Research Article, J Food Nutr Disord Vol: 6 Issue: 3
Antioxidant Activity of Natural Plant Extracts from Mate (Ilex paraguariensis), Lotus Plumule (Nelumbo nucifera Gaertn.) and Rhubarb (Rheum rhabarbarum L.)
Wu TW, Lee CC, Hsu WH, Hengel M and Shibamoto T*
Department of Environmental Toxicology, University of California, USA
*Corresponding Author : Takayuki Shibamoto
Department of Environmental Toxicology, University of California, Davis, CA 95616, USA
Tel: 530 752 4523
Fax: 530 752 3394
E-mail: tshibamoto@ucdavis.edu
Received: May 30, 2017 Accepted: June 09, 2017 Published: June 15, 2017
Citation: Wu TW, Lee CC, Hsu WH, Hengel M, Shibamoto T (2017) Antioxidant Activity of Natural Plant Extracts from Mate (Ilex paraguariensis), Lotus Plumule (Nelumbo nucifera Gaertn.) and Rhubarb (Rheum rhabarbarum L.). J Food Nutr Disord 6:3. doi: 10.4172/2324-9323.1000225
Abstract
The antioxidant activity of organic solvent extracts from green mate leaves (Ilex paraguariensis), lotus plumule (Nelumbo nucifera Gaertn.) and fresh rhubarb (Rheum rhabarbarum L.) stalks was investigated using four antioxidant assays. An extract obtained with 60% methanol solution from mate leaves exhibited antioxidant activity of 99.96 ± 1.20% at the level of 2000 g/mL (TBA assay) and of 81.80 ± 0.54% at the level of 125 g/mL (DPPH assay), respectively. In this fraction, five polyphenols, including chlorogenic acids, were identified by HPLC/MS analysis. Methanol extracts from lotus and rhubarb showed potent antioxidant activity of 98.45 ± 0.78% and 83. 93 ± 2.39%, respectively, at the level of 100 L/mL in a malonaldehyde/gas chromatography assay. Analysis of a hexane fraction of lotus and rhubarb identified several volatile antioxidants, such as 1-octen-3-ol and benzaldehyde. The results of the present study demonstrate that mate, lotus and rhubarb are excellent natural sources of antioxidants.
Keywords: Antioxidants; Lotus plumule; Mate tea; Rhubarb; Volatile chemicals
Abbreviations
BHT: Butylated Hydroxytoluene; DPPH: 1,1-Diphenyl-2- Picrylhydrazyl; FID: Flame Ionization Detector; GC/MS: Gas Chromatography/Mass Spectrometry; HPLC: High Performance Liquid Chromatography; MA/GC: Malonaldehyde/Gas Chromatography; NPD: Nitrogen Phosphorous Detector; SDS: Sodium Dodecyl Sulfate; TBA: Thiobarbituric Acid
Introduction
Throughout human evolution, people have survived against many diseases. Natural medicinal components of plants must have played an important role in their survival. Ancient era records speak of people searching for natural materials to protect themselves from various diseases. Through trial and error, it must have taken a long time for these ancient people to find safe natural medicinal plants for disease treatment. Many ancient documents exist describing the use of medicinal plants for the treatment of various diseases-a Sumerian clay slab (approx. 5000 years old), the Chinese book Pen T’Sao (2500 BC), the Ebers Papyrus (1550 BC), and others [1].
Since the development of modern chemistry in the late 19th century, many drugs have been synthesized and the focus on natural sources of medicinal compounds has decreased. Recently, however, due to the discovery of toxicity in some synthetic chemicals, the merits of medicinal compounds from non-toxic natural plants have come to be recognized once again.
It is well known that oxidative damage is initiated by reactive oxygen species (ROSs) and this subsequently causes various diseases [2]. Therefore, among the medicinal activities found in extracts and components of natural plants, antioxidant activity has been the most commonly and widely investigated over the last two decades. There are many reports on the preventive effects of natural antioxidants against various diseases, including eye diseases [3], HIV/AIDS [4], Alzheimer’s disease [5], cardiovascular disease [6], Parkinson’s disease [7], diabetes [8], and cancer [9]. We have been studying the antioxidant activities of various essences and components from natural plants, such as flavonoids from young green barley essences [10], medicinal herbs [11], essential oils [12], teas [13], and coffee [14].
In the present study, the investigation is focused on three commonly consumed natural plants-mate (Ilex paraguariensis ), lotus plumule (Nelumbo nucifera Gaertn. ), and rhubarb (Rheum rhabarbarum L. ), for their antioxidant activity to find how best to utilize these plants for medicinal purposes.
Materials and Methods
Reagents and chemicals
1,1-Diphenyl-2-picrylhydrazyl (DPPH) was purchased from TCI America (Portland, OR, USA). Cod liver oil (approx. 70% -3 fatty acid methyl esters) was bought from Sigma-Aldrich Co., Ltd. (St. Louis, MO, USA). Sodium dodecyl sulfate (SDS) was purchased from J. T. Baker Co., Ltd. (Phillipsburg, NJ, USA). Trizma hydrochloride and trizma base were bought from Sigma Chemical Co., Ltd. (St. Louis, MO, USA). All other reagents and solvents were purchased from reliable commercial sources, including Sigma-Aldrich Co. (St. Louis, MO, USA), Fisher Scientific Co., Ltd. (Fair Lawn, NJ, USA), and EMD Chemicals Inc. (Gibbstown, NJ, USA). Authentic volatile chemicals were gifts from TAKATA KORYO Co., Ltd. (Osaka, Japan).
Plant samples
Green mate leaves (Ilex paraguariensis ) were bought from Las Marias Co., Ltd. (Buenos Aires, Argentina). Dried lotus plumule (Nelumbo nucifera Gaertn. ) and fresh rhubarb (Rheum rhabarbarum L. ) stalks were purchased from local stores in Davis, CA, USA.
Sample preparation of mate leaves
Mate leaves (40 g) were extracted using a Soxhlet extractor with 500 mL hexane for 2 h to remove the chlorophyll. The hexane extract was condensed to 10 mL in volume using a rotary evaporator and then further condensed to 2 mL by purging under a purified nitrogen stream. The extract was stored at 5ºC. The residual leaves in the extractor were air-dried and then extracted for 2 h with 500 mL methanol using a Soxhlet extractor. After reducing the solvent and extract to approximately 10 mL in volume using a rotary evaporator, the extract was freeze dried at -4 6ºC and 0.05 mBar to yield 15.5 g of yellowish powder, which was kept at 5ºC. The 15.5 g of powder obtained above was fractionated into seven fractions using a 60 cmx5.5 cm i.d. glass column packed with XAD-2 resins (Aldrich Chemical Co. Inc., Milwaukee, WI, USA). A sample was eluted sequentially with a 1 L water/methanol solution (100/0, 80/20, 60/40, 80/20, 0/100) and 1 L acetone. Solvent was removed, first by rotary evaporator to approximately 10 mL in volume, then by a purified nitrogen stream to exactly 2 mL in volume. The extracts were kept at 5ºC.
Sample preparation of lotus plumule
Dried lotus plumule was ground into fine powder in a blender. The powder (40 g) was extracted using a Soxhlet extractor with 300 mL each of hexane, dichloromethane, ethyl acetate, and methanol for 6 h in series. Solvent was removed, first by rotary evaporator to approximately 10 mL in volume, then by a purified nitrogen stream to exactly 2 mL in volume. The extract samples were kept at 5ºC.
Sample preparation of rhubarb
Fresh rhubarb stalks (1 kg) were freeze dried and then blended to a white powder (13 g). The powder was extracted using a Soxhlet extractor with 300 mL each of hexane, dichloromethane, ethyl acetate, and methanol for 6 h in series. Solvent was removed, first by rotary evaporator to approximately 10 mL in volume, then by a purified nitrogen stream to exactly 2 mL in volume. The extracts were kept at 5ºC.
Antioxidant assays
The samples prepared from natural plants were tested for in vitro antioxidant assays according to previously reported methods [15]. All assays were performed in triplicate.
DPPH radical scavenging assay: This assay may be the most commonly and widely used for the investigation of antioxidant activity of plant essences [12]. One of the major drawbacks of this assay is, it is not applicable to a dark-colored sample because it uses a spectrophotometer and the UV absorption of the target sample overlaps with non-target substances [15]. In the present study, therefore, some plant extracts were diluted to lighten the color. The assay was then performed according to a previously reported method [16]. The test amounts of the samples used for this assay are shown in the Figures. This assay was used for the extracts from mate leaves, lotus plumule, and rhubarb. Antioxidant activity (%) was calculated using the following equation:
Antioxidant activity (%)=[(Absorbance of control-Absorbance of test sample)/Absorbance of control]*100
Thiobarbituric Acid (TBA) assay: This assay may be the most conventional method for antioxidant activity associated with the lipid peroxidation, which monitors the amount of malonaldehyde (MA) in a sample using a spectrophotometer [15]. This assay was conducted according to a previously reported method [17]. The test amounts of the samples used for this assay are shown in the Figures. This assay was used for extracts from mate leaves. Antioxidant activity (%) was calculated using the following equation:
Antioxidant activity (%)=[(Absorbance of control-Absorbance of test sample)/Absorbance of control]X100
Malonaldehyde/Gas Chromatography (MA/GC) assay: This assay also monitors the amounts of MA in a sample using a gas chromatograph. It is more specific and highly sensitive to MA than the TBA assay [15]. In the present study, the MA/GC assay was used for extracts from mate leaves, lotus plumule, and rhubarb. It was conducted according to a previously reported method [14] in the present study. The test amounts of the samples used are shown in the Figures. Antioxidant activity (%) was calculated using the equation used for the TBA assay.
Aldehyde/Carboxylic acid assay: This assay is mostly used in evaluating the strength of antioxidants against oxidation from an alkyl aldehyde to a corresponding acid. It is useful in examining the longterm effects of antioxidants, which can be used for things such as determining the shelf-life of foods and beverages [15]. Briefly, our analytic method is as follows. Each organic solvent extract (30 L/mL) was added to 1.94 mL dichloromethane solution containing 600 L hexanal solution (10 mg/mL), 100 L of undecane solution (4 mg/mL) as an internal standard, 10 L of 3% hydrogen peroxide and 10 mL of surfactant SDS in a 20-mL vial. The vials were kept at room temperature. The remaining hexanal was monitored by GC in the time intervals shown in Figure 5. This assay was used for extracts from mate leaves and lotus plumule.
Antioxidant activity (%) was calculated using the following equation:
Antioxidant activity (%)=[1-((Amount of Hexanal in Original Solution- Amount of Hexanal in test sample)/Amount of Hexanal in Original Solution)]*100
Instrumental
Spectrophotometric analysis: A Hewlett-Packard 8452 A diode array spectrophotometer was used with absorbance set at 517 nm and 532 nm for the DPPH and TBA assays, respectively.
Gas Chromatographic analysis: An Agilent Model 6890N GC equipped with a 30 m x 025 mm i.d. (df=0.25 m) bonded phase DB-1 capillary column and a flame ionization detector (FID) was used to monitor the amount of hexanal in extracts in the Aldehyde/Carboxylic Acid Assay.
An Agilent Model 6890N GC equipped with a 30 mx0.25 mm i.d. (df=0.25 m) DB, a nitrogen-phosphorous detector (NPD), was used to analyze the levels of MA in the samples for the MA/GC assay. The oven temperature was programmed to rise from 60ºC to 100ºC at 4ºC/min.
GC/MS analysis: An Agilent model 6890 GC interfaced to an Agilent 5973 Network Mass Selective Detector (Foster City, CA, USA) was used for mass spectral identification of the GC components in the hexane extracts from lotus plumule and rhubarb. The GC was equipped with a 30 mx0.25 mm i.d. (df=0.25 μm) DB-WAX bonded phase fused silica capillary column (J&W Scientific, Folsom, CA, USA). The oven temperature was held at 40ºC for 2 min and then programmed to rise to 200ºC at 2ºC/min. Identification of components was conducted by comparing their GC retention index and mass spectral fragmentation pattern with those of a standard chemical. The NIST02 mass spectral library (National Institute of Standards and Technology, Gaithersburg, MD, USA) was referred to for further confirmation of our identifications.
LC/MS analysis: Identification of chemicals in a fraction eluted with 60% methanol solution from mate was performed with an Agilent Model 1100 HPLC equipped with a 25 cmx4.6 mm i.d. (5 m) Phenomenex Luna C18 column and interfaced to an Applied Biosystems API 2000 MS/MS. Mobile phase A was 10 mm citric acid and mobile phase B was methanol. The gradient mode was initially set at A/B ratio of 85/15 from 0 to 5 min, and then linearly increased to 60/40 at 40 to 85 min. The flow rate was 1.0 mL/min. Chemicals were identified by comparing their retention times and MS fragmentation patterns with those of authentic chemicals.
Statistical analysis: Results are expressed as the mean ± SD (n=3). Data was analyzed by one-way ANOVA, followed by Dunnett’s test for separate comparisons with the standard. Differences were considered significant at a P value of <0.05.
Results and Discussion
Extracts from mate leaves
Mate (Ilex paraguariensis ), commonly named Mate or Yerba Mate is a tea-like beverage widely consumed in South American countries, such as Argentina, Brazil, Paraguay and Uruguay. Of these countries, Argentina is the largest producer, with an annual production of 280,000 tons. The annual production of mate around the world is estimated at $1 billion [18]. Mate has been used as a social drink and as a medicinal remedy in the countries listed above and has gradually spread into other New World countries, including the US. Traditionally, mate leaves were applied in poultices to combat anthrax poisoning and ulcers in native South American populations. They have also been used for the treatment of some diseases, such as hypocholesterolemia and geoatioritectuve [19]. Mate leaves reportedly protect DNA and LDL oxidation, suggesting that they have an antioxidant activity [20].
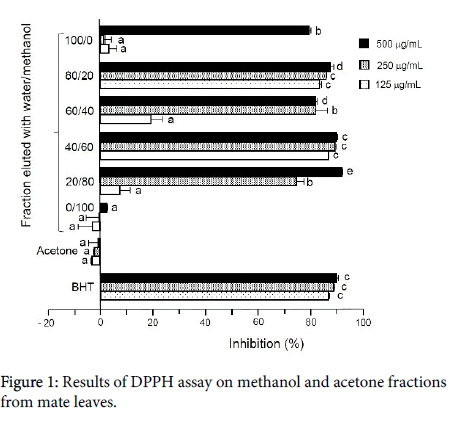
Figure 1 shows the results of the DPPH assay on the methanol and acetone fractions. The values are mean ± standard deviation (SD, n=3) exhibited nearly 95% dose-dependent antioxidant activity at all levels tested, indicating that this assay was valid. Methanol fractions showed strong antioxidant effects at the level of 125 g/mL, ranging from 81.80 ± 0.54% (water/methanol=60/40) to 91.6 ± 0.15% (water/ methanol=20/80). The results indicate that methanol extract of mate leaves contains potent antioxidants, in particular in the cases of those fractions eluted with a 60% or 80% methanol solution.
A hexane solution used to remove chlorophyll did not show any significant antioxidant activity (10.47 ± 4.47%) at the level of 20 L/mL in the DPPH assay, whereas it showed only slight activity (33.18 ± 6.28%) at the level of 20 L/mL in the TBA assay. Therefore, no further examination of this fraction was performed.
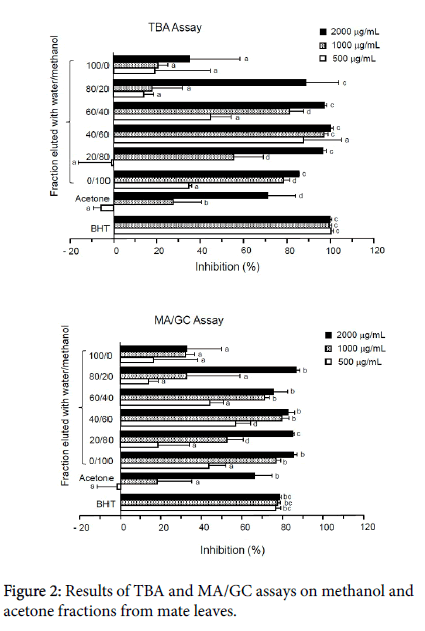
Figure 2 shows the results of the TBA and MA/GC assays on the methanol and acetone fractions. The values are mean ± SD (n=3). In the case of the TBA assay, a standard antioxidant, showed nearly 100% antioxidant activity at all levels tested, indicating that this assay was valid. All methanol fractions showed dose-dependent activity. Among methanol fractions, the fraction eluted with a 60% methanol solution exhibited the greatest antioxidant activity 99.96 ± 1.20% at 2000 g/mL, 97.19 ± 1.75% at 1000 g/mL and 67.37 ± 13.56% at 500 g/mL. The acetone fraction showed slight pro-oxidant activity, but the values are within the range of error.
In the case of the MA/GC assay, BHT showed nearly 80% activity at all levels tested, indicating that this assay is valid. All methanol fractions exhibited dose-dependent antioxidant activity. In particular, potent activities were demonstrated in the methanol fractions at the level of 2000 g/mL, ranging from 86.57 ± 2.00% (20%) to 75.48 ± 6.96% (40%).
Generally, values obtained with the MA/GC assay were about 10% less than those obtained from the TBA assay, which is reasonable because the TBA assay overestimates the amount of MA.15 However, overall responses from the two assays were quite consistent Figure 3.
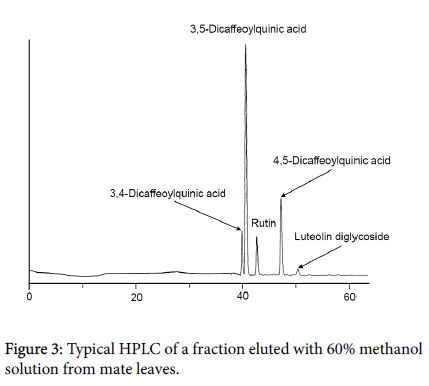
The fraction obtained with the 60% methanol solution exhibited consistent results across the three assays. Therefore, this fraction was further analyzed by LC/MS to investigate more precisely which chemicals contribute to mate’s antioxidant activity. Figure 3 shows a typical LC of this fraction. Chemicals positively identified were three chlorogenic acids (3,4- dicaffeoylquinic acid, 3,5-dicaffeoylquinic acid and 4,5-dicaffeoylquinic acid) and one flavonoid (rutin). Luteolin diglucoside (flavonoid) was only tentatively identified due to lack of an authentic chemical. All these are polyphenols. Chlorogenic acids are the major acids found in coffee beans and they are known to play an important role in the antioxidant activities of brewed coffee [21]. They were also found in mate tea [22]. A previous study reported that the antioxidant activity of mate is due to the presence of high levels of polyphenols [23].
The results of the Aldehyde/Carboxylic Acid Assay indicated that all methanol fractions, water fraction, and acetone fraction inhibited the oxidation of hexanal to hexanoic acid over 40 days at the level of 1000 g/mL. In the case of the control (without extracts), the amount of hexanal decreased by 77.35 ± 11.53% after 40 days. The results of all assays conducted indicate that mate leaves contain potent antioxidants.
Extracts from lotus plumule: Sacred lotus (Nelumbo nucifera Gaertn ) is an aquatic plant that belongs to the Nymphaeaceae family. Lotus originated from the banks of the river Nile in ancient Egypt and has spread widely and been cultivated throughout Asia, especially in Japan and China, for more than a thousand years. Most parts of the plant, including the leaves, flowers, seeds, and rhizomes are commonly consumed as food and used in traditional medicine [24]. Previous studies have reported that lotus plant possesses various medicinal effects, such as antiproliferative effects [25], antioxidative effects [26] and hypocholoesterolaemic effects [27]. However, there is only limited knowledge about “lotus plumule” the green spire in the middle of the mature lotus seed.
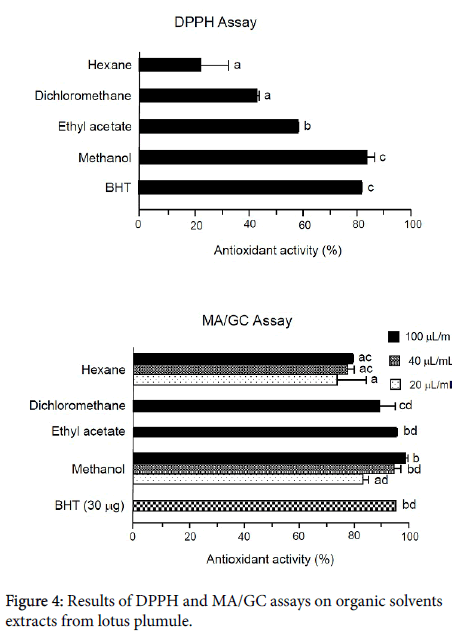
Figure 4 shows the results of the DPPH and MA/GC assays on fractions obtained from lotus plumule with organic solvents. The values are mean ± SD (n=3). The standard antioxidant BHT exhibited over 80% (81.79 ± 0.34%) antioxidant activity at the 30 g/mL level of 30 g/mL, indicating that this assay is valid. However, the color of the sample solutions was very dark and it was not possible to accurately measure antioxidant activities at levels higher than 30 L/ml by this assay. Nevertheless, the methanol fraction showed potent antioxidant activity (83.81 ± 2.87%). It is clear that lotus plumule extract contains antioxidants.
More specific details of antioxidant activity were confirmed by the MA/GC assay, the results of which are also shown in Figure 4. BHT exhibited 94.79 ± 0.01% antioxidant activity at the 30 g/mL level, indicating that this assay is also valid. The inhibitory effect of fractions ranged from 79.07 ± 0.02% (hexane) to 98.45 ± 0.78% (methanol) at the level of 100 L/mL. Hexane and methanol fractions exhibited doseresponse activity as shown in Figure 4.
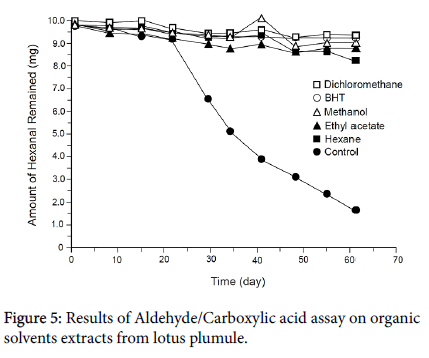
Figure 5 shows the results of the aldehyde/carboxylic acid assay on the organic solvent extracts tested at the level of 30 L/mL. The values are mean ± SD (n=3). Error bars did not appear due to the low values, ranging from 0.02 (control at 55 days) to 0.89 (control at 29 days). The standard antioxidant BHT was tested at the level of 30 g/mL and exhibited Inhibition of hexanal oxidation over 62 days, indicating that this assay is valid. After 62 days, 82.97 ± 0.18% hexanal was oxidized to hexanoic acid in the control. On the other hand, the amount of hexanal oxidized in the samples containing organic solvent extracts was 12.82 ± 0.10% by hexane extract, 4.76 ± 0.11% by dichloromethane extract, 5.65 ± 0.31% by ethyl acetate extract, and 6.59 ± 0.44% by methanol, suggesting that the organic solvent extracts from lotus plumule prevent hexanal oxidation over prolonged periods of time.
These results confirm the presence of antioxidants in lotus plumule. A previous study reported that edible lotus (Nelumbo nuficera ) extracts exhibited potent antioxidant activity due to the presence of polyphenols [26]. Therefore, the antioxidant activity of the lotus plumule extracts may be due to the presence of polyphenols, particularly in the methanol fraction. The hexane fraction, which contains volatile chemicals, was analyzed by GC/MS in the present study because there are no reports on volatile chemicals and their role in the antioxidant activity of lotus.
Extracts from Rhubarb: Rhubarb belongs to the Polygonaceae family. The vegetable rhubarb, Rheum rhabarbarum L. , grows in the wild in central Asia. In western societies, it has been used as an ingredient in jams, pies, and desserts because of its unique taste. Rheum officinale B. and Rheum palmatum L. are known as medicinal rhubarbs [28]. Medicinal rhubarb is known to have aid in combating several diseases, including diabetes [29], and HIV [30]. Moreover, a recent report indicates that rhubarb contains many polyphenols [31].
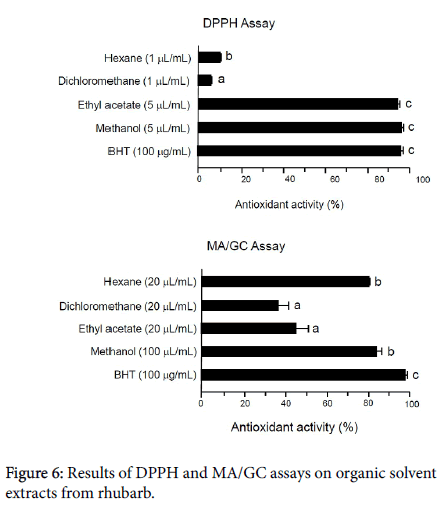
Figure 6 shows the results of the DPPH and MS/GC assays on the organic solvent extracts from rhubarb in the present study. The values are mean ± SD (n=3). In the case of the DPPH assay, standard antioxidant BHT exhibited 96.00 ± 0.94% antioxidant activity at the level of 100 g/mL, indicating that this assay is valid. Ethyl acetate and methanol extracts showed potent antioxidant activity at the 5 L/mL level, suppressing oxidation by 94.12 ± 1.17% and 96.00 ± 1.18%, respectively. On the other hand, the hexane and dichloromethane extracts did not show appreciable activity at the 1 L/mL level, and it was not possible to examine the antioxidant activity of these fractions at levels higher than 1 L/mL because the testing solution became too dark for this assay to be used.
In the case of MA/GC, the standard antioxidant BHT exhibited 97.84 ± 0.96% antioxidant activity at the level of 100 g/mL, indicating that this assay is valid.
The methanol fraction also showed potent antioxidant activity, 83.93 ± 2.39% at the 100 L/mL level. In addition, the hexane fraction showed relatively high antioxidant activity (80.34 ± 0.48%) at the 20 L/mL level. These results demonstrate that rhubarb extract, in particular in a methanol fraction, contains strong antioxidants.
Detailed analysis of the methanol fraction was not conducted for the present study, but it is obvious that this fraction contains many antioxidant polyphenols, as a previous study has reported [32]. The hexane fraction was analyzed by GC/MS because there are no reports on the analysis of volatile chemicals and their role in the antioxidant activity of rhubarb.
Volatile Chemicals Found in Lotus Plumule and Rhubarb: The major purpose of this analysis is to find volatile chemicals that contribute to the antioxidant activity of lotus and rhubarb. Table 1 shows the analytical results of the hexane fractions from lotus plumule and rhubarb in the present study. The table includes positively identified chemicals only (those for which authentic chemicals were available). There are many chemicals found in the present study for which previous studies have reported antioxidant activities. They are 1- octen-3-ol [32], pyrroles [33] furan [34], benzaldehyde [35], acetophenone [36], benzyl alcohol [37] and furanones [38]. The amounts of these volatile chemicals present in natural plants are relatively low, ranging from g/g to mg/g [39]. Even though the amounts of volatile antioxidants present in these natural plants are relatively low, their combined effect is comparable to that of polyphenols.
| GC peak area %1 | |||
|---|---|---|---|
| Chemical | I2 | Lotus | Rhubarb |
| Alcohols | |||
| 3-methyl-3-pentanol | 1117 | 1.41 | 1.61 |
| 3-hexanol | 1200 | 1.78 | 2.65 |
| 2-hexanol | 1224 | 2.42 | 3.99 |
| cyclopentanol | 1232 | 6.64 | 9.98 |
| pentanol | 1253 | 0.36 | 0.36 |
| 2-methylcyclopentanol 1st | 1316 | 0.39 | 0.56 |
| 2-methylcyclopentanol 2nd | 1341 | 1.34 | 0.49 |
| 3-methylcyclopentanol | 1343 | 2.26 | 3.99 |
| 1-octen-3-ol | 1454 | 4.47 | 0.68 |
| octanol | 1518 | 0.71 | 0.11 |
| Aldehydes and Ketones | |||
| acetone | 824 | 0.72 | 0.11 |
| butanal | 905 | -3 | 0.35 |
| pentanal | 976 | 0.67 | 0.09 |
| 3-methyl-2-pentanone | 1014 | 0.09 | 0.08 |
| 3-hexanone | 1048 | 0.54 | 0.44 |
| 2-hexanone | 1078 | 1.26 | 1.58 |
| hexanal | 1079 | 4.34 | 0.46 |
| 2-methylcyclopentanone | 1189 | 0.23 | 0.22 |
| 3-methylcyclopentanone | 1210 | 0.77 | 0.91 |
| octanal | 1287 | 0.35 | 0.10 |
| (E)-2-heptanal | 1321 | 1.23 | - |
| nonanal | 1392 | 0.99 | 0.36 |
| 3-octen-2-one | 1406 | 3.42 | 1.31 |
| (E)-2-octenal | 1427 | 0.62 | - |
| (E,E)-3,5-octadien-2-one | 1530 | 2.76 | - |
| (E,E)-2,4-nonadienal | 1658 | - | 0.80 |
| Acids and Esters | |||
| butyl acetate | 1071 | - | 0.21 |
| pentanoic acid | 1771 | 1.39 | 0.14 |
| hexanoic acid | 1867 | 16.76 | 9.56 |
| 3-hydroxy-2,2,4-trimethypentyl isobutanoate | 1876 | 4.96 | 3.40 |
| 2,2,4-trimetyl-1,3-pentanediol diisobutanoate | 1882 | - | 2.75 |
| heptanoic acid | 1970 | - | 0.69 |
| octanoic acid | 2065 | - | 0.09 |
| nonanoic acid | 2163 | - | 0.30 |
| Aromatic Compounds | |||
| benzaldehyde | 1512 | 0.61 | 0.40 |
| acetophenone | 1621 | 0.50 | 0.55 |
| trans-anetol | 1854 | 0.10 | - |
| benzyl alcohol | 1911 | 0.74 | 0.45 |
| phenetyl alcohol | 1951 | 1.13 | 0.12 |
| 1-indanone | 2006 | 0.92 | - |
| p-anisaldehyde | 2019 | 0.54 | 0.36 |
| Heterocyclic Compounds | |||
| 2,5-dimethyl-tetra-hydrofuran | 915 | 0.11 | 0.13 |
| 5-methylfurfural | 1533 | 1.19 | 4.75 |
| 2-acetylpyrrole | 1976 | - | 1.01 |
| 2-(hydroxyacetyl)furan | 2000 | 1.14 | 2.48 |
| pyrrole-2-carboxaldehyde | 2022 | - | 2.87 |
| dihydro-3-hydroxy-4,4-dimethyl-2(3H)-furanone | 2027 | - | 0.65 |
| 5-pentyl-2(5H)-furanone | 2071 | 0.91 | 1.48 |
| 3-methyl-1H-pyrrole-2-carboxaldehyde | 2100 | - | 0.94 |
| 2,3-dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one | 2269 | 0.16 | 4.59 |
| Hydrocarbons | |||
| hexane | 600 | - | 0.16 |
| cyclohexane | 701 | - | 2.63 |
| methylcyclohexane | 725 | - | 3.17 |
| tetradecane | 1400 | - | 0.07 |
3Not detected
Table 1: Chemicals identified in a hexane extract from lotus plumule and rhubarb.
The present study demonstrates that methanol extracts suggesting the possible presence of potent antioxidant polyphenols contribute to the antioxidant activity of mate, lotus, and rhubarb. In addition, some volatile chemicals found in the hexane extracts from lotus and rhubarb also possess antioxidant activity. Their antioxidant effects may be lower than those of polyphenols, but the numbers of volatile chemicals present in natural plant extracts are much greater than those of polyphenols.
Therefore, the combined activity of volatile antioxidants may be comparable to that of polyphenols. The results from the present study indicate that mate, lotus, and rhubarb are excellent sources of natural antioxidants.
References
- Petrovska BB (2012) Historical review of medicinal plants' usage. Pharmacogn Rev 6: 1-5.
- Parveen A, Akash MS, Rehman K, Kyunn WW (2016) Recent Investigations for Discovery of Natural Antioxidants: A Comprehensive Review. Crit Rev Eukaryot Gene Expr 26: 143-160.
- Akyol S, Ugurcu V, Balci M, Gurel A, Erden G, et al. (2014) Caffeic acid phenethyl ester: its protective role against certain major eye diseases. J Ocul Pharmacol Ther 30: 700-708.
- Singh G, Pai RS (2015) Dawn of antioxidants and immune modulators to stop HIV- progression and boost the immune system in HIV/AIDS patients: An updated comprehensive and critical review. Pharmacol Rep 67: 600-605.
- Wojtunik-Kulesza KA, Oniszczuk A, Oniszczuk T, Waksmundzka-Hajnos M (2016) The influence of common free radicals and antioxidants on development of Alzheimer's Disease. Biomed Pharmacother 78: 39-49.
- Pagliaro B, Santolamazza C, Simonelli F, Rubattu S (2015) Phytochemical Compounds and Protection from Cardiovascular Diseases: A State of the Art. Biomed Res Int 2015: 918069.
- Magalingam KB, Radhakrishnan AK, Haleagrahara N (2015) Protective Mechanisms of Flavonoids in Parkinson's Disease. Oxid Med Cell Longev 2015: 314560.
- Rahimi-Madiseh M, Malekpour-Tehrani A, Bahmani M, Rafieian-Kopaei M (2016) The research and development on the antioxidants in prevention of diabetic complications. Asian Pac J Trop Med 9: 825-831.
- Rafieian-Kopaei M, Nasri H (2015) On the occasion of world cancer day 2015; the possibility of cancer prevention or treatment with antioxidant: The ongoing cancer prevention researches. Int J Prev Med 6: 208.
- Kamiyama M, Shibamoto T (2015) Flavonoids with potent antioxidant activity found in young green barley leaves. J Agric Food Chem 60: 6260-6267.
- Lee KG, Shibamoto T (2001) Antioxidant property of aroma extract isolated from clove bud [Syzygiun aromaticum (L.) Merr. et Perry]. Food Chem 74: 443-448.
- Wei A, Shibamoto T (2010) Antioxidant/lipoxygenase inhibitory activities and chemical compositions of selected essential oils. J Agric Food Chem 58: 7218-7225.
- Nam S, Jang HW, Shibamoto T (2012) Antioxidant activities of extract from teas prepared from medicinal plants, Morus alba L., Camellia sinensis L., and Cudrania tricuspidata, and their volatile components. J Agric Food Chem 60: 9097-9105.
- Kamiyama M, Moon JK, Jang HW, Shibamoto T (2015) Role of degradation products of chlorogenic acid in the antioxidant activity of roasted coffee. J Agric Food Chem 63: 1996-2005.
- Moon JK, Shibamoto T (2009) Antioxidant assays for plant and food components. J Agric Food Chem 57: 1655-1666.
- Wei A, Shibamoto T (2007) Antioxidant activities and volatile constituents of various essential oils. J Agric Food Chem 55: 1737-1742.
- Lee KG, Shibamoto T (2002) Determination of antioxidant potential of volatile extracts isolated from various herbs and spices. J Agric Food Chem 50: 4947-4952.
- FAOSTAT (2017) Food and Agricultural Organization (FAO) Statistics Division.
- Filip R, Ferraro GE (2003) Researching on new species of "Mate": Ilex brevicuspis: phytochemical and pharmacology study. Eur J Nutr 42: 50-54.
- Bracesco N, Dell M, Rocha A, Behtash S, Menini T, et al. (2003) Antioxidant activity of a botanical extract preparation of Ilex paraguariensis: prevention of DNA double-strand breaks in Saccharonyces cerevisiae and human low-density lipoprotein oxidation. J Altern Complement Med 9: 379-387.
- Fujioka K, Shibamoto T (2007) Chlorogenic acid and caffeine contents in various commercial brewed coffees. Food Chem 106: 217-221.
- Bojic M, Simon Haas V, Saric D, Males Z (2013) Determination of Flavonoids, Phenolic Acids, and Xanthines in Mate Tea (Ilex paraguariensis St.-Hil.). J Anal Methods Chem 2013: 658596.
- Heck CI, Schmalko M, Gonzalez de Mejia E (2008) Effect of growing and drying conditions on the phenolic composition of mate teas (Ilex paraguariensis). J Agric Food Chem 56: 8394-8403.
- Mukherjee PK, Mukherjee D, Maji AK, Rai S, Heinrich M (2009) The sacred lotus (Nelumbo nucifera)-phytochemical and therapeutic profile. J Pharm Pharmacol 61: 407-422.
- Liu CP, Tsai WJ, Lin YL, Liao JF, Chen C Fu, et al. (2004) The extracts from nelumbo nucifera suppress cell cycle progression, cytokine genes expression, and cell proliferation in human peripheral blood mononuclear cells. Life Sci 75: 699-716.
- Hu M, Skibsted LH (2002) Antioxidative capacity of rhizome extract and rhizome knot extract of edible lotus (Nelumbo Nuficera). Food Chem 76: 327-333.
- Onishi E, Yamada K, Yamada T, Kaji K, Inoue H, et al. (1984) Comparative effects of crude drugs on serum lipids. Chem Pharm Bull (Tokyo) 32: 646-650.
- Dregus M, Engel KH (2003) Volatile constituents of uncooked rhubarb (Rheum rhabarbarum L.) stalks. J Agric Food Chem 51: 6530-6536.
- Chen H, Guo J, Pang B, Zhao L, Tong X (2015) Application of herbal medicines with bitter flavor and cold property on treating diabetes mellitus. Evid Based Complement Alternat Med 2015, 529491.
- Esposito F, Carli I, Del Vecchio C, Xu L, Corona A, et al. (2016) Sennoside A, derived from the traditional Chinese medicine plant Rheum L., is a new dual HIV-1 inhibitor effective on HIV-1 replication. Phytomedicine 23: 1383-1391.
- Hu L, Chen NN, Hu Q, Yang C, Yang QS, et al. (2014) An unusual piceatannol dimer from Rheum austral D. Don with antioxidant activity. Molecules 19: 11453-11464.
- Lee KG, Shibamoto T (2000) Antioxidant properties of aroma compounds isolated from soybeans and mung beans. J Agric Food Chem 48: 4290-4293.
- Yanagimoto K, Lee KG, Ochi H, Shibamoto T (2000) Antioxidative activity of heterocyclic compounds found in coffee volatiles produced by Maillard reaction. J Agric Food Chem 50: 5480-5484.
- Eiserich JP, Shibamoto T (1994) Antioxidative activity of volatile heterocyclic compounds. J Agric Food Chem 42: 1060-1063.
- Ullah I, Khan AL, Ali L, Khan AR, Wagas M, et al. (2015) Benzaldehyde as an insecticidal, antimicrobial, and antioxidant compound produced by photorhabdus temperate M1021. J Microbiol 53: 127-133.
- Mamadalieva NZ, Sharopov F, Satyal P, Azimova SS, Wink M (2017) Composition of the essential oils of three Uzbek Scutellaria species (Lamiaceae) and their antioxidant activities. Nat Prod Res 31: 1172-1176.
- Lee SJ, Lee KG (2005) Inhibitory effects of volatile antioxidants found in various beans on malonaldehyde formation in horse blood plasma. Food Chem Toxicol 43: 515-520.
- Miyake T, Shibamoto T (1998) Inhibition of malonaldehyde and acetaldehyde formation from blood plasma oxidation by naturally occurring antioxidants. J Agric Food Chem 46: 3694-3697.
- Lee KG, Shibamoto T (2001) Antioxidant activities of volatile components isolated from Eucalyptus species. J Sci Food Agric 81: 1573-1579.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi