Research Article, J Nanomater Mol Nanotechnol S Vol: 7 Issue: 5
Multiple Dye Doped Core-Shell Silica Nanoparticles: Outstanding Stability and Signal Intensity Exploiting FRET Phenomenon for Biomedical Applications
Pellegrino C*, Volpe A, Juris R, Menna M, Calabrese V, Sola F, Barattini C and Ventola A
Aczon s.r.l., Monte San Pietro (BO), Italy
*Corresponding Author : Pellegrino C
Aczon s.r.l., Monte San Pietro (BO), Italy
Tel: +39 051 6759711
Fax: +39 051 6759799
E-mail: chiarapellegrino@aczonpharma.com
Received: June 13, 2018 Accepted: August 09, 2018 Published: August 15, 2018
Citation: Pellegrino C, Volpe A, Juris R, Menna M, Calabrese V, et al. (2018) Multiple Dye Doped Core-Shell Silica Nanoparticles: Outstanding Stability and Signal Intensity Exploiting FRET Phenomenon for Biomedical Applications. J Nanomater Mol Nanotechnol S6: 003. doi: 10.4172/2324-8777.S6-003
Abstract
We present a one-pot synthesis of core-shell silica nanoparticles (SiNPs) as a novel fluorescent probe for biological applications. SiNPs were doped with a different number of dyes to ensure high efficiency Fluorescence Resonance Energy Transfer (FRET). Dyes are individually entrapped in the silica core without a covalent bonding between them. The strong interconnection achieved inside the core, a FRET with efficiency up to 86% was obtained. Nanoparticles called NTB530, NTB575 and NTB660 contain two, three and four different dyes respectively. Nanoparticles can be excited with a common blue laser and characterized by a long Stokes Shift up to the near-IR emission. Photostability, tested under continuous irradiation with a mercury lamp, showed higher stability of our Nanoparticles compared to commercial dyes like Fluorescein and R-Phycoerythrin. To prove the potential application of our Nanoparticle in flow-cytometry, they were conjugated with Anti-Human CD8 antibody and tested in comparison with commercial ones.
Keywords: Nanoparticles; Photostability; Biomedical applications
Introduction
Conjugation of biological molecules with fluorescent dyes has stimulated a growing interest to the huge number of exploitable biological applications. The need to monitor multiple parameters simultaneously has urged researchers to obtain dyes with different absorption and emission characteristics. There are different types of molecules from which to choose, ranging from small organic dyes, like Fluorescein Isothiocyanate (FITC) [1] to biologically sourced phycobiliproteins like R-Phycoerythrin (RPE) [2]. Both molecules show good emission intensity and quantum yield. Unfortunately, they are prone to some problems: 1) their photo lability [3] negatively influence the use in some biological applications; 2) they are significantly quenched when linked to biological molecules [4] therefore only low numbers of fluorophores per antibody are exploitable; 3) FITC fluorescence intensity strongly depends on a pH exceeding pH=8 [5]; 4) RPE is subject to a low conjugation yield [6]. The reduced numbers of exploitable molecules have to be attributed to the need to be soluble in water or most relevant buffers. Conversely, high numbers of bright fluorescent molecules (ex. Bodipy) are strongly hydrophobic. Nanoparticles are now emerging as promising to their features that overcome single molecule dye limits. Quantum dots became very famous because of the possibility to be synthetized with tuneable fluorescent intensity [7], but the high cytotoxicity and difficulties in surface modification reduce the possibility to be applied for biological applications. Moreover, the complicate decay behaviour [8] renders QDs unsuitable for applications like microscopy where high stability is needed. Indeed, even though considerable efforts have been made to improve the microscopy analysis on both reagents and instruments, the time needed by the operator to focus the samples and take pictures results in the bleaching of the fluorescent molecules and a consequent loss in signal intensity. Fluorescent nanoparticles are emerging as promising probes to high bio-compatibility and wellknown chemistry. In recent years, fluorescent silica nanoparticles (NPs), have been studied extensively to their interesting characteristics. First, synthesis of fluorescent core-shell silica nanoparticles ensures the dyes’ solubility in water. Moreover, these biocompatible systems provide the dual purpose of increasing fluorescence intensity and photostability. Multiple dyes are entrapped into each nanoparticle. Therefore, the molar extinction coefficient of fluorescent silica NPs is higher than that of common dyes. Moreover, the protection afforded by the silica core or, eventually, by an outer shell, should limit photobleaching. The flexibility of dye doped silica nanostructures opens plenty of possibilities to gain a switchable and responsive signal by only changing types and numbers of fluorochromes entrapped in the core.
Synthesis
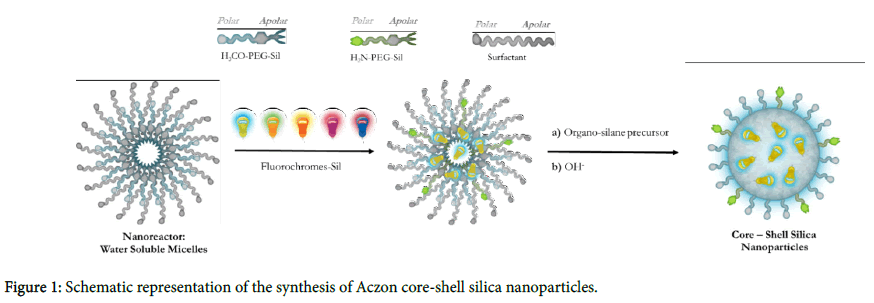
Aczon Nanoprobes are core-shell dye doped silica nanoparticles synthesized through a one-pot, two-step reaction known as the micelle-assisted method (Figure 1). First, all reagents are mixed together in a solution of water and n-butanol. In this condition, surfactant creates micelles within which all reagents, due to their hydrophobicity, arrange.
When ammonia is added to the mixture together with a silane precursor, the base-catalysed hydrolysis of all different trialkoxysilanes leads to the formation of monodisperse core-shell nanoparticles. Different kinds of surfactants (Tween, Triton, Brij etc…) have been efficiently used for the synthesis. By changing the surfactant characteristics, the micelles’ dimension can be modulated and, consequently, the NPs diameter (from 20 to 100 nm). Shell is composed of two different polyethylene glycols (Figure 2: H3CO-PEGSil, [9] H2N-PEG- Sil [10]), both terminating with a trialkoxysilane: one inducing the stability and solubility in water, while the other is functionalized to have amine reactive groups on the shell.
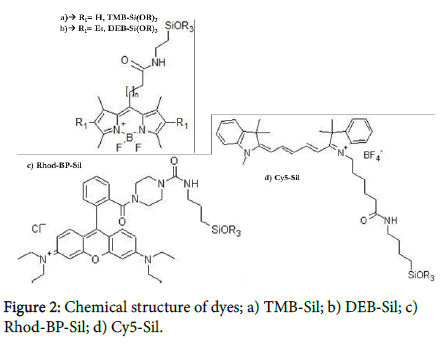
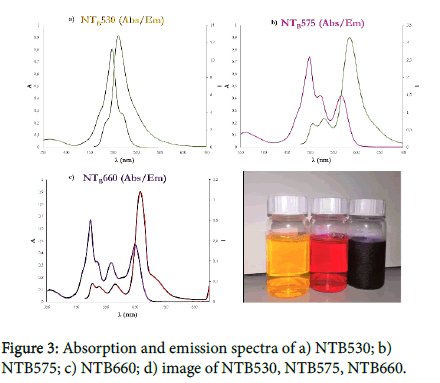
The initial mixture may contain one or more hydrophobic dyes. The solubility in an organic solvent, they will arrange inside surfactant micelles and, due to the covalent modification with a trialkoxysilane group, they will be able to react and become a component of the silica core. Being part of the structure, dyes are not released over time and are strongly interconnected inside the core. Therefore, even without a covalent bonding between them, dyes with the opportune photophysical characteristics can generate an efficient fluorescence resonance energy transfer (FRET). Nanoparticles showed in Figure 3 called NTB530, NTB575 and NTB660 contain two, three and four different dyes respectively. We chose four dyes with different chromophore groups. Following the absorption spectra, the first two dyes, known as TMB-Sil [10](ε=56800 M-1cm-1, Φ=0,63 in NPs) and DEB-Sil [10] (ε=48200 M-1cm-1, Φ=0,51 in NPs), are Bodipy-like dyes (Figure 2a,b); the third one, known as Rhod-BP-SiOR3 [11,12] (ε=45200 M-1cm-1, Φ=0,33 in NPs), is a Rhodamine-like dye (Figure 2c); the last, known as Cy5-Sil (ε=130500 M-1cm-1, Φ=0,15 in NPs), is a Cyanine-like dye (Figure 2d). This choice was made to simultaneously obtain the best fluorescent cascade while proving the versatility of the synthesis. All probes have a maximum absorption at 488 nm, the presence of TMB-Sil, while the final emission varies from 530 nm, 585 nm and 660 nm depending on the final acceptor.
Nanoparticle emissions perfectly mimic that of commercial dyes used for flow-cytometry and microscopy (FITC, RPE and RPE-Cy5). As mentioned before, other dyes can be efficiently used providing the solubility in organic solvents and the presence of a trialkoxysilane group. The amount of each fluorophore was modulated to obtain the most efficient cascade transfer without losing intensity emission induced by self-quenching. What we have discovered is that the amount of the first donor only affects the final intensity and has no significant effects on the cascade. Increasing the first donor will create a tandem with better fluorescence intensity due to the proportional increase in molar extinction coefficient that induces a major amount of energy to be absorbed. On the other hand, the last donor strongly influences the cascade efficiency together with the intensity emission. Increasing the acceptor leads to a significant increase in FRET efficiency. Unfortunately, when we reach the concentration at which self-quenching arises, the final intensity decreases? No significant differences have been noticed when the concentration of the middle donor was changed. FRET efficiency, calculated considering the emission intensity of the first donor and the last acceptor, reaches a percentage exceeding 86%. These results firstly demonstrate that dyes are at the correct distance for energy transfer. Secondly that the distribution of dyes inside each nanoparticle is homogeneous, otherwise the energy transfer won’t be so efficient.
Photostability
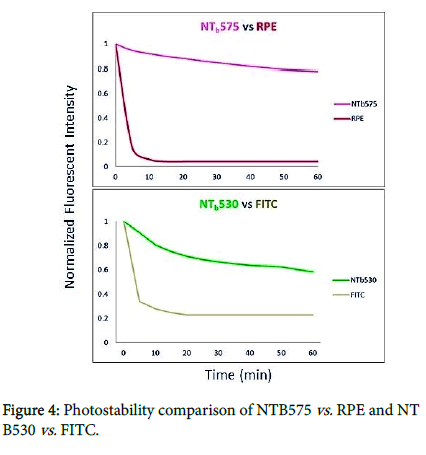
AcZon silica Nanoparticles, the protective core/shell structure, protects the sensitive fluorescent molecules increasing the stability under stressing conditions. We made a preliminary analysis using continuous irradiation with a spectrofluorometer. A photobleaching experiment in liquid phase gave us good preliminary results (data not showed). Anyway, we must consider that the energy at stake is lower and the optical pathway is higher in spectrofluorometric analysis compared to fluorescence microscopy analysis. Therefore, photostability of these dyes or Nanoprobes could be different for application with a microscope. To evaluate the performance of Aczon Nanoprobes in this field, a photobleaching experiment was performed under continuous irradiation with a fluorescence microscope. To immobilize nanoparticles and molecular fluorophores in an inert system, these dyes have been embedded in a thin Polyacrylamide (10%) gel matrix. The doped gel was then continuously irradiated under fluorescence microscope, collecting images every 5 min. Nanoparticles used for this experiment are NTB530 against the respective molecular fluorophore, FITC, and NTB575 against the respective molecular fluorophore, RPE.
The gel has been continuously irradiated by means of a fluorescent microscope working with a mercury lamp. Pictures have been taken at regular intervals (0, 20’, 40’, 60’) with a CCD camera. The fluorescence intensity has been analysed on the pictures with ImageJ software. The normalized fluorescent data have been plotted on a Cartesian graph against exposure time (Figure 4). The results underline a dramatic decrease of fluorescence intensity for the molecular fluorophores fluorescein. Fluorescein loses fluorescent intensity (≈70%) in the first 20 min which isn’t observed in the nanoparticle-based fluorophores (loss of fluorescence intensity less than 30%). The value of the results is easily derived directly from the images (Figure 5) in which the molecular fluorophore phycoerythrin signal disappears in the early moments of irradiation despite the NTB575 high stability (loss of fluorescence intensity less than 20%).
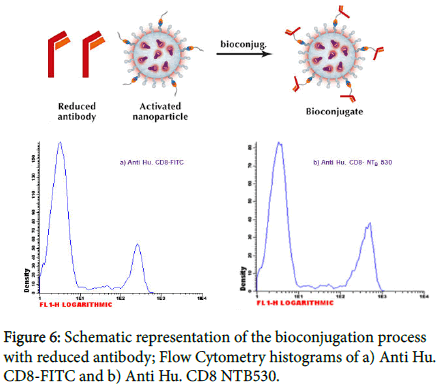
Bioconjugation Flow-cytometry
Our conjugation method (Figure 6) takes advantage of the heterobifunctional crosslinker SMCC that contains Nhydroxysuccinimide (NHS) ester and maleimide groups that simultaneously allow covalent conjugation of amine- (SiNPs) and sulfhydryl-containing molecules (antibody). The NHS moiety is used to react with nanoparticles thus changing the surface reactive groups from ammine to maleimide (activated nanoparticles).
To react with the maleimide moiety, the antibody must have free (reduced) sulfhydryl. Our procedure involves the use of 1,4- Dithiothreitol (DTT) as a reducing agent. As all reducing agents, DTT will reduce disulphides only if they are accessible. A protein, in its native conformation, is frequently protected from complete reduction. For instance, at a moderate concentration of DTT and without denaturants, limited cleavage of disulphide in antibody molecules results in reducing mainly the bonds between the heavy chains of immunoglobulin. Therefore, two half antibody molecules are produced, each one containing the antigen binding site and free sulfhydryl in the hinge region. For this reason, the method preserves the antibody’s biological activity [13]. Conjugation molar ratio depends on the biological molecules involved in the bioconjugation process (isotype and consequently sulfhydryl group available on hinge region). Generally, a fold molar excess of maleimide group is required despite the high reactivity of sulfhydryl containing proteins. The CD8 antigen is present on 20-36% of normal peripheral blood lymphocytes (suppressor/cytotoxic T cells), on 60-85% of normal thymocytes and on a NK cell subset (CD3-CD8+). The analysis of flow-cytometry data has been done on a lymphocyte population using the Infinicyt™ software. Conjugates with nanoparticles showed good statistical parameters (Table 1).
| Mean+ | CV+ | S/B | % Positive | |
|---|---|---|---|---|
| Anti Hu. CD8-NTB530 | 476.66 | 42.25 | 108.3 | 21.65 |
| Anti Hu. CD8-FITC | 192.28 | 40.00 | 56.5 | 20.82 |
| Anti Hu. CD8-NTB575 | 455.93 | 28.75 | 167.0 | 17.38 |
| Anti Hu. CD8-RPE | 1251.75 | 25.00 | 154.6 | 17.58 |
| Anti Hu. CD8--NTB660 | 654.75 | 28.12 | 135.3 | 17.80 |
| Anti Hu. CD8-RPECy5 | 703.26 | 36.00 | 133.7 | 18.29 |
Table 1: Statistical parameters of nanoparticle conjugates and respective commercial competitors.
Specificity discrimination of dim and bright [14] population and signal to background (S/B) are comparable to commercial competitors thus proving the efficiency of the conjugation procedure. The emission intensity (Mean+) of Anti Hu. CD8-NTB530 is two times more brilliant than Anti Hu. CD8-FITC (Table 1). While Anti Hu. CD8- NTB660 is comparable to Anti Hu. CD8-RPECy5, only in case of Anti Hu. CD8-NTB575 do we find lower emission intensity when compared to Anti Hu. CD8-RPE imputable to the high molar extinction coefficient of RPE compared to Rhodamine dyes.
Materials and Method
General All reagents and solvents were used as received from commercial sources without further purification. H3CO-PEG-Sil and H2N-PEG-Sil were prepared as described by I.M. Rio-Echevarria. TMB-Sil was synthesized by coupling (3-aminopropyl) triethoxysilane (APTES) with the carboxylic acid-fluorophore (TMB-COOH) as described below. The synthesis of the carboxylic acid itself has been reported by I.A. Boldyrev. DEB-Sil was prepared with the same procedure as TMB-Sil, but starting with 3-ethyl-2,4-dimetyl pyrrole instead of 2,4-dimetyl pyrrole. Rhod-BP-Sil was synthesized following previously published methods (T. Nguyen; E. Rampazzo). Cy5-Sil was synthesized from Cyanine5 NHS ester, purchased from Lumiprobe GmbH, as described below. Bio-Beads SM-2 Adsorbents were purchased from Bio-Rad Laboratories Srl. Succinimidyl 4-(Nmalimidomethyl) cyclohexane-1-1carboxylate (SMCC) was purchased from Setareh Biotech. Anti-Human CD8 (733, Ms. IgG1) and lysing solution are commercialized by our company. All the other reagents were purchased from Sigma-Aldrich Srl [15,16].
TMB-Sil and DEB-Sil A two-necked flask is dried with a heat gun under a flow of nitrogen. Next, dichloromethane (0.4 mL/mg of dye), TMB or DEB-COOH (1 eq.), APTES (2 eq.), triethylamine (2 eq.), EDC (1.5 eq.), and COMU (1.5 eq.) are mixed together and stirred at room temperature for 90 min; after that, an additional amount of EDC (1.5 eq.) and COMU (1.5 eq.) is added. Then, the solvent is removed under reduced pressure with a rotary evaporator. Finally, the powder is purified by means of flash chromatography on silica gel (cyclohexane/ ethyl acetate gradient); the appropriate fractions of the eluate are then concentrated with the rotary evaporator and desiccated in vacuum, affording a bright orange solid.
Cy5-Sil A two-necked flask is dried with a heat gun under a flow of nitrogen. Next, dichloromethane (0.4 mL/mg of dye), Cyanine5 NHS ester (1 eq.), and APTES (1.1 eq.) are mixed together and stirred at room temperature for 3 h. Then, the reaction mixture is transferred into a vial and the solvent is removed with a gentle flow of nitrogen. Finally, the resulting solid is desiccated in vacuum and used as such without further purification.
Dye-doped silica nanoparticles In a typical preparation, 843 mg of surfactant (0,75 mmol) and 172 mg of H3CO-PEG-Sil (0,075 mmol) are mixed together with the desired amount of silanized fluorophores. Then, 25 mL of distilled water and 375 μL of 1-butanol are added to the flask and mixed under magnetic stirring for 1 h at room temperature. The sample is then transferred into a heating bath set at 30-40°C and 364 μL of trimethoxymethylsilane (2,5 mmol), a solution of 178 mg of H2N-PEG-SiO3 (0,0875 mmol) 0,3 M in dimethyl sulfoxide (DMSO) and 375 μL of a 2,8% solution of ammonia in water (0,555 mmol) is added to the mixture and the reaction is kept under stirring for at least 48 h. For the purification 10 g of Bio-Beads are added and the nanoparticle suspension is left stirring for 3 h at room temperature. Bio-Beads are removed by filtration and the whole sample is dialyzed against distilled water using regenerated cellulose membrane tubing (cut-off 12 kDa) for at least 24 h. The sample is collected and passed at least twice through hydrophilic PVDF membrane filters with a 0,45, 0,22 and 0,1 μm pore size. Nanoparticles are centrifuged at 1000 rpm for 30 min at room temperature, discarding the pellet, if any. Sample is characterized by spectrophotometer and spectrofluorometer. DLS: 20±0.3 nm, PDI=0.151, Z-Pot=6.3±1.7 mV.
Bio-conjugation 5 mL of NPs in 50 mM Napi/1 mM EDTA pH=8 are made to react with 4.5 mg of SMCC (10 mM) for one hour at room temperature. The mixture is purified by Size Exclusion Chromatography using 50 mM MES/2 mM EDTA. 2 mg of Anti Hu. CD8 (733) are reduced with Dithiothreitol and made to react with activated nanoparticles for 1 h, at room temperature, under stirring. Reaction mixture is again purified by size exclusion chromatography.
Flow cytometry 100 μl of whole blood are dispended into suitable tubes. Then, 10 μl of conjugated antibody are added and mixed by vortex at low speed. The sample is incubated for at least 15 min at room temperature in the dark. 2 mL of lysing solution are added and mixed by vortex at low speed. After 10 min incubation at room temperature in the dark, sample is centrifuged for 5 min at 300 × g at 2°C. The supernatant is discarded and substituted with 1 mL of 10 mM NaPi/ 150 mM NaCl. The analysis was performed on BD FACScan and 10.000 events have been acquired for each sample.
Conclusions
We synthetized monodisperse core-shell silica nanoparticles onepot reaction, it is known as micelles-assisted method. Reaction characteristics allow us to obtain nanoparticles simultaneously doped with multiple dyes. The advantageous distance between dye molecules, reached inside the core, induced good Fluorescence Resonance Energy Transfer. Therefore, multiple doped nanoparticles produce blue laser excitable probes characterized by a long Stokes Shift up to the near-IR. The modulation of the dyes’ concentration gives rise to different photochemical characteristics that can be changed according to the final application. We proved that the success of the procedure is completely independent from dyes’ chemical structure. Therefore, the synthesis can be used with other molecules besides the one showed. That opens the possibility to create tandems that are excitable and emit at the desired wavelength. The protection ensured by the shell increased dyes’ photostability extremely compared to commercial available dyes. This is a very useful characteristic in those analyses where long-time stability is required. The presence of amino groups on the surface allows us to conjugate Nanoparticles with different biological molecules. We decided to choose a monoclonal antibody to prove the use of our nanoparticles in flow cytometric. The conjugate demonstrates good fluorescence intensity, high signal to background and good discrimination. All the aforementioned features make Aczon Nanoparticles interesting probes for both microscopy and flow cytometry.
References
- Jobbàgy A, Kiràly K (1966) Chemical characterization of fluorescein isothiocyanate-protein conjugates. Biochim Bionphys Acta 124: 166.
- Oi VT, Glazer AN, Stryer L (1982) Fluorescent phycoliprotein conjugates for analyses of cells and molecules. J Cell Biol 93: 981.
- Kawamura Jr A (1978) Fluorescent antibody techniques and their applications. Immunology 35: 571.
- Haugland (HP) Coupling of monoclonal antibodies with fluorophores. Monoclonal Antibody Protocols 45: 205-221.
- Lanz E, Gregor M, Slavik J, Kotyk A (1997) Use of FITC as Fluorescent Probe for Intracellular pH measurement. J Fluorescence 7: 317-319.
- Ma SY, Wang GC, Sun HB (2003) Characterization of the artificially covalent conjugate of B-phycoerythrin and R-phycocyanin and the phycobilisome from Porphyridium cruentum. Plant Sci 164: 253.
- Resch-Genger U, Grabolle M, Cavaliere-Jaricot S, Nitschke R, Nann T (2008) Quantum dots versus organic dyes as fluorescent labels. Nat Rev 5: 763.
- Schlegel G, Bohnenberger J, Potapova I, Mews A (2002) Fluorescence decay time of single semiconductor nanocrystals. Phys Rev Lett 88: 137401.
- Rio-Echevarria IM, Selvestrel F, Segat D, Guarino G, Tavano R, et al. (2010) Highly PEGylated silica nanoparticles: ‘‘ready to use’’ stealth functional nanocarriers. J Mater Chem 20: 2780-2787.
- Boldyrev IA, Zhai X, Momsen MM, Brockman HL, Brown RE, et al. (2007) New BODIPY lipid probes for fluorescence studies of membranes. J Lipid Res 48: 1518-1532.
- Nguyen T, Francis MB (2003) Practical Synthetic route to functionalized rhodamine dyes. Org Lett 5: 3245.
- Rampazzo E, Bonacchi S, Juris R, Montalti M, Genovese D, et al. (2010) Energy transfer from silica core-surfactant shell nanoparticles to hoisted molecular fluorophores. J Phys Chem B 114: 14605-14613.
- Bioconjugate Techniques, Elsevier.
- The Leucocyte Antigen FactsBook, Academic Press Inc. San Diego.
- Tahir MN, Ragg R, Natalio F, Sahoo JK, Daniel J, et al. (2015) Amine functionalized ZrO2 nanoparticles as biocompatible and luminescent probes for ligand specific cellular imaging. J Mater Chem 3: 2371-2377.
- Natalio F, Kashyap A, Lorenz S, Kerschbaumer H, Dietzsch M, et al. (2012) Multi-photon imaging of amine-functionalized silica nanoparticles. Nanoscale 4: 4680-4686.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi