Research Article, Dent Health Curr Res Vol: 4 Issue: 2
Motivation of Patients to use Interdental Toothbrushes for the Domiciliary Management of Peri-Implants Health: Different Compliance According to the Age
Saverio Cosola1,2*, Young Min Park3, Enrica Giammarinaro1,2, Davide Quercia1, Andrea Vecchieschi1, Simone Marconcini1,4 and Anna Maria Genovesi1,5
1Tuscan Stomatologic Institute, Foundation for Dental Clinic, Research and Continuing Education, Versilia General Hospital, Lido di Camaiore, Italy
2Department of Stomatology, Faculty of Medicine and Dentistry, University of Valencia, Valencia, Spain
3Oral Health Center, Chulwon Public Health Center, Chulwon, South Korea
4Department of Surgical, Medical, Molecular and Critical Area Pathology, University Hospital of Pisa, University of Pisa, Pisa, Italy
5Study Center for Multidisciplinar Regerative Research, Guglielmo Marconi University, Rome, Italy
*Corresponding Author : Saverio Cosola
Tuscan Stomatologic Institute, Foundation for Dental Clinic, Research and Continuing Education, Versilia General Hospital, Lido di Camaiore, Italy
E-mail: s.cosola@hotmail.it
Received: May 17, 2018 Accepted: May 31, 2018 Published: June 06, 2018
Citation: Cosola S, Park YM, Giammarinaro E, Quercia D, Vecchieschi A, et al. (2018) Motivation of Patients to use Interdental Toothbrushes for the Domiciliary Management of Peri-Implants Health: Different Compliance According to the Age. Dent Health Curr Res 4:2. doi: 10.4172/2470-0886.1000132
Abstract
Introduction: The main goal for preventing and controlling periimplantitis is the reduction of oral bacteria load, through home and professional procedures. The aim of the present study was to evaluate the impact of a professional standard non-surgical therapy and the motivation to the use of domiciliary interdental brushes (Gunchi - South Korea) in a cohort of 75 patients with at least 1 implant placed with a minimum of 2 years follow-up and maximum 5 years.
Materials and methods: Seventy-five patients were screened on the basis of specific inclusion and exclusion criteria and were grouped into three different controlled cohorts based on their age: Group 1: patients aged 25-39 years; Group 2: patients aged 40-54 years; Group 3: patients aged 55-69 years. All subjects received oral hygiene instructions and a standard professional non-surgical treatment with ultrasonic debridement, polishing and ozonated water rinse by a specific professional device for a minute (Aquolab® - Italy). After the education and motivation session patients were instructed to use interdental aids (interdental brushes, Gunchi South Korea).
Patients were monitored for a total of 2 months follow-up. The following clinical parameters were measured on each patient before dental debridement and oral hygiene motivation (T0) and at 30 days (T1) and 60 days (T2):
Probing pocket depth (PPD), Recession (Rec), Modified Plaque Index (mPI) and Modified Sulcus Bleeding Index (mSBI).
Results: Each group showed significant improvements on Probing Depth (PD), mSBI and mPI at T1 an T2 follow-up. No statistically significant differences were found in terms of soft tissue recession. The difference between T2 and T0 and between T1 and T0 were computed on each parameter (ΔPPD, ΔRec, ΔmPI, ΔmSBI) for the comparison in efficacy between groups using the T-student test (p-value<0.05). The group-2 had statistical significant improvement in PPD respect to group-3, and in mPI and mSBI then group-1 respectively in T1 and T2 (p-value<0.05).
Keywords: Interdental toothbrush; Domiciliary oral hygiene; Implant maintenance; Patient’s compliance; Age of patients
Introduction
The increasing knowledge in terms of bone biology as well as a technology more and more performing make today implant dentistry a discipline with a very high success rate [1,2]. Even-though, implants are today a common therapeutic option, the number of implants failure is increasing. The explanation for the increasing number of implant failure, must be found “in author’s opinion” among those problems due to technical mistake of the clinicians. Too often implants are placed in wrong position or they are overloaded with inadequate prosthetic rehabilitations, conditions that may affect the long-term success outcomes [3,4]. In the last years, the role of periimplant maintenance procedures has been highlighted by several researchers and clinical trials, in order to control bacteria load around implants and thus reducing the risk of peri-implantitis [5].
Peri-implantitis is an infectious-inflammatory disease affecting soft and hard tissues surrounding dental implants and it always succeed an untreated implant mucositis. Clinically, peri-implantitis is often associated with bleeding, suppuration, and progressive loss of the implant supporting bone, but in literature there are not univocal criteria of diagnosis [6,7].
Bacterial flora associated to mucositis and peri-implantitis was found to be similar to the microbiota of periodontitis, consisting mainly of gram-negative anaerobes [8].
Therefore, as in periodontal disease, the professional and patientadministered mechanical disruption of the plaque and the reduction of the oral bacteria load is considered the standard for the management of peri-implants health [9].
Nowadays, a comprehensive treatment plan of mucositis and peri-implantitis usually includes a combination of different treatment methods such as oral hygiene instructions, non-surgical debridement, local/systemic antibiotics, surgical debridement, surgery, antimicrobial gel/rinse with mouthwash containing Zn-nHAp/Zn- PCA, chlorhexidine or other substances such as Ozone [10,11].
In this scenario, the prevention of peri-implant infection needs to be addressed to the patients starting from a strong motivation, good domiciliary oral hygiene instructions, efficient therapeutic instruments and appropriate behaviors such as avoidance of tobacco, and controlling systemic diseases [12].
The compliance to the treatment plan depends on the communication between dental hygienist/dentist and patients. Therefore, patients should be carefully evaluated by the dental team to assess the presence of specific systemic conditions, type of life style, age, which could suggest a tailored approach in terms of professional and domiciliary oral hygiene. At the same time patients should be motivated according to their needs and should demonstrate a good compliance. The primary aim of this prospective clinical study was to evaluate the impact of a professional non-surgical debridement and the role of correct domiciliary oral hygiene by using interdental aids, in patients with at least 1 implants with a minimum of 2 years follow-up. The secondary aim was to compare the clinical outcome of professional non-surgical treatment and correct domiciliary oral hygiene habits in 3 different cohorts of patients grouped by age.
Materials and Methods
Seventy-five patients were recruited for the present clinical study at the Tuscan Stomatologic Institute, Dental Center Fortis, Forte dei Marmi (LU) - Italy. The study was conducted in accordance with the Helsinki declaration of 2008. Each patient included in the study was asked to sign an informed written consent form and to answer to a specific anamnestic questionnaire.
All the participants were screened according to the following inclusion and exclusion criteria.
Inclusion criteria
• Aged between 25 and 70 years old
• Patients with at least 1 osseointegrated dental implant loaded for minimum 2 years and maximum 5 years
• Compliance with the study protocol and willingness to adhere to the hygiene instructions
Exclusion criteria
• Pregnancy
• Antibiotics, non-steroidal anti-inflammatory drugs, or steroids use in the previous 3 months
• Severe systemic disease that could compromise the conduct of the study
• Uncompensated diabetes
• Chronic or aggressive periodontitis or other severe oral pathologies
• Alcohol abuse
• Drug abuse
• Positive history of osteonecrosis
• Bisphosphonate use in the previous 12 months
• Antitumoral drugs use in the previous 24 months
• History of stroke in the previous 24 months
• Psychiatric disease
• Smoking more than 20 cigarettes/day
At the end of the screening procedure, 7 patients were excluded for the exclusion criteria and 75 patients were selected for the study and grouped into 3 different cohorts according their age:
• Group 1: patients aged between 25 and 39 years
• Group 2: patients aged between 40 and 54 years
• Group 3: patients aged between 55 and 69 years;
The three groups were balanced for gender and life style habits. Anamnestic and clinic recording were collected at baseline (T0) on each patient. The clinical parameters measured on each patient were:
- Probing pocket depth (PPD), measuring the mean value of vestibular, palatal/lingual, mesial and distal sites of implants by a plastic periodontal probe;
- Recession (Rec), measured on vestibular site for each implant;
- Modified Plaque Index (mPI) by Mombelli (1987) for dental implants scales as follows (0=No plaque, 1=plaque at the cervical margin difficult to be seen, 2=Plaque can be seen by naked eye, 3=Abundance of soft matter) [13,14].
- Modified Sulcus Bleeding Index (mSBI) for dental implants scales as follows (0=No bleeding when periodontal probe is passed along the gingival margin 1= Isolated bleeding spots visible 2=Blood forms a confluent red line on the gingival margin 3=Heavy or profuse bleeding) [15].
- General instructions for domiciliary oral hygiene were given to each patient during a motivation section before the non-surgical treatment ultrasonic debridement, polish and ozonated water rinse by a specific professional device for 1 min. Subjects were instructed to use a sonic toothbrush and to clean the interproximal spaces between teeth using interdental brushes (Gunchi - South Korea).
- Brushing procedures were first shown in a dental model and then demonstrated directly on the patient in front of a mirror in order to show the correct procedure to clean interdental spaces (Figure 1). Then, the participants were asked to reproduce the correct interdental mechanical cleaning technique under the supervision of the trained dental hygienist/dentist.
- After the instructions session, all patients were treated with a non-surgical periodontal treatment by using ultrasonic devices, polishing and ozonated water rinse for 2 minutes by a specific professional orale care device (Aquolab® - Italy).
- The clinical parameters were measured before dental debridement and oral hygiene motivation (T0) and patients were re-evaluated after 30 days (T1) and 60 days (T2) (Figure 2).
As diagnostic criteria for peri-implant mucositis was used the consensus report of 2008: probing dept of 5 mm or more and bleeding on probing [16,17].
Results
The present study is a prospective clinical study conducted on seventy-five patients with at least 1 dental implant. Patients were recruited at the Stomatological Tuscan Institute and were divided in 3 groups according their age: 25-39 years (Group 1); 40-54 years (Group 2); 55-69 years (Group 3).
Each group had 25 patients:
- Group 1: 27 implants on 25 patients with the mean age of 33,64 ± 3,91 years;
- Group 2: 32 implants on 25 patients with the mean age of 46,64 ± 3,99 years;
- Group 3: 37 implants on 25 patients with the mean age of 61,88 ± 4,03 years;
- Complete anamnestic data of the 3 groups of patients are reported in Table 1.
| Mean ± SD | Group 1 (n=25) | Group 2 (n=25) | Group 3 (n=25) |
|---|---|---|---|
| Age | 33,64 ± 3,91 years | 46,64 ± 3,99 years | 61,88 ± 4,03 years |
| Implants | 27 | 32 | 37 |
| Type of implants | 2 Prama® 8 Khono® 17 Premium® |
3 Prama® 11 Khono® 18 Premium® |
3 Prama® 12 Khono® 22 Premium® |
| Follow-up of implants | 3,32 ± 1,11 years | 3,64 ± 1,08 years | 3,72 ± 1,10 years |
| Treated diabetes | 1 patient | 3 patients | 6 patients |
| Cardiovascular disease or hypertension | 2 patients | 4 patients | 7 patients |
| Smoking<10 cigarettes | 3 patients | 4 patients | 4 patients |
| Professional oral hygiene per year | 1,76 ± 0,43 | 2,08 ± 0,64 | 2,16 ± 0,62 |
Table 1: Complete anamnestic data of 3 groups of patients.
- The 3 groups were balanced for gender and life style habits. The mean follow-up of dental implants was 3.56 ± 1.09 years. In the group 1, mean follow-up was 3.32 ± 1.11 years; in the group-2 was 3.64 ± 1.08 years and in the Group 3 was 3.72 ± 1.10 years.
- A complete descriptive and analytic statistics of the analyses clinical parameters is reported in Table 2. Before treatment (T0), no significant statistical differences were recorded in clinical parameters (PPD, Rec, mPI and mSBI) among the 3 groups.
| Variable | Time point | Differences between groups (p-value<0.05) | |||||
|---|---|---|---|---|---|---|---|
| Group 1 | Group 2 | Group 3 | G1G2 | G1G3 | G2G3 | ||
| Mean ± SD | Mean ± SD | Mean ± SD | |||||
| PPD | T0 | 2.56 ± 1.08 | 3.36 ± 1.11 | 3.40 ± 1.35 | |||
| T1 | 2.14 ± 1.02 | 2.64 ± 1.07 | 2.96 ± 1.06 | ||||
| T2 | 1.80 ± 0.96 | 2.48 ± 1.08 | 2.88 ± 1.01 | ||||
| ∆PPD | T0-T1 | 0.42 ± 0.49 | 0.72 ± 0.68 | 0.44 ± 0.51 | 0,0800 | 0,8881 | 0,1047 |
| T0-T2 | 0.76 ± 0.72 | 0.88 ± 0.72 | 0.52 ± 0.51 | 0,5609 | 0,1815 | 0,0480 | |
| Rec | T0 | 0.42 ± 0.59 | 0.96 ± 0.75 | 1.38 ± 0.87 | |||
| T1 | 0.32 ± 0.50 | 0.76 ± 0.68 | 1.30 ± 0.85 | ||||
| T2 | 0.30 ± 0.50 | 0.72 ± 0.66 | 1.28 ± 0.82 | ||||
| ∆Rec | T0-T1 | 0.10 ± 0.20 | 0.20 ± 0.38 | 0.08 ± 0.19 | 0,2539 | 0,7196 | 0,1647 |
| T0-T2 | 0.12 ± 0.26 | 0.24 ± 0.39 | 0.10 ± 0.20 | 0,2036 | 0,7643 | 0,1149 | |
| mPI | T0 | 1.04 ± 0.89 | 1.72 ± 1.10 | 1.96 ± 1.14 | |||
| T1 | 0.56 ±0.77 | 0.84 ± 0.69 | 1.40 ± 0.87 | ||||
| T2 | 0.20 ± 0.41 | 0.52 ± 0.65 | 0.84 ± 0.75 | ||||
| ∆mPI | T0-T1 | 0.48 ± 0.71 | 0.88 ± 0.66 | 0.56 ± 0.58 | 0,0460 | 0,6663 | 0,0769 |
| T0-T2 | 0.84 ± 0.69 | 1.20 ± 0.96 | 1.12 ± 0.97 | 0,1334 | 0,2453 | 0,7706 | |
| SBI | T0 | 0.80 ± 0.76 | 1.52 ± 1.04 | 1.84 ± 0.85 | |||
| T1 | 0.32 ± 0.55 | 0.80 ± 0.71 | 1.08 ± 0.76 | ||||
| T2 | 0.12 ± 0.33 | 0.16 ± 0.37 | 1.00 ± 1.94 | ||||
| ∆ mSBI | T0-T1 | 0.48 ± 0.59 | 0.72 ± 0.61 | 0.76 ± 0.43 | 0,1638 | 0,0612 | 0,7916 |
| T0-T2 | 0.68 ± 0.69 | 1.36 ± 0.99 | 0.84 ± 2.17 | 0,0072 | 0,7273 | 0,2821 | |
Table 2: Mean and standard deviation of each clinical parameter and the respective change during the follow-up: pocket probing depth (PPD and ΔPPD), Gingival recession (Rec and ΔRec), Modified plaque index (mPI and ΔmPI), modified sulcus bleeding index (mSBI and ΔmSBI). The column on the right reports p-values of the inter-group analysis. Significant statistic is reported in bold, setting a p-value <0.05.
- At baseline (T0), in the group 1, mean PPD was 2.56 ± 1.08 mm; in the group-2 was 3.36 ± 1.11 mm and in the group-3 was 3.40 ± 1.35 mm.
- Mean Rec in group 1 was 0.42 ± 0.59 mm, in the group-2 was 0.96 ± 0.75 mm and in the group 3 was 1.38 ± 0.87 mm.
- Mean mPI in the group 1 was 1.04 ± 0.89, in the group-2 was 1.72 ± 1.10 and in the group-3 was 1.96 ± 1.14. Mean mSBI in the group 1 was 0.80 ± 0.76, in the group-2 was 1.52 ± 1.04 and in the group-3 was 1.84 ± 0.85.
- At one month (T1) and two months evaluations (T2), all groups showed a significant improvement of PPD, mPI and mSBI. Minimal clinical improvements were observed also in Rec, but there was no statistical significance (Table 2).
- The difference between T2 and T0 and between T1 and T0 were computed on each parameter (ΔPPD, ΔRec, ΔmPI, ΔmSBI) for the comparison in efficacy between groups using the T-student test (p-value<0.05). The group-2 had a statistical significant improvement in PPD respect to group 3, and in mPI and mSBI then group-1 respectively in T1 and T2 (p-value<0.05) as depicted in Table 2. No change of Rec was recorded between groups.
- No statistical differences were reported in mucositis between groups at any times of the follow-up.
Discussion
The present study is a non-randomized control prospective clinical study with a total of 2 months follow-up period. The aim of the present prospective study was to evaluate the clinical effects of a standard non-surgical periodontal treatment with an appropriate domiciliary oral hygiene based on the use of interdental toothbrushes in patients with different ages. Several studies published in literature suggested the importance of a correct maintenance protocol of dental implants, nevertheless, too often clinicians do not keep the right attention to this crucial aspect of implant therapy [18].
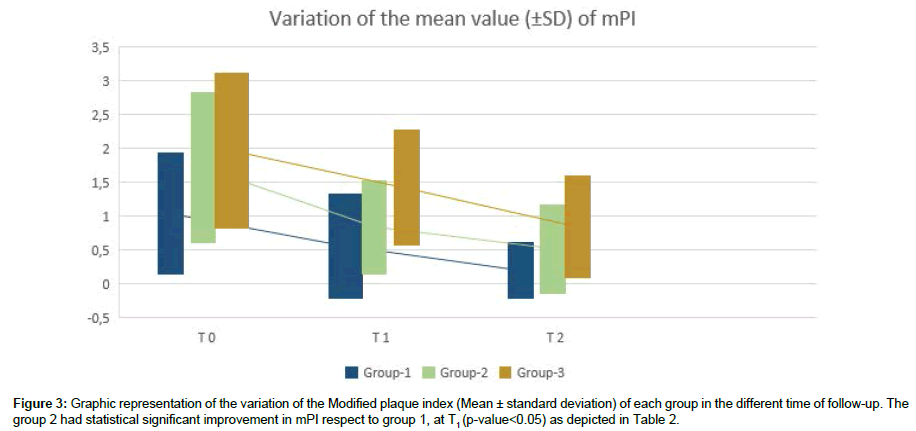
As represented in Table 2, the modified Plaque Index (mPI) and Modified Sulcus Bleeding Index (mSBI) registered at T1 have lower values then T0 (before professional treatment) and the outcomes continue to decrease in T2 showing also the good impact of domiciliary therapy.
The data of the present study are in accordance with the most part of other studies in literature highlighting the importance of domiciliary oral hygiene aids and motivations of patients [19,20].
Two recent reviews published by De Jager et al. [21] and Deacon et al. [22] recommended sonic or rotatory toothbrush instead of manual due to a major clinical efficacy, less risk of recession and abfractions on natural teeth. It is well known that the correct use of a sonic toothbrush determines improvements of plaque control and gingival inflammation, especially if it is used with an interdental manual toothbrush reducing also interdental plaque and preventing interdental caries and papilla inflammation.
Interdental toothbrushes are reported to be easier to use for patients and more efficient to reduce gingival bleeding comparing to toothpicks and floss [23,24].
The interdental toothbrush used in the present study showed good results in terms of clinical parameters analysed and good feedback furnished by the patients because of their ease. Vantages of these domiciliary instruments could explain the good compliance and clinical results of all three groups. In the present study the cohort of patients were assigned to one of the 3 groups based on their age. Group 1 (25-39 years), Group 2 (40-54 years) and group 3 (55-69 years) were respectively considered as young adults, adults and old patients. It is well known that these different ages have different style of life and different habits. Young patients have often irregular eating habits and irregular domiciliary oral hygiene control [25,26]. Thorstensson [27] highlighted the presence of a significant association between loosing teeth, poorer education, and lower quality of life, especially among older individuals.
Old patients, are often characterized for a less attention and motivation to oral hygiene and moreover their tissue, including, gingiva, show worse healing properties compared to younger subjects [27].
Regarding clinical outcomes changes, there is statistical significance between adults and young adults at T1 for mPI and at T2 for mSBI, but also in other timing patients aged between 40 to 54 years (adults) are reported to have better improvements in clinical outcomes on their implants. A graphic representation of the tendency of the Modified plaque index (Mean ± standard deviation) of each group in the different time of follow-up is reported in Figure 3.
Even thought, young adults have a worse domiciliary oral hygiene, reporting at T0 a high bleeding on probing (SmBI) and this aspect could be a bias of the present results.
These results of the present clinical study seem to agree with the previous consideration addressing to a better compliance and clinical outcomes in the adults group compared to young adults and old subjects [28].
A recent paper of Li et al. [29] focused its topic on the effects of App and technology to simplify and improve the motivation patients with orthodontic treatment.
Alternative ways to instruct patients for domiciliary maintenance of their natural teeth and their implants must be used. These methods need to be customized on patients considering age and style of life.
Conclusion
Non-surgical periodontal therapy has a good impact on clinical outcomes of all patients enrolled in the present study. All clinical parameters analyzed in the study decrease during the follow up period in presence of a good patient’s compliance. Although the intrinsic limitations of this analysis, the compliance and clinical effects seems to be better in patients of meddle age then young or old ones and remark the importance of a strong motivation considering the special needs of each patients.
References
- Chiappelli F, Kasar VR, Balenton N, Khakshooy A (2018) Quantitative Consensus in Systematic Reviews: Current and Future Challenges in Translational Science. Bioinformation 14: 86-92.
- Andreescu CF (2015) Survival Rate of Immediately Loaded Implants Restored using the Intraoral Welding Technique: A Literature Review. Dent Health Curr Res 1: 2.
- Ribes LN, Monreal BA, Fuster TMA, Penarrocha OD, Penarrocha DM (2017) Periimplant soft-tissue and bone levels around dental implants with different neck designs and neck surface treatments: A retrospective cohort study with 3-year follow up. J Oral Sci Rehabil 3: 16-23.
- Barone A, Toti P, Marconcini S, Derchi G, Saverio M, et al. (2016) Esthetic Outcome of Implants Placed in Fresh Extraction Sockets by Clinicians with or without Experience: A Medium-Term Retrospective Evaluation. Int J Oral Maxillofac Implants 31: 1397-1406.
- Canullo L, Penarrocha M, Monje A, Catena A, Wang HL, et al. (2017) Association Between Clinical and Microbiologic Cluster Profiles and Peri-implantitis. Int J Oral Maxillofac Implants 32: 1054-1064.
- Heydenrijk K, Meijer JA, Van der RWA, Raghoebar GM, Vissink A, et al. (2002) Microbiota around root-form endosseous implants: A review of the literature. Int J Oral Maxillofac Implants 17: 829-38.
- Canullo L, Penarrocha OD, Covani U, Botticelli D, Serino G, et al. (2016) Clinical and microbiological findings in patients with peri-implantitis: a cross sectional study. Clin Oral Implants Res 27: 376-382.
- Ata AJ, Flichy FAJ, Alegre DT, Candel MME, Penarrocha D, et al. (2011) Analysis of the peri-implant microbiota in 90 dental implants and its relationship to crevicular fluid volume. Med Oral Patol Oral Cir Bucal 16: 944-947.
- Salvi GE, Ramseier CA (2015) Efficacy of patient-administered mechanical and/or chemical plaque control protocols in the management of peri-implant mucositis. A systematic review. J Clin Periodontol 42: 87-201.
- Marconcini S, Giammarinaro E, Giampietro O, Giampietro C, Birgitta S, et al. (2017) Oxidative stress and periodontal disease in diabetic patients: a 3-month pilot study. Dent Oral Craniofac Res 3: 1-5.
- Cosola S, Marconcini S, Giammarinaro E, Marchisio O, Lelli M, et al. (2017) Antimicrobial efficacy of mouthwashes containing zinc-substituted nanohydroxyapatite and zinc L-pyrrolidone carboxylate on suture threads after surgical procedures. J Oral Science Rehabilitation 3: 24-30.
- Papathanasiou E, Finkelman M, Hanley J, Parashis AO (2016) Prevalence, etiology and treatment of peri-implant mucositis and peri-implantitis: A survey of periodontists in the United States. J Periodontol 87: 493-501.
- Ferreira SB, Figueiredo CM, Almeida AL, Assis GF, Dionisio TJ, et al. (2009) Clinical, histological, and microbiological findings in peri-implant disease: a pilot study. Implant Dent 18: 334-344.
- Mombelli A, van OMA, Schurch E, Land NP (1987) The microbiota associated with successful or failing osseointegrated titanium implants. Oral Microbiol Immunol 2: 145-151.
- Salvi GE, Lang N (2004) Diagnostic parameters for monitoring peri-implant conditions. Int J Oral Maxillofac Implants 19: 116-127.
- Lindhe J, Meyle J (2008) Group D of European Workshop on Periodontology. Peri-implant diseases: Consensus Report of the Sixth European Workshop on Periodontology. J Clin Periodontol 35: 282-285.
- Mombelli A, Lang NP (2000) The Diagnosis and Treatment of Peri-implantitis. Periodontol 17: 63-76.
- Riben GC, Norderyd O, Andre U, Renvert S (2015) Treatment of peri-implant mucositis using a glycine powder air-polishing or ultrasonic device: a randomized clinical trial. J Clin Periodontol 42: 462-469.
- Genovesi A, Barone A, Toti P, Covani U (2017) The efficacy of 0.12% chlorhexidine versus 0.12% chlorhexidine plus hyaluronic acid mouthwash on healing of submerged single implant insertion areas: a short-term randomized controlled clinical trial. Int J Dent Hygiene 15: 65-72.
- Graziani F, Palazzolo A, Gennai S, Karapetsa D, Giuca MR, et al. (2017) Interdental plaque reduction after use of different devices in young subjects with intact papilla: A randomized clinical trial. Int J Dent Hyg.
- De JM, Rmaile A, Darch O, Bikker JW (2017) The Effectiveness of Manual versus High-Frequency, High-Amplitude Sonic Powered Toothbrushes for Oral Health: A Meta-Analysis. J Clin Dent 28: 13-28.
- Deacon SA, Glenny AM, Deery C, Robinson PG, Heanue M, et al. (2010) Different powered toothbrushes for plaque control and gingival health. Cochrane Database Syst Rev 8: 4971.
- Kotsakis GA, Lian Q, Ioannou AL, Michalowicz BS, John M, et al. (2018) A network meta-analysis of interproximal oral hygiene methods in the reduction of clinical indices of inflammation. J Periodontol.
- Ricci M, Marchisio O, Genovesi AM, Gelpi F, Marconcini S, et al. (2014) Comparison between oscillating-twisting rotating of brush head vs a characteristic sweeping bristles motion on reducing oral inflammation. Minerva Stomatol.
- Lyle DM (2011) Use of a water flosser for interdental cleaning. Compend Contin Educ Dent 32: 78.
- Cozzani M, Ragazzini G, Delucchi A, Mutinelli S, Barreca C, et al. (2016) Oral hygiene compliance in orthodontic patients: a randomized controlled study on the effects of a post-treatment communication. Prog Orthod 17: 41.
- Thorstensson H (2010) Why do some people lose teeth across their lifespan whereas others retain a functional dentition into very old age? Gerodontology 27: 19-25.
- Reisine S, Schensul JJ, Goldblatt R, Radda K, Foster BC, et al. (2016) A bi-level intervention to improve oral hygiene of older and disabled adults in low-income housing: results of a pilot study. Community Dent Health 33: 127-132.
- Li X, Xu ZR, Tang N, Ye C, Zhu XL, et al. (2016) Effect of intervention using a messaging app on compliance and duration of treatment in orthodontic patients. Clin Oral Investig 20: 1849-1859.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi