Research Article, J Clin Nutr Metab Vol: 2 Issue: 2
The Impact of Nutrition on Hashimoto Thyroiditis Patients: An Overview
Noboru Motohashi1, Jyothirmayi Vadapalli2, Anuradha Vanam3 and Rao Gollapudi4*
1Meiji Pharmaceutical University, 2-522-1 Noshio, Kiyose-shi, 204-8588 Tokyo, Japan
2Acharya Nagarjuna University, Nagarjunanagar, AP, India
3Sri Venkateswara University, Tirupathi, AP, India
4University of Kansas, Lawrence, Kansas-66045, USA
*Corresponding Author : Rao Gollapudi
University of Kansas, Lawrence, KS-66045, USA
E-mail: gollapudirao@ku.edu
Received: April 16, 2018 Accepted: May 09, 2018 Published: May 17, 2018
Citation: Motohashi N, Vadapalli J, Vanam A, Gollapudi R (2018) The Impact of Nutrition on Hashimoto’s Thyroiditis Patients: An Overview. J Clin Nutr Metab 2:2.
Abstract
Thyroid hormone is important for the regulation of body temperature, heart rate, blood pressure and metabolism. Thyroid abnormalities affect a large number of populations more in the elderly than in children. Hashimoto’s thyroiditis happens to be the most common cause of hypothyroidism. Women are more likely affected by Hashimoto’s disease than men. Hypothyroidism causes multiple symptoms upsetting many body functions resulting in the slowdown and may lead to fatigue, dry hair & skin and memory problems. Thyroid hormones are essential for the regulation of body energy, optimum use of other hormones and vitamins in the body as well as for the growth of body tissues. The common symptoms of hypothyroidism are hair loss, memory loss, constipation, depression, appetite loss, feeling cold, mild weight gain, irritability, worsening menstrual periods and cramps, goiter and growth delay (in children). Thyroid inflammation is hereditary where the familial predisposition to the disease is most common factor. A variety of nutritional factors are essential in optimizing thyroid function. Occasionally, conservative treatment of levothyroxine might not benefit Hashimoto’s thyroiditis patients for whom thyroidectomy is recommended. However, nutrient deficiencies and their excess might activate or aggravate the symptoms. Goitrogens in cruciferous vegetables adversely affect the function of thyroid gland by reducing the production of thyroxin hormones. Nonetheless, foods like dulse, seaweed, or kelp containing higher amounts of iodine might cause or worsen hypothyroidism. Since certain foods, drugs and calcium supplements antagonize levothyroxine (synthetic T4) function, caution is advised while taking levothyroxine along with those foods, drugs and supplements.
Keywords: Hashimoto’s thyroiditis; Autoimmune thyroiditis; Hypothyroidism; Triiodothyronine; Thyroxin; Mono-iodo tyrosin; Di-iodo tyrosin; Calorigenic effect; Thermogenic effect; Gluconeogenesis; Goiter; Vitamin B12; Selenium; Glucosinolates; Isothiocyanates; Progoitrin; Goitrin; Genistein; Chromium picolinate; Wobenzyme; Rutin
Introduction
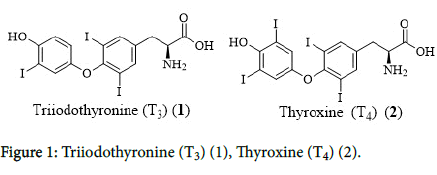
Hashimoto’s thyroiditis is also known as chronic lymphocytic thyroiditis or autoimmune thyroiditis which is the most occurring thyroid disease in the United States, the most common cause of hypothyroidism. It is a hereditary disorder distressing nearly 14 million Americans and is about 7 times more common in the middleaged women than in men [1]. The body erroneously recognizes its own tissues as an invader and attacks them through cell signaling processes like autophagy or apoptosis until the organ is destroyed. Ultimately, this prolonged attack prevents the thyroid gland from producing and releasing sufficient levels of the hormones of triiodothyronine (T3) (3, 3’, 5’-tri-iodo thyroxin) (1) and thyroxin (T4) (3, 5, 3', 5'-tetra-iodo thyroxin) (2), necessary to keep the body performing its functions accurately. The lack T3 (1) and T4 (2) hormones could slow down metabolism and cause weight gain, fatigue, dry skin and hair, and difficulty in concentrating. Hashimoto’s thyroiditis was associated with autoimmune disorders, like type 1 diabetes, lupus, rheumatoid arthritis and celiac disease. Nearly in all cases hypothyroidism is the outcome of inadequate T4 production, rather than inadequate T3 (1) (Figure 1) [2].
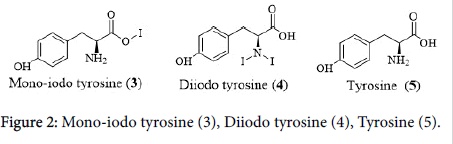
The thyroid gland is located in neck which secretes hormones into blood that are carried into the body tissues. Thyroid gland absorbs and concentrates iodine, accelerated by thyroid stimulating hormone or thyrotropin (TSH). In thyroid cells, the oxidation of iodine is catalyzed by thyroperoxidase enzyme. Later, thyroglobulin, synthesized by thyroid follicular cells gets idonized facilitating the production of mono-iodo tyrosin (3) and di-iodo tyrosine (4) from tyrosine (5) in the periphery of follicular cells. Afterwards, the two di-iodo tyrosine molecules couple to form T4 and the de-iodonization from T4 produces T3 (1). T4 (4) is stored as colloid in thyroglobulin of the thyroid acini. Subsequently, thyroid hormone gets intercalated to proteins such as globulin, transthyretin and albumin followed by the transport through plasma. The bound forms of thyroid hormones are biologically inactive. However, active T3 (1) and T4 (2) are swiftly released, when required. The thyroglobulin is transferred from acinar colloid to the cells by pinocytosis. Moreover, T4 (2) is released through hydrolysis of proteins by certain proteases, ameliorated by TSH. The T4 (2) is released into bloodstream and majority of T3 is produced by deiodination at 5’ position of T4 (2) which is more potent than T4 (2). After binding to DNA, thyroid hormones are attached to specific nuclear receptors. The attachment of T3 to DNA facilitates a higher transcription rate [3].
Function of thyroid hormone
Thyroid hormones are major endocrine regulators of metabolic rate with hyper metabolic effects and induce considerable alterations in mitochondrial inner membrane protein as well as lipid compositions (Figure 2).
Increased uncoupling might have been responsible for some of the hyper-metabolic effects of thyroid hormones. Besides, adenosine triphosphate (ATP) synthesis and turnover reactions were affected. However, there is still a necessity to understand the thyroid hormone effects in other tissues [4].
Thermogenesis is a major calorigenic effect of thyroid hormone where 1 mg T4 (2) yields an excess of 1000 kcal. The thermogenic effect is facilitated through the separation of oxidative phosphorylation. Thyroxins up surged cellular metabolism with the growth in basal metabolic rate. Additionally, T4 (2) stimulated RNA synthesis and subsequently promoted protein synthesis. Higher concentrations of T3 (1) resulted in protein catabolism and negative nitrogen balance [5].
Gluconeogenesis is a metabolic activity associated with generation of glucose from the breakdown of proteins, glucogenic amino acids and lipids. Gluconeogenesis is one of the main mechanisms involved in maintaining normal blood glucose levels. Gluconeogenesis is a combination of carbohydrate oxidation and fatty acid metabolism which is regulated by thyroid hormones [6].
Traditionally, treatment for thyroid hormone deficiency was addressed by prescribing synthetic thyroid hormone (T3). Even though, this treatment resulted in the balancing of thyroid hormones levels in Hashimoto’s thyroiditis patients, they continued to suffer from symptoms of hypothyroidism [7]. In order to understand the root causes of Hashimoto’s thyroiditis and autoimmune reactions, it is important to develop an effective solution like gene therapy to address Hashimoto’s thyroiditis, not only for the symptoms of hypothyroidism, but also the cause of Hashimoto’s thyroiditis.
Risk factors associated with hypothyroidism
Cardiovascular: Patients with hypothyroidism are at a greater risk of cardiovascular diseases than with obesity. Low levels of thyroid hormones result in increased blood pressure, higher blood lipid profile and higher levels of homocysteine and the inflammatory marker Creactive protein [8] Thyroid hormones modulate cholesterol synthesis, cholesterol receptors, and the rate of cholesterol degradation, alter lipid profile as well as cardiovascular diseases [9]. Hypothyroidism affects carbohydrate metabolism with a great effect on glucose control Hypothyroidism patients show abnormal weight gain and find it difficult to lose weight until hormone levels are stabilized. Hashimoto’s thyroiditis normally occurs around menopause, which complicates the weight gain issue in women [10].
Diabetes: 30% of people with type1 diabetes, 12.5% of those with type2 diabetes and 6.6% of those without diabetes have autoimmune thyroid disease. The thyroid hormone therapy is essential for patients with thyroid enlargement or hypothyroidism with a daily dose of levothyroxine. Elderly patients with underlying heart disease are usually given a low dose of levothyroxine and gradually increased. However, younger patients are started with full replacement doses. Since, thyroid hormone acts very slowly, it might take several months to sight improvements in symptoms or goiter shrinkage after commencing the treatment. Ideal adjustment of thyroid hormone dosage is necessary based on laboratory tests rather than symptoms, since the body is very sensitive to even small changes in thyroid hormone levels. If the dose is too low, thyroid gland may continue to enlarge and symptoms of hypothyroidism will persist which is associated with increased serum cholesterol levels, possibly increasing the risk for atherosclerosis and heart disease. However, if the dosage quantity is too high, it can cause symptoms related to hyperthyroidism, creating excessive strain on heart and an increased risk in the development of osteoporosis [10].
Pregnancy: During pregnancy, hypothyroidism is commonly caused by Hashimoto’s disease and occurs in three to five out of every 1,000 pregnancies. Uncontrolled hypothyroidism increases the chance of miscarriage, premature birth, stillbirth, and preeclampsia (a dangerous rise in blood pressure in late pregnancy). Untreated hypothyroidism during pregnancy may also affect baby’s growth and brain development and thyroid medications are safe and effective in preventing these problems [11].
Hashimoto’s thyroiditis
Hashimoto’s thyroiditis is characterized by the production of immune cells and auto-antibodies by body’s immune system. These antibodies damage thyroid cells and compromise their ability to synthesize thyroid hormone. When the amount of thyroid hormone produced is insufficient, it results in hypothyroidism where thyroid gland may enlarge, results in the formation of goitre. Goitre can be caused by hypothyroidism; hyperthyroidism resulted from excessive or inadequate intake of iodine through the diet, or thyroid cancer which is the most common endocrine cancer [12]. Some patients have autoimmune antibodies and however, maintain adequate thyroid function for many years without any requirement for intervention. Thyroid hormone supplementation medication is necessary to correct the hormonal imbalances associated with hypothyroidism, when body fails to produce sufficient amounts of thyroid hormone, necessary for the physiological functions. Hashimoto’s thyroiditis patients with hypothyroidism are more likely to experience the symptoms like fatigue, constipation, difficulty with learning, drowsiness, dry, brittle hair and nails, dry, itchy skin, forgetfulness, heavy menstrual flow, increased frequency of miscarriages, increased sensitivity to many medications, puffy face, sore muscles and weight gain [2]. Although multiple environmental factors have been researched, none of the factors are conclusive to establish the cause of Hashimoto’s thyroiditis.
Hashimoto’s thyroiditis patients and their genetic family members will never experience any other autoimmune conditions but at the risk of developing Addison’s disease (the adrenal gland provides cortisol to manage stress and ailment) Grave’s disease (goiter and hyperthyroidism), lupus erythematosus (autoimmune disorder that affects skin, heart, lungs, kidneys), pernicious anemia (inability to absorb vitamin B12 (6) initiating anaemia and neurologic problems ), premature ovarian failure (early menopause), rheumatoid arthritis, thrombocytopenic purpura (bleeding disorder by inadequate platelets in the blood), vitiligo (patchy loss of skin pigmentation) and Type 1 diabetes mellitus (insulin-dependent) (Figure 3) [13].
Minerals and vitamins supplements: A variety of nutritional elements are helpful in maintaining thyroid function. However, both nutrient deficiencies and excesses can onset or aggravate the symptoms.
Iodine: Iodine is a vital nutrient in the body, essential for thyroid function and thyroid hormones contain iodine. Globally, iodine deficiency is the primary cause of thyroid dysfunction whereas autoimmune disease is the primary cause of thyroid dysfunction in the United States [14]. Since 1920s in the United States, iodine deficiency is considered rare, mainly due to the extensive use of iodized salt, along with fish, dairy, and grains (main source of iodine in a standard American diet). Conversely, iodine intake has dropped during the past few decades and Americans get approximately 70% of their salt intake from processed foods that usually lack iodine [15]. The iodine deficiency or excess pose significant risks, supplementation should be approached with great caution. Supplemental iodine and consumption of dulse, seaweed, or kelp containing higher amounts of iodine may cause symptom flare-ups in people with Hashimoto’s disease because it fuels autoimmune antibodies [16].
Vitamin D: Vitamin D (7) deficiency is linked to Hashimoto’s thyroiditis and more than 90% of patients studied exhibited deficiency in vitamin D (7) levels. Nonetheless, it is not established whether the lower vitamin D (7) levels were direct cause to develop Hashimoto’s thyroiditis or the result of the disease process itself [17]. Fatty fish, milk, dairy, eggs, and mushrooms are essential dietary sources of vitamin D (7). Sunlight is an important source for the biosynthesis of vitamin D (7), but the amount of vitamin formation depends on the seasons and latitude. However, patients with low vitamin D (7) levels require with the supplementation of Vitamin D (Figure 4) (7).
Selenium: The optimum concentration of selenium in thyroid gland is necessary for essential enzymes in thyroid gland to function [18]. Selenium, an essential trace element has a profound effect on the immune system, cognitive function, fertility and mortality rate in both men as well as women. A meta-analysis of haphazard, placebocontrolled studies demonstrated the benefits of selenium in both thyroid antibody titers and mood in patients with Hashimoto’s. However, this effect was more distinct in people with a selenium deficiency or insufficiency. An excessive intake of selenium can result in gastrointestinal distress or even increase the risk of type 2 diabetes and cancer. Therefore, it is essential to get selenium levels tested before incorporating healthful, selenium-rich foods in the diets, such as Brazil nuts, tuna, crab, and lobster [19].
Vitamin B12: Vitamin B12 (6) deficiency is observed in over 30% of people with autoimmune thyroid disease. Food sources of B12 include molluscs, sardines, salmon, and organ meats such as liver, muscle meat, and dairy. The primary sources of vitamin B12 (6) in vegan diets include fortified cereals, nutritional yeast [20].
Nutritional factors
Brassica and drumstick vegetables in diet exhibit beneficial and adverse health effects. Following enzymatic breakdown, some glucosinolates in brassica vegetables produce sulforaphane, phenethyl, and indolylic isothiocyanates that possess anticarcinogenic activity. Cruciferous vegetables (broccoli, cauliflower, cabbage, brussels sprouts etc.) drumstick leaves and are rich source of glucosinates and isothyocyanates some of which are converted to progoitrin (8) and goitrin (9) by the enzymes, glucosinolate oxidase, glucosinolate and glucosinolate hydroxylation followed by the nonenzymatic reactions [21,22]. Goitrogens are compounds that impair the function of thyroid gland and interfere with the production of thyroxin hormones. Progoitrin (8) and goitrin (9) interfered with iodine utilization in the synthesis of thyroid hormones which is a concern only when associated with an iodine deficiency [21].
Heating cruciferous vegetables altered the potential goitrogenic effect. Soy is another potential goitrogen and the isoflavone, genistein (10) in soy inhibited the synthesis of thyroxin which could result in goitre. Nonetheless, multiple studies revealed that hypothyroidism is not associated with soy intake in people with adequate amounts of iodine [23,24]. On the other hand, goitrogens containing vegetables and fruits may be helpful in controlling hyperthyroidism like Grave’s disease. However, further research is necessary to determine the beneficial effects of goitrogens to hyperthyroid patients (Figure 5).
While moderate soy intake is recommended for patients with preexisting hypothyroidism, a high-dose soy supplementation, is still a distress. A randomized, double-blinded study suggested a threefold increase in the development of clinical hypothyroidism among women with hypothyroidism when supplemented with high doses of soy. Iodine levels did not appear to be a factor in the production of thyroid hormones (Figure 6) [25].
The cooked crucifers and soy consumption is usually safe in people with adequate iodine, the potential exception is millet, a nutritious gluten-free grain, which may suppress thyroid function even in people with adequate iodine intake [26]. Since, cruciferous vegetables are extremely rich in nutrients; it’s certainly not advisable to cut them out of the diet completely. Hypothyroidism was induced in rats with a continuous diet of progoitrin (8), present in brassica vegetables and defatted rapeseed meal (154 lmol per 100 g), negatively affecting glutathione S-transferase activities which is responsible for the conjugation and subsequent detoxification of multiple toxic and electrophilic substances [27]. The rapeseed meal in animal food supplement is limited due to the goitrogenic effect of 5- vinyloxazolidine-2-thione that accounts for up to 80% of total glucosinolates in rapeseed [28]. Progoitrin (8) - containing diet ameliorated the concentrations of progoitrin (8) in thyroid and lung of lambs. Whereas, a higher goitrin (9) concentration diet markedly attenuated repressed weight gain and increased thyroid and liver masses in pigs [29,30]. The intake of broccoli and broccoli rabe, each containing less than 10 lmol progoitrin (8) per 100-g serving, resulted in minimal risk of thyroidal toxicity. Hence, single serving of brassica vegetables with less than 70 lmol of progoitrin (8) would not decrease thyroid hormone production in humans [31]. Daily consumption of raw bok choy (1.5 kg) for several months in the elderly presented with myxedema coma, the most severe and a life-threatening form of hypothyroidism [32]. Animal feeding studies investigating the safety of rapeseed meal revealed that progoitrin (8) in rapeseed whose degradation product is goitrin (9) displayed antithyroid effects [33]. Thus, goitrin (9) and thiocyanates content in brassica vegetables exerted a negative effect on thyroid hormone production. The rate of increase in intracellular concentrations is attributed to the diffusion of isothiocyanates across cell membrane and inside the cell. The isothiocyanates spontaneously form adducts with cysteine thiol of glutathione. These conjugates are unstable and readily disassociate from their parent compounds [34].
Consequently, conjugate concentrations of isothiocyanates in the millimolar range would expose tissues to the same range of isothiocyanate concentrations in thyroid slices, thereby decreasing iodine uptake [35]. Metabolic experiments in rats suggested the relation between excretory pattern of iodine and thiocyanate, with thyroid gland weight and circulating levels of thyroxine, in response to moderate and high intake of iodine and under conditions of goitrogen induced altered thyroid status. The moderate intake of iodine (by depriving diet of KI) 25 mg of thiocyanate or substitution of 1/3rd proportion of casein based diet with dry cabbage; markedly reduce plasma thyroxin levels after 60 days. The exposure to goitrogen did not affect body weight or the weights of liver, kidney, heart or spleen. A significant increase in thyroid gland weight as well as higher excretion of iodine and thiocyanate were evident in goitrogen-fed rats. Semiquantitative evaluation of thyroid gland, indicated hypo-functioning of thyroid with follicular hyperplasia in thiocyanate fed rats. These results suggested that moderate intake of required iodine may not ensure normal functioning of thyroid in the presence of goitrogens [36].
Adverse effects of certain medications and diets
Chromium picolinate (10) (a medication used for blood sugar control, weight loss) and calcium supplements impair the absorption of thyroid medications. Studies recommend appropriate spacing of four hours should be followed in taking calcium supplements as well as chromium picolinate (10) with thyroid medications (Figure 7) [37,38].
Coffee and fiber supplements lower the absorption of thyroid medication suggesting a one hour gap between the both with thyroid medication [39,40]. Flavonoids in fruits, vegetables and tea exhibited a potential cardiovascular benefit; however, high-dosages of flavonoid supplements attenuated thyroid function [41].
Recently, thyroid inflammation is the most frequent thyroid illness, including morphological and hormonal modifications. In an open controlled study, Wobenzyme formulated with bromelian (45 mg), chymotriptin (1 mg), pancreatin (100 mg), papain (60 mg), rutin (12) (50 mg), trypsin (24 mg) was found to be effective in controlling the symptoms of Hashimoto’s thyroiditis alone or in combination with thyroid hormone. Wobenzyme was effective on inflammation which was beneficial in improving thyroid morphology and function.
Recently, thyroid inflammation is the most frequent thyroid illness, including morphological and hormonal modifications. With administration of thyroid hormones alone, the symptoms decrease was slower and the effects on antibody concentrations were surprisingly deficient. However, the effect of Wobezyme on TSH levels was much lower than thyroid hormones, suggesting that its activity was mainly on thyroid inflammation but not on TSH. Additional research is required to substantiate efficacy and the underlying mechanisms of Wobenzyme in treating autoimmune thyroid disorders (Figure 8) [42].
Conclusion
An underactive thyroid is known as hypothyroidism when the gland fails to produce enough thyroid hormone. Hashimoto’s thyroiditis is an autoimmune disease that leads to hypothyroidism, the most common in the Western world. Conventional treatment for an underactive thyroid is the prescription of synthetic T4 (levothyroxine). Hashimoto’s thyroiditis has a strong genetic link and is associated with other autoimmune disorders such as type 1 diabetes, rheumatoid arthritis, lupus, and celiac disease. Iodine is an essential element for biosynthesis of thyroxin by thyroid gland. However, people with Hashimoto’s disease might be sensitive to the harmful side effects from iodine intake. However, consumption of foods containing greater amounts of iodine such as dulse, seaweed, or kelp might cause or worsen hypothyroidism. Goitrogens present in the cruciferous vegetables interfere with function of thyroid gland by reducing the production of thyroxin hormones. The cooking process destroys goitrogens. Hence, consuming cooked cruciferous vegetables might be safer for hypothyroidism patients [26]. Certain medications and foods reduce the efficacy of thyroid medication and hence, proper spacing should be maintained while on thyroid medication.
References
- Golden SH, Robinson KA, Saldanha I, Anton B, Ladenson PW (2009) Clinical review: prevalence and incidence of endocrine and metabolic disorders in the United States: a comprehensive review. J Clin Endocr Metab 94: 1853-1878.
- Bethesda (2013) Hypothyroidism: National endocrine and metabolic diseases information service. Department of health and human services, NIH publication, US 13-6180.
- Ma Y, Sih CJ (1999) Mechanism of thyroid hormone biosynthesis. Enzymatic oxidative coupling of 3,5-diiodo-L-thyrosine derivatives. Tetrahedron Lett 40: 9211-9244.
- Harper ME, Seifert EL (2008) Thyroid hormones effects on mitochondrial energetics. Thyroid 18: 145-156.
- Melanie D (1995) Thyroid hormones and their thermogenic properties. J Restorative Med 5: 481-492.
- McCulloch AJ, Johnston DG, Baylis PH, Paylor PK (1983) Evidence that thyroid hormones regulate gluconeogenesis from glycerol in man. Clin Endocrinology 19: 67-76.
- Sheremet MI (2015) Hashimoto’s thyroiditis: surgical treatment. Akademos 2: 99-102.
- Biondi B, Klein I (2004) Hypothyroidism as a risk factor for cardiovascular disease. Endocrine 24: 1-13.
- Duntas LH, Brenta G (2012) The effect of thyroid disorders on lipid levels and metabolism. Med Clin North Am 96: 269-281.
- Johnson JL (2006) Diabetes control in thyroid disease. Diabetes Spectrum 19:148-153.
- Galofre J, Davies TF (2018) Autoimmune thyroid disease in pregnancy: a review. J Womens Health 18: 1847-1856.
- Aschebrook-Kilfoy B, Ward MH, Sabra MM, Devesa SS (2011) Thyroid cancer incidence patterns in the United States by histologic type, 1992-2006. Thyroid 21: 125-134.
- Boelaert K, Newby PR, Simmonds MJ, Holder RL, Franklyn JA, et al. (2010) Prevalence and relative risk of other autoimmune diseases in subjects with autoimmune thyroid disease. Am J Med 123: 183 -183.
- Dietary supplement fact sheet: iodine. Office of dietary supplements, NIH, US.
- Centers for Disease Control and Prevention (2012) Second report on biochemical indicators of diet and nutrition in the US population. Department of Health and Human Services Georgia, USA.
- Dean S (2008) Krause’s food, Nutrition & Diet Therapy. Medical nutrition therapy for thyroid and related disorders. WB Saunders, Philadelphia.
- Tamer G, Arik S, Tamer I, Coksert D (2011) Relative vitamin D insufficiency in Hashimoto’s thyroiditis. Thyroid 31: 891-896.
- Rayman MP (2012) Selenium and human health. Lancet 379: 1256-1268.
- Toulis KA, Anastasilakis AD, Tzellos TG, Goulis DG (2010) Selenium supplementation in the treatment of Hashimotos’s thyroiditis: a systematic review and a meta-analysis. Thyroid 20: 1163-1173.
- Sworczak K, Wisniewski P (2011) The role of vitamins in the prevention and treatment f thyroid disorders. Endokrynol Pol 62: 340-344.
- Felker P, Bunch R, Leung AM (2016) Concentrations of thiocyanate and goitrin in human plasma, their precursor concentrations in brassica vegetable and associated potential for hypothyroidism. Nutr Rev 74: 248-258.
- Gollapudi R, Gallagher R, Motohashi N (2017) Occurrence, biosynthesis and health benefits on their evidences of medicinal and phytochemicals in vegetables and fruits. Nova Science Publishers, USA.
- Divia RL, Changa HC, Doerge DR (1997) Anti-thyroid isoflavones from soy bean: isolation, characterization and mechanisms of action. Biochem Pharmacol 54: 1087-1096.
- Messina M, Redmond G (2006) Effects of soy protein and soy bean isoflavones on thyroid function in healthy adults and hypothyroid patients: a review of the relevant literutare. Thyroid 16: 249-258.
- Sathyapalan T, Manuchehri AM, Thatcher NJ, Rigby AS, Atkin SL, et al. (2011) The effect of soy phytoestrogen supplementation on thyroid status and cardiovascular risk markers in patients with subclinical hypothyroidism: a randomized, double-blind crossover study. J Clin Endocrinol Metab 96: 1442-1449.
- Elnour A. Hambraeus L, Eltom M, Dramaix M, Bourdoux P (2000) Endemic goitre with iodine sufficiency: a possible role for the consumption of pearl millet in the etiology of endemic goiter. Am J Clin Nutr 71: 59-66.
- Kelley MK, Bjeldanes LF (1995) Modulation f glutathione S-transferase activity and isozyme pattern in liver and small intestine of rats fed goitrin-and T3-suplimented diets. Food Chem Toxicol 33: 129-137.
- Chens A (2001) Update on glucosinolate metabolism and transport. Plant Physiol Biochem 39: 743-758.
- Mabon N, Mandiki SNM, Derycke G, Bister JL, Paquay R (2000) Chemical changes and influences of rapeseed antinutritional factors on lamb physiology and performance. 3. Ntinutritinal factors in plasma and organs. Animal Feed Sci Technol 85: 111-120.
- Thomke S, Petterson H, Neil M, Hakansson J (1998) Skeletal muscle goitrin concentration and organ weights in growing pigs fed diets containing rapeseed meal. Animal Feed Sci technol 73: 207-215.
- Langer P, Michailovskij N, Sedlak J, Kulka M (1971) Studies on the antithyroid activity of naturally occurring L-5-vinyl-2-thiooxazolidone in man. Endokrinologie 57: 225-229.
- Chu M, Seltzer TF (2010) Myxederma coma induced by ingestion of raw bok choy. New Engl Med 362: 1945-1946.
- Tripathi MK, Mishra AS (2007) Glucosinolates in animal nutrition: a review. Animal Feed Sci 132: 1-27.
- Zhang Y (2012) The molecular basis that unifies the metabolism, cellular uptake and chemoprevetive activities of dietary isothiocyanates. Carcinogenesis 33: 2-9.
- Ermans AM, Bourdoux P (1989) Antithyroid sulfurated compounds. In: Environmental Goitrogenesis. CRC Press, Boca Raton, FL.
- Lakshmy R, Rao PS, Sesikeran B, Suryaprakash P (1995) Iodine metabolism in response to goitrogens induced altered thyroid status under conditions of moderate and high intake of iodine. Horn Metab Res 10: 450-454.
- John-Kalarickal J, Pearlman G, Carlson HE (2007) New medications which decrease levothyroxine absorption. Thyroid 17: 763-765.
- Neafsy PJ (2004) Levothyroxin and calcium interaction-timing is everything. Hom Health Nurse 22: 338-339.
- Mazokopakis EE, Giannakopoulos TG, Starakis (2008) Interaction between levothyroxine and calcium carbonate. Can Fam Physician 54: 39
- Benvenga S, Bartolone L, Pappalardo MA, Lapa RA, Trimarchi F, et al. (2008) Altered intestinal absorption of L-thyroxine cause by coffee. Thyroid 18:293-301.
- Egert S, Rimbach G (2011) Which sources of flavonoids: complex diets or dietary supliments? Adv Nutr 2: 8-14.
- Nordio M, Basciani S (2015) Efficacy of a food supplement in patients with Hashimoto’s thyroiditis. J Biol Regulators & Homeostatic Agents 29: 63-72.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi