Research Article, J Nucl Ene Sci Power Generat Technol Vol: 7 Issue: 2
Synthesis of Silver-Activated Zinc Sulfide (Zns:Ag) Powder by Thermal Diffusion Method for Production of Thin Film Alpha Scintillators
Mehdi Sohrabi*, Fereshteh Saheli and Kheirollah Mohammadi
Department of Energy Engineering and Physics, Health Physics and Dosimetry Research Laboratory, Amirkabir University of Technology, Tehran, Islamic Republic of Iran
*Corresponding Author : Mehdi Sohrabi
Department of Energy Engineering and Physics, Health Physics and Dosimetry Research Laboratory, Amirkabir University of Technology, Tehran, Islamic Republic of Iran
Tel: +98 912 1097485
E-mail: drmsohrabi@yahoo.com
Received: March 15, 2018 Accepted: March 23, 2018 Published: March 29, 2018
Citation: Sohrabi M, Saheli F, Mohammadi K (2018) Synthesis of Silver-Activated Zinc Sulfide (Zns:Ag) Powder by Thermal Diffusion Method for Production of Thin Film Alpha Scintillators. J Nucl Ene Sci Power Generat Technol 7:1. doi: 10.4172/2325-9809.1000182
Abstract
Silver-activated zinc sulfide (ZnS:Ag) powder was successfully synthesized by a physical thermal diffusion method in flux of NaCl to produce this film scintillators for alpha particle detection. The synthesized and commercial ZnS:Ag powders were coated uniformly on Plexiglas disc substrates by a spin coating method. Alpha counting efficiency of the films produced from the synthesized and commercial powders are respectively ~56% and ~67% while they are ~65.6% and ~80% relative to that of the commercial Eljen440 scintillator film. Alpha counting efficiency of the synthesized scintillator film is ~82.8% of the commercial powder film which has itself ~80% efficiency relative to that of the Eljen440 scintillator film. The physical thermal diffusion method applied is rather simple and efficient in particular, if a higher purity material is used which can be simply applied in any developing laboratory.
Keywords: Synthesis; Silver-activated zinc sulfide; ZnS: Ag; Powder; Thermaldiffusion method
Introduction
Scintillators are solid, liquid or gaseous materials used for detection of different types of ionizing radiation. Zinc sulfide doped with silver (ZnS:Ag) is one of the oldest inorganic scintillators usually used for alpha counting [1]. This type of scintillator also is used as thin screens and has played a key role in the early Rutherford experiments [1,2]. This scintillator is available in polycrystalline powder forms primarily used as thin films mounted on photomultiplier tubes for alpha particle or other heavy ion detection [3,4], in Lucas cells for radon detection [5-7], or to a lesser extent in thermal neutron detection when enriched with Li6 [8,9]. If its thickness is approximately >25 mg.cm-2, the scintillator is not usable due to the opacity of the multicrystalline layer to its own luminescence [8].
Various chemical methods are applied to produce ZnS:Ag powder such as hydrothermal [10], co-precipitation [11-15] and sol-gel methods [16]. The solubility constant of Ag2S (6 × 10-50) is far less than that of ZnS (1.1 × 10-21) when used for production, thus it is difficult for Ag+ and Zn2+ ions to co-precipitate with S2- [10-12], leading to a low photoluminescence efficiency of ZnS:Ag powder relative to pure zinc sulfide powder [12]. These three chemical methods usually suffer from problems such as poor crystallization requiring in some cases annealing at a high temperature, high cost of equipment, and particle agglomeration, which significantly hinders their potential applications [17]. To avoid such problems, the physical thermal diffusion method was applied provided superior characteristics in terms of control of impurity diffusion on the host lattice by tuning time and temperature, good crystallization and low cost procedure [18-22].
The physical thermal diffusion method applied in this study is basically a mixture of the main phosphor (ZnS) with a dopant compound (AgCl) which was physically mixed to increase an effective surface diffusion and heating the mixture according to a planned protocol. The heating often is slightly below the melting point of the host material [21,22]. This thermal diffusion method has a onestage procedure and produces satisfactory ZnS:Ag powder with an acceptable efficiency, although its production is not free of challenges [18-22].
Commercial ZnS: Ag powders are usually limited in availability specially in developing laboratories, have a high cost and some with a low quality and low efficiency. Therefore, due to the high academic interest, need to developing a simple method for laboratories in particular in a university environment, and in particular to fulfill a high need of our Health Physics and Dosimetry Research Laboratory to such phosphors, this research and development study was performed. Accordingly, it is the purpose of this study to:
• Synthesize ZnS: Ag powder by developing a thermal diffusion method by doping silver in ZnS crystal lattice in a flux of NaCl,
• Produce thin film ZnS:Ag scintillators from the synthesized powder and also from a commercial powder by a spin coating method,
• Optimize the thickness of thin scintillator films produced by an alpha counting method,
• Determine the alpha counting efficiency of the scintillator film produced compared with those of two types of commercial films, and
• Discuss with an educational view the methodology developed so that it can easily be produced in any developing laboratory.
Material and Methods
Synthesis of ZnS:Ag scintillator powder in flux of NaCl
The synthesis of ZnS:Ag powder was performed by using zinc sulfide (ZnS) (Aldrich Co. with particle sizes <10 μm and a purity higher than 99.99%), sodium chloride (NaCl 99.5% pure, Merck Co.), silver chloride (AgCl; analytical grade without extra purification) and sulfur.
In order to synthesize ZnS:Ag powder, several steps were followed:
1. A primary mixed powder consisting of one mole (M) zinc sulfide powder, 10-3 M of silver chloride, 10-3 M sodium chloride and 10-2 M sulfur was produced.
2. The powder was thoroughly mixed, grinded, poured into a quartz crucible and then covered with a quartz cap to be placed in a furnace.
3. Another quartz crucible containing an optimum amount of sulfur (determine with try and error until synthesize done in sulfide atmosphere, as an alter way we can done synthesize under purging H2S gas) was placed in the tube furnace so that the thermal regime applied later changes the sulfur into SO2; the first step for making the furnace oxygen free.
4. In order to completely make the furnace oxygen free, oxygen was removed by applying the following steps:
a. The furnace was heated up to 120°C with a rate of 20°C. min−1 and was simultaneously vacuumed down to 10-2 Torr and was kept for 2 h.
b. The vacuum was broken by purging continuous flow of argon gas into the furnace while the vacuum pump was turned off so that the gas pressure was allowed to reach 760 Torr pressure. Then the furnace was vacuumed again to 10-2 Torr. This process was repeated for five times.
c. Final step to make the furnace oxygen free was performed during a thermal regime.
5. Thermal regime was performed by applying the following steps [22]:
a. The furnace was heated again from 25 to 400°C at a rate of 13°C. min−1 and was maintained at this temperature for 90 min.
b. The sulfur (with melting point 144°C and boiling point 444°C, in large particle sizes of ~100 μm) was melt down in the heating mixture in the crucible. As a result, the melted sulfur spreads into the pores and gaps of the primary mixture powder and is uniformly distributed throughout the mixture.
c. The furnace temperature was further increased from 400°C to 600°C with a rate of 3.3°C. min−1 and was maintained for 30 min; the excess sulfur that has not been chemically reacted evaporates in the pores. The evaporated sulfur reacts with O2 in the pores to form SO2 which is harmless to the production of ZnS phosphor particles. Therefore, the oxygen in the blended mixture is converted into SO2 gas in the heating mixture before the start of the growth of ZnS particle crystals (as mentioned in papa 4c above).
d. Then, the crucible is heated up to 950°C at a rate of 3°C. min−1 and is maintained for 60 min.
e. Heat treatment was applied in an argon gas environment at atmospheric pressure after which the furnace was cooled down in the argon gas atmosphere. Then the powder was taken out, washed with distilled water to eliminate excess salt and the final powder was dried at 70l°C. In these experiments, the glassware used was always washed by a solution of hydrochloric acid and nitric acid with a 3:1 ratio.
Production of ZnS:Ag scintillator thin films on substrates
The ZnS:Ag scintillator powder is in general polycrystalline and its use is limited to films with desired thin thicknesses for detection of alpha particles or other heavy ions. Thin ZnS:Ag scintillator films were produced from the powder samples synthesized in this study on 2 mm thick Plexiglas substrates. Scintillator films were also produced by using a commercial powder (Keyan Phosphor Technology Co. Ltd., China). The two thin film types produced were used for comparative studies with a commercial Eljen440 ZnS:Ag scintillator film.
For the thin film production, several steps were followed:
1. Transparent Plexiglas dough with a suitable viscosity was prepared using 450 mg polymer chips dissolved in 10 ml dimethylformamide (DMF).
2. ZnS:Ag powder (3.5 g) was then added slowly to the dough. The final dough was stirred to produce uniform dough for coating on the 2 mm thick Plexiglas substrate.
3. The dough was spread on the surface of the Plexiglas substrate by a spin-coating method at 1000 rpm for 30 s [23].
4. In order to produce scintillator films with a specific thickness, a suitable amount of dough was spread on a 4 cm diameter area of the substrate. The final scintillator films produced had density thicknesses from 3.5 to 25 mg.cm-2. The density and thicknesses for each sample was calculated as the ratio of the weight of the samples (by subtracting the weight of the substrate) to its area.
Alpha Counting by Scintillator Films
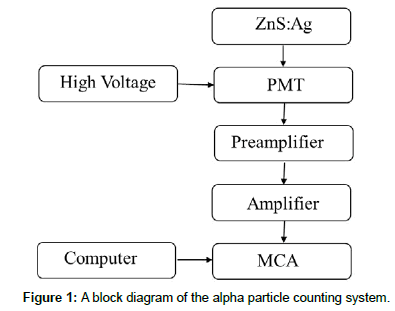
A photomultiplier tube (PMT XP2020) with 5.08 cm diameter coupled to a counting system was used in this study for alpha counting with a scintillator film, as shown in Figure 1. Alpha counting efficiency of the produced ZnS:Ag scintillator films of different thicknesses and the commercial Eljen440 films were determined using an 241Am source with an activity of 3.3 kBq with a 2 cm2 active area, by the ratio of recorded counts to source fluence in the same time intervals and considering solid angle and subtracting background counts.
Results and Discussion
The scintillator powder produced under a UV light exposure showed a light glow color which indicates successful doping of the silver in the crystal lattice. Figure 2 shows the fluorescent glow of the synthesized powder under 366 nm UV light excitation wavelength. As observed in Figure 2, the color glow seems to be an indication that the activator has been doped in the ZnS crystal lattice.
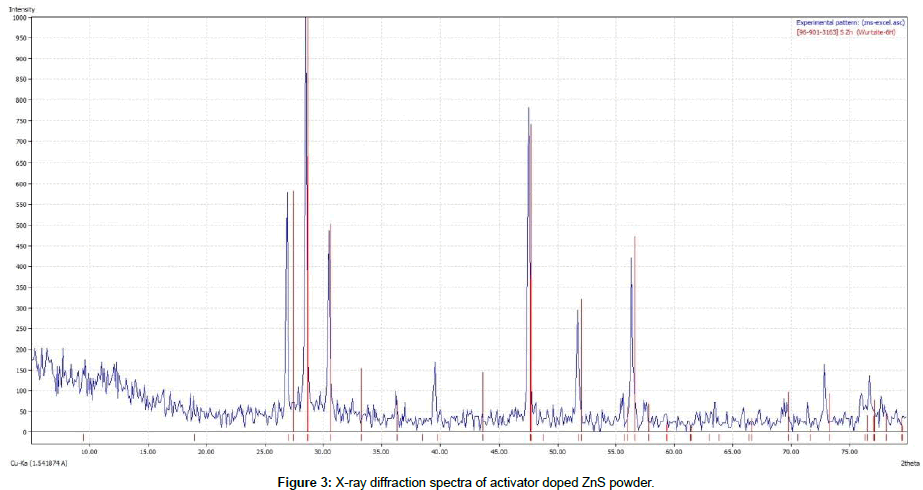
Figure 3 shows X-ray diffraction spectra (XRD) of the activator doped ZnS powder which is synthesized in this study. This analysis show that the particles are in cubic and hexagonal structure of ZnS. Also, in this figure shift peaks are observed which are related to the diffusion of the Ag atoms in the host lattice crystal.
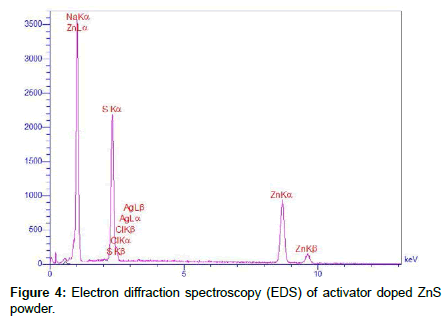
Figure 4 shows the electron diffraction spectroscopy (EDS) of the activator doped synthesized ZnS powder. This analysis shows that the NaCl particles are diffused in structure of ZnS, without appearing any peak of oxygen or ZnO in the structure of produced powder. It seems that the sintered material has been identified as NaCl under the assumption that the melted NaCl has the flux action for diffusing Ag in the ZnS structure and the growth of ZnS:Ag particles. Then one must also consider the removal of NaCl [22]. But a little amount of NaCl diffusion in the host lattice crystal even after several times dried the synthesized powder.
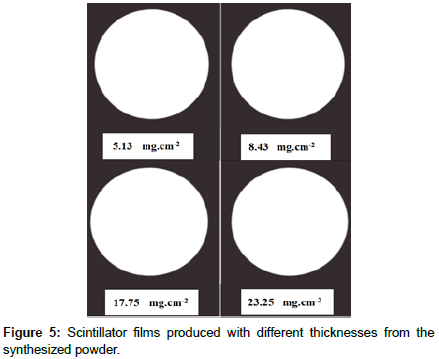
As stated above, scintillator films of different density thicknesses were successfully made. Figure 5 shows the ZnS:Ag films with different density thicknesses coated on Plexiglas substrates after being dried up in air. The scintillator films produced from the synthesized powder and from the commercial powder performed having a high quality and were compared with the Eljen440 scintillator films available in the laboratory.
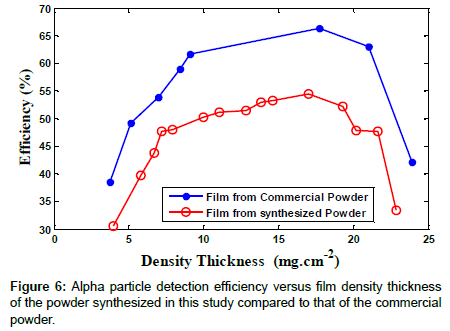
Figure 6 shows alpha particle detection efficiency as a function of the ZnS:Ag scintillator film density thickness produced from the powder synthesized in this study and the commercial powder. In both types of film produced, the efficiency has approximately the same trend for ZnS:Ag film density thicknesses from 9 to 20 mg.cm-2. At a density thickness of about 17.75 mg.cm-2, higher efficiencies for both of the ZnS:Ag scintillator films were obtained which was used as an optimized value in our studies which is in good agreement with that of another study [24].
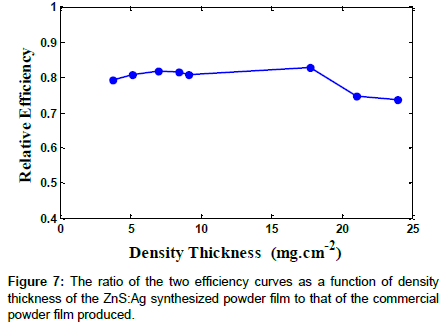
By obtaining the ratio of the efficiency of the synthesized to commercial powders, the relative efficiency of the two produced films was obtained. Figure 7 shows the ratio of the two efficiency curves as function of the film density thickness of the two types of film used. As it can be seen, the ratios of the two efficiencies are rather relatively flat up to the density thickness where the efficiencies are maximum. Therefore, it can be stated that the produced powder has the same counting characteristics of the commercial powder but it requires further improvement in terms of production and material consideration.
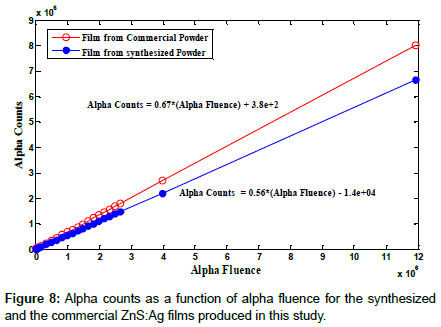
In order to obtain dependence of the alpha counting rate and in turn the detection efficiency of the two types of ZnS:Ag scintillator films produced, alpha counting rate as a function of alpha fluence was studied for counting durations from 10 to 7200 s for alpha fluences up to 1.2 × 107 alpha particles using an 241Am source, as shown in Figure 8. The slope of each response represents the alpha counting efficiency of each of the scintillators; it is 55.7% and 67.25% for the scintillator films produced from the synthesized powders and the commercial scintillator film respectively. The results show that the alpha counting efficiency of the scintillator film produced from the synthesized powder film of this study compared to that of the commercial powder film is 82.8%.
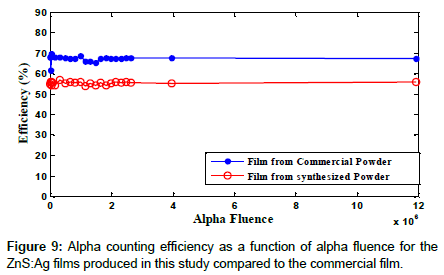
Figure 9 shows the alpha counting efficiency as a function of alpha fluence for the films produced from the powder synthesized in this study and the commercial ZnS:Ag films. As can be seen, the efficiencies are constant over a broad range of fluences up to 1.2 × 107 alpha particles.cm-2 with efficiencies of 55.7% and 67.25% for the two types of films which are actually the slopes of the responses in (Figure 9).
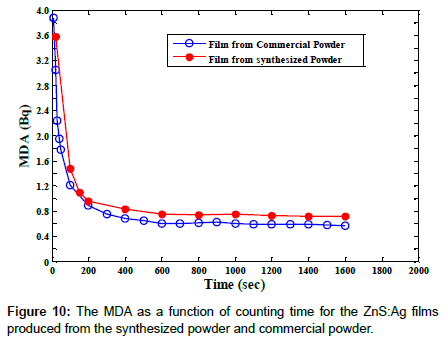
The minimum detectable activity (MDA) of the two types of scintillator films produced from the synthesized and commercial powders were obtained by applying the minimum detection limit (MDA) relationship given by Altshuler and Pasternack [25,26]. The MDA for different counting times were calculated for a confidence level of 99.99%. Figure 10 shows the MDA as a function of counting time for the two types of film produced from the synthesized and commercial powders. The results obtained are in good agreement with the MDA methods studied by others [26,27].
In order to ensure further the quality of the ZnS:Ag scintillator films made on Plexiglas substrates as produced in this study from the synthesized and commercial powders, alpha counting efficiency of the two scintillator films produced in this study were compared with that of the commercially made Eljen440 scintillator film. This commercial ZnS:Ag film has a very uniform thickness on a transparent 0.25 mm thick polyester substrate film. The alpha counting efficiency of the Eljen440 scintillator film was found to be 84% (relative to the source fluence) which is the highest among the three scintillators films discussed. The alpha counting efficiencies of the two ZnS:Ag films produced from the synthesized and commercial powders were found to be 56% and 67% (relative to the exposed alpha fluence) and 65.6% and 80% relative to the that of Eljen440 scintillator film respectively. The alpha counting efficiency of the scintillator film of the synthesized powder produced in this study is about 82.8% of that of the film made from the commercial powder which itself has an efficiency of 80.0% relative to the Eljen440 commercial ZnS:Ag scintillator film.
Conclusion
Silver-activated zinc sulfide (ZnS:Ag) powder was successfully synthesized in this study by a physical thermal diffusion method in a flux of NaCl. Thin film scintillator layers with different density thicknesses were coated uniformly by a spin coating method on Plexiglas discs. The highest alpha counting efficiency from an 241Am source determined corresponds to a density thickness of 17.75 mg.cm-2. The alpha counting efficiency of the Eljen440 scintillator commercial film was found to be 84% being the highest among the three scintillator films discussed. The alpha counting efficiencies of the films of the synthesized and commercial powders produced in this study were found to be 56% and 67% (relative to the alpha fluence) and 65.6% and 80% (relative to that the Eljen440 scintillator film) respectively. The alpha counting efficiency of the scintillator film of the synthesized powder is about 82.8% of the commercial powder film which has itself an efficiency of 80% relative to the ZnS:Ag scintillator commercial film (Eljen440). Applying the physical thermal diffusion method was found to be a practical method to be recommended for production of ZnS:Ag scintillator in developing laboratories as well as for educational purposes.
Acknowledgement
The research was supported by the Amirkabir University of Technology under the current budget.
References
- Knoll GF (2010) Radiation detection and measurement. John Wiley & Sons, New York, USA.
- Tsoulfanidis N (1995) Measurement and detection of radiation. Taylor & Francis CRC Press, Florida, USA.
- McElhaney S, Ramsey J, Bauer M, Chiles M (1990) A more rugged ZnS:Ag alpha scintillation detector. IEEE T Nucl Sci 37: 868-872.
- Collinson A, Haque AA (1963) Scintillation counter for the measurement of radon concentration in air. J Sci Instrum 40: 521-523.
- Lucas HF (1957) Improved low-level alpha scintillation counter for radon. Rev Sci Instrum 28: 680-683.
- Quindos-Poncela L, Fernandez P, Sainz C, Arteche J, Arozamena J, et al. (2003) An improved scintillation cell for radon measurements. Nucl Instrum Meth A 512: 606–609.
- Sohrabi M, Solaymanian AR (1988) Some characteristics of AEOI Passive Radon Diffusion Dosimeter. Nucl. Tracks and Rad Meas 15: 605-608.
- Saint-Gobain Crystals (2014) ZnS:Ag product data sheets. Advanced Engineered Materials and Sciences.
- Stedman R (1960) Scintillator for thermal neutrons using Li6F and ZnS(Ag). Rev Sci Instrum 31: 1156-1156.
- Qin D, Yang G, He G, Zhang L, Zhang Q, et al. (2012) The investigation on synthesis and optical properties of Ag-doped ZnS nanocrystals by hydrothermal method. Chalcogenide Lett 9: 441-446.
- Qu H, Cao L, Su G, Liu W, Sun Y, et al. (2009) Effect of ultraviolet irradiation on luminescence properties of undoped ZnS and ZnS:Ag nanoparticles. J Appl Phys 106: 093506.
- Ramasamy V, Praba K, Murugadoss G (2012) Synthesis and study of optical properties of transition metals doped ZnS nanoparticle. Spectrochim Acta A 96: 963-971.
- Porambo M, Marsh A (2009) Synthesis and photoluminescent properties of doped ZnS nanocrystals capped by poly (vinylpyrrolidone). Opt Mater 31: 1631-1635.
- Murugadoss A, Chattopadhyay A (2008) Tuning photoluminescence of ZnS nanoparticles by silver. B Mater Sci 31: 533-539.
- Jian W, Zhuang J, Zhang D, Dai J, Yang W, et al. (2006) Synthesis of highly luminescent and photostable ZnS:Ag nanocrystals under microwave irradiation. Mater Chem Phys 99: 494-497.
- Huang F, Pengand Y, Lin C (2006) Synthesis and characterization of ZnS:Ag nanocrystals surface capped with thiourea. Chem Res Chinese U 22: 675-678.
- Mu J, Gu D, Xu ZZ (2005) Effect of annealing on the structural and optical properties of non-coated and silica-coated ZnS:Mn nanoparticles. Mater Res Bull 40: 2198-2204.
- Ozawa L, Makimura M, Itoh M (2006) Eradication of oxygen contamination in production of ZnS:Ag:Cl blue phosphor powder. Mater Chem Phys 96: 511–516.
- Marking G, Warren C, Payne B (2014) ZnS:Ag, Al phosphor and method of making same.
- Yen W, Shionoya S, Yamamoto H (2007) Practical applications of phosphors (1st edtn), CRC Press, Florida, USA.
- Yen W, Weber M (2004) Inorganic phosphors: compositions, preparation, and optical properties. (1st edtn), CRC Press, Florida, USA.
- Ozawa L (2007) Cathodoluminescence and photoluminescence, theories and practical applications. (1st edtn), CRC Press, Florida, USA.
- Pauleau Y (2001) Chemical physics of thin film deposition processes for micro- and nano-technologies. Springer, The Netherlands.
- Lee SK, Kang SY, Jang DY, Lee CH, Kang SM, et al. (2011) Comparison of new simple methods in fabricating ZnS(Ag) scintillators for detecting alpha particles. Prog Nucl Sci Tech 1: 194-197.
- Altshuler B, Pasternack B (1963) Statistical measures of the lower limit of detection of a radioactivity counter. Health Phys 9: 293-298.
- Sohrabi M, Hakimi A, Soltani Z (2016) Background track density reduction of 50 Hz–HV ECE-processed thick polycarbonate detectors to improve lower detection limit. Radiat Prot Dosim 171: 470-476.
- ASTM (2009) International standard practices for the measurement of radioactivity. Designation, Practice for use of a polymethylmethacrylate dosimetry system D 3648-3654.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi