Short Communication, J Nanomater Mol Nanotechnol Vol: 8 Issue: 3
Contrast of Ultrasonic Assisted Phase-Separation of RodSphere Mixture with Static Deposits in a Coffee-Stain Rin
Imtiaz Ahmad1*, Hidayat Ullah Khan1 and Rahim Jan2
1Department of Physics, University of Peshawar, Pakistan
2School of Chemical and Materials Engineering, National University of Sciences and Technology, Islamabad 44000, Pakistan
*Corresponding Author : Imtiaz Ahmad
Department of Physics, University of Peshawar, Pakistan
E-mail: imtiaz@uop.edu.pk
Received: December 05, 2018 Accepted: May 21, 2019 Published: May 31, 2019
Citation: Ahmad I, Khan HU, Jan R (2019) Contrast of Ultrasonic Assisted Phase-Separation of Rod-Sphere Mixture with Static Deposits in a Coffee-Stain Ring. J Nanomater Mol Nanotechnol 8:3. doi: 10.4172/2324-8777.1000269
Abstract
The effect of ultrasonic vibrations on the self-assembly and phase separation of nanorods and nanospheres is investigated. Such deposits of rod-sphere mixture on SiO2 surface, dried during ultrasonic vibrations, are compared with static nanoparticle deposits. Phaseseparation at various positions of the coffee-stain ring for both of the cases is evaluated. The ensuing phase separation under ultrasonic agitation is found more noticeable than the one ensemble through static drying. Also, shape separated nanoparticle deposits are studied at various positions of the coffee-stain ring and discuss in relation with steric interaction.
Keywords: Ultrasonic vibration; Contact-line; Self-assembly; Evaporation; Coffee-stain ring; Phase separation
Introduction
Self-assembly of nano entities is a prevailing methodology for building colloidal crystals, where different shapes can build up several joint structures. The optical, electrical and chemical properties of these structures are mainly determined by their nature and arrangement of phases. The onset of these phenomena is also possible in biological problems such as micro-compartmentalization [1,2]. Similarly study of the interaction of various shaped nanoparticles with cell membranes is necessary in treatment and gene transport [3-6]. In view of importance of these structures, the phase behavior of rod-sphere mixture has been investigated theoretically [7-11] experimentally [12-18] and via simulations [19,20]. Despite these efforts, however, the complete behavior of such effects still needs to be thoroughly understood.
In droplet evaporation technique, convictive flow towards three phase contact line, induced by the faster evaporation at the edges than the center, happens [18]. This phenomenon not only causes transport of concentration of suspended nonentities, but also assists the spatial rearrangement of their deposits [18,21-24]. Droplet drying is a multifaceted process and, therefore, challenging to understand and control it [22]. Despite the non-equilibrium nature of the drying suspension, the consequential deposits have key role in printing [22], biological patterning [25,26] self-assembly [18,27] and related fabrications [28,29]. For rod-sphere mixtures, the conviction flow, aforementioned, somehow sets up shape-depended forces [18]. Such convictive forces and the related droplet dynamics may assist further the shape-separation effect.
Mutual interactions between large sized (˃ ~ 100 nm) particles are albeit fairly understood but one still needs to understand the formation of bigger superstructures for which the interaction between nano entities plays a key role. Such knowledge is helpful in knowing the nature of forces (electrostatic, van der Waals, steric, and entropic) at Nano scale [30]. In phase separation mechanism, besides these forces, entropy also plays a significant role [31-34]. In diluted systems, for instance, entropy can affect both attractive and repulsive interactions; whereas in dense systems, the same can induce driving force to facilitate order [30]. However, in case of surfactant coated nanoparticles it is believed [18] that the short range steric interaction is the most dominant agent to part different shapes from each other in suspension. As such, the interaction of surfactant molecules around the nanoparticles at their closest approach is recognized as steric interaction [30].
Ultrasonic vibrations act as a mechanical disturbance in the form of pressure waves through a medium. In case of liquid this disturbance generates energy which induces fluctuations that tend to enhance interactions among the stabilizing layers around nanoparticles in suspension. To understand the role of this interaction particularly in shape separation, it is important to study the suspensions’ deposits under fluctuations. Vibrating assisted deposition is thus important to unveil features that could possibly be used to understand shape separation phenomenon. This technique is also used in fabrication of transistors [35] and nanowires [36] and is crucial in medical diagnostic techniques and related applications [37-41]. Recently, it has also been used [42] in thin-film and photovoltaic solar cell devices. Steric interaction, therefore, can play an important role in shape-separation of various nanostructures.
In the present work, the effect of ultrasonic vibrations on the gold nanoparticles deposition particularly the rod-sphere mixture at various locations of the coffee-stain ring is investigated. The scenario is compared with the one without vibration (static). The phase-separation of rods and spheres are observed almost in a similar fashion at various positions of the coffee-stain ring as observed previously [18]. Under ultrasonic vibrations, we have observed even more prominent shape dependent separation of rod-sphere deposits at various places of the coffee-stain ring.
Experimental Techniques
Materials
Cetyltrimethyl Ammonium Bromide (CTAB, Sigma Aldrich, 98%), Hydrogen tetrachloroaurate (HAuCl43H2O, 99.999%, Aldrich), silver nitrate (AgNO3, 99%, Acros), ascorbic acid (AA, 99%, Merck), sodium borohydrate (NaBH4, 99%, Aldrich), hydrochloric acid (HCl, 37%, Merck), were all used as received without further purification. Water, used for synthesis, was of Milli-Q quality (18.2 cm), produced in a Simplicity 185 system (Millipore).
Synthesis and deposition
The 2-step seed mediated protocol explained by Nikoobakht and El-Sayed [43] was used to make gold nanorods. First CTAB coated seed particles were synthesized by mixing 25 μL of HAuCl4 (0.1M) with 10 mL of CTAB (0.1M). Then 60 μL of ice cold NaBH4 (0.1M) was added with uninterrupted stirring for 5 minutes. The resulting solution abruptly turned light brown displaying the creation of gold seeds. The solution was kept at room temperature for at least one hour to achieve saturated seed growth.
For the synthesis of Aspect Ratio (AR) 6 gold nanorods, the growth solution was made by mixing 50 μL of HAuCl4 (0.1M) with 10 mL of CTAB (0.1M). This solution was placed at 33°C for at least one hour to completely dissolve the CTAB with continuous stirring. Henceforth, while keeping the solution at room temperature, 20 μL of AgNO3 (0.1M) was introduced. Then 70 μL of ascorbic acid (0.1M) was added followed by 100 μL of HCl (1M). Finally, 25 μL of seed solution was introduced into the growth solution. The resulting solution was left uninterrupted overnight at room temperature.
On the next day, the nanoparticles suspensions were centrifuged at 15000 rpm for 10 minutes to eliminate the excess CTAB. Likewise, the similar growth solution was centrifuged again at 5600 rpm for 5 minutes to separate some spheres from nanorods. The supernatant comprising of mainly nanorods was separated carefully from the precipitate at the bottom of the centrifuge tube; which contained mostly spheres. The nano rod suspensions were stored in refrigerator.
For drop casting, SiO2 substrates were cleaned in ultrasonic bath in distilled water for 10 minutes. The substrates were rinsed with distilled water and dried in nitrogen flow. Droplets (5 μL) for each case were placed on the clean SiO2 substrate. For static drying the droplet was placed on substrate and allowed to dry at room temperature. The solvent was completely evaporated within two hours leaving a 'coffee-stain'-like ring on the substrate. In the second experiment, the substrate was first placed in petri dish and a droplet of nanoparticles suspension was deposited on it. The droplet gradually dried under ultrasonic agitated condition at a frequency of 60 kHz. This ultrasonic agitation was induced in a HBM 2.5 L Ultrasonic Cleaner filled with suspension was deposited on SiO2 substrate and evaporated during water.
Characterization techniques
Ultraviolet-visible (UV-Vis) spectroscopy technique was used to determine optical absorbance of ultra-violet and visible light on passage through a material. All measurements were carried out using Ocean Optics HR 2000+ spectrometer operated using the Spectra-Suite software package combined with a Mikropack UV- VIS light source (model DH-2000-BAL). Since water with suspended nanoparticles was used, standard semi-micro UV cuvettes with dimensions 12.5 mm×12.5 mm×45 mm for the optical analysis was utilized; the distance through the liquid was 10 mm. The capacity of cuvettes used was 1.5 mL enabling analysis of smaller volumes of suspensions.
Imaging of deposits were performed using high resolution Scanning Electron Microscope (SEM; on a Merlin Zeiss 1550 system, for our samples. Typical voltages were in the range 0.1-30 kV to accelerate and electromagnetically focus electrons onto the sample. Part of electron beam back scattered off sample to produce secondary electrons. The relevant detector converted electrical signal from these electrons into an image. Images were taken at accelerating voltage of 2 kV.
Results
On hydrophilic surface, the droplet of nanoparticles suspension left behind ring like deposits, coffee-stain effect [18,44]. Initial deposits of rod-sphere mixture at the outer edge of the coffee stain ring are shown Figure 1. Due to the low concentration of nanoparticles close to the contact line, deposits looked more systematic and almost oriented in the same direction. Figures 2 (A) and 2 (C) display initial deposits resulted from static drying at the outer edge of the coffee stain ring. Deposits in this case seemed largely directed by convictive flow towards the contact line. Long side-by-side ribbons of gold nanorods were separated by mostly spheres and some rods. This effect is evident from 3-4 micron area of coffee stain ring. Figures 2 (B) and 2 (D) show similar deposits but this time induced/assisted by ultrasonic vibrations. Broken short ribbons of nanorods were observed arranged side-by-side and oriented in all possible directions. No substantial existence of nanospheres was observed within inter-ribbon space.
Figure 2: SEM images gold nano rod-sphere mixture deposits obtained from the drying of droplets. Outer edge of the coffee-stain ring results from the static drying (A,B) and from ultrasonic vibrations (C,D).
The calculated value of rods and spheres concentration is 1.8×1011/cm3 and 5.3×1010/cm3 respectively.
The portion of a whole contact line with deposits at initial, intermediate, and vanishing evaporative droplet locations is displayed in detail for both static and vibrated systems, Figures 3 and 4. A patch of a coffee-stain ring for static droplet along with some magnified portions is shown in Figure 3 (A). The outer region of the coffee-stain ring enclosed by red line, Figure 3 (B), displays stripes of alternating aligned rods separated by spheres. The yellow rectangle, Figure 3 (C), is the middle region showing dense superstructures breaking the stripes-like structures. The phase separation of rods and spheres, however, still remains intact. In the inner region, dodger blue rectangle, Figure 3 (D), massive three-dimensional columnar phases of rods and hexagonal close packed spheres can be clearly observed. This result is easily reproducible and is similar to the one reported previously [18] with the sole difference of deposit structures in the inner region of the ring. Deposits in the present case are multilayered and very dense. This might be due to the higher density (24% higher than the one reported earlier [18] of nanoparticles in the solution of the present work.
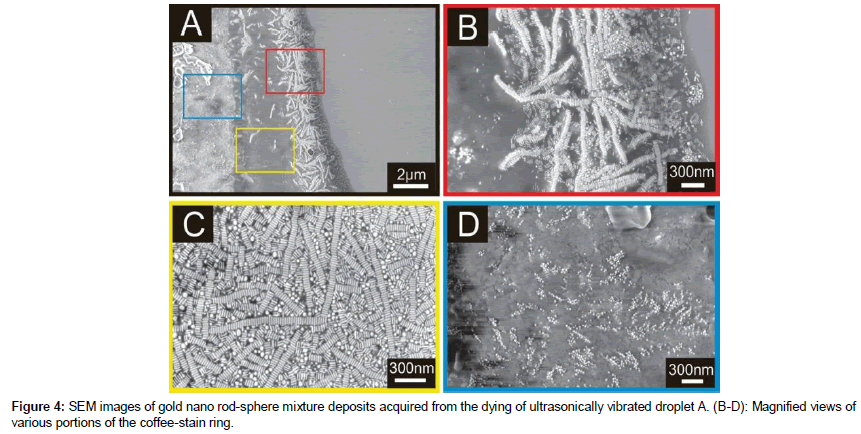
Interesting results were acquired when similar gold nanoparticles suspension was deposited on SiO2 substrate and evaporated during ultrasonic vibrations. Figure 4 (A) shows a section of the coffee-stain ring like deposits. Ribbon-like arrays of nanorods, bright regions, assembled side-by-side nanorods are clearly visible at the periphery of the outer portion, Figure 4 (B), red rectangle. N.B these ribbons are shorter in length than the ones formed statically, Figure 3 (B).
Comparing the two deposits (static, Figure 3, and ultrasonic vibrating, Figure 4), following important conclusions may readily be drawn:
Ribbons in Figure 3 (B) are longer than the ones in Figure 4 (B).
Rod-sphere stripes are present at the outer edge, Figure 3 (B), contrary to the ultrasonic assisted deposits, Figure 4 (B).
There are more spheres at the outer edge in static case, Figure 3 (B), than in the ultrasonic assisted case, Figure 4 (B).
In addition, we observed rather close packed ribbons with some spheres in between the arrays, Figure 4 (C). No preferential orientation of the ribbons was observed. Interestingly, at the inner end of the coffee-stain ring were found large clusters of spheres, Figure 4 (D).
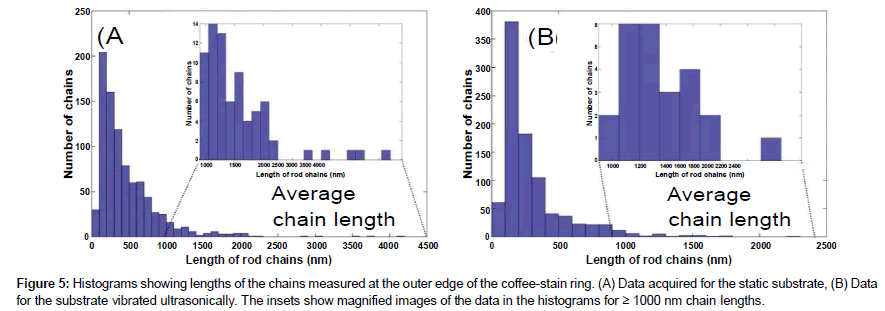
The lengths of the chains of gold nanorods were measured for solutions of rod-sphere mixture by analyzing their SEM images. Figures 5 (A) and 5 (B) show the data acquired for the static and ultrasonically vibrated substrate respectively. This is evident from the figures that the average static chain length (473 nm) is longer than average vibrating length (302 nm) supporting our previous observations, Figure 4 (A) and 4 (B). Furthermore, the insets show a larger image of the data in the histograms for chain lengths ≥ 1000 nm, clearly showing that the maximum length of the chains for the case of no ultrasonic agitation is much larger than the maximum length of the chains otherwise, consistent with earlier observations.
Discussion
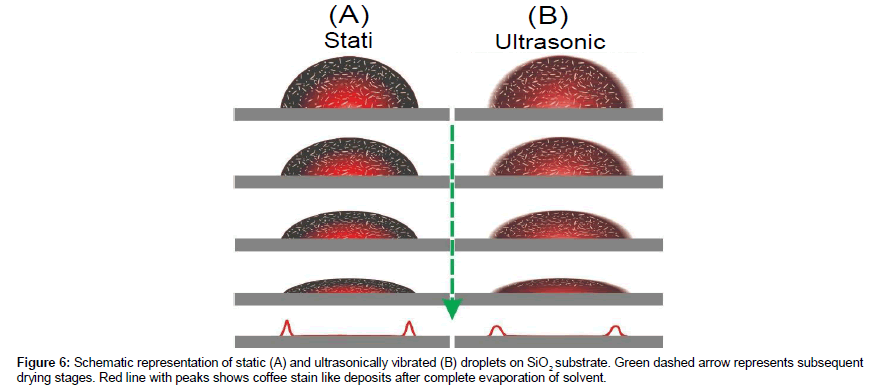
Droplets of gold rod-sphere mixture were dried under static and ultrasonically vibrating conditions. The drying mechanism is schematically shown in Figure 6. In dried deposits, phase separation of rod-sphere mixture was repeatedly observed in all samples. The suspension used in this work is a bit more concentrated than one reported previously [18].
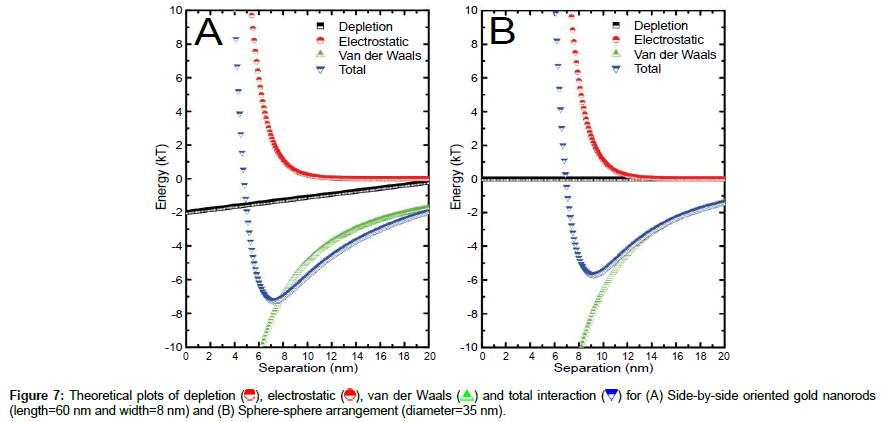
In dense suspension, it is difficult to imagine so systematic self- separated phases. We believe that mere convictive flow can induce such effects between different shapes. Using theoretical models of nano scale forces [18], it has been shown that the energetically favorable (lowest energy~7.2 kT) position for nanorods in suspension is the side-by-side aligned formation as depicted in Figure 7 (A) for nanorods of length (60 nm) and width (8 nm). Owing to such strong attractive interaction in suspension, nanorods are found everywhere in the form of closed packed ribbons as revealed in all SEM images. Likewise, the lowest energy (~5.7 kT) position for a sphere is possible only with the identically curved surface in the vicinity of that sphere as shown in Figure 7 (B). In this case, the average diameter of sphere like particles is 35 nm.
It is even more fascinating to observe such shape dependent deposits in stripes, Figure 3 (B). Formation of these stripes in the absence of external forces/vibrations indicated that the flow rate was induced due to the convictive gradient. Moreover, the current towards the three phase contact line did not influence the flouting rate of various shapes suspended in water. If it was not so, we would not be able to see alternating stripes of rods and spheres so orderly around the coffee-stain deposits. For that reason one expects such superstructures of either shape approaching the contact line probably at the similar rate, and eventually assembling and settling on the surface. A thorough examination of various micro/nanographs revealed that first pinning particles are always nanorods. This, owing to the long length of nanorods, might be due to the maximum pinning length to hold the contact line. The lowest energy arrangement for nanorods is to orient themselves parallel to the contact line.
An important aspect considered for the shape separation effect is the role of self-crafted surfactant molecules at the nanoparticles surface. Such arrangements of stabilizing CTAB layers around the gold nano rod and sphere like structure plays decisive role as soon as these nanoparticles approaches close to each other in suspension. It is well-known that on the side facets the CTAB molecules are much more closely placed compared to their outer ends, owing to the crystallographic orientation [45]. In fact, this is assumed to effectively guide the anisotropic growth of the nanorods during their synthesis [46]. As a result, when radially arranged surfactant molecules around the nanospheres interact sterically with the densely covered surfactant molecules at the flat surface of the nanorods. Such arrangement of sphere and rod is less probablesterically [18]. Furthermore, in a rod- sphere mixture mechanical disturbance can assist such interaction repeatedly, which allows to push even those spheres that are trapped deeply among the dense population of nanorods.
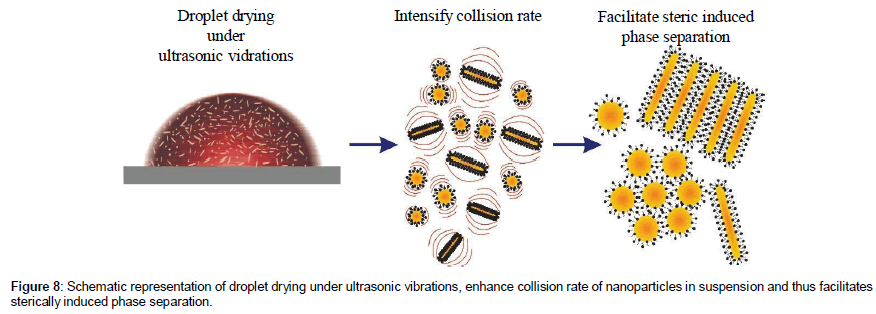
For the ultrasonically agitated droplets, interesting phenomena may arise from propagation of waves in the liquid medium. Key importance can be given to the process known as acoustic streaming. Such streaming waves originate from the dissipation of acoustic energy which produces gradients in momentum that cause fluid currents45. Ultrasonic vibrations enhance the effect of sterically induced phase separation due to the collision of dissimilar arrangement of the stabilizing layers around nanoparticles [18]. To be brief, steric interaction is strongest in the presence of ultrasonic vibrations. The dictating phase separation effect is observed in the present work, demonstrating pronounced steric interactions especially in the case vibrating droplet. The wave current prompted by the ultrasonic vibration will also, in addition, provide enough energy to break the ribbons like superstructure of nano rods formed in suspension.
It has been reported recently [47] that ultrasonic vibrations enhance the effect of crystallization in pharmaceutical constituents. The vibrations induced by ultrasonic agitation will generate fluctuations which increases collision frequency of nanoparticles within the droplet. As a result, the stabilized layers of surfactant molecules interact more efficiently. Consequently, spheres push out from the areas where rods are present in high concentration. The whole mechanism described above is illustrated in Figure 8.
Conclusion
Rod-sphere mixture of gold nano rods is investigated under ultrasonic and static conditions upon complete evaporation. The static self-assembled phase separation at the outer, middle, and inner regions of the coffee-stain like deposits agrees well with the previous work. In case of ultrasonic vibrations, the effect of steric interaction of adsorbed surfactant molecules at various facets of nano-crystals (stabilizing layers around nanoparticles) will stimulate phase separation effect with the adjacent particles. These observations are important and significant to assess the claim reported elsewhere18, where it is emphasize that steric interaction induces phase separation of nanoparticles. In this work, we observed that ultrasonic vibration allows nanoparticles to interact repeatedly in suspension during ultrasonic fluctuations, consequently induces distinct shape separation effect. These observations can be helpful in nano/micro shape-separation as well as in many other relevant prospective applications.
References
- Walter H, Brooks DE (1995) Phase separation in cytoplasm, due to macromolecular crowding, is the basis for microcompartmentation. FEBS Letters 361: 135-139.
- Welch GR, Clegg JS (Eds.) (1986) The Organization of Cell Metabolism, Plenum Press, New York.
- Xia Y (2008) Nanomaterials at work in biomedical research. Nature Mater 7: 758–760.
- Service RF (2005) Nanotechnology Takes Aim at Cancer. Science 310: 1132–1134.
- Andre EN, Lutz M, Darrell V, Tian X, Eric MVH (2009) PonisserilSomasundaran, Fred Klaessig, Vince Castranova & Mike Thompson, Understanding biophysicochemical interactions at the nano–bio interface. Nature Mater 8: 543–557.
- Mitragotri S, Lahann G (2009) Physical approaches to biomaterial design. Nature Mater 8: 15–23.
- Onsager L (1949) The effects of shape on the interaction of colloidal particles. Ann NY Acad Sci 51: 627–659.
- Rosenfeld Y (1994) Phase Separation of Asymmetric Binary Hard-Sphere Fluids: Self-Consistent Density Functional Theory. Phys Rev Lett 72: 3831.
- Poon WCK , Warren PB (1994) Phase Behaviour of Hard-Sphere Mixtures. Europhys Lett 28: 513.
- Hosino M, Nakano H, Kimura H N (1979) Ematic-Smectic Transition in an Aligned Rod System. J Phys Soc Jpn 46: 1709–1715.
- Schmidt M (2001) Density functional theory for colloidal rod-sphere mixtures. Phys Rev E 63: 050201
- N (1979) Ematic
- Smectic Transition in an Aligned Rod System
- Density functional theory for colloidal rod-sphere mixtures
- Wenyan L, Nathan AM, Oleg G, Athanassios Z P, Sanat KK, et al. (2017) Directionally Interacting Spheres and Rods Form Ordered Phases. ACS Nano 5: 4950–4959.
- Tang J , Fraden S (1996) Nonmonotonic temperature dependence of the flexibility of bacteriophage fd . Biopolymers 39: 13–22.
- Sato T, Teramoto A (1996) Concentrated solutions of liquid-crystalline polymers. Adv Polym Sci 126: 85–161.
- Kaplan PD, Rouke JL, Yodh AG, Pine DJ (1994) Entropically Driven Surface Phase Separation in Binary Colloidal Mixtures. Phys Rev Lett 72 582-585.
- Dinsmore AD, Yodh AG, Pine D (1995) Phase diagrams of nearly-hard-sphere binary colloids. J Phys Rev E 52: 40-45.
- Dogic Z, Fraden S (1997) Smectic phase in a colloidal suspension of semiflexible virus particles. Phys Rev Lett 78: 2417–2420.
- Concentrated solutions of liquid
- crystalline polymers
- Ahmad HJW, Zandvliet ES (2014) Kooij,Shape-Induced Separation of Nanospheres and Aligned Nanorods. Langmuir 30: 7953.
- Yang K, Ma YQ (2010) Computer simulation of the translocation of nanoparticles with different shapes across a lipid bilayer. Nat Nanotechnol 5: 579–583.
- Umi Y, Kenneth SS (2013) Theory of Entanglements and Tube Confinement in Rod–Sphere Nanocomposites. ACS Macro Lett 2: 955–959.
- Imtiaz A, Floor D, Pantelis B, Tim B, Harold JW, et al. (2017) Step-edge Induced Orientation of Nanorods in Evaporative Self-assembly onHOPG.Colloid and Interface Science Communications 19: 25–30.
- Terry PB, Xiao ML, Toan TN, Eric IC, Thomas A (2006) Witten & Heinrich M. Jaeger. Kinetically driven self-assembly of highly ordered nanoparticle monolayers. Nature Mater 5: 265–270.
- Yunker PJ, Still T, Lohr MA, Yodh AG (2011) Suppression of the coffee-ring effect by shape-dependent capillary interactions. Nature 476: 308–311.
- Still T, Yunker PJ ,Yodh AG (2012) Surfactant-induced Marangoni eddies alter the coffee-rings of evaporating colloidal drops. Langmuir 28: 4984–4988.
- Wu L (2015) Printing Patterned Fine 3D Structures by Manipulating the Three Phase Contact Line. Adv Funct Mater 25: 2237–2242.
- Zhang D, Jiang Y, Xiaohui W, Linxi Z (2013)Phase separation and crystallization of binary nanoparticles induced by polymer brushes. Soft Matter 9: 1789-1797.
- MacBeath G , Schreiber SL (2000) Printing proteins as microarrays for high-throughput function determination. Science 289: 1760–1763.
- Sempels W, Dier R, Mizuno H, Hofkens J, Vermant J (2013) Auto-production of biosurfactants reverses the coffee ring effect in a bacterial system. Nature Commun. 4: 1757.
- Zhiliang Z, Xingye Z , Zhiqing X, Mengmeng D , Yongqiang W, et al. (2013) Controlled inkjetting of a conductive pattern of silver nanoparticles based on the coffee-ring effect. Adv Mater 25: 6714–6718.
- Shimoni A, Azoubel S, Magdassi S (2014) Inkjet printing of flexible high-performance carbon nanotube transparent conductive films by “coffee ring effect”. Nanoscale 6: 11084-11089.
- Kyle JMB, Christopher E ,Wilmer SS, Bartosz AG (2009) Nanoscale forces and their uses in self-assembly. Small 14: 1600-1630.
- Adams M, Dogic Z, Keller LS, Fraden S (1998) Entropically driven microphase transitions in mixtures of colloidal rods and spheres. Nature 393: 349-352.
- Bushong S (1999) Diagnostic Ultrasound. McGraw-Hill, New York, USA.
- Asakura S, Oosawa F (1958) Interaction between Particles Suspended in Solutions of Macromolecules. J Polym Sci 33:183-192.
- Vliegenthart GA (1999) Phase behavior of colloidal rod-sphere mixtures. J Chem Phy 111: 4153-4157.
- Diemer PJ, Lyle CR, Mei Y, Sutton C, Payne MM, et al. (2013) Vibration-assisted crystallization improves organic/dielectric interface in organic thin-film transistors. Adv Mater 25:6956-6962
- Kim BG, Kim MS, Kim J (2010) Ultrasonic-assisted nanodimensional self-assembly of poly-3-hexylthiophene for organic photovoltaic cells. ACS Nan 4:2160-2116.
- Gallego-Ju´arez JA, Corral RG, Moraleda JCG, Yang TS (1999) A new high-intensity ultrasonic technology for food dehydration. Drying Technol 17: 597-608.
- Garc´Ã„±a-P´erez JV, C´arcel JA, Riera E, Mulet A (2009) Influence of the applied acoustic energy on the drying of carrots and lemon peel. Drying Technol 27: 281–287.
- Zhen Y, Sohei M, Goto H, Matsumotob M, Ryutaro M (2001) Ultrasonic micromixer for microfluidic systems. Sensors and Actuators A: Physical 93: 266–272.
- Eslamian M, Zabihi F (2015) Ultrasonic Substrate Vibration-Assisted Drop Casting (SVADC) for the Fabrication of Photovoltaic Solar Cell Arrays and Thin-Film Devices. Nano Res Letters 10:462
- Bjorn G, Jeroen J, Thomassen LCJ, Braeken L, Gerven TV (2017) Agglomeration Control during Ultrasonic Crystallization of an Active Pharmaceutical Ingredient. Crystals 7: 1-20.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 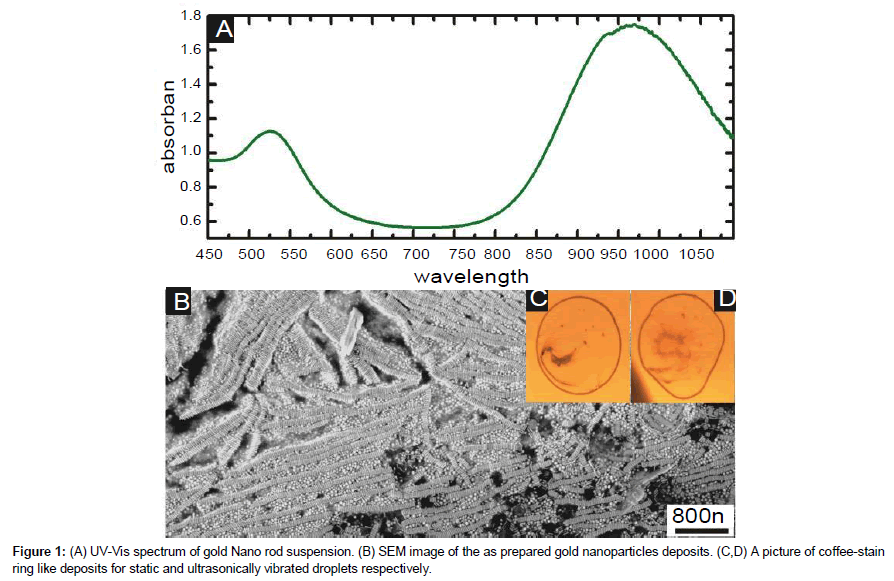


 , electrostatic
, electrostatic  , van der Waals
, van der Waals  and total interaction
and total interaction  for (A) Side-by-side oriented gold nanorods (length=60 nm and width=8 nm) and (B) Sphere-sphere arrangement (diameter=35 nm).
for (A) Side-by-side oriented gold nanorods (length=60 nm and width=8 nm) and (B) Sphere-sphere arrangement (diameter=35 nm).