Research Article, J Clin Nutr Metab Vol: 3 Issue: 1
Transferrin Receptor 1 and Ferroportin Expression in the Full-Term Human Placenta and its Association with Maternal and Neonatal Iron Status: A Pilot Study
Vanessa Corrales-Agudelo1, Julio C Bueno-Sanchez2, Beatriz Pena- Arboleda2, Arturo Cardona-Ospina3, Ernesto Lopez-Rojas4, Luis Escobar5, Juan G Maldonado-Estrada6 and Beatriz E Parra-Sosa1*
1Food and Human Nutrition Research Group, School of Nutrition and Dietetics, University of Antioquia, Medellin, 050010474, Colombia
2Reproduction Program, School of Medicine, University of Antioquia, Medellin, 050010474, Colombia
3Clinical el Prado, Medellin, 05001000, Colombia
4Group of biological CES-EIA, Universidad del CES-Escuela de Ingeniería de Antioquia, Medellin, 050021000, Colombia
5Empresa Social del Estado Hospital General de Medellin, Medellin, 05001000, Colombia
6Centauro Group, School of Veterinary Medicine, University of Antioquia, Medellin, 050010474, Colombia
*Corresponding Author : Beatriz E Parra-Sosa
Carrera 53 #61 30, Laboratorio 413, Medellin Colombia, 050010474
Tel: (+574) 2199236
E-mail: beatriz.parra@udea.edu.co
Received: 08 August 2018 Accepted: 30 November 2018 Published: 07 December 2018
Citation: Corrales-Agudelo V, Bueno-Sanchez JC, Pena-Arboleda B, Cardona-Ospina A, Lopez-Rojas E (2018) Transferrin Receptor 1 and Ferroportin Expression in the Full-Term Human Placenta and its Association with Maternal and Neonatal Iron Status: A Pilot Study. J Clin Nutr Metab 3:1.
Abstract
Background: Maternal iron deficiency negatively affects the iron status of the newborn and could have adverse effects during childhood and adult life, particularly on cognitive and productive processes. Because few studies have related the
level of iron receptor expression in the placenta with maternal and/or newborn iron status, the objective of this study was to evaluate the association between the iron statuses in the mother and the newborn and the expression of transferrin
receptor-1 (TfR1) and ferroportin (FPN) in the full-term human placenta.
Materials and methods: A pilot study, cross-sectional design, we selected full-term pregnant women based on their antepartum iron status: iron deficiency with anemia (IDA, n=5), iron deficiency without anemia (IDNA, n=9) and normal iron
status (NIS, control group, n=10). All newborns were delivered by caesarean section (between weeks 37 and 39 of pregnancy). Blood samples were taken from the newborn by umbilical vein sampling for serum ferritin measurements. The
expression of TfR1 and FPN in placental villous tissue and isolated trophoblast cells was evaluated by immunohistochemistry (IHC) and flow cytometry, respectively.
Results: The expression levels of TfR1 and FPN in villous tissue were significantly correlated, with no significant differences between groups. TfR1 and FPN expression was predominantly found in the apical membrane of the syncytiotrophoblast and villous stroma, respectively. Neonatal ferritin serum levels significantly correlated with FPN expression.
Keywords: Transferrin receptor; Ferroportin; Anemia; Maternal-fetal relations; Placenta
Abbreviations
BMI: Body Mass Index; CELL DYN: Cyanmethemoglobin Method; CK7: Cytokeratin-7; CRP: C-Reactive Protein; Ct: Cytotrophoblast; DMSO: Dimethyl Sulfoxide; DNAse: Deoxyribonuclease; ESE: Empresa Social del Estado; ECLIA: Electro Chemiluminescence Immune Assay; EDTA: Anticoagulant Ethylenediaminetetraacetic Acid; FCS: Fetal Calf Serum; FITC: Fluorescein Isothiocyanate; FPN: Ferroportin; Hb: hemoglobin; hCG: Human Chorionic Gonadotropin; IDA: Iron Deficiency with Anemia; IDNA: Iron Deficiency without Anemia; IHC: Immunohistochemistry; IUGR: Intrauterine Growth Restriction; IV: Intervillous Space; VS: Villous Stroma; MFI: Mean Fluorescence Intensity; mRNA: Messenger Ribonucleic Acid; NIS: Normal Iron Status; OCT: Optimal Cutting Temperature; PBS: Phosphate-Buffered Saline; SGA: Small For Gestational Age St: Syncytiotrophoblast; TfR1: Transferrin Receptor-1.
Introduction
Iron deficiency and ferropenic anemia are two of the most important nutritional conditions affecting pregnant women in developing countries [1-3]. They also affect the health and wellbeing of the newborn(1-2). In countries such as Colombia, nutritional anemia and ferropenia occur in 20% and 39% of pregnant women, respectively. Among women affected by anemia, 50% also suffer from ferropenia [4].
Studies focused on the relationship between maternal iron status and fetal iron status has produced no conclusive results. Some authors have argued that, even if iron placental deficiency does exist because of iron maternal status, it has negligible effects on the fetal iron status because the iron levels in the fetus are compensated by its intestinal regulation, as suggested by O’Brien et al. [5]. However, other authors have provided evidence of the detrimental effect of maternal iron deficiency on fetus development, reporting that impaired iron supply to the fetus results in neurological developmental anomalies and increased risk of postnatal iron deficiency in the newborn[6,7](8,9).
Dietary and endogenous iron in the mother constitutes the fetal iron sources. Many proteins could be involved in iron uptake and utilization by the placenta, and some have hypothesized that these proteins may also be expressed in enterocytes [8,9]. Two of the proteins most involved in iron transport into placental cells are transferrin receptor type-1 (TfR1) [9-15] and ferroportin (FPN) [9-11,13-15]. The choriocarcinoma cell line BeWo has been widely used as a model of the syncytiotrophoblast because of its expression of human chorionic gonadotropin (hCG) and syncytin, and as a result, it has been used to study of the contributions of TfR1 and FPN to placental iron transport in vitro [9,11,16]. In BeWo cells, TfR1 is predominantly expressed in the apical membranes of the cells, whereas FPN is predominantly expressed in the basolateral membranes.
For this reason, FPN appears to be responsible for iron export from the placenta to the fetal circulation [9,11]. Studies in pregnant rats suggest that placenta. TfR1 and FPN expression varies according to the iron availability in the maternal circulation [14,17]. To date, only a few reports have examined the expression of TfR1 and FPN in the human placenta. Li et al. [18] found no relationship between maternal iron status and TfR1 and/or FPN expression, as evaluated by mRNA and protein analyses. In contrast, Young et al. [19] found an inverse relationship between TfR1 expression, evaluated by Western blot, and the iron reserves of the mother and fetus [19].
Because iron deficiency in the newborn is related to adverse outcomes in cognitive activities and productivity during childhood and adult life [20,21], a study of the mechanisms responsible for iron transport in the full-term human placenta could provide insights into the modulation of iron receptor expression in cases of maternal iron deficiency. Based on the working hypothesis that the expression of the proteins (TfR1 and FPN) in the placenta could be inversely related to the iron statuses of the mother and fetus (as compensatory mechanisms in cases of iron deficiency), the present pilot study aimed to evaluate the relationship between maternal and fetal iron status and the placental expression of TfR1 and FPN in human full-term villous placentas and trophoblast cells.
Materials and Methods
Ethics
The Institutional Board on Subject Experimentation of the University of Antioquia approved this study (Act#08026187, November 27, 2009). All patients received complete information about the aim of the study and signed an informed consent form.
Study design
A pilot cross-sectional study, including three groups of individuals defined according to their iron storage and hemoglobin (Hb) values. We bled patients during the third trimester of pregnancy near term to determine their iron status. Similarly, we bled the fetus by umbilical blood sampling before sectioning of the umbilical cord. Finally, within 30 min after delivery, the placenta was brought to the laboratory under sterile conditions and used to isolate the placental villi and trophoblast cells.
The sample size was defined according to formulae for quantitative variables [22], in which the minimum difference for TfR1 mRNA expression between normal pregnant women and pregnant women with moderate anemia was considered (0.28) [18]. The standard error was 0.188, 95% confidence intervals were used, and the statistical power was 85%. In addition, we defined an additional sample size of 10%. Thus, the total sample size was 27 pregnant women. The study was performed with women from the metropolitan area of the Aburra Valley who attended three health care centers, as shown below.
Experimental groups:
A: The control group (normal iron status [NIS], n=10) included pregnant women with ferritin and Hb levels of ≥15 μg/L and ≥11.0 g/dL, respectively.
B: The iron deficiency without anemia group (IDNA, n=9) included pregnant women with ferritin and Hb levels of <15 μg/L and ≥11.0 g/dL, respectively.
C: The iron deficiency with anemia group (IDA, n=5) included pregnant women with ferritin and Hb values of <15 μg/L and <11.0 g/dL, respectively [23] C-reactive protein (CRP) values of ≤1.5 mg/dL were considered to be normal with no evidence of infection or inflammatory conditions [24,25]. In contrast, CRP values of>1.5 mg/dL were considered to reflect an acute infection or inflammation, and in this case, ferritin values of <30 μg/L were considered to be indicative of iron deficiency. Hb levels in pregnant women were adjusted according to the altitude at which the mother resided [26] because the altitude of the metropolitan area of the Aburra Valley varies, reaching as high as 1,500 meters over sea level (m.o.s.l).
Inclusion criteria: The inclusion criteria of the study were as follows.
A: Full-term pregnancy with no obstetrical complications
B: Age ranging from 19 to 40 years
C: Monofetal pregnancy
D: Normal or overweight body mass index (BMI)
E: Absence of systemic disease other than anemia and/or iron deficiency
F: No drug or tobacco abuse
G: Other indicators of iron status considered in mother's state of anemia with iron deficiency (serum level <40 μg/dL, transferrin iron saturation index <15%, or transferrin iron-binding capacity ≥410 μg/dL) [27].
Patients from three different locations in the city of Medellin were included.
A: A third-level public hospital (E.S.E. Hospital General de Medellin)
B: A second-level health care center (E.S.E. Metrosalud, Unidad Hospitalaria de Manrique)
C: A private practice obstetric clinic (Clinica del Prado, Medellin)
Exclusion criteria: The exclusion criteria of the study were as follows.
A: Maternal underweight BMI
B: Intrauterine growth restriction (IUGR)
C: Small for gestational age (SGA) fetus
D: Pre-term newborn
E: Vaginal delivery
F: Adolescent mother (women under 19 years)
G: Older mother (women over 40 years)
H: General disease
I: Multiple pregnancy
J: Addiction and/or drug abuse diseases.
Loss of subject for data analysis: The sample size for certain procedures was affected because of several difficulties affecting sample collection and/or sample storage as follows. A: In the IDA group (n=5), it was not possible to complete the original sample size because most patients in this group concomitantly suffered low weight, and thus, their babies suffered from IUGR or were born SGA; these clinical conditions were considered exclusion criteria.
B: Data from the flow cytometry analyses performed to evaluate the expression of TfR1 and FPN were obtained from five patients in the IDA group, eight patients in the IDNA group and 10 patients in the control group.
C: The maternal total iron-binding capacity was calculated in 22 samples: 5 in the IDA group, 9 in the IDNA group and 8 in the control group.
D: The percent saturation of maternal transferrin was determined in 20 patients: three in the IDA group, nine in the IDNA group and eight in the control group. Sampling the umbilical cord in one neonate was impossible because the newborn was cyanotic at the time of sampling, and he/she was considered to be at high risk. Consequently, neonatal ferritin levels were calculated in 23 newborn babies: five in the IDA group, eight in the IDNA group, and ten in the control group.
Methods
Measurement of maternal anthropometry: Pregnant women were engaged in the study at week 36 of pregnancy. All patients received complete clinical and nutritional examinations performed by a gynecologist and a nutritionist, respectively. Body weight was measured on a Tanita HD327 electronic scale (Arlington Heights, Illinois, USA; 0.1 kg sensitivity, 150 kg capacity), and body height was determined with a Seca portable measuring rods (Hamburg, Germany; 0.1-cm sensitivity). All patients received complete information about the aim of the study and signed an informed consent form.
Quantification of biochemical markers of maternal iron status: After 10 h of fasting, 5 ml blood samples were taken by peripheral vein puncture using sterile anticoagulant ethylenediaminetetraacetic acid (EDTA)-containing 3 K glass tubes, and 10 ml samples were collected in sterile tubes without anticoagulant. All disposable materials used for blood sampling were iron free, and the materials used for sample processing were sterilized in 50% (weight/volume) nitric acid in deionized water.
An automated BD-hemocytometer was used for the complete hemoleucogram obtained from coagulated blood samples. Ferritin levels were measured by the Electro Chemiluminescence Immune Assay (ECLIA) (Roche Modular Analytics E170); Hb was measured with the modified cyanmethemoglobin method (CELL DYN 3700; Abbott), and CRP levels were measured by immunoturbidimetry using an automated analyzer (Modular P800; Roche). All these procedures were performed in the PROLAB Clinic Laboratory, Medellín. The CRP values were used as indicators of acute inflammation and as correction factors for maternal ferritin levels. Serum iron levels and total ironbinding capacity were calculated using the photo colorimetric method with a commercial kit (Fe-ferrozina, BioSystem) and read using semi-automated RA-50-Bayer equipment. The transferrin serum iron saturation index was calculated based on the resulting values.
Quantification of neonatal iron status: Umbilical cord samplings were performed according to the protocol established by the Congenital Neonatal Hypothyroidism Screening Study (Ministry of Health, Republic of Colombia) [28] and the Colombian guidelines for newborn care [29]. Neonatal serum ferritin levels were calculated by the ECLIA method as mentioned above in the PROLAB Clinical Laboratory. The cut-off value for neonatal iron deficiency was a ferritin level of <60 μg/L.
Isolation of placental villous tissue: Immediately after caesarean section, the placenta was removed under sterile conditions, packed in a sterile plastic bag, and transported to the laboratory within 30 min. The placenta of each patient was processed individually for villous tissue dissection and trophoblast cell isolation using trypsin+DNAse digestion, as previously reported [30]. Villous tissue fragments with diameters of 3 mm to 5 mm were washed in phosphate-buffered saline (PBS), placed in optimal cutting temperature (OCT) compoundcontaining Tissue-Tek® II Brand Cryomold® molds and frozen at -80°C until being processed for immunohistochemistry (IHC). Triplicate samples of whole trophoblast cells isolated after trypsin-DNAse digestion as previously reported [30,31] were washed, frozen in 10% fetal calf serum (FCS)-containing dimethyl sulfoxide (DMSO) and stored at -196°C until being processed.
TfR1 and FPN expression in trophoblast cells by flow cytometry: Trophoblast cells were thawed at 37°C for 30 sec and washed three times in 10% FCS-containing PBS to eliminate excess DMSO. The cells were kept at 4°C for 12 h to allow them to recover after thawing. They were then processed for double staining with TfR1 and cytokeratin-7 (CK7) or FPN and CK7 monoclonal antibodies by immunofluorescence, as previously reported [31]. CK7 staining was performed using a mouse anti-human CK7 monoclonal antibody [32] (Dako, Denmark), followed by incubation with secondary fluorescein isothiocyanate (FITC)-coupled goat anti-mouse polyclonal antibodies (Millipore). CK7 immunofluorescence was performed after solubilizing the cell membrane using 0.03% Triton-X detergent [31].
After CK7 staining, the cells were fixed with 0.1% paraformaldehyde. TfR1 or FPN was stained by incubation with a primary mouse anti-human TfR1 or mouse anti-human FPN monoclonal antibody, followed by incubation with a secondary Cy3- coupled goat anti-mouse monoclonal antibody. As a positive control for CK7 or TfR1 and FPN expression, Jeg3 choriocarcinoma cells cultured to 80% to 90% confluence and human adherent monocytederived macrophages cultured under standard conditions for 72 h, respectively, were used. For immunofluorescence staining, trophoblast cells (105/ml) were processed in 2 ml polystyrene tubes and read by a FACS Canto II instrument (Beckton-Dickinson). Measurements were performed in duplicate, and data from each marker and its specific isotype control were recorded over 50,000 events. TfR1 and FPN expression data were calculated based on the CK7 positive-gated events.
TfR1 and FPN expression in villous tissues assessed by immunohistochemistry: Cryomolds were used to process villous tissue samples. Five-millimeter samples were obtained and fixed onto microscopic slides (n=10 slides/sample): One slide was used for hematoxylin and eosin staining, and the remaining nine slides were used for CK7 [31], TfR1 and FPN staining in triplicate. IHC was performed using the EnVision+System-HRP (AEC) kit (Dako, K4005). For TfR1 expression, rabbit anti-human polyclonal antibodies were used (1 mg/mL, Millipore) at 10:200 dilution. To evaluate FPN expression, a rabbit anti-mouse metal transporter protein (1 mg/mL, Alpha Diagnostic International) was used at a 10:200 dilution.
For CK7 expression, the monoclonal antibody indicated above for immunofluorescence was used at a 1:200 dilution and as a positive control for flow cytometry and IHC [31]. The slides were processed according to the manufacturer’s instructions, and measurements were taken using images from a Nikon Eclipse E200 microscope. Microphotographs were recorded using an Aiptek z600 camera adapted to a 100X objective. Duplicate samples were processed. IHC was performed on 15 of 25 samples (5 samples per group), and the samples were paired according to the anemia and ferropenia group. The images were analyzed using the ImageJ software as follows: To evaluate each marker, whether the CK7 signal was positive was determined, and an area corresponding to the entire syncytiotrophoblast layer was recorded; a similar area was then evaluated on those slides corresponding to TfR1. For FPN expression, the area was recorded between the syncytiotrophoblast layer and the fetal capillaries corresponding to the villous stroma, in which the FPN signal was always observed. Experiments were performed in triplicate. For statistical analyses, the data were normalized to the isotype control values. The TfR1 or FPN expression was then further normalized to CK7 expression.
Statistical analysis: The normality of the data was assessed by the Shapiro-Wilk test. TfR1 and FPN expression data were evaluated by the mean fluorescence intensity (MFI) and percentage of expression. Gestational age, maternal and neonatal serum ferritin, CRP and the iron-binding capacity of transferrin did not exhibit normal distributions; the other variables were normally distributed. In accordance with the distribution of variables, the mean and standard deviation or median and interquartile range were used, as appropriate. In addition, relative frequencies were determined to clarify the information when necessary.
Comparisons between the three study groups were performed using analysis of variance (ANOVA) or the Kruskal-Wallis test according to the normality of the variables, and comparisons between two groups were made using the Mann-Whitney U test when the variable was nonparametric and Student’s t test for normally distributed variables. Similarly, the Gaussian distribution was considered to determine the association between the variables of state of maternal and neonatal iron by the Spearman or Pearson rank correlation coefficient, as appropriate. Statistical significance was set at p <0.05. Variables from in vitro assays were analyzed with GraphPad Prism 5 and FlowJo 7.6.1; for other variables, SPSS V 18 was used.
Results
Anthropometric measures
The median gestational age was 38 weeks for all groups. Pregnant women with ferropenia and anemia tended to exhibit lower pregestational and gestational weights than women with ferropenia without anemia and women from the control group (p=0.104 and p=0.089, respectively). BMI values revealed that 71% (n=17) and 29% (n=7) of the women had appropriate and overweight nutritional statuses, respectively. The body weight and size of the neonates did not significantly differ between groups (p>0.05).
Biochemical parameters
The serum iron status, transferrin iron-binding capacity and percent saturation of maternal transferrin were complementary indicators of maternal iron status. When the associations among maternal iron biochemical parameters were evaluated, a significant relationship was apparent between maternal Hb concentration and maternal serum ferritin levels (r=0.6; p<0.01), maternal serum iron levels (r=0.6; p<0.01) and the percent saturation of maternal transferrin (r=0.6; p<0.001) (data not shown). In contrast, when biochemical parameters were compared between the mothers and their newborns, maternal serum ferritin levels tended to correlate with fetal serum ferritin levels (r=0.3; p>0.05) (data not shown). Twenty-two percent of neonates (n=5) had serum ferritin values lower than 60 μg/L, the cut-off point for a determination of neonatal iron deficiency, and no statistically significant differences were found among the groups (p>0.05) (data not shown).
TfR1 and FPN expression in the full-term human placenta
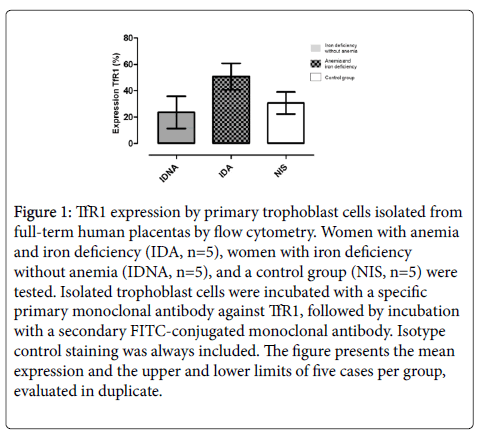
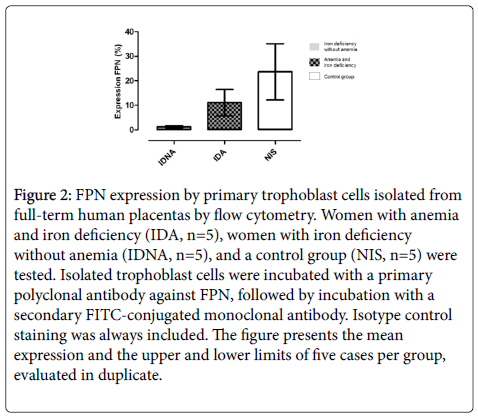
TfR1 and FPN expression in isolated trophoblast cells, as measured by flow cytometry, was not statistically significantly different among groups (Figures 1 and 2) (p>0.05). Similarly, no statistically significant differences were found when TfR1 and FPN expression was evaluated by IHC in villous tissues isolated from full-term human placentas (p>0.05). These data were corroborated by semi-quantitative analyses (p>0.05).
Figure 1: TfR1 expression by primary trophoblast cells isolated from full-term human placentas by flow cytometry. Women with anemia and iron deficiency (IDA, n=5), women with iron deficiency without anemia (IDNA, n=5), and a control group (NIS, n=5) were tested. Isolated trophoblast cells were incubated with a specific primary monoclonal antibody against TfR1, followed by incubation with a secondary FITC-conjugated monoclonal antibody. Isotype control staining was always included. The figure presents the mean expression and the upper and lower limits of five cases per group, evaluated in duplicate.
Figure 2: FPN expression by primary trophoblast cells isolated from full-term human placentas by flow cytometry. Women with anemia and iron deficiency (IDA, n=5), women with iron deficiency without anemia (IDNA, n=5), and a control group (NIS, n=5) were tested. Isolated trophoblast cells were incubated with a primary polyclonal antibody against FPN, followed by incubation with a secondary FITC-conjugated monoclonal antibody. Isotype control staining was always included. The figure presents the mean expression and the upper and lower limits of five cases per group, evaluated in duplicate.
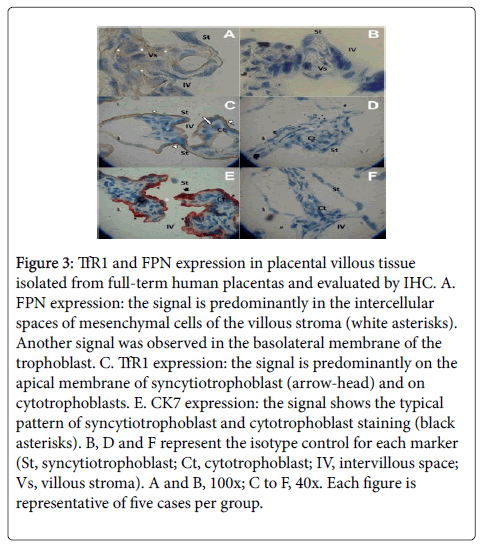
CK7 expression was included in the analysis as a co-variable for TfR1 and FPN expression (p<0.01 in the model) (data not shown). Finally, TfR1 expression showed significant correlations with FPN expression evaluated by IHC (correlation coefficient=0.740982, Rsquared= 54.905%; p<0.01). The expression of TfR1 was predominantly detected in the apical membrane of the syncytiotrophoblast layer, although some signals were also observed in zones of contact between the syncytium and cytotrophoblasts (Figure 3C). A strong TfR1 signal was observed in protruding syncytiotrophoblast microparticles in several villous tissues (data not shown). In contrast, FPN expression was predominantly found in the basal membrane of the syncytia facing the villous stroma (e.g., the fetal side of the interface). The FPN signal was consistently weaker than the TfR1 signal (Figure 3A). In all cases, CK7 expression showed the classical pattern at the level of the syncytiotrophoblasts (Figure 3E), and its signal was stronger than those of TfR1 (Figure 3C) and FPN (Figure 3A).
Figure 3: TfR1 and FPN expression in placental villous tissue isolated from full-term human placentas and evaluated by IHC. A. FPN expression: the signal is predominantly in the intercellular spaces of mesenchymal cells of the villous stroma (white asterisks). Another signal was observed in the basolateral membrane of the trophoblast. C. TfR1 expression: the signal is predominantly on the apical membrane of syncytiotrophoblast (arrow-head) and on cytotrophoblasts. E. CK7 expression: the signal shows the typical pattern of syncytiotrophoblast and cytotrophoblast staining (black asterisks). B, D and F represent the isotype control for each marker (St, syncytiotrophoblast; Ct, cytotrophoblast; IV, intervillous space; Vs, villous stroma). A and B, 100x; C to F, 40x. Each figure is representative of five cases per group.
Discussion
Our pilot study provided the first results on the expression of TfR1 and FPN in trophoblast cells isolated from pregnant women with IDA, IDNA and NIS whose neonates were delivered by caesarean section between weeks 37 and 39 of pregnancy. No statistically significant relationship was found between the expression of these iron receptors and transporters and the maternal gestational consumption of iron supplements, its frequency or the duration of supplementation or the mother’s iron status (data not shown). In agreement with the report by Bastin et al. [33], TfR1 expression occurred predominantly in the syncytiotrophoblast brush border and the entire syncytia, including in several cytotrophoblast cells (Figure 3C). In contrast, FPN expression was predominantly found in areas of the villous stroma facing the fetal capillary, and weak signals were observed in some isolated cytotrophoblast cells (Figure 3A), similar to previous reports [33]. These expression patterns are in agreement with previous reports in human placenta [33,34], rat placenta [17] and BeWo cells [11], a choriocarcinoma cell line considered to be a cellular model of syncytiotrophoblasts [35,36].
The weak FPN signal found by IHC in villous stroma (Figure 3A) and its low level of expression in trophoblast cells observed by flow cytometry (Figure 2) suggest that this weak expression is common in the human placenta. The flow cytometry signals of TfR1 and FPN were evaluated in CK7 cells only, providing evidence of their expression in trophoblast cells. Interestingly, the flow cytometry results indicated that the expression levels of FPN and TfR1 were significantly correlated, confirming that these receptors play a key role in iron transport from the mother to the fetus. Similar results were reported by Mando et al. [37], who observed the same pattern of TfR1 expression in normal pregnant women and in women with newborns affected by IUGR. Finally, the pattern of TfR1 and FPN expression reported here and elsewhere [9,38] suggests that iron transport in pregnant women follows a pattern similar to those reported previously in the rat model [17] and in BeWo cells [11,35,36].
The iron serum values of women from the IDA group (average: 36.0 μg/dL) were below the cut-off point for a diagnosis of iron deficiency (40 μg/dL). In women from the IDNA and the control groups, these values were 51.9 μg/dL and 78.4 μg/dL, respectively. The percent saturation of maternal transferrin was lower in women from the IDA and IDNA groups (15.7% and 26.8%, respectively), and no statistically significant difference was found between the groups. However, Basu et al. [39] noted significant differences in the serum ferritin in the cord blood of newborns from non-anemic mothers and anemic mothers. Similar to Li et al. [40] we found no significant differences in the iron status of newborns from pregnant women with different iron statuses however, newborns from the IDA and IDNA groups had lower iron levels (99.1 μg/L and 69.2 μg/L, respectively) than those from the control group (112.8 μg/L). Instead, most biochemical parameters indicative of maternal iron status were significantly correlated with maternal Hb levels, although such a relationship was not confirmed for maternal ferritin levels (data not shown).
Interestingly, a statistically significant relationship was found between placental FPN expression and neonatal ferritin levels, suggesting that pregnant women with higher degrees of iron deficiency exhibited higher FPN expression as a possible mechanism for protecting their fetuses from iron deficiency, although neonatal ferritin levels were lower in newborns from pregnant women with impaired iron status. A similar result was reported by Li et al. [40] who measured mRNA levels and found that the expression of FPN mRNA in the placenta was upregulated in pregnant women with severe iron deficiency. Prenatal iron deficiency has been shown to predispose newborns to childhood anemia if they are further exposed to risk factors, such as a lactation period shorter than six months, low intake of iron-rich food after six months of age, and/or late inclusion of iron sources in the diet after six months of age [41]. Our results also suggest that other markers of iron transport from the mother to the fetus must be included in further studies.
The sample size was one of the biggest limitations of our study, particularly in the IDA group, as most pregnant women with elective caesarean section and IDA also presented undernutrition, IUGR or babies of small size for their gestational age; these conditions made them ineligible for enrollment in this study. This limited the analysis and therefore potentially important clinical findings in iron status of the mother, the neonate or expression of TfR1 and FPN.
Our results provide evidence supporting the importance of balanced maternal nutrition during pregnancy for fetal wellbeing, showing a possible respond from mothers to the stressor of iron deficiency to guarantee adequate passage of iron to the fetus through the placental barrier. In fact, in the human full-term placenta, TfR1 and FPN exhibit similar patterns of expression as those reported for animal models with gestational anemia. The results of this pilot study contribute to expand the knowledge of a possible modulating the expression of proteins necessary for the maternal-fetal iron transport according to the maternal iron state in humans. Even if this regulation seems to favor the passage of iron from the placenta to the fetus, infants from mothers that suffered from anemia or iron deficiency during pregnancy have lower levels of ferritin in umbilical cord blood than newborns from mothers with normal iron status. This highlights the importance of iron supplementation and the monitoring of the nutritional status of this micronutrient during pregnancy to ensure adequate transport to the fetus and the maintenance of normal iron status in the mother.
Conclusions
Immunohistochemistry shows that TfR1 expression is correlated with FPN expression.
In response to iron deficiency, a maternal mechanism assures the passage of iron to the fetus.
Increased FPN expression protects the fetus from iron deficiency.
Balanced maternal nutrition during pregnancy supports fetal wellbeing.
Funding Statement
This work was supported by the “Vicerrectoría de Investigación” of the University of Antioquia [Sustainability Strategy for Research Groups, CODI-2013-2014 for the Food and Human Nutrition Research Group, School of Nutrition and Dietetics, University of Antioquia; “Fundación Banco de la República” [Ref: Proyecto No. 3.032]; and Laproff S.A. Laboratories.
The funding sources had no involvement in study design; the collection, analysis and interpretation of data; the writing of the report; or the decision to submit the article for publication.
Acknowledgment
The authors thank the “E.S.E. Hospital General de Medellín”, “Clínica del Prado” and “Unidad Hospitalaria de Manrique (E.S.E. Metrosalud)” for enrolling patients. Special thanks are also due to Dr. Alba Osorio, MD, and the nursing personnel of each center for helping with patient recruitment and selection. We gratefully appreciate the cooperation of the pregnant women who signed the informed consent for inclusion in the study. Special thanks are extended to Dr. Mauricio Rojas (head of the Flow Cytometry Unit) of the “Sede de Investigación Universitaria (University of Antioquia)” for providing technical support.
Conflicts of interest: none.
References
- Perez EM, Hendricks MK, Beard JL (2005) Mother-infant interactions and infant development are altered by maternal iron deficiency anemia. J Nutr 135: 850-855.
- Darnton-Hill I, Webb P, Harvey PW, Hunt JM, Dalmiya N, et al. (2005) Micronutrient deficiencies and gender: social and economic costs. Am J Clin Nutr 81: 1198-1205.
- Jemal H (2001) Prevalence and epidemiology of iron deficiency. In: WHO (ed) Iron Defic. anaemia assessment. Prev. Control. A Guid. Program. Manag. World Health Organization, Geneva, 15-21.
- Instituto Colombiano de Bienestar Familiar, Profamilia, Instituto Nacional de Salud, Ministerio de la Protección social, Organización Panamericana de la Salud, Programa Mundial de Alimentos, Acofanud, DANE, Coldeportes (2010) Distribución de la concentración de Hemogobina y prevalencia de anemia en gestantes de 13-49 años según caracteristicas demográficas, socioeconómicas, y consumo de cigarrillo. In: ICBF (ed) Encuesta Nac. la Situación Nutr. en Colomb. 2010 ENSIN. Bogotá, D.C., p 163,172,203.
- O’Brien KO, Zavaleta N, Abrams SA, Caulfield LE (2003) Maternal iron status influences iron transfer to the fetus during the third trimester of pregnancy. Am J Clin Nutr 77: 924-930.
- Jaime-Perez JC, Herrera-Garza JL, Gomez-Almaguer D (2005) Sub-optimal fetal iron acquisition under a maternal environment. Arch Med Res 36: 598–602.
- Rao R, Georgieff MK (2007) Iron in fetal and neonatal nutrition. Semin Fetal Neonatal Med 12: 54-63.
- Cao C, O’Brien KO (2013) Pregnancy and iron homeostasis: An update. Nutr Rev 71:35-51.
- Cao C, Fleming MD (2016) The placenta: the forgotten essential organ of iron transport. Nutr Rev 74: 421-431.
- Pattillo RA, Gey GO (1968) The establishment of a cell line of human hormone-synthesizing trophoblastic cells in vitro. Cancer Res 28: 1231-1236.
- Heaton SJ, Eady JJ, Parker ML, Gotts KL, Dainty JR, et al. (2008) The use of BeWo cells as an in vitro model for placental iron transport. Am J Physiol Cell Physiol 295: 1445-1453.
- Gómez-Gutiérrez AM, Parra-Sosa BE, César Bueno-Sánchez J (2013) Papel del receptor 1 de transferrina en la captación del hierro y su relación con la deficiencia gestacional de hierro y la preeclampsia. Rev Cuba Obstet y Ginecol 39: 33-42.
- McArdle HJ, Gambling L, Kennedy C (2014) Iron deficiency during pregnancy: The consequences for placental function and fetal outcome. Proc Nutr Soc 73: 9-15.
- Sangkhae V, Nemeth E (2018) Placental iron transport: the mechanism and regulatory circuits. Free Radic Biol Med.
- Fisher AL, Nemeth E (2017) Iron homeostasis during pregnancy. Am J Clin Nutr 106: 1567-1574.
- Van der Ende A, du Maine A, Schwartz AL, Strous GJ (1989) Effect of ATP depletion and temperature on the transferrin-mediated uptake and release of iron by BeWo choriocarcinoma cells. Biochem J 259: 685-692.
- Gambling L, Danzeisen R, Gair S, Lea RG, Charania Z, et al. (2001) Effect of iron deficiency on placental transfer of iron and expression of iron transport proteins in vivo and in vitro. Biochem J 356: 883-889.
- Li YQ, Yan H, Bai B (2008) Change in iron transporter expression in human term placenta with different maternal iron status. Eur J Obs Gynecol Reprod Biol 140: 48-54.
- Young MF, Pressman E, Foehr ML, McNanley T, Cooper E, et al. (2010) Impact of maternal and neonatal iron status on placental transferrin receptor expression in pregnant adolescents. Placenta 31: 1010-1014.
- Hay G, Refsum H, Whitelaw A, Melbye EL, Haug E, et al. (2007) Predictors of serum ferritin and serum soluble transferrin receptor in newborns and their associations with iron status during the first 2 y of life. Am J Clin Nutr 86: 64-73.
- Agarwal K, Gupta V, Agarwal S (2013) Effect of maternal iron status on placenta, fetus and newborn. IJMMS 5: 391-395.
- Martínez-Gonzalez MA, Alonso A, Bes-Rastrollo M (2006) Estimación del tamaño muestral. In: Martínez-González MA, Sánchez-Villegas A, Faulín FJ (eds) Bioestadistica amigable, 2da ed. Diaz de santos, España, 373-417.
- WHO/UNICEF/UNU (2001) Variations in haemoglobin and haematocrit levels. In: WHO (edtn) Iron Defic. anaemia assessment. Prev. Control. A Guid. Program. Manag. World Health Organization, Geneva, 99-101.
- Cunningham GF, Leveno KJ, Bloom SL, Spong CY, Dashe JS, Hoffman BL, Casey BM, Sheffield JS (2014) Maternal Physiology. In: Cunningham GF, Leveno KJ, Bloom SL, Spong CY, Dashe JS, Hoffman BL, Casey BM, Sheffield JS (eds) Williams Obstet. 24th Ed., 24th ed. Mc Graw Hill Education, New York 46-77.
- Parra BE, Manjarres LM, Gomez AL, Alzate DM, Jaramillo MC (2005) Assessment of nutritional education and iron supplement impact on prevention of pregnancy anemia. Biomedica 25: 211-219.
- Dirren H, Logman MHGM, Barclay DV, WB F (1994) Altitude correction for hemoglobin. Eur J Clin Nutr 48: 625-632.
- Gropper S, Smith J, Groff J (2005) Microminerals. In: Gropper S, Smith J, Groff J (eds) Adv. Nutr. Hum. Metab., 4th edtn. Thomson Wadsworth, Estados Unidos, 433.
- Ministerio de la Protección Social (2004) Tamizaje neonatal de hipotiroidismo congenito 1-36.
- Ministerio de la Proteccin Social (2007) Guías de promoción de la salud y prevención de enfermedades en la salud pública. Bogotá, D.C.
- Moussa M, Roques P, Fievet N, Menu E, Maldonado-Estrada JG, et al. (2001) Placental cytokine and chemokine production in HIV-1-infected women: trophoblast cells show a different pattern compared to cells from HIV-negative women. Clin Exp Immunol 125: 455-464.
- Maldonado-Estrada J, Menu E, Roques P, Barre-Sinoussi F, Chaouat G (2004) Evaluation of Cytokeratin 7 as an accurate intracellular marker with which to assess the purity of human placental villous trophoblast cells by flow cytometry. J Immunol Methods 286: 21-34.
- Blaschitz A, Weiss U, Dohr G, Desoye G (2000) Antibody reaction patterns in first trimester placenta: implications for trophoblast isolation and purity screening. Placenta 21: 733-741.
- Bastin J, Drakesmith H, Rees M, Sargent I, Townsend A (2006) Localisation of proteins of iron metabolism in the human placenta and liver. Br J Haematol 134: 532-543.
- Yang A, Zhao J, Lu M, Gu Y, Zhu Y, et al. (2016) Expression of hepcidin and ferroportin in the placenta, and ferritin and transferrin receptor 1 levels in maternal and umbilical cord blood in pregnant women with and without Gestational Diabetes. Int J Environ Res Public Heal.
- Speeg Jr. KV, Azizkhan JC, Stromberg K (1976) The stimulation by methotrexate of human chorionic gonadotropin and placental alkaline phosphatase in cultured choriocarcinoma cells. Cancer Res 36: 4570-4576.
- Bilban M, Tauber S, Haslinger P, Pollheimer J, Saleh L, et al. (2010) Trophoblast invasion: assessment of cellular models using gene expression signatures. Placenta 31: 989-996.
- Mandó C, Tabano S, Colapietro P, Pileri P, Colleoni F, et al. (2011) Transferrin receptor gene and protein expression and localization in human IUGR and normal term placentas. Placenta 32: 44-50.
- Haigh T, Chen CP, Jones CJP, Aplin JD (1999) Studies of mesenchymal cells from 1st trimester human placenta: Expression of cytokeratin outside the trophoblast lineage. Placenta 20: 615-625.
- Basu S, Kumar N, Srivastava R, Kumar A (2016) Maternal and Cord Blood Hepcidin Concentrations in Severe Iron Deficiency Anemia. Pediatr Neonatol.
- Li S, Liu Y, Wang Y, Qi P, Wang D (2015) The role of serum hepcidin and ferroportin1 in placenta on iron transfer from mother to fetus. Zhonghua Xue Ye Xue Za Zhi 36: 307-311.
- Pizarro F, Calvo E (2009) El significado a mediano y largo plazo de la deficiencia de hierro y zinc durante los primeros dos años de vida, para asegurar un buen crecimeinto temprano. In: Uauy R, Carmuega E, Barker B (eds) Impacto del Crecim. y Desarro. Tempr. sobre la salud y bienestar la población. Perspect. y reflexiones desde el Cono Sur. Instituto Danone del Cono Sur, Argentina, Chile y Uruguay 49–55.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi