Research Article, J Nucl Ene Sci Power Generat Technol Vol: 5 Issue: 1
Separation of Vanadium Using Dispersive Liquid-Liquid Extraction
| Mohammad Hossein Barani Beiranvand1, Mohammad Hassan Mallah2* and Sohrab Ali Ghorbanian1 | |
| 1School of Chemical Engineering, College of Engineering, University of Tehran,Tehran, Iran | |
| 2Nuclear Fuel Cycle Research School, Nuclear Science & Technology Research Institute, End of North Karegar Ave. Tehran, Iran | |
| Corresponding author : Mohammad Hassan Mallah Nuclear Fuel Cycle Research School, Nuclear Science & Technology Research Institute, End of North Karegar Ave., Tehran, Iran E-mail: mmallah@aeoi.org.ir |
|
| Received: August 11, 2015 Accepted: November 12, 2015 Published: November 17, 2015 | |
| Citation: Beiranvand MHB, Mallah MH, Ghorbanian SA (2015) Separation of Vanadium Using Dispersive Liquid-Liquid Extraction. J Nucl Ene Sci Power Generat Technol 5:1. doi:10.4172/2325-9809.1000140 |
Abstract
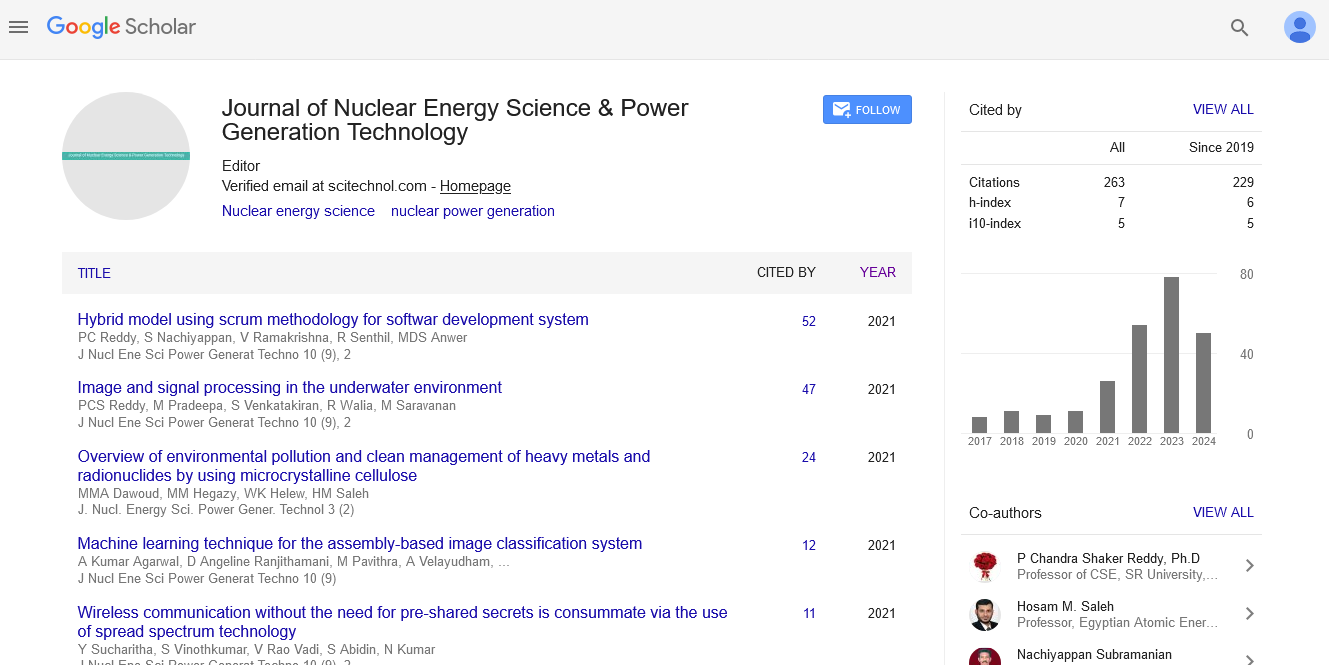
In this study, the conditions of vanadium separation from wastewater by dispersive liquid-liquid extraction in batch and continuous states have been investigated. The best extractant solvents and dispersive or diluents have been selected and then the effect of pH and, the best time of separation have been optimized. Also, effect of foreign ions was tested and at last vanadium of wastewater has been extracted by Di-(2- ethylhexyl) phosphoric acid and amines (Tri-C8-C10–alkyl amine). The results show that amines better than Di-(2- ethylhexyl) phosphoric acid in the present foreign ions can extract vanadium. Under the best optimized conditions, the extraction amounts increased from 34.67% to 92.7%. Methanol with Di-(2-ethylhexyl) phosphoric acid is the best dispersive solvent for extraction of vanadium and, acetonitrile is as a dispersive solvent for amines. Diagram of the McCabe-Thiel shows nine theoretical stages at flow ratio of 0.5:5 (organic to aqueous) with a residence time of about 100s need for yield 90%.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi