Research Article, J Nanomater Mol Nanotechnol Vol: 7 Issue: 2
The Brain Cells on the Carbon Nanotubes: Morphological and Functional Changes in Neurons and Glia
Won-Seok Lee1 and Bo-Eun Yoon1,2*
1Department of Molecular Biology, Dankook University, Cheonan 31116, Korea
2Department of Nanobiomedical science, Dankook University, Cheonan 31116, Korea
*Corresponding Author : Bo-Eun Yoon
Department of Molecular Biology and Department of Nanobiomedical science, Dankook University, Dandaero 119, Cheonan 31116, Korea
Tel: +82-41-550-3694
E-mail: boeunyoon@dankook.ac.kr
Received: March 26, 2018 Accepted: April 04, 2018 Published: April 10, 2018
Citation: Lee W, Yoon B (2018) The Brain Cells on the Carbon Nanotubes: Morphological and Functional Changes in Neurons and Glia. J Nanomater Mol Nanotechnol 7:2. doi: 10.4172/2324-8777.1000241
Abstract
Carbon Nanotubes (CNT) are promising material for research and medical application. Because of the electrochemical nature of CNT, it is considered as a potentially effective nanomaterial in neuroscience. By the way, properties of these CNT are dependent on how they are synthesized or which functional groups they have. As the nature of CNT varies, the effect on brain cells can have different features from cell to cell. Also, the diversity should be concerned to not only neurons also glia in the brain. Therefore, we focus on studies for understanding the functional and morphological changes of neurons and glia in the effects of CNT.
Keywords: Carbon nanotubes; Neurons; Glia; Astrocytes; Microglia
Abbreviations
CNT: Carbon Nanotubes; SW-CNT: Single- Walled CNT; MW-CNT: Multi-Walled CNT; CVD: Chemical Vapor Deposition; AP-MW-CNT: as-Prepared MW-CNT; EN: Ethlene Diamine; PABS: Poly-M-Amino Benzene Sulphonic Acid; Peis: Polyethyleneimines; PEG: Polyethylene Glycol; DRG: Dorsal Root Ganglia; SFM: Serum-Free Media; PA: Phosphatidylserine; Anx V: Annexin V; PI: Propidium Iodide; LDH: Lactate Dehydrogenase; Pscs: Postsynaptic Currents; FF: Form Factor; GFAP: Glial Fibrillary Acidic Protein; GFAP-Ir: GFAP Immunoreactivity; LPS: Lipopolysaccharide; Brdu: Bromodeoxyuridine; FCO: Frontal Cortex; ST: Striatum
Introduction and Backgrounds
Nanomaterials such as carbon nanotubes (CNT), hold promise for biomedical application, particularly in the nervous system [1,2]. CNT are mainly classified into two types depending on the number of layers: single-walled CNT (SW-CNT) and multi-walled CNT (MWCNT). SW-CNT consists of a single cylinder of graphene, whereas MW-CNT is formed from multiple coaxial cylinders [3,4]. These CNT can affect tissues and cells, causing lung toxicity, acting as a skin stimulus, and exerting cytotoxicity. However, the effects of CNT on neurons seem to have more positive effects, such as increased growth or better cell viability [5]. Because of their electrical properties, CNT offer advantages for electron transport [2]. Plane-layered CNT can modulate neuronal growth and neurite outgrowth on cultured cells and affect the electrical characteristics of neurons [6,7]. However, cytotoxicity can occur by MW-CNT on neurons [8].
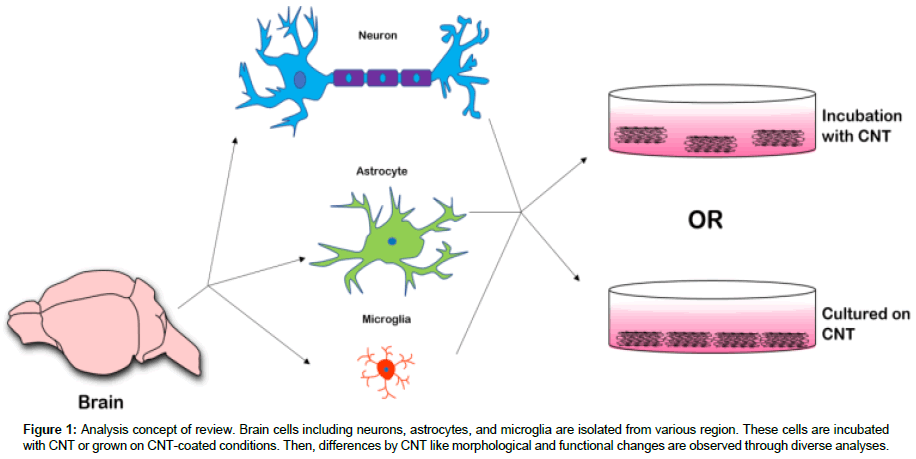
CNT can also affect other brain cells in addition to neurons, astrocytes, which is the most abundant glial cell type [1,9]. In addition, CNT inhibits microglia-mediated inflammatory responses or can be degraded by microglia through internalization [10]. The reason that CNT has a different effect on each cell is the type and functionalization method [11]. SW-CNT and MW-CNT can be manufactured by using several processes, including laser vaporization, electric arc vaporization, gas phase catalytic growth from carbon monoxide, and chemical vapor deposition (CVD) from hydrocarbons [5]. There are various functionalization groups as well as various methods of synthesizing CNT. Since CNT are mostly insoluble and cytotoxic, it is important to modify them through the functional groups such as carboxyl groups or ethylene diamines [6]. Functionalized CNT are reduced in cytotoxicity and biologically available [5]. Finally, we speculate on the effects of neurons and glial cells by different type of CNT (Figure 1) and describe some of the obstacles to overcome.
Figure 1: Analysis concept of review. Brain cells including neurons, astrocytes, and microglia are isolated from various region. These cells are incubated with CNT or grown on CNT-coated conditions. Then, differences by CNT like morphological and functional changes are observed through diverse analyses.
Changes in Neuronal Growth and Shape
Because neurons exchange large amounts of information with other neurons or glial cells, proper neuronal growth and process formation are crucial to brain function [12]. Therefore, it is important to understand what changes in neurons are made by CNT and whether these changes can have positive effects. Neurons grown on multi-walled carbon nanotubes functionalized with the aldehyde 4-hydroxynonenal (4-HNE) that can lead to increase of intracellular Ca2+ levels [13] and modify cytoskeletons [14], signaling mechanisms that modulate neurite outgrowth in cultured embryonic hippocampal neurons [15] had more delicate neuronal processes than neurons grown on control nanotubes. Whereas neurons grown on nonfunctionalized MW-CNT generated only one or two neurites, those grown on 4-HNE-functionalized nanotubes elaborated 4-6 neurites. Total neurite length was increased more than two-fold and number of branches/neurite were increased approximately three-fold, in neurons grown on 4-HNE-MW-CNT.
This means a remarkable influence of MW-CNT, functionalized with 4-HNE on neurite outgrowth and establish the suitability of using chemically modified carbon nanotubes as a tool for studying and manipulating neurite outgrowth [16,17]. Conversely, as prepared- MW-CNT was not as good a substrate as MW-CNT-PEI, but allowed nerve growth characterized by the presence of growth cone, neurite outgrowth, and branching. Nanotubes diminished the initiation of neurite outgrowth as characterized by the reduction in the numbers of growth cones and neurites per neuron as well as neurite branching. Interestingly, no difference in mean neurite length was observed between cultured neurons on PEI and AP-MW-CNT, indicating that MW-CNT did not affect the length of neurites that succeeded in their outgrowth.
Next whether the surface charge of the MW-CNT can modulate the neurite outgrowth and branching was studied. To approach this issue, neurons are cultivated on MW-CNT functionalized with polym- aminobenzene sulfonic acid (PABS) or ethylenediamine (EN). Although there was no difference in neurite number per neuron among cells grown in differentially functionalized MW-CNT, the average length of neurites was longer for neurons cultured onto MWCNT- EN substrates, which are positively charged, than on negatively charged MW-CNT-PABS or MW-CNT-COOH substrates. Neurons grown on MW-CNT-PABS or MW-CNT-EN substrates had larger growth cones than neurons grown on MW-CNT-COOH. In culture media (pH=7.35), the carboxyl groups of MW-CNT-COOH are deprotonated (pKa∼4.5) and become negatively charged whereas MW-CNT-EN is positively charged due to the high pKa of amine groups (pKa∼9.9). MW-CNT-PABS, which contains both amine and sulfonic groups, is almost an amphoteric ion at pH 7.35 and thus has similar effects on neurite growth as when neurons are plated on APMW- CNT.
The results indicate that the surface charge of the MW-CNT positively forms a larger number of growth cones, the mean neurite length is longer and neurite branching is more elaborate compared with neutral or negatively charged MW-CNT [6]. The effect of MWCNT on regenerative axonal growth of dorsal root ganglia (DRG) neurons, measuring the number of axon branches and axon length following treatment with 10 μg/ml of MW-CNT was also tested [18]. Significant differences between cells incubated with MW-CNT and the vehicle alone. DRG neurons incubated with MW-CNT exhibited hampered neurite outgrowth with fewer processes and branches. The mean length of neurites decreased in neurons incubated with MWCNT. The number of neurons treated with vehicle only was 330 ± 23 μm, and that of neurons treated with MW-CNT was 69 ± 10 μm.
Both the lengths of axons and the number of branches were greatly reduced by this concentration of MW-CNT. A subsequent concentration-response analysis confirmed that effects were greatest at 10 μg /ml, but showed that axon length and number of branches were also significantly reduced by treatment with 1 μg /ml MW-CNT; at a lower dose (0.1 μg /ml), MW-CNT had no significant effect. Moreover, when primary neurons were incubated with MW-CNT at different concentrations (0.1~10 μg /ml), the DRG neurons showed a dose-depended deficiency in their regenerative ability. Interestingly, the treatment with MW-CNT lowered the elongation and branching of axons, but did not generate apoptosis of those cells. They suggest that exposure to unmodified MW-CNT may have harmful effect on regenerative axon growth and may potentially trigger axonal pathology [18]. To investigate the application of different types of CNT to neurons, single-walled carbon nanotubes (SW-CNT) added polyethylene glycol (PEG) to CNT to make them soluble in aqueous solution which increases their biocompatibility. And then, SW-CNT was produced with thickness of 10nm, 30nm and 60nm which had conductivities of 0.3, 28 and 42 S/cm, respectively. Roughness also increased as thickness increased [7,19].
The total number of neurites generated by cell cycling of each neuron remained the same, regardless of the conductivity of the CNT. Although the lengths of neurites were not significantly different between neurons grown on coverslips coated with PEI, a smoother surface used as a control, and those grown on any SW-CNT, the total outgrowth, combined length of all processes and their branches were greatest when grown on 10-nm SW-CNT-PEG. This suggests that the properties of certain SW-CNT-PEG promote neurite outgrowth compared to those of other SW-CNT films. The number of neurites showed little change among all SW-CNT, but total outgrowth increased, with mean neurite length exhibiting the greatest increase on 10-nm SW-CNT. Moreover, the number of growth cones was greatest for neurons grown on 60-nm CNT compared with other CNT, whereas cell body area was highest for neurons grown on 10- and 30-nm CNT. There are no differences in the number of branches among SW-CNT with different thicknesses.
The straightness of neurites, measured as the ratio of the distance from the starting point to the end point of the process, was also unaffected by CNT [7]. These results have many meanings. First, it was confirmed that if the CNT were not functionalized, they could adversely affect neuronal cells. The axon regeneration ability of DRG neurons was inhibited as well as neuronal outgrowth was inhibited [18]. However, unmodified MW-CNT actively forms the process, increasing neurite outgrowth. Of course, it does not affect the length of the neurite [6,18]. If CNT are chemically functionalized in various ways such as surface charge is positive [6] or functionalized with 4-HNE [18], they will increase the length of neurite, increase growth cones and neuronal outgrowth [6,18]. In addition, morphological changes of neurons due to the conductivity of CNT were also confirmed [7]. Therefore, proper functionalization of CNT can lead to positive effects such as increasing outgrowth in neuron and forming more processes through branching or neurite.
The Viability and Electrical Properties of Neurons
Neuronal growth naturally induces changes in shape. Because structure and function are closely related, morphological changes can lead to functional changes. Due to functional changes, there was a change in the viability and electrical properties of the neurons. Accordingly, we next consider effects of CNT on the viability and electric properties of neurons, the latter being one of the most prominent features of this cell type. The effects of CNT on cell viability using NS-1 (neuronal screening) cells, a sub clone of the PC12 pheochromocytoma cell line, as a neuronal cell model. CNT, designated CNT-CA and CNT-D, were synthesized by CVD with Fe or Co. CNT-CA was >99% pure with 10-20nm diameter and 10-30 microns length. CNT-D contained <0.1% Fe as judged by inductively coupled plasma (ICP) spectroscopy. They were treated on cultured cells with two different MW-CNT-ones with a concentration of soluble MW-CNT of 190 ppm (CNT-CA) and the other with a concentration of 295 ppm (CNT-D)-and cell viability was measured after 24 and 48 hours. Compared with controls treated with serumfree media (SFM), cell viability was reduced by about 40% after 24- hour treatment and by almost 70% after 48 hours.
Cell viability assay was monitored through the translocation of phosphatidylserine (PS) phospholipids from the cytoplasmic surface to the extracellular surface of the cell membrane, an early apoptotic event, by double-stain flow cytometry using the protein annexin V (Anx V), which has a high affinity for PS, and propidium iodide (PI), which stains nuclei. These analyses revealed an increase in both apoptotic and necrotic cells. Consistent with this, CNT induced a sustained increase in the activity of the pro-apoptotic proteins, caspase-3 and -7. Their investigation of mitochondrial membrane depolarization using flow cytometry in conjunction with JC-1, which stains mitochondria, showed that CNT decreased membrane potential ~10-20% compared with controls, suggesting degradation of the mitochondrial membrane by CNT. Taken together, MWCNT toxicity occurred at a slower rate than the control and required relatively long exposures to induce cell death. Cell death occurs via apoptosis with serine translocation by 24 hours, mitochondrial membrane depolarization by 48 hours, DNA fragmentation by 48-72 hours and moderate caspase 3 activation by 72 hours [8].
The effects of different concentrations (0.1, 1, 10, and 100 μg/ ml) of PEG-functionalized (SW-CNT-PEG) and non-functionalized SW-CNT on PC-12 cells was studied. SW-CNT was first coated with carboxyl groups. After they consecutively washed and dried, SOCl2 was added and SW-CNT was mixed with the CNT-COOH powders. This process generated COCl-functionalized SW-CNT, which were further mixed with PEG. The PEG functionalization served to convert SW-CNT from an insoluble to a soluble form. Cell viability was measured through mitochondrial function using MTT [3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide] and XTT [2,3-bis-(2-methoxy-4-nitro- 5-sulfophenyl)-2H-tetrazolium-5-carboxanilide] assays after treatment with SW-CNT or SW-CNT-PEG for 24 hours.
MTT assays revealed that both SW-CNT and SW-CNT-PEG induced a concentration-dependent decrease in mitochondrial activity. XTT assays also showed an increase in mitochondrial cytotoxicity following treatment with CNT, but only at the highest concentration tested (100 g/ml). SW-CNT-PEG was found to be significantly less toxic than uncoated SW-CNT in high concentration, suggesting that PEG-functionalized SW-CNT indicated a higher biocompatibility compared to the unmodified SW-CNT. Using lactate dehydrogenase (LDH) release to measure membrane damage, a characteristic of necrosis, they found that SW-CNT-PEG caused less LDH release than uncoated SW-CNT, indicating that SWCNT coated with PEG are less cytotoxic than uncoated SW-CNT. Significant LDH release was noted after 24 hours of exposure to 10- 100 μg/mL. Consistent with MTT and XTT assay, SW-CNT-PEG had a trend to decrease LDH release after functionalization, indicating the less cytotoxic effect of the SW-CNT-PEG nanotubes compared to the uncoated SW-CNT.
The lower cytotoxicity of SW-CNT-PEGs suggested that functionalization of carbon nanotubes with PEG appears to increase the biocompatibility of these nanomaterials. These mitochondrial effects and membrane damages would be explained by the surface modification of these nanotubes and their biological interaction with the cellular systems [20]. The influence of CNT on neuronal electrical signaling by using MW-CNT functionalized with pyrrolidine groups, which notably increases the solubility in organic solvents. Pyrrolidinefunctionalized MW-CNT were then dissolved in dimethylformamide, applied to coverslips, and heated at 350°C to eliminate the organic solvent, leaving a carbon network coating. Effects of the CNT surface was tested on the neural network activity of plated hippocampal neurons by measuring spontaneous postsynaptic currents (PSCs), an indicator of functional synapse formation, using single-cell patchclamp recordings.
The frequency of spontaneous PSCs in neurons grown on CNT was increased 6-fold compared with that for neurons grown on peptide-free borosilicate glass coverslips, used as a control. However, amplitude was not significantly different between CNT and control groups, a result that reflects a mixture of inhibitory and excitatory PSCs. To eliminate inhibitory synapse currents and provide an estimate of total excitatory PSCs, they performed measurements again at -40 mV. These results confirmed that there is no difference between controls and CNT-treated groups. The frequency of spontaneous PSCs is dependent on both action potentials and quantitative spontaneous release, and should primarily reflect firing of local neurons and enhancing effects of CNT on neural network function. Using current-clamp recordings, they measured resting membrane potential, and found that action potentials in neurons grown on CNT increased from 0.22 ± 0.01 to 1.34 ± 0.69 Hz, strongly suggesting that CNT greatly increase network electrical activity [21,22].
Taken together, it was confirmed that the effect on the viability of neurons differs depending on the type of CNT and how it was functionalized. When exposed to CNT too long [8] or at high concentrations [20], it was rather toxic to cells and inhibited cell viability. Therefore, it was confirmed that the proper concentration and exposure time are important when using these nanomaterials on neurons to expect positive effects of CNT. In addition, CNT also affects the electrical properties of neurons. Through spontaneous postsynaptic current measurement, CNT increases the activity of the neuronal network [21]. These results suggest that these effects are caused by biological interactions between the functionalized group of CNT and cells [6-8,16,20].
Morphological and Functional Changes in Astrocytes
Astrocytes in the brain interact with neurons; these interactions serve a number of functions, including regulation of the concentration of extracellular K+ ions. This relationship suggests that astrocytes are also likely to be affected by CNT [23]. As noted above, the effects of CNT of various thicknesses on neurons were confirmed8. Extending these findings, SW-CNT can affect astrocytes, which are poorly understood [1]. To study the effect of SW-CNT on astrocytes, SWCNT was covalently linked with PEG to create the soluble SWCNT- PEG form, and compared the effects of SW-CNT-PEG–coated coverslips on astrocyte morphological parameters, measured using the acetoxymethyl ester of calcein, with those of PEI-coated coverslips. Morphological parameters were measured using cell perimeters and area to calculate the form factor (FF), defined by the equation, FF=4π [area (μm2)]/[perimeter (μm)]. The 10-nm SW-CNT did not cause significant changes in cell area or perimeter values of astrocytes compared with PEI, but did significantly increase FF.
However, 30-and 60-nm SW-CNT did significantly increase cell area, but without changing perimeter values, resulting in a significant increase in the FF. Because glial fibrillary acidic protein (GFAP) contributes to astrocyte form and can inhibit their proliferation [24], changes in the morphology of astrocytes have been linked to changes in the level of GFAP, which can affect neuronal excitability. Following on these observations, changes in astrocytes induced by CNT were monitored by using immunocytochemistry. In these experiments, astrocytes were stained with the dipeptide, β-Ala-Lys conjugated to 7-amino-4-methylcoumarin-3-acetic acid (AMCA), the latter of which serves as a detector of the total area of cells because it localizes to the cytoplasm of astrocytes, after which, the density (average fluorescence intensity per pixel of the total cell area), content (density × total cell area) and occupancy (positive cell/total cell area) of GFAP immunoreactivity (GFAP-ir) was assessed. Density tended to decrease as the thickness of CNT increased, with the most significant difference observed at a thickness of 60 nm GFAP-ir content and occupancy were not significantly different between CNT and PEI; however, GFAP-ir occupancy was significantly increased for the 10- nm SW-CNT-PEG than for the 60-nm SW-CNT-PEG.
These results show that different thicknesses of SW-CNT have different effects on astrocytes, and are consistent with previous reports that changes in GFAP may affect proliferation as well as morphofunctional characteristics of astrocytes [25,26]. These researchers’ next cultured astrocytes on PEI- or CNT-coated coverslips, stained them with calcein and Hoechst 33342 so they were detectable, and exposed them to CNT for 4 hours or 4 days. These experiments showed that, regardless of the thickness of the CNT substrate, the number of live cells was higher than that observed following culture on a PEI substrate. Thus, the specific thickness of CNT modulates various properties of astrocytes in culture, reducing GFAP-ir and increasing adhesion and proliferation [7]. And then, how CNT cause other cellular changes depending on methodological differences in exposure conditions. Specifically, they cultured astrocytes on PEIcoated coverslips and then treated them with SW-CNT-PEG (5 μg/ ml), or cultured cells on coverslips coated with a 60-nm SW-CNTPEG substrate.
Variation in astrocyte GFAP-ir density, content and occupancy, and astrocyte cell area, perimeter and FF were monitored. Cell area and perimeter of astrocytes were increased by treatment with SWCNT, whereas FF was decreased. Finally, experiments designed to determine if GFAP modulates the proliferative characteristics of astrocytes showed more dead cells following treatment with SWCNT than after culturing cells on coverslips coated with SW-CNT. Taken together, these results indicate that differences in the thickness of SW-CNT not only affect neurons, they also affect astrocytes. They further confirm that CNT have different effects depending on whether cells are treated with exogenously added CNT or are cultured on a CNT substrate [24].
To investigate the application of different types of CNT to astrocytes as in neurons, whether MW-CNT alter the shape, function and/or GABA characteristics of astrocytes was tested. Highly soluble multi-walled carbon nanotubes functionalized with carboxyl group (MW-CNT-COOH of lengths ~50 and ~1000 nm (MW-CNT-50 and -1000) were prepared by CVD; the acid oxidative method. Scanning electron microscopy was used to determine whether MW-CNT affected astrocyte morphology, assessed by calculating a roundness factor. MW-CNT-50 increased the roundness factor of astrocytes compared with controls, whereas MW-CNT-1000 had no significant effect. Astrocytes can interact with surrounding cells by forming gap junctions [27].
In this context, an assessment of cell-to-cell interactions showed that growth on MW-CNT induced an increase in gap junctions compared with controls, an effect that was not significant different between MW-CNT-50 and MW-CNT-1000. Cell viability was also assessed by CCK-8 assay on days 1 and 4 after treatment. The viability of astrocytes was increased on day 1 following treatment with MW-CNT-50 or MW-CNT-1000 compared with controls, and saw an even greater effect of MW-CNT-1000 on day 4. A subsequent immunocytochemical examination showed that astrocytes that underwent morphological changes in response to MW-CNT exposure also differed in intracellular GABA-ir distribution.
Using GFAP expression as an indicator of damage (i.e., neuronal loss), they determined the effect of MW-CNT length on GFAP-ir. MW-CNT significantly increased GFAP-ir intensity in astrocytes compared with controls, although this effect was unaffected by differences in the length of MW-CNT. Thus, these results show that the length of MW-CNT may have different effects, altering not only morphological feature, but also functional properties [9].
Taken together, these results indicate that the type and functionalization of CNT have different effects on astrocytes. SWCNT affects not only the area of the astrocyte but also its shape and density [7]. In addition, astrocytes incubated with SW-CNT and grown on nanotubes showed differences [24]. SW-CNT as well as MW-CNT could affect astrocyte, resulting in both morphological and functional changes. By nanotube, astrocyte formed more process, increased cell to cell interaction and spread GABA distribution. Also, increased GFAP intensity decreases tissue damage and neuronal loss and demyelination [9]. Overall, astrocyte, as well as its effect on neurons, suggests that biological interactions by these nanomaterials have increased and have also been functionally altered. These effects proposed that astrocyte mitigates neuronal damage and may protect through interaction with other cells.
Microglial Immune Responses
Microglia, the representative immune cell of the brain, may also respond to nanomaterials. CNT are foreign substances and can cause immune reactions. Thus, whether CNT affect microglia, and if so how, should be seriously considered when using CNT as a drugdelivery system. First, the effects of MW-CNT on microglia function were tested. MW-CNT was synthesized through CVD [10,28,29]. Among the various competences of microglia, cell migration has been intensively studied because it is one of the main features of the immune response of microglia. To assess this, microglia was incubated with MW-CNT for 70 hours, and measured cell velocity and motility using time-lapse video microscopy. After 3.5 hours of incubation, MW-CNT had significantly decreased the cell-migration capacity of microglia compared with controls. The effects of CNT on microglia were investigated by inducing bacterial infection and assessing inhibitory effects of microglia on bacterial growth. Bacterial growth kinetics, calculated from growth curves, showed that MW-CNT had no effect at any concentration on microglia inhibition of bacterial growth, confirming that CNT interfere with microglia cell migration [10,28]. Possible effects of SW-CNT on microglia cell function were monitored by using the microglia cell lines, N9 and BV2, and SWCNT synthesized using the arc discharge technique [29-31].
Specifically, whether the cytotoxic properties of CNT reduce microglia activation, which causes neuronal damage, was assessed. In these experiments, microglia were activated with lipopolysaccharide (LPS), enriched from Gram-negative bacteria, and effects of CNT on microglia proliferation were studied using bromodeoxyuridine (BrdU) labeling. In the absence of LPS treatment, the percentage of BrdU-positive N9 cells increased over time under control conditions, but decreased following culture with SW-CNT. By contrast, LPS treatment sharply increased the number of BrdU-positive N9 cells compared with untreated controls, an effect that was significantly reduced in microglia cells cultured with SW-CNT. Consistent with this, they further found that the number of mitotic cells increased with time, reflecting the need for somatic cells to undergo mitosis in order to proliferate, but the percentage of mitotic microglial cells decreased following incubating with SW-CNT, with or without LPS treatment, compared with controls.
The cell cycle using N9 cells was examined, and found that SWCNT– treated microglia accumulated in G1 phase, an effect that was much larger in N9 cells treated with LPS than in controls. These results were supported by XTT assays, which showed that SW-CNT decreased the growth of both LPS-treated and -untreated microglia, although the effect was much greater in the case of microglia treated with LPS. Similar to the results of XTT assays, CCK-8 assays showed that SW-CNT decreased the viability of N9 cells compared with controls, an effect that was much greater for LPS-treated microglia. Moreover, SW-CNT induced a greater degree of apoptosis in LPSactivated N9 microglia compared with controls, as measured by testing the ratio of annexin-V–positive cells. As was the case for previous experiments, the proportion of microglial cells induced to undergo apoptosis was increased by LPS treatment.
These results indicate that SW-CNT inhibit the growth and proliferation of activated microglia by affecting their cell cycle or inducing apoptosis [29,31]. Microglia is responsible for locally specific cytotoxic responses to chemically functionalized CNT [11]. Various functionalized MW-CNT was used to monitor change of microglia by CNT, including pristine MW-CNT; carboxylated MW-CNT (ox- MW-CNT) [32,33]; amino-functionalized, oxidized MW-CNT (ox- MW-CNT-NH3+) [34], prepared by 1,3-dipolar cycloaddition reaction after an initial oxidation; amino-functionalized MW-CNT (MWCNT- NH3+) [35,36], prepared following the 1,3-dipolar cycloaddition reaction of pristine MW-CNT; and amino-functionalized MW-CNT (ox-MW-CNT-am-NH3+) [37]. Experiments were conducted on primary cultures of cells obtained from the frontal cortex (FCO) and striatum (ST) regions, and cell viability was measured by using LDH assays.
The MW-CNT used in this experiment was variously specified at a concentration 5-100 μg/mL. MW-CNT decreased the viability of mixed cultured cells from the ST region compared with mixed glia derived from the FCO region. To confirm the results from LDH assay, cell viability was also tested by propidium iodide/ 40,6-diamidino-2- phenylindole (PI/DAPI) staining. PI is an impermeable dye that can only stain dead cells but not living cells. Nuclei of all cells (live and dead) were counterstained by the permeable dye DAPI. Mixed glial cultures of both regions were incubated with MW-CNT at 10 μg/mL for 24 h. Similar to LDH results, only mixed glial cells dissociated from the ST were clearly PI positive, whereas FCO mixed glial cultures appeared mostly PI negative in comparison to both negative and positive controls.
DMSO was used as positive control in this experiment and shown to be toxic to both ST and FCO mixed glial cells. So, ST-derived mixed glial cultures seem to be more sensitive to MW-CNT exposure than FCO-derived mixed glial cultures. Immunostaining confirmed that microglia, which are CD11b/c-positive, were more abundant in the ST region than the FCO region. Microglia is known for their capacity to envelope foreign particles as a mechanism of maintaining brain homeostasis and protection against damage [38,39]. Mixed glial cultures were exposed to 10 μg/mL MW-CNT for 24 h followed by CD11b/c marker staining. High co-localization of the CD11b/c marker with cells that contained large amounts of internalized MWCNT was observed. CD11b/c positive cells appeared to engulf large amounts of nanotubes irrespective of MW-CNT type. Moreover, various MW-CNT internalized in microglia lessened cell viability to a greater extent at the ST site than at the FCO site, suggesting that observed region-specific cytotoxicity depends on microglia [11].
As was shown for neurons and astrocytes, the effects of CNT on microglia varied depending on the type of CNT and its functionalization. It has been reported that CNT mainly exert cytotoxic effects on microglia [10,11,29]. However, this does not mean that CNT-induced cytotoxicity has a completely negative effect, because it can also prevent neuronal damage caused by activated microglia and reduce damage from neuroinflammatory responses [40,41].
Conclusion
CNT have shown therapeutic effects in nanomedicine applications and have attracted attention as a feasible drug-delivery system. Their potential benefit in neuroscience is particularly noticeable [4,6]. In this review, we have discussed the varied effects of CNT on neurons and glia in the brain. We have emphasized the influence of CNT on astrocytes, the most abundant glial cell, and microglia, which are involved in immune responses in brain. These analyses have shown that CNT have differing action through their type and functional group on brain cells. For example, CNT affect the growth and viability of neurons, as well as their morphological properties, increasing the lengths of neurites and outgrowth of neurites, and decreasing axon length and number of branches [6,7,16,18]. Nanotubes work on not only the growth and viability of cells but also their electrical properties that reflect the fact that they are electrically conductive [22].
The various effects of CNT on astrocytes are also important. Using different nanotubes including SW-CNT and MW-CNT, researchers have reported distinct effects on astrocytes that reflect both CNT type and methodological differences. Morphologically, the major change in astrocytes produced by CNT is induction of a round shape [1,9,24]. As with neurons, the effects of CNT on astrocytes varied depending on how cells were exposed. For example, cytotoxicity was higher when cells were treated with exogenous CNT than when they were grown on a CNT substrate [24]. Functional characteristics were identified by monitoring GFAP, a representative marker of astrocytes and an indicator of neuronal damage or demyelination [9,24,26]. GFAPir was generally increased by CNT, suggesting that the effects of nanotubes on astrocytes could be used medically to mitigate neuronal loss or damage. Like neurons, astrocytes were differentially affected by CNT depending on their type and functionalization, as well as the method of cellular exposure [1,9,24-26].
Because microglia is a type of brain immune cell, it is important to investigate how CNT affect them. CNT have been shown to inhibit the growth and proliferation of microglia, at least in part by causing cell cycle arrest [29], indicating that CNT are primarily toxic towards microglia. However, this is not necessarily a negative effect, because activation of microglia or increases in their number may cause inflammation and neuronal damage [10]. This suggests that CNT could feasibly be used to reduce that activated microglia that can cause such brain damage [11,40,41]. In most cases of neurodegenerative diseases, brain cells including neuron, astrocyte and microglia are damaged and irreproducible [42]. However, changes by CNT increase the growth, viability and process formation of neurons and astrocytes. Thus, CNT are suggested that restore damaged brain cells and restore normal function. By reducing the migration and viability of microglia, CNT minimize loss of normal brain cells. In addition, CNT interact with various brain cells, producing effects that vary depending on the method of CNT synthesis and functional groups. These various influences are summarized in Table 1.
| CNT type | Synthesis | Functional groups | Cell type | Properties | References |
|---|---|---|---|---|---|
| MW-CNT | Catalytic decomposition | Chemically modified with 4-hydroxynonenal | Neuron | Biological moderation of neurons by CNTs: more extensive neurites and branching | [16] |
| MW-CNT | CVD | Chemically modified with COOH groups | Neuron | Dependence of neurite outgrowth on the outer charge of MW-CNTs | [6,21] |
| MW-CNT | CVD | Chemically modified with PABS | Neuron | ||
| MW-CNT | CVD | Chemically modified with EN | Neuron | ||
| MW-CNT | CVD | Functionalized with Fe, used as a single metal catalyst | Neuron | Concentration-dependent decrease in axonal growth | [18] |
| SW-CNT | CVD | Chemically modified with PEG | Neuron | Other effects depending on CNT thickness (0, 30, 60 nm); maximally increased outgrowth at 30 nm | [7] |
| MW-CNT | CVD | Functionalized with Fe or Co | Neuron | Cytotoxicity of MW-CNTs functionalized with Fe or Co | [8] |
| SW-CNT | CVD | Chemically modified with PEG | Neuron | Concentration-dependent reduction of cell viability by SW-CNTs | [20] |
| MW-CNT | CVD | Chemically modified with pyrrolidine groups | Neurons | Increased spontaneous synaptic activity and neuronal firing | [21,22] |
| SW-CNT | CVD | Chemically modified with PEG | Astrocyte | Increased area and decreased GFPA density with thickness; reduced cell viability with CNT treatment | [1] |
| MW-CNT | CVD | Chemically modified with COOH groups | Astrocyte | Increases in both cell viability and GFAP intensity | [9] |
| MW-CNT | CVD | As-prepared CNT | Microglia | Disturbed cell migration | [10,28] |
| SW-CNT | Direct current arc discharge | X | Microglia | Inhibition of microglia growth and proliferation | [29] |
| MW-CNT | Catalytic decomposition | Various functionalization methods (Refer to the text) |
Microglia | Difference in cytotoxicity depending on brain region | [11] |
| MW-CNT | Catalytic decomposition | Microglia | |||
| MW-CNT | Catalytic decomposition | Microglia | |||
| MW-CNT | Catalytic decomposition | Microglia |
Table 1: Summary of the effects of various CNTs.
Although the various findings discussed here suggest that CNT can have positive effects on brain cells, it is difficult to predict whether the effects of CNT are good or bad, because the data recently obtained are based on experiments on each cell type in isolation. When each cells are combined with other brain cells, such as neurons and astrocytes or neurons and microglia, the cells can interact with each other and form communities that can have totally different-and more realistic-responses to CNT than those reported using a single type of cell. Therefore, an important direction of future studies is to use cocultivation of several cell types to create an environment similar to that of cells in the brain. In addition, depending on the region of the brain, the same cells have different characteristics. So, it is necessary to study whether the cells by brain area are undergone different impacts by the CNT.
Acknowledgements
This research was supported by the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2016M3C7A1905074).
References
- Gottipati MK, Samuelson JJ, Kalinina I, Bekyarova E, Haddon RC, et al. (2013) Chemically functionalized single-walled carbon nanotube films modulate the morpho-functional and proliferative characteristics of astrocytes. Nano Lett 13: 4387-4392.
- Smart SK, Cassady AI, Lu GQ, Martin DJ (2006) The biocompatibility of carbon nanotubes. Carbon 44: 1034-1047.
- Baughman RH, Zakhidov AA, de Heer WA (2002) Carbon Nanotubes-the Route toward Applications. Science 297: 787-792.
- Eatemadi A, Daraee H, Karimkhanloo H, Kouhi M, Zarghami N, et al. (2014) Carbon nanotubes: properties, synthesis, purification, and medical applications. Nanoscale Res Lett 9: 393.
- Tran PA, Zhange L, Webster TJ (2009) Carbon nanofibers and carbon nanotubes in regenerative medicine. Adv Drug Deliv Rev 61: 1097-1114.
- Hu H, Ni Y, Montana V, Haddon RC, Parpura V (2004) Chemically functionalized carbon nanotubes as substrates for neuronal growth. Nano Lett 4: 507-511.
- Malarkey EB, Fisher KA, Bekyarova E, Liu W, Haddon RC, et al. (2009) Conductive single-walled carbon nanotube substrates modulate neuronal growth. Nano Lett 9: 264-268.
- Bang J, Yeyeodu S, Gilyazova N, Witherspoon S, Ibeanu G (2011) Effects of Carbon Nanotubes on a Neuronal cell model in vitro. Atlas J Biol 1: 70-77.
- Min JO, Kim SY, Shin US, Yoon BE (2015) Multi-walled carbon nanotubes change morpho-functional and GABA characteristics of mouse cortical astrocytes. J Nanobiotechnology 18: 92.
- Villegas JC, Álvarez-Montes L, Rodríguez-Fernández L, González J, Valiente R, et al. (2014) Multiwalled Carbon Nanotubes Hinder Microglia Function Interfering with Cell Migration and Phagocytosis. Adv Healthc Mater 3: 424-432.
- Bussy C, Al-Jamal KT, Boczkowski J, Lanone S, Prato M, et al. (2015) Microglia Determine Brain Region-Specific Neurotoxic Responses to Chemically Functionalized Carbon Nanotubes. ACS Nano 9: 7815-7830.
- Sainath R, Gallo G (2015) Cytoskeletal and signaling mechanisms of neurite formation. Cell Tissue Res 359: 267-278.
- Mark RJ, Lovell MA, Markesbery WR, Uchida K, Mattson MP (1997) A role for 4-hydroxynonenal, an aldehydic product of lipid peroxidation, in disruption of ion homeostasis and neuronal death induced by amyloid beta-peptide. J Neurochem 68: 255-264.
- Mattson MP (1988) Neurotransmitters in the regulation of neuronal cytoarchitecture. Brain Res 472: 179-212.
- Mattson MP, Kater SB (1987) Calcium regulation of neurite elongation and growth cone motility. J Neurosci 7: 4034-4043.
- Mattson MP, Haddon RC, Rao AM (2000) Molecular functionalization of carbon nanotubes and use as substrates for neuronal growth. J Mol Neurosci 14: 175-182.
- Andrews R, Jacques D, Rao AM, Derbyshire F, Qian D, et al. (1999) Continuous production of aligned carbon nanotubes: a step closer to commercial realization. Chem Phys Lett 303: 467-474.
- Wu D, Pak ES, Wingard CJ, Murashov AK (2012) Multi-walled carbon nanotubes inhibit regenerative axon growth of dorsal root ganglia neurons of mice. Neurosci Lett 507: 72-77.
- Bekyarova E, Ni Y, Malarkey EB, Montana V, McWilliams JL, et al. (2005) Applications of Carbon Nanotubes in Biotechnology and Biomedicine. J Biomed Nanotechnol 1: 3-17.
- Zhang Y, Xu Y, Li Z, Chen T, Lantz SM, et al. (2011) Mechanistic Toxicity Evaluation of Uncoated and PEGylated Single-Walled Carbon Nanotubes in Neuronal PC12 Cells. ACS Nano 5: 7020-7033.
- Sucapane A, Cellot G, Prato M, Giugliano M, Parpura V, et al. (2009) Interactions Between cultured Neurons and Carbon Nanotubes: a Nanoneuroscience Vignette. J Nanoneurosci 1: 10-16.
- Lovat V, Pantarotto D, Lagostena L, Cacciari B, Grandolfo M, et al. (2005) Carbon nanotube substrates boost neuronal electrical signaling. Nano Lett 5: 1107-1110.
- Leif Hertz (2014) Book review: “Glial physiology and pathophysiology” by Alexei Verkhratsky and Arthur Butt, Wiley-Blackwell, 2013. Front Syst Neurosci 8: 17.
- Manoj K, Gottipati EB, Michael B, Robert CH, Vladimir P (2014) Changes in the morphology and proliferation of astrocytes induced by two modalities of chemically functionalized single-walled carbon nanotubes are differentially mediated by glial fibrillary acidic protein. Nano Lett 14: 3720-3727.
- Gadea A, Schinelli S, Gallo V (2008) Endothelin-1 regulates astrocyte proliferation and reactive gliosis via a JNK/c-Jun signaling pathway. J Neurosci 28: 2394-2408.
- Vázquez R, Blanco E, Sánchez F, Juanes JA, Rubio M, et al. (2001) Characterization of GFAP expression and cell proliferation in the rat median eminence following hypophysectomy. Histol Histopathol 16: 1107-1116.
- Lee SH, Kim WT, Cornell-Bell AH, Sontheimer H (1994) Astrocytes exhibit regional specificity in gap-junction coupling. Glia 11: 315-325.
- Rodriguez-Fernandez L, Valiente R, Gonzalez J, Villegas JC, Fanarraga ML (2012) Multiwalled carbon nanotubes display microtubule biomimetic properties in vivo, enhancing microtubule assembly and stabilization. ACS Nano 6: 6614-6625.
- Li L, Zhang J, Yang Y, Wang Q, Gao L, et al. (2013) Single-wall carbon nanohorns inhibited activation of microglia induced by lipopolysaccharide through blocking of Sirt3. Nanoscale Res Lett 8: 100.
- Wang X, Katwa P, Podila R, Chen P, Ke PC, et al. (2011) Multi-walled carbon nanotube instillation impairs pulmonary function in C57BL/6 mice. Part Fibre Toxicol 18: 24.
- Nan L, Zhiyong W, Keke Z, Zujin S, Zhennan G, et al. (2010) Synthesis of single-wall carbon nanohorns by arc-discharge in air and their formation mechanism. Carbon 48: 1580-1585.
- Li SP, Wu W, Campidelli S, Sarnatskaia V, Prato M, et al. (2008) Adsorption of Carbon Nanotubes on Active Carbon Microparticles. Carbon 46: 1091-1095.
- Flahaut E, Laurent CH, Peigney A (2005) Catalytic CVD synthesis of double and triple-walled carbon nanotubes by the control of the catalyst preparation. Carbon 43: 375-383.
- Gaillard C, Cellot G, Li SP, Toma FM, Dumortier H, et al. (2009) Carbon Nanotubes Carrying Cell-Adhesion Peptides Do Not Interfere with Neuronal Functionality. Adv Mater 21: 2903-2908.
- Georgakilas V, Kordatos K, Prato M, Guldi DM, Holzinger M, et al. (2002) Organic Functionalization of Carbon Nanotubes. J Am Chem Soc 124: 760-761.
- Georgakilas V, Tagmatarchis N, Pantarotto D, Bianco A, Briand JP, et al. (2002) Amino Acid Functionalisation of Water Soluble Carbon Nanotubes. Chem Commun (Camb) 21: 3050-3051.
- Sun YP, Fu K, Lin Y, Huang W (2002) Functionalized Carbon Nanotubes: Properties and Applications. Acc Chem Res 35: 1096-1104.
- Neumann H, Kotter MR, Franklin RJM (2009) Debris Clearance by Microglia: An Essential Link between Degeneration and Regeneration. Brain 132: 288-295.
- Napoli I, Neumann H (2009) Microglial Clearance Function in Health and Disease. Neuroscience 158: 1030-1038.
- Kateb B, Van Handel M, Zhang L, Bronikowski MJ, Manohara H, et al. (2007) Internalization of MWCNTs by microglia: Possible application in immunotherapy of brain tumors. Neuro image 37: S9-17.
- Bussy C, Hadad C, Prato M, Bianco 3, Kostarelos K (2016) Intracellular degradation of chemically functionalized carbon nanotubes using a long-term primary microglial culture model. Nanoscale 8: 590-601.
- Amor S, Puentes F, Baker D, Valk PV (2010) Inflammation in neurodegenerative diseases. Immunology 129: 154-169.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi