some visual improvement after 6 months, patients are often debilitated by their vision loss and bilateral NAION significantly affects quality of life. Whether PAP therapy can reduce the risk of sequential NAION in patients who suffer unilateral NAION remains unclear. But with limited interventions to treat NAION and the many other benefits to early recognition and treatment of OSA, providers should maintain a low threshold for OSA testing in NAION patients. Our patient was diagnosed with NAION in the right eye in 2009 and unfortunately suffered sequential NAION in 2016. He likely harbored undiagnosed OSA especially with symptoms suggestive of OSA back in 2009 which could have contributed to his vision loss in the second eye. We report this case to encourage ophthalmologists, pulmonologists, neurologists and primary care providers to remain cognizant of the association between OSA and NAION and consider screening of these patients.
References
- Huon LK, Liu SY (2016) The association between ophthalmologic diseases and obstructive sleep apnea: a systematic review and meta-analysis. Sleep Breath 20: 1145-1154.
- McNab AA (2005) The eye and sleep. Clin Exp Ophthalmol 33: 117-125.
- Mojon DS, Hedges TR 3rd, Ehrenberg B, Karam EZ, Goldblum D, et al. (2002) Association between sleep apnea syndrome and nonarteritic anterior ischemic optic neuropathy. Arch Ophthalmol 120: 601-605.
- Buono LM, Foroozan R, Sergott RC, Savino PJ (2002) Nonarteritic anterior ischemic optic neuropathy. Curr Opin Ophthalmol 13: 357-361.
- Aptel F, Khayi H, Pépin JL, Tamisier R, Levy P, et al. (2015) Assocoaition of nonarteritic ischemic optic neuropathy with obstructive sleep apnea syndrome. JAMA Ophthalmol 133: 797-804.
- Palombi K, Renard E, Levy P, Chiquet C, Deschaux, Ch, et al. (2006) Non-arteritic anterior ischemic optic neuropathy is nearly systematically associated with obstructive sleep apnea. Br J Ophthalmol 90: 879-882.
- Stein JD, Kim DS, Mundy KM, Talwar N, Nan B, et al. (2011) The association between glaucomatous and other causes of optic neuropathy and sleep apnea. Am J Ophthalmol 152: 989-999.
- Behbehani R, Mathews MK, Sergott RC, Savino PJ (2005) Nonarteritic anterior ischemic optic neuropathy in patients with sleep apnea while being treated with continuous positive airway pressure. Am J Ophthalmol 139: 518-521
- Hayreh SS, Podhajsky P, Zimmerman MB (1999) Role of nocturnal arterial hypotension in optic nerve head ischemic disorders. Ophthalmologica 213: 76-96.
- Dhillon S, Shapiro CM, Flanagan J (2007) Sleep-disordered breathing and effects on ocular health. Can J Ophthalmol 42: 238-243.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi