Research Article, J Nanomater Mol Nanotechnol Vol: 8 Issue: 3
Simple and Customizable Gelatin Nanoparticle Encapsulation System for Biomedical Applications
Jewel Cary1, Frank William Pierson2 and Abby R Whittington1,2,3*
1Department of Biomedical Engineering and Mechanics, Virginia Polytechnic Institute and State University, 325 Stanger Street, 220 Kelly Hall, Blacksburg, Virginia, 24061, United States of America
2Department of Biomedical Sciences and Pathobiology, Virginia Polytechnic Institute and State University, 14210 Prices Fork Road, Blacksburg, Virginia, 24061, United States of America
3Department of Chemical Engineering, Department of Material Science and Engineering, Virginia Polytechnic Institute and State University, 400 Stanger St, 117 Surge Building, MC-0237, Blacksburg, Virginia, 24061, United States of America
*Corresponding Author : Abby R Whittington
Departments of Chemical Engineering, Material Science and Engineering, Virginia Polytechnic Institute and State University, 400 Stanger St, 117 Surge Building MC-0237, Blacksburg, VA, 24061, United States of America
Tel: 540-231-0665
E-mail: awhit@vt.edu
Received: June 10, 2019 Accepted: June 10, 2019 Published: June 28, 2019
Citation: Cary J, Pierson FW, Whittington AR (2019) Simple and Customizable Gelatin Nanoparticle Encapsulation System for Biomedical Applications. J Nanomater Mol Nanotechnol 8:3.
Abstract
Objective: The double desolvation technique has been used to encapsulate small, hydrophilic drugs with protein affinity in gelatin nanoparticles for many years. Expanding the types of materials that can be encapsulated would allow the double desolvation method to be used for a wider range of biomedical applications, including biological delivery. Methods: Here, we use the double desolvation technique to encapsulate two different sizes of polystyrene beads as a first step toward encapsulating biologics like viruses and nucleic acids of similar size, shape, zeta potential, and functional groups in a new delivery system. Drug delivery systems that are easy to produce and customizable to different biomedical applications are in demand. With these parameters in mind, we created a simple gelatin nanoparticle encapsulation system with the potential for chemical modification for targeting purposes and encapsulation of different materials. Results: Matching the encapsulation material to the size and shape of the empty nanoparticles resulted in encapsulated nanoparticles of ideal narrow size distribution with stable storage parameters at room temperature over a 1-month period in distilled water. Additionally, the encapsulation system was shown to be most stable at pH 3-4 compared to other physiological pH ranges. Transmission electron microscopy verified the size ranges found using dynamic light scattering and revealed the inert material was encapsulated, partially encapsulated, and non-encapsulated nanoparticles in each formulation. Conclusion: This matched encapsulation material fabrication method may decrease the need for additional filtration after biologics are added and the encapsulation range would be ideal for sustained biologic release over time.
Keywords: Material encapsulation; Biomedical engineering; Gelatin nanoparticles
Introduction
Major advances in modern-day drug delivery systems have been controlled release formulations and specific targeting to the tissues of interest. Formulations with these characteristics have increased therapeutic effectiveness, reduced administration frequency, and reduced side effects related to repeat dosing [1-11]. Delivery systems must be simple to produce, customizable, and inexpensive to be viable alternatives to the conventional delivery methods already in use in healthcare [5,6,12,13]. To date, the majority of biologics encapsulated in gelatin for therapeutic purposes have been small drugs of <5 nm diameter [14-16]. Our goal in this project was to encapsulate larger materials in a gelatin nanoparticle (NP) system as a first step toward a new delivery system of larger biologic materials. To ensure the system was simple to produce and easily adaptable to customer needs, inexpensive materials were used and the lab equipment required was kept simple, so the system could be produced in a wide range of laboratories at a minimal cost [3,5,7].
NPs can be used to deliver a variety of encapsulated materials, including drugs, polyphenols, RNA, genes, proteins, and viruses allowing for NP delivery systems to be used across the medical field in different biomedical applications [1,3,4,17,18]. Using nanoscale particles can lead to enhanced uptake by cell populations of interest, such as macrophages, M cells, and tumor cells [3-7,9,11,19-21]. Gelatin was chosen to form the NP polymer shell because it is a natural polymer that is widely biodegradable into benign components, Generally Recognized As Safe (GRAS)-certified, already in use in the medical field for various applications, inexpensive, and has many functional groups that can be modified for targeted drug delivery [1,5,7,9,19,20,22,23]. NPs can be used to prolong the delivery of encapsulated materials with otherwise low bioavailability or with short half-lives [5,8,18]. NPs may also be targeted to tissues that historically have been difficult to reach and can achieve sufficient therapeutic drug doses using systemic drug delivery. We chose to use polystyrene beads of two different sizes, 100 nm, and 200 nm, as inert encapsulated materials for ease of initial characterization of the system. In later work, these stand-in materials will be replaced by biologics of similar size, shape, zeta potential, and containing similar functional groups as the system is modified for different biomedical applications. Controlling the size of NPs allows for targeting the delivery system to particular tissues or cells and should further targeting ability be required, the reactive groups on the gelatin may be modified [1,5,7,9]. Overall, this system has the potential to expand NP delivery to biologics larger than conventional drugs. Our hypothesis is that matching the encapsulated material diameter to the empty nanoparticle diameter will produce the best combination of encapsulated nanoparticle parameters: a diameter close to the encapsulated material diameter, a narrow and monodisperse size distribution (polydispersity index, PDI), and sufficient particle charge (zeta potential, ZP) to avoid particle aggregation.
The ideal specifications for NPs in this study were to have encapsulated NPs of similar initial average diameter to the encapsulation material (100 nm and 200 nm) in order to facilitate cellular uptake, to have the initial average size distribution less than 0.1 for highly controlled encapsulations or at most 0.2 for less controlled encapsulations, and to have an initial average ZP close to ± 30 mV at a storage pH of 3-4 to avoid NP aggregation [5,7,8,17,19]. Additionally, the NPs should no statistically significant changes in diameter or size distribution for at least one month at room temperature in distilled water as an indicator of product storage stability.
Materials and Methods
Two types of gelatin, Type A 300 Bloom (Sigma-Aldrich; St. Louis, MO) and Type B 275 Bloom (Vyse Gelatin Company; Schiller Park, IL) were used in the two-step desolvation NP fabrication process. These polymers have been characterized in previous literature [24]. Polystyrene PolyBead beads (100 nm average diameter) and Fluoresbrite beads (200 nm average diameter) were used as encapsulation materials (PolySciences, Inc.; Warrington, PA) as they have anionic charges from their sulfate ester groups similar to those found in biologics like proteins and nucleic acids. The two gelatin types were selected because their NP sizes could be consistently and repeatedly produced [24]. Glutaraldehyde (25%, Sigma-Aldrich; St. Louis, MO) was used for all NP crosslinking. All other materials were analytical grade and used as received.
Nanoparticle preparation
A two-step desolvation method was used to make the nanoparticles in which an organic desolvating agent caused aggregation of the gelatin molecular chains around the encapsulation material of interest with chemical crosslinking for stability [1,10,24].
Briefly, gelatin was dissolved in distilled water with stirring at 600 rpm and heated until a solution was clear and light yellow (42ºC for Vyse 275 Bloom and 39.5ºC for Sigma 300 Bloom). The solution was removed from heat and acetone was added to allow separation of high molecular weight (HMW) and low molecular weight (LMW) gelatin chains to decrease the variability in gelatin used to create the NP suspension-After 24hr at room temperature, the top LMW liquid gelatin layer was decanted and the bottom HMW solid gelatin layer was redissolved in distilled water with the same stirring and heating parameters as above. The pH was adjusted to 2.5 using 1M HCl and then 9.1 × 1012 polystyrene beads (100 nm or 200 nm) were added. The stirring rate was increased to 1000 rpm to ensure homogeneous dispersion of the insoluble beads. Acetone (70 mL) was added dropwise using a syringe pump (3 mL/min) while the solution temperature was maintained (37-39.5ºC for Sigma 300 Bloom and 40-42ºC for Vyse 275 Bloom). An iridescent blue color change from the Tyndall effect was observed, indicating nanoparticle formation as the gelatin encapsulated the polystyrene beads. Excess acetone (5mL) was added and then the beaker was moved to a stir plate without heat for 5 min (600 rpm). Glutaraldehyde (25 wt% in water, 250 μL) was added in a single injection and the colloidal suspension was left to crosslink over 24 hr. After crosslinking, the sample was concentrated by rotary evaporation using an RV 10 digital V (IKA; Wilmington, NC) to remove acetone until 25-30 mL of suspension at pH 3-4 remained over 40-60 min. An aliquot of the sample suspension (100 μL) was diluted into deionized water (900 μL) in a plastic cuvette and three measurements were taken to estimate the average diameter, size distribution and ZP of each sample using cumulants analysis with a Zetasizer-Nano-ZS (Malvern instruments; Southborough, UK). The remainder of the sample suspension was stored at room temperature.
Nanoparticle stability characterization
NPs fabricated from the initial diameter studies, were used to determine stability under room temperature storage, freeze/thaw cycle conditions, and under varying pH conditions. For the storage study, samples from each batch of NP formulation were stored at room temperature in distilled water and aliquots measured weekly for average diameter and PDI over the course of a month. For the freeze/ thaw study, each suspension was measured as above and frozen for 48 hr, and then thawed completely prior to measuring the diameter and PDI again to determine the effect of a single freeze/thaw cycle. A short 5-day study on the effect of a physiological pH range was also investigated. One molar hydrochloric acid and 0.5 M NaOH were used to adjust the NP suspensions from a baseline pH of 3-4 to the desired pH ranges of 1-2, 5-6, and 8-9. The average diameter, PDI, and ZP were measured daily for each of the pH ranges.
Nanoparticle imaging
Transmission electron microscopy (TEM) was used to determine the types of encapsulation present in the NP suspensions. A blue FD and C dye was used to stain the gelatin during fabrication just prior to the addition of drop-wise acetone to aid sample visualization. The NPs were dehydrated in 100% ethanol for 4 hr, filtered onto a nitrocellulose membrane, and dried with a LADD critical point dryer overnight. The NPs were stained with lead citrate prior to imaging with a JEOL JEM-1400 Transmission Electron Microscope (JEOL Ltd.; Japan).
Statistical analysis
One-way ANOVA analysis was used to determine whether significant differences existed between sample groups. When significance was present, Tukey’s HSD Test was used to reveal which samples were significantly different. JMP Pro Software (v.12.0.1, SAS Institute Inc., Cary, NC) was used for all statistical analysis. Data are reported as averages ± standard error with three replicates.
Results
Initial nanoparticle characterization
For initial NP fabrication, two polystyrene bead sizes and two types of gelatin were used for encapsulation. The basic properties of the 100 nm and 200 nm polystyrene beads designated as PS100 and PS200, respectively, in deionized water at pH 5 are shown below in Table 1. If the encapsulated material diameter closely matched the empty NP diameter, then the encapsulated NP is considered matched for the purposes of this work. Sigma Type A 300 Bloom gelatin is designated as G300 while Vyse Type B 275 Bloom gelatin is designated as G275. G300 and G275 designated empty NP fabrications, G300PS200 and G275PS100 were matched encapsulated NPs, and G300PS100 and G275PS200 were non-matched encapsulated NPs. Three batches of each NP type were averaged to calculate the as-fabricated NP parameters in Table 2.
| Polystyrene Bead | Average Diameter (nm) | Average PDI | Average ZP (mV) |
|---|---|---|---|
| PS 100 | 108.07 ± 1.70 | 0.022 ± 0.021 | -57.50 ± 0.72 |
| PS 200 | 215.63 ± 0.93 | 0.019 ± 0.005 | -56.80 ± 0.82 |
Table 1: Properties of 100 nm (PS100) and 200 nm (PS200) polystyrene beads. One sample was measured three times for each polystyrene bead type and values are reported as the average ± standard deviation (n=3 measurements).
| Nanoparticle Type | Average Diameter (nm) | Average PDI | Average ZP (mV) |
|---|---|---|---|
| G300 | 253.16 ± 15.5 | 0.071 ± 0.013 | 24.59 ± 1.6 |
| G300PS100 | 252.26 ± 27.8 | 0.11 ± 0.035* | 20.33 ± 5.1§ |
| G300PS200 | 226.67 ± 9.05• | 0.071 ± 0.018 | 27.23 ± 2.8 |
| G275 | 83.43 ± 23.0 † | 0.22 ± 0.056 | 31.74 ± 3.8 |
| G275PS100 | 104.63 ± 24.5 | 0.17 ± 0.046 | 28.94 ± 4.2 ǂ |
| G275PS200 | 122.66 ± 7.36 | 0.22 ± 0.14 | 40.59 ± 1.9 |
Table 2: As fabricated properties of NP types measured using DLS. Sigma Type A 300 Bloom (G300), and Vyse Type B 275 Bloom (G275) gelatin were used to encapsulate 100 nm (PS100) and 200 nm (PS200) polystyrene beads. Three samples of each fabrication type were measured three times each and the values are reported as the average ± standard deviation (n=3). The asterisk, section sign, diamond, cross, and double dagger (*, §,•,†,‡) indicate significant differences between fabrications (p<0.05).
For the three Sigma Type A gelatin NP types, G300PS200 NPs were significantly smaller in diameter (~226 nm) from either the G300 and G300PS100 NPs (~253 nm), despite encapsulating a larger material (Table 1), •p<0.05). The G300PS100 NP PDI was significantly higher than the G300 and G300PS200 PDI values (0.11 versus 0.071, respectively), reflecting a larger size distribution (*p<0.05). The G300PS100 ZP (~20mV) was statistically smaller than that of G300 (~24 mV), indicating the particles were less positively charged when encapsulating the unmatched PS100 material (§p<0.05).
For the three Vyse Type B NP types, the G275 NPs were significantly smaller in diameter (~83 nm) from the G275PS200 NPs (~122 nm) (†p < 0.05) and nearly significantly smaller from the G275PS100 NPs (~104 nm) (p=0.08), suggesting the addition of an encapsulation material increased the size of the gelatin type B NPs. None of the PDI values were significantly different for G275 (~0.22), G275PS100 (~0.17) or G275PS200 (~0.22). G275PS100 was significantly smaller in ZP (~28 mV) from G275 (~31 mV) and G275PS200 (~40 mV) (‡p < 0.05), suggesting these encapsulated G275PS100 particles are less stable.
Nanoparticle stability
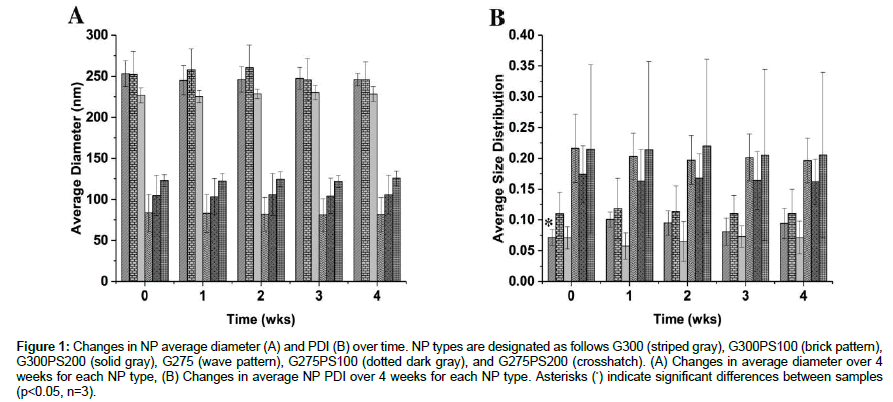
Once fabricated, the same batches were characterized over a 4-week period to determine the stability of the NPs in distilled water (pH 3-4) at room temperature for storage conditions (Figure 1). For the purposes of this paper, stability is defined as insignificant changes in diameter, size distribution (PDI), or ZP. Overall, none of the NP types changed significantly in size or PDI over the 4-week period.
Figure 1: Changes in NP average diameter (A) and PDI (B) over time. NP types are designated as follows G300 (striped gray), G300PS100 (brick pattern), G300PS200 (solid gray), G275 (wave pattern), G275PS100 (dotted dark gray), and G275PS200 (crosshatch). (A) Changes in average diameter over 4 weeks for each NP type, (B) Changes in average NP PDI over 4 weeks for each NP type. Asterisks (*) indicate significant differences between samples (p<0.05, n=3).
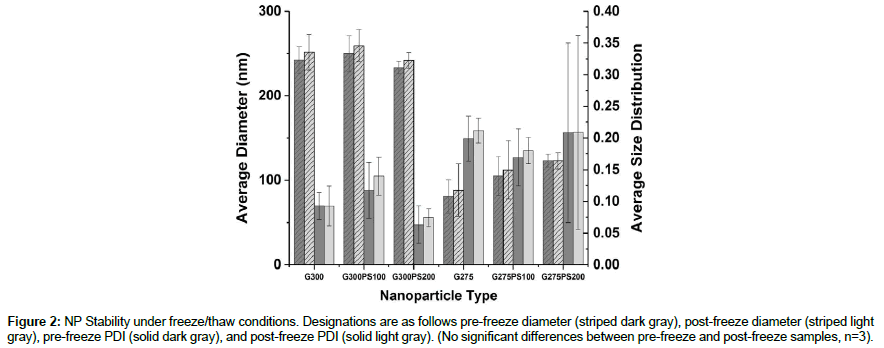
As shown in Figure 2, the NP suspensions were cycled through one freeze/thaw in order to determine their stability under a standard storage condition. All of the NP types were able to survive the freeze/ thaw cycle without significant changes in diameter or PDI.
Figure 2: NP Stability under freeze/thaw conditions. Designations are as follows pre-freeze diameter (striped dark gray), post-freeze diameter (striped light gray), pre-freeze PDI (solid dark gray), and post-freeze PDI (solid light gray). (No significant differences between pre-freeze and post-freeze samples, n=3).
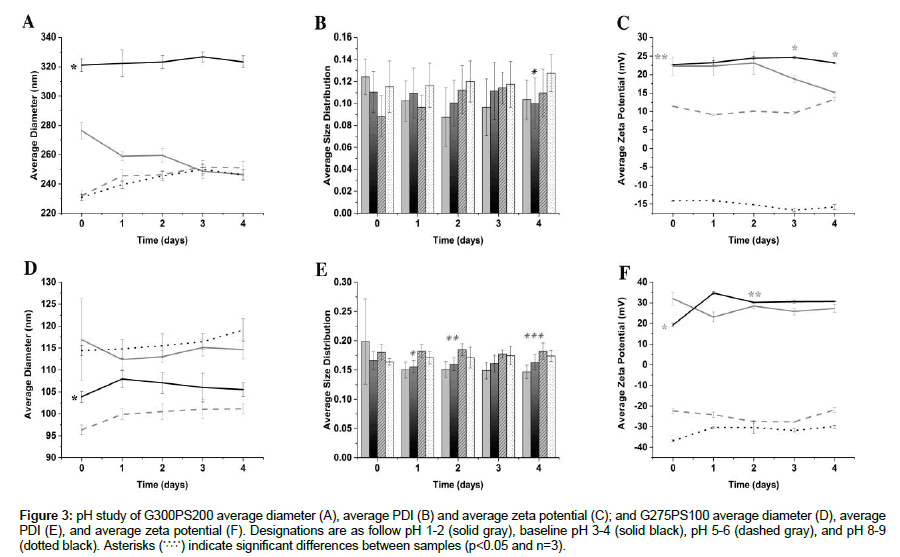
G300PS200 and G275PS100 were identified as the NP types with the preferred overall combination of initial average diameter close to the encapsulation material, initial average PDI between 0.1 and 0.2, and initial ZP close to +30 mV. These two matched NP types were further tested to determine stability over a physiological pH range of 1-9 during a 5-day-period. Both NP types had a baseline pH of 3-4 in distilled water prior to being adjusted to the desired pH values for the study (Figure 3).
Figure 3: pH study of G300PS200 average diameter (A), average PDI (B) and average zeta potential (C); and G275PS100 average diameter (D), average PDI (E), and average zeta potential (F). Designations are as follow pH 1-2 (solid gray), baseline pH 3-4 (solid black), pH 5-6 (dashed gray), and pH 8-9 (dotted black). Asterisks (*,*,*,*) indicate significant differences between samples (p<0.05 and n=3).
The average NP diameter, size distribution, and ZP for the G300PS200 NPs over a pH range of 1-9 are shown in Figure 3A-3C. The baseline pH 3-4 G300PS200 NP average diameter (Figure 3A, solid black line) was significantly larger in diameter throughout the 4-day-period than the G300PS200 diameters at the other pH values (*indicate p<0.05). There were no significant changes in size distribution until day 4 where the baseline pH 3-4 size distribution (Figure 3B, black solid bar) was significantly smaller than the pH 8-9 size distribution (Figure 3B, black dotted bar), indicating instability at the higher pH range (*indicate p <0.05). The NPs also maintain a larger positive charge at pH 3-4 for days 0,1,2 (Figure 3C, black solid line), which is significantly larger than that of pH 5-6 (dashed gray line) and 8-9 (black dotted line) by days 3 and 4, pH 3-4 ZP is significantly larger than that of all the other pH values (*indicates p<0.05).
The average NP diameter, PDI, and ZP for the G275PS100 NPs over a pH range of 1-9 are shown in Figure 3D-3F. The baseline pH 3-4 G275PS100 NP average diameter (Figure 3D, solid black line) was significantly larger in diameter than that of pH 5-6 and significantly smaller than that of pH 1-2 and pH 8-9 throughout the 4-day- period (*indicates p<0.05). The baseline pH 3-4 size distribution (Figure 3E, black solid bar) was significantly smaller than that of pH 5-6 and pH 8-9 (Figure 3E, dashed gray bar and black dotted bar, respectively) for day 1 (*indicates p<0.05) significantly smaller than that of pH 5-6 (dashed gray line) for day 2 (**indicates p<0.05) and significantly different from that of pH 1-2 (solid gray line) and 5-6 (dashed gray line) for day 4 (***indicates p<0.05). The NPs also maintain a significantly different positive charge at pH 3-4 for days 0,1,3,4 (Figure 3F, black solid line) than that of all of the other pH values (* indicates p<0.05). On day 2, pH 3-4 ZP is significantly larger than that pH 5-6 (dashed gray line) and 8-9 (dotted black line) indicating storage in acidic conditions causes a strong positive NP charge (**indicates p<0.05).
Nanoparticle imaging
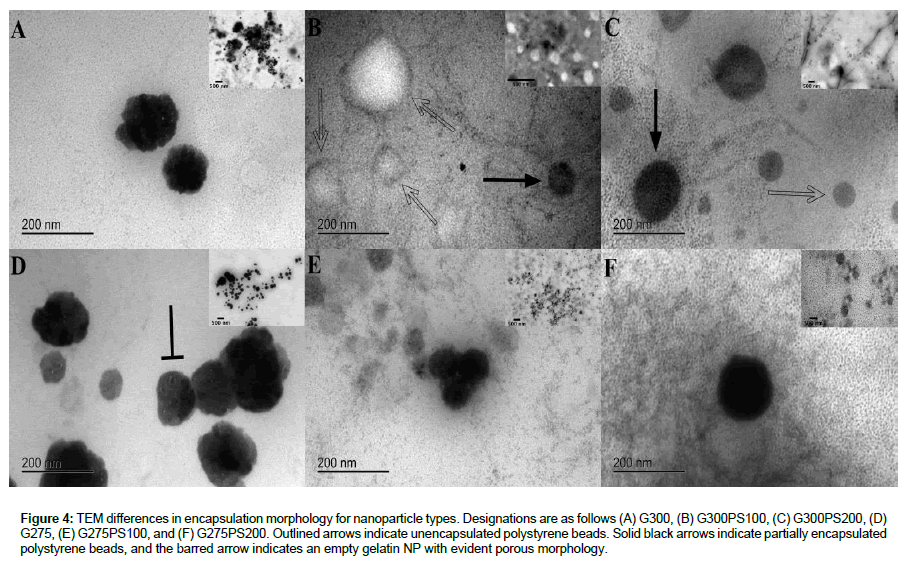
TEM imaging was used to determine whether the NP suspensions contained unencapsulated beads, partially encapsulated beads, completely encapsulated beads or, as expected, a mixture of all three types; as well as to confirm the Zetasizer dynamic light scattering size range measurements from Tables 1 and 2.
Figure 4 is a composite image of all NP types with insets showing the overall sample morphology. The expected range of NP sizes and types of encapsulation present were present, even with some coagulation artifact due to sample processing. The empty unencapsulated gelatin NPs were dark in color with a large size range, scalloped edges, and porous morphology (barred black arrow) as shown in Figure 4A and 4D. The unencapsulated beads on TEM imaging were smooth edged and lightest in color (Figure 4B and 4C outlined arrows), the partially encapsulated NPs were intermediate in color and still have fairly smooth edges (Figure 4B and 4C solid black arrows), and the fully encapsulated beads were darkest in color and have fuzzy edges (Figure 4E and 4F). Additionally, a smaller size range was observed when the matched encapsulation material is used as shown in the TEM images of G300PS200 and G275PS100 in Figure 4C and 4E, respectively and the insets.
Figure 4: TEM differences in encapsulation morphology for nanoparticle types. Designations are as follows (A) G300, (B) G300PS100, (C) G300PS200, (D) G275, (E) G275PS100, and (F) G275PS200. Outlined arrows indicate unencapsulated polystyrene beads. Solid black arrows indicate partially encapsulated polystyrene beads, and the barred arrow indicates an empty gelatin NP with evident porous morphology.
Discussion
Inclusion of an encapsulated material does affect the average diameter, size distribution, and ZP of the NPs (Table 2). When the material to be encapsulated was of similar size and shape to the empty NPs, the overall preferred combination of diameter, size distribution, and ZP was observed, as evidenced by the G300PS200 and G275PS100 encapsulated nanoparticles. The significant reduction in size observed with the matched G300PS200 NPs, despite having the larger encapsulation material, as well as the significantly higher size distribution seen with the unmatched G300PS100 NPs suggests that choosing an encapsulation material that closely matches the morphology of the empty gelatin NPs is a good strategy for controlling the fabrication process and leads to a 1:1 NP to encapsulation material ratio. This 1:1 ratio is further supported by the TEM results discussed below. Additionally, the matched material with its narrow distribution and ideal size parameters allows for consistency in NP in vitro release and cellular uptake studies. Using a material that does not match the size of the empty gelatin NPs led to more variation in encapsulation, causing a more variable average diameter, a larger average size distribution, and a more variable average ZP as seen with the G300PS100 and G275PS200 encapsulated nanoparticles types. Over time, this may lead to less stable nanoparticles. The average size of the encapsulated G275PS200 NPs (~122 nm) being smaller than the encapsulation material (200 nm polystyrene beads) can be attributed to the formulation including both partial encapsulations and empty encapsulations, leading to a smaller average size. Both G300PS100 and G275PS100 had the lowest ZP values, which may be due to the negatively charged PS1 particles having a wider size range than indicated by the manufacturer (Figure 4B) leading to variation in the positively charged gelatin shell thickness and therefore a lower overall ZP [5,22]. Regardless, all formulations had an average ZP sufficiently large enough to avoid aggregation or dissolution in suspension for the 4-week period as evidenced by the lack of significant changes in diameter and size distribution in Figure 1.
In general, adding a “matched” encapsulation material appears to minimize the variation in the NP fabrication parameters compared to the empty NP fabrication results seen with G300 and G275.
It was determined all NP batches were stable in distilled water (pH 3-4) at room temperature with regard to maintaining the same bulk average diameter and PDI up to 4 wk (Figure 1). For G300, the significant difference between week 0 and week 1 PDI values can be attributed to residual acetone evaporation. In addition, each NP suspension was also put through a freeze/thaw cycle to determine if freezing was a viable option to potentially extend NP storage life. This test revealed that each NP type was able to endure a freeze/thaw cycle without significant changes to the average diameter or PDI. As the NPs can be frozen to extend the product lifespan prior to use, the results from Figures 1 and 2 combined meets and exceed the product specification for the NPs to be stable in suspension at room temperature up to 1-month.
When G300PS200 and G275PS100 were tested under varying pH conditions, pH 3-4 served as the baseline for comparison of average diameter, size distribution, and ZP. The significant differences seen in the study can be attributed to the effect of pH on polymer chain size within the NP and the surface charge. As the pH moves away from the gelatin polymer pI (Type A pI~pH 7-9; Type B pI~pH 4.7-5.4), the hydration state and charge of gelatin molecules will change. When the pH is above or below the pI, the polymer chains will swell or collapse thus increasing or decreasing the average NP diameter, respectively [1,7,25]. Concurrently, the charge on the gelation molecules will increase, leading to increased NP repulsion as the pH moves away from the gelatin’s pI.
For G300PS200, the diameter of the NPs was consistently larger at pH 3-4 compared to the other pH values over the 4-day-period. Polymer chain collapse leading to a decreased diameter is expected at pH 1-2, which is below the pI of Type A gelatin, even with high NP charge. The smaller sizes of the NPs at the pH values close to the Type A gelatin pI at pH 5-6 and 8-9 can be attributed to these being the least stable NPs due to having low surface charge, leading to NP falling apart. There were no significant differences in size distribution until day 4 when the pH 3-4 PDI was significantly smaller than that of pH 8-9. This suggests that although the average diameter was significantly different over the entire 4-day-period, the overall spread of the NP sizes were similar for the first three days, but the effect of the higher pH eventually caused the pH 8-9 NPs to be more variable. A similar pattern where pH 3-4 is significantly different from both pH 5-6 and 8-9 is seen in Figure 3C as well. The pH 3-4 ZP is significantly different from pH 5-6 and 8-9 for days 0,1,2 and then significantly different from all pH values for days 3 and 4. Since pH 3-4 maintained the highest charge across all four days compared to the other pH values, this suggests that pH 5-6 and 8-9 are less stable due to the gelatin chains being less charged close to the Type A gelatin pI and that the NPs are able to survive in pH 1-2 for the first two days but eventually the acidic pH leads to a decrease in NP charge and then a decrease in average diameter as the NPs grow unstable and break apart. Ahsan and Rao’s work contains a possible explanation as to the effect of pH on NP charge. Ahsan and Rao found the peak zeta potential values for Type A and Type B gelatin were pH 4 and 3.5, respectively. They hypothesized the increase in zeta potential in both gelatin Type A and Type B to a peak value of ~20 mV in the pH range 3.5-4 is due to protonation of aspartic acid and glutamic acid. Lowering the pH below 3.5-4 lead to an unexpected decrease in zeta potential, which they potentially attributed to the ion-pair formation and Debye-Huckel screening [25].
For G275PS100, the diameter of the NPs was consistently smaller at pH 3-4 compared to pH 1-2 and 8-9 and larger than pH 5-6 over the 4-day-period. As the closest to the pI of the Type B gelatin (4.7- 5.4), the pH 5-6 diameter values represent polymer chains that are neither swollen nor collapsed. It is expected that the polymer chains will be swollen above the Type B gelatin’s pI at pH 8-9, leading to a larger diameter provided, as there is in this case, sufficient NP charge to avoid agglomeration. The pH 1-2 diameter is larger than the expected base on the predicted polymer chain collapse below the gelatin pI but may be the product of a slightly lower zeta potential at the more acidic pH, leading to the ion-pair formation and Debye- Huckel screening as previously mentioned. For size distribution, the increased variability seen with pH 1-2, 5-6, and 8-9 suggests they are less stable over the 4-day-period compared to that of pH 3-4. For the ZP, all the pH values are close to either +30 mV or -30 mV, suggesting these NPs are more stable at different pH values due to Type B gelatin used.
Our data agree with Ahsan and Rao as the overall peak ZP values were found for both gelatin Type A and B at pH 3-4 and storage of the NPs in this pH range led to the preferred combination of average diameter close to the encapsulation material size, small size distribution, and large ZP over the four-day-period [25]. This indicated both NP types G300PS200 and G275PS100 were stable at pH 3-4 but begin to fall apart as the suspension pH approaches each gelatin’s respective pI ranges or significant ion interactions come into play at extremely acidic or basic pH. The optimal storage pH for Type A and Type B gelatin NP suspensions was therefore at pH 3-4 and suggests the NPs would be ideal for delivery in the body, where the acidic pH of the stomach or neutral pH of the blood would facilitate the release of the encapsulated material.
The TEM imaging revealed that all three types of encapsulation were present in each NP sample and confirmed the DLS average size for each formulation (Table 2).
The 1:1 NP to encapsulation material ratio should reduce the need for multiple filtration steps to achieve the desired particle size range and PDIs, thus reducing the loss of encapsulated material during processing. All of these results taken together suggest matching the encapsulation material size to the initial empty gelatin NP size does result in a more uniform, stable particle for biological delivery.
Conclusion
The encapsulation system developed here demonstrates that different NP types can be consistently produced to meet desired specifications with matched encapsulation material NPs having the preferred overall parameters. This proves the encapsulation system is customizable to encapsulating different nanometer-sized materials and were stable both at room temperature and after freeze/thaw conditions for storage. The encapsulation system is most stable at pH 3-4 but begins to fall apart outside that pH range. This system shows promise for being easily adaptable for sustained delivery of biologics to patients due to the multiple types of encapsulation present in each formulation. While we have used a stable particle stand-in for initial fabrication and characterization of the basic encapsulation system, the next step will be to modify the system for different biomedical applications. Future studies include encapsulating spherical biologics such as viruses to determine their characteristic NP profiles and release profiles; lyophilization of different NP formulations to determine if an alternative dry formulation can be created; modification of the reactive groups on the gelatin polymer encapsulation shell for targeting purposes; and studying NP uptake and cytotoxicity for the cell populations of interest given different biomedical applications.
Acknowledgments
Authors would like to thank the VT Regenerative Medicine Interdisciplinary Graduate Education Program (IGEP) and the VT Biomedical Engineering and Mechanics Department for support, the Virginia-Maryland College of Veterinary Medicine Morphology Lab for their expertise with TEM imaging, and Andre Stevenson, Jr for his expertise on the double desolvation method for NP fabrication.
References
- Azarmi S, Huang Y, Chen H, McQuarrie S, Adams D, et al. (2006) Optimization of a two-step desolvation method for preparing gelatin nanoparticles and cell uptake studies in 143B osteosarcoma cancer cells. J Pharm Sci 9: 124-132.
- Dash S, Murthy PN, Nath L, Chowdhury P (2010) Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm 67: 217-223.
- Reis CP, Neufeld RJ, Ribeiro AJ, Veiga F (2006) Nanoencapsulation I methods for preparation of drug-loaded polymeric nanoparticles. Nanomed Nanotechnol 2: 8-21.
- Farokhzad OC, Langer R (2009) Impact of nanotechnology on drug delivery. ACS Nano 3: 16-20.
- Jahanshahi M, Babaei Z (2008) Protein nanoparticle: a unique system as drug delivery vehicles. Afr J Biotechnol 7: 4926-4934.
- Sahoo SK, Labhasetwar V (2003) Nanotech approaches to drug delivery and imaging. Drug Discov Today 8: 1112-1120.
- Weber C, Coester C, Kreuter J, Langer K (2000) Desolvation process and surface characterization of protein nanoparticles. Int J Pharm 194: 91-102.
- Mudshinge SR, Deore AB, Patil S, Bhalgat CM (2011) Nanoparticles: emerging carriers for drug delivery. Saudi Pharm J 19: 129-141.
- Mohanraj VJ, Chen Y (2006) Nanoparticles-a review. Trop J Pharm Res 5: 561-573.
- Roser M, Fischer D, Kissel T (1998) Surface-modified biodegradable albumin nano-and microspheres, II: effect of surface charges on in vitro phagocytosis and biodistribution in rats. Eur J Pharm Biopharm 46: 225-263.
- Shafer V, Von BH, Andreesen R, Steffan AM, Royer C, et al. (1992) Human immunodeficiency virus (HIV)-infected macrophages: a possibility for antiviral drug targeting. Pharm Res 9: 541-546.
- Sastry SV, Nyshadham JR, Fix JA (2000) Recent technological advances in oral drug delivery-a review. Pharm Sci Technolog Today 3: 138-145.
- Del EM, Urtti A (2008) Current and future ophthalmic drug delivery systems a shift to the posterior segment. Drug Discov Today 13: 135-143.
- Yadav R, Balasubramanian K (2016) Bioabsorbable engineered nanobiomaterials for antibacterial therapy In: Grumezescu, A editor Engineering of Nanobiomaterials: Application of Nanobiomaterials. 78-83.
- Kumari A, Yadav SK, Yadav SC (2010) Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf B 75: 1-18.
- Bhatia S (2016) Natural polymer drug delivery systems: nanoparticles, plants, and algae. 1st ed. Switzerland: Springer International Publishing.
- Shutava TG, Balkundi SS, Vangala P, Steffan JJ, Bigelow RL, et al. (2009) Layer-by-layer-coated gelatin nanoparticles as a vehicle for delivery of natural polyphenols. ACS Nano 3: 1877-1885.
- Brzoska M, Langer K, Coester C, Loitsch S, Wagner TO, et al. (2004) Incorporation of biodegradable nanoparticles into human airway epithelium cells-in vitro study of the suitability as a vehicle for drug or gene delivery in pulmonary diseases. Biochem Biophys Res Commun 318: 562-570.
- Coester CJ, Langer K, Briesen HV, Kreuter J (2000) Gelatin nanoparticles by two step desolvation-a new preparation method, surface modifications and cell uptake. J Microencapsul 17: 187-193.
- Leo E, Vandelli MA, Cameroni R, Forni F (1997) Doxorubicin-loaded gelatin nanoparticles stabilized by glutaraldehyde: involvement of the drug in the cross-linking process. Int J Pharm 155: 75-82.
- Jung T, Kamm W, Breitenbach A, Kaiserling E, Xiao JX, et al. (2000) Biodegradable nanoparticles for oral delivery of peptides: is there a role for polymers to affect mucosal uptake? Eur J Pharm Biopharm 50: 147-160.
- Gaihre B, Khil MS, Lee DR, Kim HY (2009) Gelatin-coated magnetic iron oxide nanoparticles as carrier system: drug loading and in vitro drug release study. Int J Pharm 365: 180-189.
- Narayani R, Rao KP (1993) Preparation, characterization, and in vitro stability of hydrophilic gelatin microspheres using a gelatin-methotrexate conjugate. Int J Pharm 95: 85-91.
- Stevenson AT, Jankus DJ, Tarshis MA, Whittington AR (2018) The correlation between gelatin macroscale differences and nanoparticle properties: providing insight into biopolymer variability. Nanoscale 10: 1094-1108.
- Ahsan SM, Rao CM (2017) The role of surface charge in the desolvation process of gelatin: implications in nanoparticle synthesis and modulation of drug release. Int J Nanomed 12: 795-808.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi