Research Article, J Med Toxicol Res Vol: 1 Issue: 1
Effects of Differently Processed Rehmannia Roots on Dexamethasone-induced Side Effects in Rats
Zhang FF1, Wen XS1*, Lu YF1, Luo SF1, Lv C1 and Xian CJ2
1Institute of Pharmacognosy, School of Pharmaceutical Sciences, Shandong University, Jinan 250012, China
2Sansom Institute for Health Research, School of Pharmacy and Medical Sciences, University of South Australia, Adelaide, SA 5001, Australia
*Corresponding Author : Xue-sen Wen
Institute of Pharmacognosy, School of Pharmaceutical Sciences, Shandong University, Jinan 250012, China
Tel: (+86) 0531 88382028
E-mail: x.s.wen@163.com
Received: January 11, 2018 Accepted: January 22, 2018 Published: January 29, 2018
Citation: Zhang FF, Wen XS, Lu YF, Luo SF, Lv C, et al. (2018) Effects of Differently Processed Rehmannia Roots on Dexamethasone-induced Side Effects in Rats. J Med Toxicol Res 1:1.
Abstract
Background: There are three differently processed rehmannia roots, namely fresh, dried and steamed rehmannia roots (FRR, DRR and SRR), which have different indications according to the theory of traditional Chinese medicine (TCM).
Objective: To confirm the effects of different rehmannia roots on dexamethasone (Dex) treatment induced side effects, which belong to Yin Deficiency Syndrome in TCM.
Materials and Methods: Rats were subcutaneously injected with Dex (0.1m g/kg) and orally administrated with different roots (2.16 g/kg) for 28 days, with metformin (Met, 0.25 g/kg) as a reference substance. Food intake, blood cell count, and organ weight were recorded, and hepatic glycogen and serum insulin, glucose, free fatty acids, total cholesterol, and malondialdehyde contents were
assayed.
Results: Dex treatment led to decreases in food intake, white blood cell count, and spleen index, and increases in hemocrit, heart and liver indexes, hepatic glycogen, and serum contents of insulin, glucose, free fatty acids, total cholesterol, and malondialdehyde. The alterations in food intake, spleen index, hematocrit and serum insulin were significantly relieved by FRR but not DRR and SRR; and on the contrary, heart index was significantly reduced by DRR and SRR but not FRR.
Conclusions: Dex treatment results in insulin resistance, high blood viscosity, oxidative damage and immunosuppression in rats, and FRR is better than DRR and SRR in attenuating Dex-induced insulin resistance and high blood viscosity. These findings have thus firstly provided evidence that postharvest processing has a profound influence on the biological activities of Rehmannia roots.
Keywords: Dexamethasone; Insulin resistance; Metformin; Postharvest processing; Rehmannia glutinosa; Side effect
Introduction
Traditional Chinese medicinal materials must be processed before being prescribed for patients. The processing technologies include drying, cutting, roasting, steaming, calcining, fermenting and so on, which not only facilitate storing, making up a prescription and enabling better solubility of active components, but also reduce side effects, strengthen or modify actions. Most of the claimed advantages of the traditional processing methods have not been systemically examined up to now, such as processing methods for the roots of Rehmannia glutinosa (Gaertn.) DC. (The family of Plantaginaceae).
In China, there are three differently processed rehmannia roots, namely fresh, dried and steamed rehmannia roots (FRR, DRR and SRR). According to the traditional practices, FRR is collected as the aerial parts die down, and then dried at a temperature of 70-80ºC and used as DRR. SRR is derived from DRR by steaming for several days. In TCM theory, FRR and DRR have a cold property, being used to clear away pathogenic heat, while SRR is slightly warm and has a tonic effect. Previously, we have verified that significant chemical changes occurred during the postharvest drying and steaming. For example, stachyose and catalpol, the two main components of FRR, were found largely decomposed, while 5-hydromethyl furfural, one of the intermediate products of Maillard reaction, was generated [1,2]. However, the relationship between these chemical changes and their different traditional applications remains unknown.
Dexamethasone (Dex) is a synthetic glucocorticoid with potent anti-inflammatory and immunosuppressive actions, which is commonly used to treat rheumatic problems, allergic disorders, ulcerative colitis, psoriasis, and cancer etc. [3,4]. Unfortunately, a wide spectrum of side effects of Dex usage has been observed, such as anxiety, agitation, palpitation, insulin resistance, high blood viscosity, dislipidemia, cardiotoxicity, oxidative stress and bone defects [5-17]. In the view of traditional Chinese medicine (TCM), most of Dex’s side effects belong to the Syndrome of Yin Deficiency, and an animal model of Yin Deficiency has been established by Dex treatment [18- 20]. Both of DRR and SRR have the action to nourish Yin and have been clinically used to suppress the side effects of glucocorticoids [21]. In the meantime, DRR has been proved having the capacity to avoid pituitary and adrenal gland atrophy induced by Dex [22-23], and Liuwei Dihuang Wan, a well-known traditional Chinese formula with SRR as the main ingredient, could improve Dex-induced side effects [18-20]. According to the theory of TCM, Yin deficiency will lead to internal heat, and accordingly, we have proposed that FRR or DRR should be better than SRR in attenuating Dex-induced pathogenic heat. In this study, therefore, the effects of three rehmannia roots on Dex-induced side effects were compared to confirm whether postharvest processing has an influence on the biological activities of these roots. Due to the lack of satisfactory drugs available for the treatment of all side effects of Dex, metformin (Met) was selected as a reference substance in this study as it was shown to relieve some of Dex’s side effects [16,17].
Materials and Methods
FRR, DRR or SRR and their extraction
Fresh tuberous roots of Rehmannia glutinosa ‘Beijing 3’ was obtained from Shandong Baiweitang TCM Co., Ltd. (Jinan, China) in December 2016. Some roots were cut into slices (3mm thick) and dried at 30ºC; the resulting slices were used as FRR. The other roots were hot-air dried at 80ºC and used as DRR. One half of DRR was further steamed for 48h at atmospheric pressure and then dried at 80ºC, which was used as SRR. The slices of FRR were extracted 5 times with boiling water for 40 min each, and the combined extracts were concentrated under vacuum at 80°C to a concentration corresponding to 0.432 g crude drug per milliliter. DRR and SRR were steamed for 1 min and cut into slices (about 2 mm thick), and then extracted as for FRR above. All extracts were diluted to 0.216g/ml when used. The samples of FRR, DRR and SRR (with reference No. 20161129, 21061205 and 20161220, respectively, as shown in Figure S1) were deposited at Institute of Pharmacognosy, Shandong University, Jinan, China, and the typical chromatograms of their RP-HPLC analyses for catalpol and 5-hydromethyl furfural (5-HMF) were shown in Figure S2.
Animals
Forty eight adult male Wistar rats weighted 300 ± 10g were obtained from Jinan Pengyue Experimental Animal Breeding Co., Ltd., (Jinan, China). These rats were housed in a room with a 12h-light-dark cycle under a controlled temperature (21 ± 2°C) and a relative humidity (50 ± 10%), and had free access to normal rodent pellet food and tap water. Animal care and experimentation strictly complied with the guidelines of the Ministry of Science and Technology of the People’s Republic of China (No. 398).
Experimental design, treatment and sampling
After being acclimatized to lab environment for one week, the rats were randomly assigned into six groups (control, Dex, FRR, DRR, SRR and Met) (n = 8 rats/group). The rats received subcutaneous injection of Dex (0.1 mg/kg body weight/day, Shandong Xinhua Pharmaceutical Co., Ltd., Zibo, China) in saline once daily at 6:00 p.m. for 28 consecutive days except for those in control group, which received equal volume of saline [9,15,24]. Rats of the FRR, DRR and SRR groups were administered with 10ml extract/kg (equivalent to 2.16g crude drug/kg) by gavage once daily at 8 a.m., and those of the Met group were given Met (0.25 g/kg). Rats in the control and Dex groups received the same volume of water gavage. During the experiment, the animals were weighed daily, and the dosage was adjusted according to body weight changes. On day 14 and 21, food intake was recorded individually.
After an overnight fasting, the rats were anesthetized by 3.5 ml/ kg of 10% chloral hydrate 1h after oral gavage on day 29. Lead II electrocardiogram was recorded for 90s using the BL-420S biological data acquisition and analysis system (Chengdu Techman Software Co., Ltd., Chengdu, China), and then venous blood was collected from the inferior vena cava. One milliliter of the blood was anticoagulated with EDTA for complete blood counting, and the remnant clotted blood was centrifuged (4°C, 3500 rpm/min for 10 min) to collect serum. Spleen, heart and liver were removed and weighed immediately.
Serum biochemical parameters and liver glycogen contents
Fasting serum contents of glucose, total cholesterol, free fatty acids, creatine kinase isoenzyme MB, total protein, and liver glycogen were determined using commercial kits (Nanjing Jiancheng Biomedical Engineering Co., Ltd., Nanjing, China). Serum insulin was measured by using the 125I-radioimmunoassay kit (Beijing Northern Bioengineering Institute, Beijing, China). Serum malondialdehyde (MDA, a product of lipid peroxidation) was determined by the method of Ohkawa et al. [25].
Statistical analyses
The data were expressed as mean ± SEM. The significance of difference between groups was determined by Student’s t-test at three levels, p<0.05, p<0.01 and p<0.001.
Results
Body and organ weight, and food intake
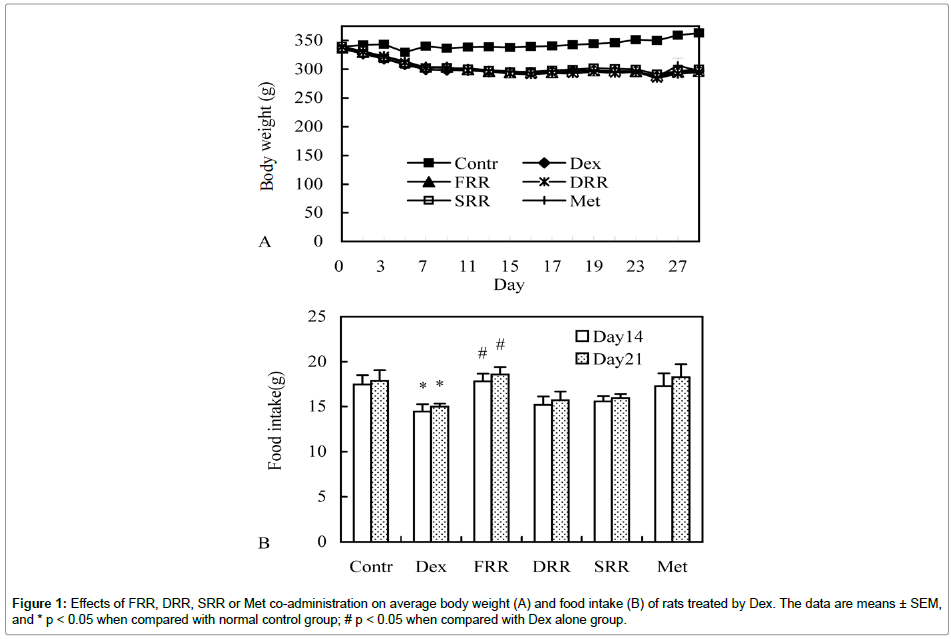
To examine the differences of fresh, dried and steamed rehmannia roots, their effects in countering Dex treatment-induced side effects were compared. In Dex alone-treated rats, significant weight loss was observed from day 2 onwards, which could not be attenuated by co-administration with rehmannia roots and metformin (Figure 1A). On day 14 and day 21, food intake was found decreased in Dex alone group, but nearly returned to normal level by supplement of FRR, while the effects of the other tested materials did not reach a statistical significance (Figure 1B). Heart and liver indexes in Dex alone-treated rats were significantly higher than those in the control group, while spleen index was just the opposite. The increased heart index was significantly reduced by DRR and SRR supplementation, and the reduced spleen index was significantly increased by FRR administration (Table 1).
| Group | Control | Dex | FRR | DRR | SRR | Met |
|---|---|---|---|---|---|---|
| Heart | 0.24 ± 0.00 | 0.30 ± 0.00 *** | 0.29 ± 0.01 | 0.28 ± 0.00 ## | 0.28 ± 0.01 ## | 0.30 ± 0.01 |
| Liver | 2.32 ± 0.03 | 2.91 ± 0.06 *** | 3.04 ± 0.09 | 2.96 ± 0.09 | 2.97 ± 0.13 | 3.21 ± 0.08 # |
| Spleen | 0.14 ± 0.01 | 0.09 ± 0.00 *** | 0.10 ± 0.00 # | 0.09 ± 0.01 | 0.08 ± 0.00 | 0.09 ± 0.00 |
Table 1: Effects of FRR, DRR, SRR or Met co-administration on organ index (g/100g body weight) a.
Complete blood cell counting
It is well known that Dex is a potent immunosuppressive agent, and it also enhances erythropoiesis [26-27], leading to polycythemia and high blood viscosity. The results of complete blood cell counting shown in Table 2 are as expected, with the total number of white blood cells being significantly decreased and the number of red blood cells being significantly increased. All the parameters in the Dex alone group were significantly decreased when compared with those of the control group except for the neutrophil count (with the difference being insignificant) and the basophil count (being significantly increased). The red blood cell count, hemoglobin content and hematocrit nearly returned to the normal levels in the FRRsupplemented group, while most of the parameters were not obviously improved by co-administration of DRR, SRR or Met (Table 2).
| Groups | Control | Dex | FRR | DRR | SRR | Met |
|---|---|---|---|---|---|---|
| White blood cells (1x109/L) | 6.14 ± 0.45 | 2.27 ± 0.22 *** | 2.57 ± 0.14 | 2.18 ± 0.27 | 2.35 ± 0.33 | 2.24 ± 0.15 |
| Neutrophil (1x109/L) | 0.34 ± 0.07 | 0.17 ± 0.04 | 0.14 ± 0.02 | 0.14 ± 0.04 | 0.17 ± 0.04 | 0.14 ± 0.04 |
| Lymphocytes (1x109/L) | 4.70 ± 0.36 | 1.12 ± 0.20 *** | 1.32 ± 0.14 | 1.06 ± 0.15 | 1.17 ± 0.20 | 1.09 ± 0.06 |
| Monocytes (1x109/L) | 0.40 ± 0.05 | 0.19 ± 0.04 ** | 0.21 ± 0.02 | 0.18 ± 0.04 | 0.19 ± 0.06 | 0.24 ± 0.06 |
| Eosinophil (1x109/L) | 0.09 ± 0.01 | 0.01 ± 0.00 *** | 0.01 ± 0.00 | 0.01 ± 0.00 | 0.02 ± 0.01 | 0.01 ± 0.00 |
| Basophil (1x109/L) | 0.48 ± 0.05 | 0.74 ± 0.03 *** | 0.88 ± 0.07 | 0.79 ± 0.13 | 0.80 ± 0.08 | 0.76 ± 0.09 |
| Red blood cells (1x109/L) | 8.01 ± 0.23 | 8.66 ± 0.19 * | 7.76 ± 0.16 ## | 8.18 ± 0.16 | 8.38 ± 0.19 | 8.44 ± 0.17 |
| Hematocrit (%) | 40.66 ± 0.85 | 45.20 ± 1.30 ** | 41.06 ± 0.69 ## | 42.41 ± 0.80 | 44.00 ± 0.84 | 43.48 ± 0.94 |
| Hemoglobin (g/L) | 144.13 ± 3.12 | 157.14 ± 4.45 * | 145.00 ± 2.41 # | 151.88 ± 3.03 | 156.75 ± 3.26 | 155.13 ± 2.94 |
| Platelets (1x1012/L) | 831.13 ± 17.52 | 734.14 ± 14.31 * | 730.38 ± 27.94 | 656.7 ± 32.60 | 669.3 ± 32.80 | 676.25 ± 33.83 |
| Platelet-large cell ratio (%) | 16.43 ± 0.99 | 12.97 ± 0.77 * | 12.28 ± 0.40 | 12.13 ± 0.73 | 13.76 ± 1.29 | 13.36 ± 0.80 |
| Thrombocytocrit (%) | 0.74 ± 0.01 | 0.61 ± 0.03 ** | 0.60 ± 0.02 | 0.54 ± 0.03 | 0.56 ± 0.03 | 0.57 ± 0.03 |
| Mean platelet volume (fL) | 8.84 ± 0.11 | 8.37 ± 0.11 ** | 8.29 ± 0.04 | 8.20 ± 0.11 | 8.44 ± 0.16 | 8.39 ± 0.11 |
Table 2: Effects of FRR, DRR, SRR or Met co-administration on complete blood cell counting of rats treated with Dex a.
Serum biochemical parameters and liver glycogen content
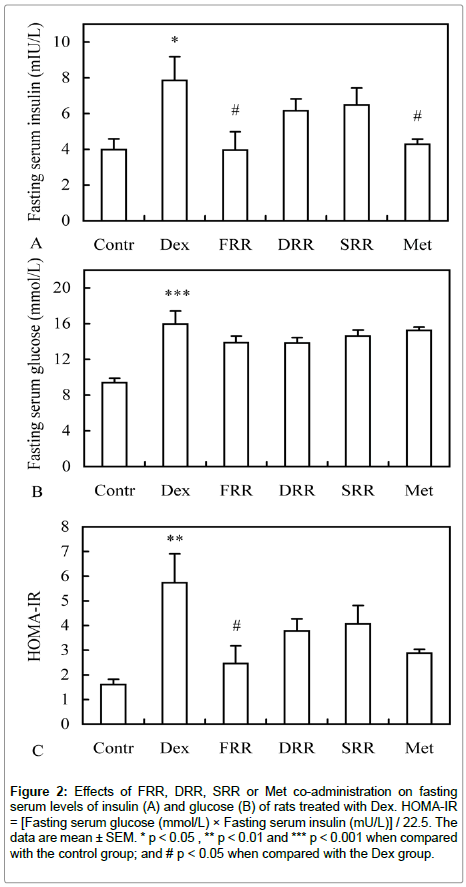
To understand the effects of different rehmania roots on abnormal energy metabolism, heart toxicity and oxidative damage induced by Dex treatment, serum biochemical parameters were measured. In Dex alone- treated rats, fasting serum glucose, insulin, free fatty acid contents were found significantly elevated comparing with the untreated control rats, and all three rehmannia roots and Met supplementation can reduce these parameters. However, only insulin concentration was reduced to a statistically significant level (p<0.05) by both FRR and Met supplementation (Figure 2A and 2B, Table 3). According to the derived parameter, HOMA-IR (homeostatic model assessment for insulin resistance), insulin resistance were induced by Dex treatment and a significant reduction was observed only in the FRR supplemented group (Figure 2C). In addition, liver glycogen content was also found significantly increased by Dex treatment, which was reduced by FRR, but further increased by DRR, SRR and Met co-treatments, although the differences did not reach a significant level (Table 3). Similarly, serum total cholesterol (TC), total protein, urea nitrogen, and malondialdehyde also significantly increased, while creatine kinase isoenzyme MB significantly decreased by Dex treatment. However, none of the tested materials could restore these alterations except for serum protein in the SRR supplemented group (Table 3).
Figure 2: Effects of FRR, DRR, SRR or Met co-administration on fasting serum levels of insulin (A) and glucose (B) of rats treated with Dex. HOMA-IR = [Fasting serum glucose (mmol/L) × Fasting serum insulin (mU/L)] / 22.5. The data are mean ± SEM. * p < 0.05 , ** p < 0.01 and *** p < 0.001 when compared with the control group; and # p < 0.05 when compared with the Dex group.
|  Groups | Control | Dex | FRR | DRR | SRR | Met |
|---|---|---|---|---|---|---|
| TC (mmol/L) | 2.26 ± 0.13 | 2.90 ± 0.21 * | 2.80 ± 0.13 | 2.95 ± 0.30 | 2.77 ± 0.21 | 3.03 ± 0.20 |
| MDA (nmol/ml) | 5.47 ± 0.59 | 10.26 ± 0.78 *** | 9.57 ± 0.54 | 9.28 ± 0.44 | 8.65 ± 0.45 | 9.67 ± 0.67 |
| CK-MB (U/L) | 355.10 ± 55.54 | 219.75 ± 16.96 * | 170.45 ± 19.19 | 179.14 ± 14.46 | 179.14 ± 21.17 | 250.80 ± 18.30 |
| Total protein (mg/ml) | 52.03 ± 2.21 | 66.06 ± 1.60 *** | 64.71 ± 2.12 | 66.55 ± 1.83 | 59.66 ± 2.48 # | 62.59 ± 1.33 |
| BUN(mmol/L) | 10.77 ± 0.17 | 12.21 ± 0.36** | 12.61 ± 0.29 | 11.90 ± 0.38 | 11.56 ± 0.25 | 11.94 ± 0.37 |
| TC (mmol/L) | 2.26 ± 0.13 | 2.90 ± 0.21 * | 2.80 ± 0.13 | 2.95 ± 0.30 | 2.77 ± 0.21 | 3.03 ± 0.20 |
| FFA (µmol/L) | 412.71 ± 30.73 | 773.875 ± 74.23** | 640.75 ± 75.12 | 644.86 ± 66.17 | 693.43 ± 66.65 | 632.00 ± 67.23 |
Table 3: Effects of FRR, DRR, SRR or Met co-administration on serum biochemical parameters and liver glycogen content of rats treated with Dex a
Discussion
In the present study, we made an attempt to confirm the influence of postharvest processing on the claimed actions of different rehmannia roots by comparing their effects in countering Dex-induced side effects in rats, which include (1) insulin resistance based on the parameters of foot intake, hepatic glycogen content, and serum concentrations of insulin, glucose, free fatty acids, and total cholesterol; (2) high blood viscosity by two primary determinants of blood viscosity: hematocrit and serum protein; and (3) heart toxicity and oxidative damage according to the changes in serum creatine kinase isoenzyme MB and malonaldehyde content.
There is no doubt that the severity of Dex’s side effects is associated with its dosage, administration route, duration, and individual responses. In the rat models established to study Dex-induced side effects, various protocols have been applied, including Dex’s dosage being ranged from 0.005 mg/kg to 5 mg/kg, being given by drinking water, intraperitoneally or subcutaneously injection, for the duration of several days or several weeks [9,11,14-16,28]. In the current study, a relatively low dose (0.1 mg/kg) of Dex subcutaneously injected for 28 days was applied. The sustained weight loss (Figure 1A) indicates that the rat response to this medication is fairly strong.
Dex-induced insulin resistance was estimated by HOMA-IR, derived from fasting serum glucose and insulin contents in this study. Although it is less accurate than the hyperinsulinemic-euglycemic clamp technique, the gold standard for investigating and quantifying insulin resistance, the value of HOMA-IR in Dex alone-treated group increased more than three times compared with that in the control group, which clearly demonstrated that insulin resistance has been successfully established by Dex treatment in this study. A reduced food intake should also relate with insulin resistance as insulin signaling in hypothalamus could be reduced by Dex treatment [29]. The impaired insulin sensitivity may be attributed to Dex’s inhibitory effect on insulin signaling, leading to reduced insulin-stimulated glucose uptake and glycogen synthesis in fat and muscle [30]. The high level of fasting serum insulin should be the net effect of Dex’s suppression on insulin synthesis and secretion [31-33] and insulin degradation in liver [28]. The high level of liver glycogen (Table 3) indicates that glycogen synthesis was strongly enhanced by Dex treatment [34]. According to the results of serum insulin, HOMA-IR, and food intake, the effect of FRR in improving Dex-induced insulin resistance is close to Met, and better than that of DRR and SRR. Clearly, the decreased capacity of DRR and SRR in alleviating insulin resistance in Dex-treated rats is related to postharvest processing, during which catalpol and stachyose, the two main active components in ameliorating insulin resistance and lowering blood glucose [35-38], are gradually decomposed as shown in Figure S2A and in our previous reports [1,39].
Dex treatment could lead to a significant increase in whole blood viscosity, especially at low shear rates[40-41]. High blood viscosity can greatly retard blood circulation because of increased resistance, and this will lead to decreased oxygen delivery [42-43]. Hematocrit, the strongest impact factor on whole blood viscosity, was significantly higher in Dex alone-treated rats than in control ones (Table 2), and this result is consistent with the known fact that Dex could enhance erythropoiesis [26,27]. The level of serum total proteins, the main determinant of plasma’s viscosity, was also significantly raised (Table 3). Although the rat blood viscosity was not measured in the current study (due to insufficient amounts of blood samples available), our result indirectly confirmed that blood viscosity was increased by Dex treatment. In FRR-supplemented rats, hematocrit returned to the normal level, which indicated that FRR could enhance blood flow despite having no effects on the content of serum total proteins. On the contrary, SRR had no effects on hematocrit but brought the level of serum total proteins down, and therefore, it might also have the effect to promote blood circulation in Dex-treated rats. In addition, the result of peripheral blood cell counting also verified the immunosuppression effect of Dex as total white blood cells and spleen index were significantly decreased in Dex alone-treated rats (Tables 1 and 2). Among the subsets of white cells, lymphocytes dropped mostly, which is consistent with the report of Ohkaru et al. [44]. However, basophil numbers did not decrease but significantly increase in Dextreated rats, which may be due to the dosage and duration of Dex treatment, and the result may be attributed to Dex’s inhibition on basophil migration and degranulation [45-46]. Unfortunately, none of the tested materials can improve Dex-induced immunosuppression.
It is well known that creatine kinase isoenzyme MB (CK-MB) is highly expressed in heart muscle cells, and that its increased release into blood indicates myocardial damage. As a means to reflect Dex’s cardiotoxity as reported by de Salvi Guimarães et al. [5], the serum CK-MB level was assayed in this study. However, this parameter did not rise but significantly decreased in Dex alone-treated rats, and the difference may be due to the lower dosage of Dex treatment used in our experiment (0.1 vs. 3.5 mg/kg). The result suggests that low dose of Dex has a cardioprotective effect. Comparing with Dex alone group, different rehmannia roots could further reduce serum CK-MB levels. Although the differences did not reach to statistically significant levels, the results indicate that rehmannia roots may also have a cardioprotective effect. As shown in Table 1, Dex treatment led to cardiac hypertrophy, which may be the adaptive response to hypertension induced by Dex [5,16,47]. The intriguing point is that Dex-induced cardiac hypertrophy is significantly alleviated by DRR and SRR, but not FRR, and this finding suggests Dex-induced hypertension might be relieved by DRR or SRR administration.
Dex treatment also leads to severe oxidative damage as shown by the high level of serum MDA, one of the end products of lipid peroxidation (Table 3). The decreasing degree of serum MDA was greater in SRR group than in FRR and DRR groups, which is in accordance with the fact that SRR has a higher antioxidative effect due to catalpol decomposition and Maillard reaction during steam processing [2,48-50]. The increased blood concentrations of small molecular substances, such as glucose, free fatty acids, urea, and malonaldehyde will enhance blood osmotic pressure, leading to thirst, one of the main symptoms of Yin deficiency. These parameters were reduced by rehmannia root administrations although the difference did not reach a statistical significance (p>0.05). The result indicates that all rehmannia roots have the potential to nourish Yin, which warrants further investigations.
In conclusion, following once daily subcutaneous injection of Dex for 28 days at a dose of 0.1 mg/kg, the rats have suffered from insulin resistance, high blood viscosity, oxidative damage and immunosuppression, and FRR supplementation is much better than DRR and SRR in alleviating Dex-induced insulin resistance and high blood viscosity. This difference may be attributed to catalpol and stachyose decomposition in rehmannia roots during postharvest processing. Our data have provided evidence, for the first time, that postharvest processing has a profound influence on the biological activities of rehmannia roots.
Acknowledgements
This work was financially supported by China–Australia Centre for Health Sciences Research, and Shandong Adiministration of Traditional Chinese Medicine (2017-515). CJX is supported by National Health and Medical Research Council (NHMRC) of Australia Senior Research Fellowship (1042105).
References
- Guo YX, Wen XS, Zhao Y, Wei GD, Sun P (2012) Maillard reaction contributing to chemical changes during the processing of Shu Dihuang, the steamed root of Rehmannia glutinosa. Asian J Tradit Med 7: 50-58.
- Wei GD, Wen XS (2014) Characteristics and kinetics of catalpol degradation and the effect of its degradation products on free radical scavenging. Pharmacogn Mag 10: S122-129.
- Jeong Y, Han HS, Lee HD, Yang J, Jeong J, et al. (2016) A pilot study evaluating steroid-induced diabetes after antiemetic dexamethasone therapy in chemotherapy-treated cancer patients. Cancer Res Treat 48: 1429-1437.
- Inui N (2017) Antiemetic therapy for non-anthracycline and cyclophosphamide moderately emetogenic chemotherapy. Med Oncol 34: 77.
- de Salvi Guimarães F, de Moraes WM, Bozi LH, Souza PR, Antonio EL, et al. (2017) Dexamethasone-induced cardiac deterioration is associated with both calcium handling abnormalities and calcineurin signaling pathway activation. Mol Cell Biochem 424: 87-98.
- Fan CM, Foster BK, Wallace WH, Xian CJ (2011) Pathobiology and prevention of cancer chemotherapy-induced bone growth arrest, bone loss, and osteonecrosis. Curr Mol Med 11: 140-151.
- Georgiou KR, Hui SK, Xian CJ (2012) Regulatory pathways associated with bone loss and bone marrow adiposity caused by aging, chemotherapy, glucocorticoid therapy and radiotherapy. Am J Stem Cell 1: 205-224.
- Ghaisas M, Navghare V, Takawale A, Zope V, Tanwar M, et al. (2009) Effect of Tectona grandis Linn. on dexamethasone-induced insulin resistance in mice. J Ethnopharmacol 122: 304-307.
- He JH, Xu C, Kuang JY, Liu QH, Jiang HF, et al. (2015) Thiazolidinediones attenuate lipolysis and ameliorate dexamethasone-induced insulin resistance. Metabolism 64: 826-836.
- Kim DS, Kim TW, Park IK, Kang JS, Om AS (2002) Effects of chromium picolinate supplementation on insulin sensitivity, serum lipids, and body weight in dexamethasone-treated rats. Metabolism 51: 589-594.
- Kimura M, Daimon M, Tominaga M, Manaka H, Sasaki H, et al. (2000) Thiazolidinediones exert different effects on insulin resistance between dexamethasone-treated rats and wistar fatty rats. Endocr J 47: 21-28.
- Kraaij MD, van der Kooij SW, Reinders ME, Koekkoek K, Rabelink TJ, et al. (2011) Dexamethasone increases ROS production and T cell suppressive capacity by anti-inflammatory macrophages. Mol Immunol 49: 549-557.
- Martínez BB, Pereira ACC, Muzetti JH, de Paiva Telles F, Mundim FGL, et al. (2016) Experimental model of glucocorticoid-induced insulin resistance. Acta Cir Bras 31: 645-649.
- Nicastro H, Zanchi NE, da Luz CR, de Moraes WM, Ramona P, et al. (2012) Effects of leucine supplementation and resistance exercise on dexamethasone-induced muscle atrophy and insulin resistance in rats. Nutrition 28: 465-471.
- Okumura S, Takeda N, Takami K, Yoshino K, Hattori J, et al. (1998) Effects of troglitazone on dexamethasone-induced insulin resistance in rats. Metabolism 47: 351-354.
- Severino C, Brizzi P, Solinas A, Secchi G, Maioli M, et al. (2002) Low-dose dexamethasone in the rat: a model to study insulin resistance. Am J Physiol Endocrinol Metab 283: E367-E373.
- Thomas CR, Turner SL, Jefferson WH, Bailey CJ (1998) Prevention of dexamethasone induced insulin resistance by metformin. Biochem Pharmacol : 1145-1150.
- Ni L, Zhang B, Liu XQ, Wang XY, Chen B, et al. (2012) Empirical study of yin asthenia state of rats induced by dexamethasone sodium phosphate. Chin J Tradit Chin Med Pharm 27: 355-358.
- Ni L, Zhang B (2009) Re-recognition of animal model of both yin and yang deficiency induced by glucocorticoid. J Beijing Univ Tradit. Chin Med 32: 327-330.
- Xia BJ, Tong PJ, Sun Y, Zhou LY, Jin HT (2014) Methods and evaluations on the sterioid induced osteoporosis mice model with the type of Kidney Yin deficiency. China J Orthop Trauma 27: 673-679.
- Wu B (2010) Glucocorticoid side effects treated by traditional Chinese medicine, a review. Lishizhen. Med Mater Med Res 21: 719-721.
- Zha LL, Shen ZY, Zhang XF, Zhang P, Shi FY, et al. (1988) Experimental studies of Rehmannia glutinosa on pituitary and adrenal cortex in glucocorticoid inhibition model of rabbits. Chin. J Integr Tradit West Med 2: 95-97.
- Zha LL, Shen ZY, Shi FY, Huang ST, Chen RQ (1990) An experimental rearch on glucocorticoid-induced side effects antagonised by Kidney-tonifying herbs. J Tradit Chin Med 2: 1-3.
- Rafacho A, Cestari T, Taboga SR, Boschero AC, Bosqueiro JR (2009) High doses of dexamethasone induce increased -cell proliferation in pancreatic rat islets. Am J Physiol Endocrinol Metab 296: E681-E689.
- Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction.Anal Biochem 95: 351-358.
- Golde DW, Bersch N, Quan SG, Cline MJ (1976) Inhibition of murine granulopoiesis in vitro by dexamethasone. Am.J Hematol 1: 369-373.
- Malgor LA, Torales PR, Klainer TE, Barrios L, Blanc CC (1974) Effects of dexamethasone on bone marrow erythropoiesis. Horm Res 5: 269-277.
- Protzek AO, Rezende LF, Costa-Júnior JM, Ferreira SM, Cappelli AP, et al. (2016) Hyperinsulinemia caused by dexamethasone treatment is associated with reduced insulin clearance and lower hepatic activity of insulin-degrading enzyme. J Steroid Biochem Mol Biol 155: 1-8.
- Chruvattil R, Banerjee S, Nath S, Machhi J, Kharkwal G, et al. (2017) Dexamethasone alters the appetite regulation via induction of hypothalamic insulin resistance in rat brain. Mol. Neurobiol 54: 7483-7496.
- Burén J, Lai YC, Lundgren M, Eriksson JW, Jensen J (2008) Insulin action and signalling in fat and muscle from dexamethasone-treated rats. Arch. Biochem. Biophys 474: 91-101.
- Jeong IK, Oh SH, Kim BJ, Chung JH, Min YK, et al. (2001) The effects of dexamethasone on insulin release and biosynthesis are dependent on the dose and duration of treatment. Diabetes Res. Clin. Pract 51: 163-171.
- Lambillotte C, Gilon P, Henquin JC (1997) Direct glucocorticoid inhibition of insulin secretion-An in vitro study of dexamethasone effects in mouse islets. J Clin Invest 99: 414-423.
- Zawalich WS, Tesz GJ, Yamazaki H, Zawalich KC, Philbrick W (2006) Dexamethasone suppresses phospholipase C activation and insulin secretion from isolated rat islets. Metabolism 55: 35-42.
- Klein HH, Ullmann S, Drenckhan M, Grimmsmann T, Unthan-Fechner K, et al. (2002) Differential modulation of insulin actions by dexamethasone: studies in primary cultures of adult rat hepatocytes. J. Hepatol 37: 432-440.
- Guo XN, Zhang RX, Jia ZP, Li MX, Wang J (2006) Effects of Rehmannia glutinosa oligosaccharides on proliferation of 3T3-L1 adipocytes and insulin resistance. China J. Chin. Mater. Med 31: 403-407.
- Huang WJ, Niu HS, Lin MH, Cheng JT, Hsu FL (2010) Antihyperglycemic effect of catalpol in streptozotocin-induced diabetic rats. J. Nat. Prod 73: 1170-1172.
- Li X, Xu ZM, Jiang ZZ, Sun LX, Ji JZ, et al. (2014) Hypoglycemic effect of catalpol on high-fat diet/streptozotocin-induced diabetic mice by increasing skelet al muscle mitochondrial biogenesis. Acta. Bio. Biophys Sin 46: 738-748.
- Zhou J, Xu G, Ma S, Li F, Yuan M, et al. (2015) Catalpol ameliorates high-fat diet-induced insulin resistance and adipose tissue inflammation by suppressing the JNK and NF-κB pathways. Biochem. Biophys. Res. Commun 467: 853-858.
- Zhao Y, Wen XS, Cui J, Wu WH (2008) Separation, purification and property study of β-glucosidase andα-galactosidase from fresh roots of Rehmannia glutinosa. Food and Drug 10: 12-15.
- Miao MS, Wu W, Chen YP (2010) Effect of curcumine on whole blood viscosity, cerebral homogenate ATP enzyme and morphology of brain tissue in rat cerebral ischemia model with blood stasis. Tradit Chin Drug Res Pharmacol 21:150-152.
- Lv DH, Hao SJ, Li J, Su F (2013) Impacts of Xinnaoning tablets on blood stasis symptom score and whole blood viscosity of rat blood stosis model. Prac.J. Med & Pharm 30: 332-334.
- Lenz C, Rebel A, Waschke KF, Koehler RC, Frietsch T (2008) Blood viscosity modulates tissue perfusion: sometimes and somewhere. Transfus Altern Transfus Med 9: 265-272.
- Kwon O, Krishnamoorthy M, Cho YI, Sankovic JM, Banerjee RK (2008) Effect of blood viscosity on oxygen transport in residual stenosed artery following angioplasty. J Biomech Eng 130: 011003.
- Ohkaru Y, Arai N, Ohno H, Sato S, Sakakibara Y, et al. (2010) Acute and subacute effects of dexamethasone on the number of white blood cells in rats. Journal of Health Science 56: 215-220.
- Andrade MV, Hiragun T, Beaven MA (2004) Dexamethasone suppresses antigen-induced activation of phosphatidylinositol 3-kinase and downstream responses in mast cells. J. Immunol 172:7254-62.
- Yamaguchi M, Hirai K, Nakajima K, Ohtoshi T, Takaishi T et al. (1994) Dexamethasone inhibits basophil migration. Allergy 49: 371-375.
- Bal MP, de Vries WB, Steendijk P, Homoet-van der Kraak P, van der Leij FR, et al. (2009) Histopathological changes of the heart after neonatal dexamethasone treatment: studies in 4-, 8-, and 50-week-old rats. Pediatr. Res 66: 74-79.
- Wang HY, Qian H, Yao WR (2011) Melanoidins produced by the Maillard reaction: structure and biological activity. Food Chem 128: 573-584.
- Shi CJ, Wen XS, Gao HF, Liu ZH, Xu XK, et al. (2016) Steamed root of Rehmannia glutinosa Libosch (Plantaginaceae) alleviates methotrexate-induced intestinal mucositis in rats. J. Ethnopharmacol 183: 143-150.
- Zhang D, Wen XS, Wang XY, Shi M, Zhao Y (2009) Antidepressant effect of Shudihuang on mice exposed to unpredictable chronic mild stress. J. Ethnopharmacol 123: 55-60.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi