Research Article, J Nanomater Mol Nanotechnol Vol: 7 Issue: 6
On the Potential for Tuning the Longitudinal Plasmon Band of a Distribution of Gold Nanorods Using a Tunable Laser
Shah A 1,2*, Jaeger M1,2, Harris-Birtill D2, deSouza NM1 and Bamber JC1,2
1Cancer Research UK Cancer Imaging Centre, The Institute of Cancer Research, London and The Royal Marsden NHS Foundation Trust, Sutton, London, SM2 5PT, United Kingdom.
2Joint Department of Physics, Division of Radiotherapy and Imaging, The Institute of Cancer Research, London and The Royal Marsden NHS Foundation Trust, Sutton, London, SM2 5PT, United Kingdom.
*Corresponding Author : Anant J Shah
Joint Department of Physics, The Institute of Cancer Research and the Royal Marsden NHS Foundation Trust, Sutton, London, United Kingdom
Tel: +74 01407475
E-mail: Anant.Shah@icr.ac.uk
Received: August 13, 2018 Accepted: September 11, 2018 Published: September 17, 2018
Citation: Shah AJ, Jaeger M, Harris-Birtill D, deSouza N, Bamber JC (2018) On the Potential for Tuning the Longitudinal Plasmon Band of a Distribution of Gold Nanorods Using a Tunable Laser. J Nanomater Mol Nanotechnol 7:6. doi: 10.4172/2324-8777.1000255
Abstract
Gold nanorods hold great potential for bio sensing, imaging and therapy applications. The seed-mediated approach is the most widely used technique for the synthesis of gold nanorods. However, batches of nanorods synthesized using this method have a broadened longitudinal plasmon (LP) peak and uncertainty in the wavelength of the peak. This paper describes a technique to tune the synthesized nanorod distributions by using laser pulses at specific wavelengths that either reduce the width of the population LP peak or control its position. The overlap between the LP peaks of pairs of batches of gold nanorods was successfully reduced by at least 9%. Batches of nanorods with a sharper LP peak and/or a more desirable LP resonance wavelength could eventually be utilized in molecular biosensing and imaging applications requiring the simultaneous detection and differentiation of multiple aspectratio gold nanorods.
Keywords: Laser-induced chemistry; Nanomaterials; Photoacoustics
Introduction
In recent times, gold nanorods (AuNRs) have attracted immense interest for biomedical applications such as drug delivery [1,2], diagnostic imaging [3,4], therapy [5,6] and biosensing assays [7]. This surge in interest can be attributed to their biocompatibility [8], ease of conjugation to moieties that permit binding to biologically interesting molecular targets [9,10] and optical properties, particularly their surface plasmon resonance [11], which arises due to the collective coherent oscillation of the electrons on the metallic surface. When the frequency of the incoming photons matches that of the oscillation of surface conduction electron density, resonance occurs. At this resonance frequency, light is more strongly absorbed by the plasmonic nanoparticle than at other frequencies, with a size normalized absorption cross-section that is five orders of magnitude greater than any known dye molecule [12]. AuNRs, because of their rod-like shape, exhibit two plasmon resonances, a transverse plasmon (TP) resonance, arising due to the oscillation of the electrons around the circumference of the nanorod’s circular cross-section, and a longitudinal plasmon (LP) resonance in the visible to near-infrared region due to the oscillation of the electrons along the length of the nanorod. Using the most commonly employed method of synthesizing AuNRs, their cylindrical diameter is approximately constant, fixing their TP resonance at a peak wavelength close to 520 nm in the visible range, whereas their length may be controlled to create AuNRs of a specific aspect ratio (length to diameter ratio). AuNRs of a given aspect ratio thus has a characteristic optical absorption spectrum, with an LP resonance that potentially allows them to be detected spectroscopically in a background of other optical absorbers, or to be distinguished from AuNRs of another, sufficiently different, aspect ratio.
Among the applications utilising AuNRs, researchers have proposed the use of multiple aspect ratio AuNRs having different LP plasmon resonance wavelengths for multiplexed biosensing [7], potentially resulting in assays that are capable of simultaneously detecting (by optical spectroscopy) a plenty of different biomolecules in vitro, and multiplexed spectroscopic molecular imaging, such as optoacoustic imaging [13,14], which can potentially simultaneously detect whether multiple receptors are expressed on the cell surface in vivo. The capacity to sensitively detect biomarker molecules in vitro and in vivo, and the number of such molecules that could be simultaneously distinguished, would depend on the sharpness of the LP resonance peak of the population of synthesized AuNRs used in the study.
Additionally, the success of multiplexed biosensing, molecular imaging and other applications of AuNRs, would depend on the certainty with which the wavelength of the LP resonance peak of a population of synthesized AuNRs can be controlled. For a given LP resonance sharpness, the wavelength of the peak will ascertain the capacity to identify the AuNRs in the presence of other chromophores or separate them from AuNRs from another population. Furthermore, optoacoustic imaging [15] and photo thermal therapy [5,16] are examples of techniques which constrain the use of AuNRs having a plasmon resonance peak matching the wavelength of the laser being employed, which is often chosen to penetrate as deep as possible in tissue.
For a bulk synthesis of AuNRs, the seed-mediated approach, developed by Nikoobakht, et al. [16] is the method preferred by the majority of researchers, probably because of its low cost and simplicity. Although use of this method does not allow the TP resonance of the AuNRs to be altered, their LP resonant wavelength may be controlled, within the so-called tissue optical spectral window of 650-1100 nm. This is done by altering the concentration of silver nitrate in the growth solution, which controls the length to which the AuNRs are grown. However, the aspect ratio of the end product depends upon many parameters, such as the temperature of the reaction [17], speed of stirring [18], the concentration of reactants [19], type of surfactant [20], pH [21] and possibly other unknown variables. The existences of so many sources of variation means that the properties of the AuNRs synthesized using this method are not easy to control accurately or precisely. This leads to bias and random uncertainty in the position of the LP peak of each AuNR and, since the absorption spectrum of a population of AuNRs is a superposition of the absorption spectra of the individual NRs, to two problems when such AuNRs are used in applications such as those mentioned above: the batch population LP peak may not be exactly at the wavelength desired, and its width may be broader than desired.
To synthesize AuNRs to a desired LP resonance peak wavelength, Wang, et al. [22] used a chemical approach adding an excess of a chemical inhibitor, sodium borohydride, to deplete the residual gold concentration in the growth solution, which terminated the growth process at the desired stage of the reaction. However, the addition of this, inhibitor decreases all the unreacted gold in the solution, so that an excess of gold clusters is formed, and yield of AuNRs is decreased. Another approach is to resize the AuNRs after synthesis. Chemical methods for achieving this include cyanide etching [23] and mild oxidation [24,25]. These chemical-induced reshaping approaches tend to be somewhat inflexible. Each shifts the LP peak to either a shorter or a longer wavelength and at least two methods are required to have the flexibility of both options. To our knowledge, the issue of batch population LP peak being wider than desired, i.e poor individual AuNR synthesis precision has still to be resolved.
To summarise the problem, there has been a limited success using chemical synthesis-control and resizing methods. Control at synthesis results in a reduced yield. Post-synthesis chemical resizing requires a different method to shift the LP peak of the AuNRs to a longer wavelength, than that used to shift it to a shorter wavelength. None of the chemical methods are able to influence the width of the population LP resonance. Here we describe a new method which can be used both to control the LP peak wavelength of an AuNR population, and to narrow the population LP resonance. It does so by using laser irradiation to resize AuNRs after they have been synthesised.
Laser irradiation increases the surface temperature of AuNRs causing partial melting and hence changes in their morphology [26- 28]. It is known for example, that a suspension of AuNRs in water begins to demonstrate changes in its optical absorption spectrum at a fluence of 8 mJ/cm2 with as few as 300 laser pulses (7 ns pulse duration, 10 Hz repetition rate) [29]. However, although these effects have been observed the possibility of using controlled heat with a tunable laser to deliberately tune the LP peak of a population of already synthesized AuNRs has not been investigated. In principle, the size distribution associated with a given population of AuNRs in suspension could be altered by irradiating the suspension at wavelengths corresponding to the LP resonance of unwanted AuNRs, partially melting them and hence eliminating their contribution to the LP peak.
The aim of the present study is to determine the feasibility of such approach and to determine the extent to which it can narrow the width, and control the wavelength of the LP resonance peak of a population of AuNRs.
Methods
Overall approach
The preliminary experiments described here were conducted in three steps. A first experiment was aimed at demonstrating that it is possible to selectively destroy AuNRs of a chosen size. It had to be shown that this is possible before attempting to use the phenomenon to tune the properties of a population and it was shown by mixing two AuNR suspensions that are having LP spectral peaks that differed by more than 200 nm, and demonstrating that one of the AuNR suspensions may be selectively removed from the mixture using high energy laser pulses at the wavelength of the LP maximum of the AuNR population to be removed. A second experiment aimed to employ this phenomenon, by demonstrating that the full width at half maximum (FWHM) of an LP spectral peak for a population of AuNRs can be decreased by a technique developed which involved irradiating the AuNR suspension sequentially, at wavelengths starting from either tail of the spectral resonance and moving towards the LP maximum. Finally, the last experiment was designed to demonstrate that using a similar sequential irradiation approach it was also possible to shift the LP resonance peak of a given population of AuNRs to any desired wavelength within the original LP resonance band.
Nanorod synthesis
AuNRs were synthesized using the three-step seed-mediated protocol developed by Nikoobakht, et al. [16], wherein gold nanospheres synthesized using a strong reducing agent with gold chloride were used as seeds in growth solutions to produce AuNRs having an expected aspect ratio of 2.6, 3.2, 3.8 and 5.5, and corresponding expected LP maxima of 696 nm, 715 nm, 770 nm and 910 nm.
Laser irradiation and measurement of optical extinction spectra
A Q-switched Nd:YAG laser (Quantel, Les Ulis), made tunable in the wavelength range of 700 nm-950 nm using a ‘magic prism’ optical parametric oscillator (Opotek, California) was used for delivering a 6 (± 0.5) nanosecond pulse every 0.1 seconds. For each pulse, the spectral line width (FWHM) was 9 (± 7) nm and the laser energy was between 23 (± 1.7) mJ and 35 (± 3.6) mJ, depending on the wavelength, as measured using a power meter (SOLO 2™, Gentec, Quebec). Linear profile fibre optic bundles were connected to the laser for delivering the laser pulse.
An optically transparent spectrophotometer cuvette having a width and depth of 1 cm, was filled with 3 ml of AuNR suspension, and fibre optic bundles delivered a diverging beam covering the cuvette cross-sectional area of 2 cm2. The minimum laser fluency, to which the AuNR suspension was exposed, was calculated to be 11 (± 1) mJ/cm2, well above the threshold of 8 mJ/cm2 required for partial melting of the AuNRs [30]. The maximum laser fluence was calculated to be 18 (± 1) mJ/cm2, which was well below the threshold of 1 J/cm2 required for a complete melting of all the AuNRs into spheres [27]. As the cuvette used was optically transparent with a wall thickness of 2 mm, the attenuation of the laser pulse by the cuvette, before reaching the AuNR suspension, was assumed to be negligible. Each sample was irradiated for 100 sec at 10 Hz pulse repetition rate, at specific wavelengths corresponding to the spectral resonances of undesired AuNRs within the distribution. Further details of the specific laser irradiation wavelengths are provided in the results section. Following each laser irradiation step, the cuvette was transferred to a spectrophotometer (6705, Jenway, Staffordshire) for measurement of the change in the extinction spectrum, after which it was returned for the next irradiation step until completion of the experiment.
Statistical analysis methods
The effects of the laser irradiation technique on the widths of the population LP peaks in the extinction spectra of each of the three different AuNR samples having expected aspect ratios 2.6, 3.8 and 5.5, before and after the laser irradiation, were assessed by the following four methods of analyses of the spectra:
i. The FWHM of the spectra were calculated.
ii. The areas of the overlaps of the normalised extinction spectra were calculated.
iii. Student’s t-test was applied for testing the null hypothesis that there is no difference between the means of the spectral distributions of any pair of AuNR populations, using the t-test statistic to provide a spectral discrimination factor (SDF) that could be used to assess the practical impact of any spectral sharpening.
For methods i) and ii), the FWHM and the areas of overlap of the extinction spectra of the different AuNR samples, before and after laser irradiation, were calculated using a spectral analysis Matlab™ tool (UV-Vis-IR Spectral Software 1.2, FluorTools, www.fluortools.com)..
For iii), the SDF for any pair of AuNR spectra, before and after laser irradiation, was calculated as:
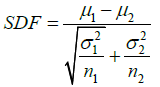
Where μ, σ2 and n are respectively the mean, variance and area under the spectrum and the subscripts 1 and 2 refer to the first and second AuNR samples under comparison.
Results
Selective removal of gold nanorods from a mixture
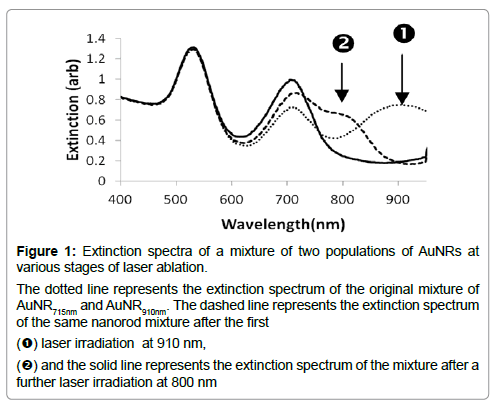
Selective removal of AuNRs of a preferred size was tested by using a mixed suspension of two AuNRs, one with an LP maximum at 715 nm (AuNR715 nm), the other at 910 nm (AuNR910 nm). The extinction spectrum of the mixture, Figure 1 (dotted curve) shows both these peaks, together with that due to the TP resonance at 520 nm. The mixture was irradiated with 1000 laser pulses (10 Hz for 100 seconds), at 910 nm. The resultant AuNR mixture showed LP peaks near 700 nm and 800 nm, (Figure 1 dashed curve), but the peak at 910 nm was no longer present, as intended. The incidental shoulder in the spectrum at 800 nm suggests that the longer AuNRs had been reshaped, with a corresponding shift of the mean LP resonance from 910 nm to 800 nm. The irradiation method was consequently repeated, this time at 800 nm to conclude whether the new population of AuNRs could be selectively removed. As a result, only a single LP peak at 715 nm was observed, with enhanced absorbance relative to the original peak at 715 nm (Figure 1, solid curve), proposing an increase in the number of AuNRs with this LP resonance. This is compatible with more reshaping of AuNRs and resulting in a shift of their LP maxima to a shorter wavelength. Consequent to this, as described in sections 3.2 and 3.3 below, we utilized sequential irradiation at different wavelengths and got evidence of the reshaping from electron microscopy.
Figure 1: Extinction spectra of a mixture of two populations of AuNRs at various stages of laser ablation.
The dotted line represents the extinction spectrum of the original mixture of AuNR715nm and AuNR910nm. The dashed line represents the extinction spectrum of the same nanorod mixture after the first
(1) laser irradiation at 910 nm,
(2) and the solid line represents the extinction spectrum of the mixture after a further laser irradiation at 800 nm
Tuning the width of the LP spectral resonance of a population of nanorods
For tuning the width of the LP spectral peak of a population of AuNRs, the two-stage sequential irradiation technique found to be useful above was extended. Each sample was irradiated with intense laser pulses in a sequence of groups of 1000 pulses, with the first group starting from a wavelength at the long wavelength end of the spectral resonance and moving towards the LP spectral peak wavelength at intervals of 25 nm, measuring the extinction spectrum after each irradiation group. Irradiating the sample with laser pulses was stopped at a wavelength that resulted in any shift of the LP spectral peak.
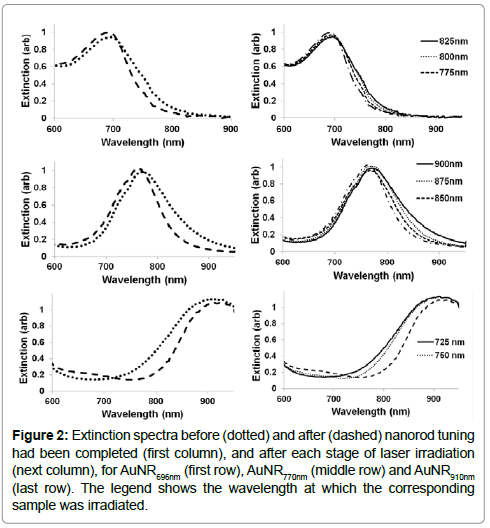
The extinction spectrum of an AuNR sample with an LP maximum at 770 nm (AuNR770 nm) before irradiation is shown in Figure 2 (middle row), along with the spectra after sequential irradiation at wavelengths from 900 nm to 850 nm at 25 nm intervals. The spectra showed that the AuNRs with plasmon resonances in this range underwent reshaping with each irradiation, resulting in a continuous sharpening of the population LP spectral peak, as well as small shifts of the peak from its original real value to shorter wavelengths. The latter topic is discussed in section 3.3 below; here the focus is on sharpening the LP spectral peak of the population. After three sequential irradiations of this sample, the right and left half-maximum points of the LP peak had shifted left by 27 nm and 5 nm, respectively. The decrease in FWHM of the LP peak was estimated to be 22 nm.
Figure 2: Extinction spectra before (dotted) and after (dashed) nanorod tuning had been completed (first column), and after each stage of laser irradiation (next column), for AuNR696nm (first row), AuNR770nm (middle row) and AuNR910nm (last row). The legend shows the wavelength at which the corresponding sample was irradiated.
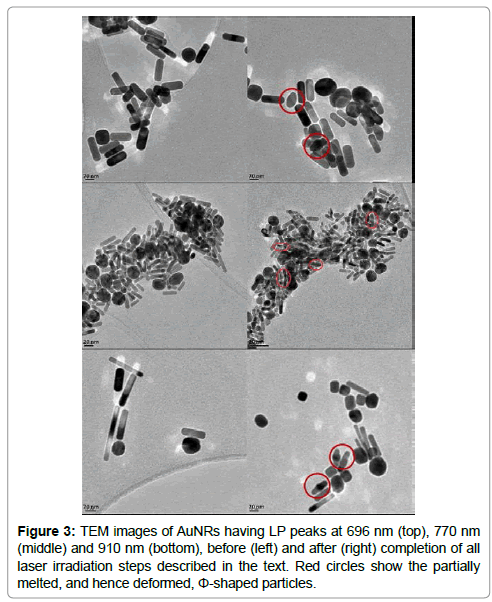
Similar results were obtained for sharpening the LP spectral peak of AuNR suspensions with pre-irradiation LP maxima at 696 nm (AuNR696 nm) and 910 nm (AuNR910 nm), using sequential irradiation at wavelengths between 825 nm and 775 nm for AuNR696 nm, and 725 nm and 750 nm for AuNR910 nm, again at intervals of 25 nm. Extinction spectra measured after each step of laser irradiation are shown in Figure 2 (top and bottom rows). The FWHM of the LP peak of each sample of AuNRs decreased in all three cases, as shown in Table 1, which suggests that the observed decrease is significant. Characteristically deformed (partially melted) phi-shaped particles, formed by selective light absorption by the undesired AuNRs in the population, were observed in the TEM images as indicated by red circles in Figure 3.
| lpeak before tuning | lpeak after tuning | Difference in lpeak due to tuning | FWHM before tuning | FWHM after tuning | Difference in the FWHM of LP peak due to tuning |
|---|---|---|---|---|---|
| 696 nm | 691 nm | 5 nm | 112* | 96 | -16 nm |
| 770 nm | 765 nm | 5 nm | 124 | 102 | -22 nm |
| 910 nm | 920 nm | -10 nm | 204* | 183 | -21 nm |
*The values of FWHM were estimated by assuming symmetry of the extinction spectrum in Figure 2.
Table 1: The wavelength (λpeak) and the width (FWHM) of the extinction spectral peak for samples of AuNRs before and after tuning them with a laser.
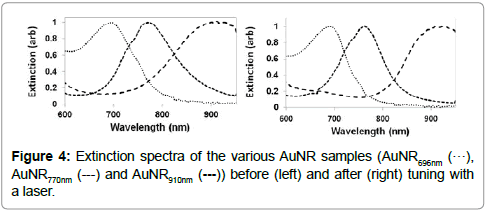
The area of overlap of the LP peaks in the normalizsed extinction spectra of the AuNR770 nm and AuNR910 nm samples was calculated between 650 nm and 950 nm. Before laser irradiation, the area of the intersected region was 76.6 square units, which reduced to 47 square units after laser-based tuning. A slightly smaller reduction in the area of overlap was observed for the LP spectral peaks of the AuNR696 nm and AuNR770 nm samples, and the AuNR696 nm and AuNR910 nm samples, as shown in Table 2. A visual judgment of the overlap before and after laser-based tuning can be done by examining the extinction spectra, which are plotted together in Figure 4.
| Pre-irradiation LP maximum of the AuNR sample under comparison | Wavelength range considered for calculation of the area of overlap (nm) | Area of overlap before laser irradiation (sq. units) | Area of overlap after laser irradiation (sq. units) | Percentage reduction |
|---|---|---|---|---|
| 696 nm - 770 nm | 650 nm - 950 nm | 55 | 49.6 | 9.8% |
| 696 nm - 910 nm | 650 nm - 950 nm | 30 | 25.4 | 15.3% |
| 770 nm - 910 nm | 650 nm - 950 nm | 76.6 | 47 | 38.6% |
Table 2: Comparison of the area of overlap of LP spectral peaks, for each pair of AuNR samples, before and after laser tuning.
The spectral discrimination factors, calculated using Equation 1, are shown in Table 3 for pairs of AuNR samples before and after laser irradiation. Tuning with laser irradiation improved discrimination in all three cases.
| Pre-irradiation LP maxima of the AuNR samples under comparison | Spectral discrimination factor before laser-irradiation | Spectral discrimination factor after laser-irradiation |
|---|---|---|
| 696 nm and 770 nm | 11.97 | 13.43 |
| 696 nm and 910 nm | 30.16 | 32.33 |
| 770 nm and 910 nm | 18.61 | 21.11 |
Table 3: Comparison of the spectral discrimination factors before and after laser-based tuning for pairs of nanorod samples with different pre-irradiation LP spectral peaks.
Tuning the LP spectral peak wavelength of a population of nanorods
To explore this capability, AuNR samples were sequentially irradiated with groups of laser pulses, starting either at long wavelengths and stepping down in 25 nm intervals (to produce a blue shift of the population spectral peak) or at short wavelengths and stepping up in 25 nm intervals (to produce a redshift of the population spectral peak). However, it may be seen from Figure 2 that irradiation of the AuNR770 nm sample at 850 nm caused an alteration of the LP maximum wavelength of only 5 nm, because of the closeness of 850 nm to the LP maximum prior to irradiation. Irradiating the sample at wavelengths even closer to the peak would have resulted in a large jump of the entire LP maximum of the AuNR suspension, as can be seen between the dotted curves of the Figure 1, which is undesirable. These conditions were consequently withdrawn when exploring the process of tuning the LP spectral peak wavelength, which was conducted using only a sample of AuNRs with a preirradiation LP maximum at 910 nm.
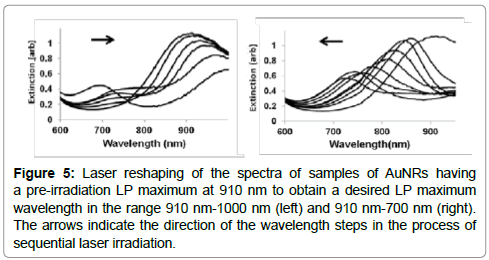
Sequential irradiation starting at 950 nm and ending at 725 nm resulted in a shift of the LP peak to a shorter wavelength at each stage, as expected until an LP maximum of 700 nm was reached (Figure 5, right). This indicated that longer AuNRs within a sample could be selectively managed to form a final distribution of AuNRs with a smaller mean aspect ratio. Shorter AuNRs were also selectively managed with sequential groups of laser pulses, starting from 700 nm and ending at 910 nm, to raise the mean aspect ratio in the sample and therefore shift its LP maximum to a longer wavelength (Figure 5 left) with a final value near 1000 nm.
Figure 5: Laser reshaping of the spectra of samples of AuNRs having a pre-irradiation LP maximum at 910 nm to obtain a desired LP maximum wavelength in the range 910 nm-1000 nm (left) and 910 nm-700 nm (right). The arrows indicate the direction of the wavelength steps in the process of sequential laser irradiation.
In both cases, it may be seen in Figure 5 that a consequence using laser irradiation to move the LP maximum of an AuNR sample to a different wavelength was a successive broadening of the peak, which in the extreme took the form creating a new absorbance peak at 700 nm (Figure 5 left). This secondary absorbance peak may be connected to the creation of phi-shaped particles illustrated in Figure 3. An increase in absorbance value at 700 nm was observed with each step of subsequent laser irradiation, as more phi-shaped particles were produced after each step.
Discussion
It is well known that a range of parameters affect the aspect ratio and LP maxima of AuNRs synthesized using the seed-mediated approach, resulting in poor reproducibility and a broad LP peak, which might make them unfavourable for applications involving identification of different aspect ratio AuNRs, such as multiplexed imaging and biosensing. This study reports the development of a laser-based method and demonstrates its ability both to narrow the synthesized AuNRs’ LP resonance peak slightly and to enable the populations with a controlled position of the absorbance peak to be produced.
As demonstrated in Figure 1, the results of laser irradiation of an AuNR sample at a wavelength chosen by spectrophotometry, followed by further irradiation at a new wavelength selected by spectrophotometry after the previous irradiation, and so on, established the feasibility of a novel consecutive method that provides complete controlled reshaping of the absorbance spectrum of an AuNR population. It was possible to completely remove the spectral peak of the AuNR910 nm sample from the two-peaked extinction spectrum of the mixture of two AuNR samples, transmitting a single spectral peak at 715 nm, which was the wavelength of the LP spectral peak of the other sample in the mixture. The lack of a significant increase in absorption at 525 nm indicated that AuNRs had been converted to nanoparticles rather than nanospheres, which was later confirmed using the TEM. This provided the potential purification process, which was then evaluated.
The FWHM (Figure 2) and areas of overlap (Table 2) between different LP spectral peaks of three different AuNRs were found to decrease. A notable improvement was observed for differentiating LP peaks of AuNR770 nm and AuNR910 nm, characterized by a decrease of the area of overlap by approximately 39%. The LP peak of AuNR770 nm was tuned starting at the long wavelength end of the right spectral tail, whereas the LP peak of AuNR910 nm was tuned from the short wavelength end of the left spectral tail.
The LP peak of AuNR770 nm could not be tuned using shorter wavelengths, as at the lowest applicable wavelength, 700 nm, there was an undesirable shift in the LP peak to a longer wavelength. Similarly, for tuning the LP peak of AuNR910 nm, wavelengths only up to 750 nm (160 nm away from the maxima) could be utilised before causing a shift in the Plasmon resonance peak to a higher wavelength. Work is underway to understand this finding. The LP peak of the AuNR910 nm was not tuned from the right spectral tail because the laser wavelength was limited to 950 nm, which is very close to the peak. Since in this case, the LP peak of AuNR770nm was tuned from the right and the LP peak of AuNR910nm, from the left, a major improvement in the spectral discrimination was observed, due to a reduction in the overlapping area.
In comparison to the improvement in discrimination between the LP peaks of the AuNR770 nm and the AuNR910 nm samples after laser tuning, no improvement was observed for discriminating between the LP peaks of the AuNR696 nm and the AuNR770 nm samples. The latter samples were tuned using longer wavelengths, from the ends of the right spectral tails. They could not be tuned from the left spectral tail because the shortest wavelength of the laser was 700 nm. The full potential of this laser tuning approach, therefore, needs to be explored in future experiments using tunable lasers that cover a wider range of wavelengths.
The LP peaks of the AuNR696 nm and AuNR910 nm samples were easily discriminated before laser tuning; correspondingly, there was no improvement in the spectral discrimination of the LP peaks of these two AuNR suspensions after laser tuning.
The tuning of AuNR910 nm sample resulted in a formation of a secondary absorbance peak (600 nm-700 nm), as also observed in Figure 5, which could be attributed to the partially deformed Φ-shaped particles that are produced as a result of partial melting of AuNRs [27]. This effectively reduced the spectral discrimination between the two AuNR spectral peaks, the two models of LP peaks FWHM decreased. The laser, which is more than the pulse force currently used in this study, can prevent the formation of such a secondary peak by breaking unwanted Nanomaterials, which form nanospheres that are absorbed in 520 nm, [27] which is more spectral discrimination in such a scenario AuNRs.
It was also shown that the LP maximum of the AuNRs could be shifted from an undesired spectral resonance to a desired one, by preserving a sub-population. This technique could also be further improved by using a laser with a wide wavelength tuning range. Its main application would be in combination with the spectral narrowing method, to further improve spectral discrimination by positioning the LP peaks to minimise overlap.
The theoretical limit of a tuning method will in principle occur when a mono disperse population of AuNRs has been produced. The FWHM of the LP peak in AuNRs varies as a function of aspect ratio. It has been reported that the FWHM of the LP peak of AuNRs increases in a non-linear way with increasing aspect ratio [30]. A single AuNR, depending on its aspect ratio, has a Plasmon resonance peak with a FWHM in the range 50 nm to 200 nm, as predicted by Zhu, et al. [30] using simulations. This implies that even with a truly monodispersed population, it is unlikely that the number of AuNRs that could be simultaneously detected would be more than about four or five, in the spectral range of 700 nm to 950 nm. The LP peaks of the synthesized AuNRs for this study, and also reported elsewhere, have similar FWHMs for the corresponding aspect ratio. The laser tuning approach employed here reduced the FWHM of the LP peaks of all population of AuNRs by at least 16 nm. After further optimisation employing a tunable laser with wider bandwidth and higher power, it would be worth assessing the practical impact of this spectral sharpening for applications involving simultaneous detection of multiple AuNRs.
The laser irradiation technique has limitations in its ability to tune the LP peak of AuNRs to a desired wavelength because it can only alter the distribution of sizes within the synthesized population of AuNRs. Shifting to a different wavelength, therefore, results in a lower yield of desired AuNRs and a reduced extinction, as can be observed in Figure 5. Chemical methods for controlling the LP peak of AuNRs are far superior in this case, as they can accurately shift the LP peak to the desired wavelength with an accuracy of a few nanometers, without causing a reduction in the extinction of the sample. However, to our knowledge, the LP peaks of the AuNRs synthesized using these chemical approaches have similar FWHM to those reported in this paper before tuning, making this laser tuning method worthwhile. A combination of a chemical approach for shifting the LP peak and a laser irradiation approach for narrowing the spectral peak would be worth investigating.
Conclusion
A laser-based AuNR tuning method has been developed which has been determined to show potential both to control the wavelength and to decrease the width of the LP resonance peak of an AuNR population. This combination of capabilities cannot be achieved by any single chemical method. Appropriate use of these capabilities, possibly in combination with a chemical method for controlling the wavelength of the LP resonance peak, could be applied to reduce the overlap between optical spectra from different populations of AuNRs so that in future, simultaneous bio sensing and molecular imaging of AuNRs functionalized to multiple molecular markers would be possible.
Acknowledgements
The research was supported by The Institute of Cancer Research CRUK and EPSRC Cancer Imaging Centre, in association with the MRC and Department of Health (England) grant #C1060/A10334. The purchase of the OPO laser was funded by the Department of Health, National Institute for Health Research. Also thanks are due to Heather Davies from The Open University, who helped to image the gold nanorods using an electron microscope.
References
- Mirza AZ (2015) A novel drug delivery system of gold nanorods with doxorubicin and study of drug release by single molecule spectroscopy. J Drug Target 23: 52-58.
- Liu J, Detrembleur C, De Pauwâ€ÂGillet MC, Mornet S, Jerome C, et al. (2015) Gold nanorods coated with mesoporous silica shell as drug delivery system for remote near infrared light-activated release and potential phototherapy. Small 11: 2323-2332.
- Li W, Chen X (2015) Gold nanoparticles for photoacoustic imaging. Nanomedicine 10: 299-320.
- de la Zerda A, Prabhulkar S, Perez VL, Ruggeri M, Paranjape AS, et al. (2015) Optical coherence contrast imaging using gold nanorods in living mice eyes. Clin Experiment Ophthalmol 43: 358-366.
- Kang X, Guo X, Niu X, An W, Li S, et al. (2017) Photothermal therapeutic application of gold nanorods-porphyrin-trastuzumab complexes in HER2-positive breast cancer. Sci Rep 7: 42069.
- Heidari Z, Salouti M, Sariri R (2015) Breast cancer photothermal therapy based on gold nanorods targeted by covalently-coupled bombesin peptide. Nanotechnology 26: 195101.
- Yu C, Irudayaraj J (2007) Multiplex biosensor using gold nanorods. Anal Chem 79: 572-579.
- Wan J, Wang JH, Liu T, Xie Z, Yu XF, et al. (2015) Surface chemistry but not aspect ratio mediates the biological toxicity of gold nanorods in vitro and in vivo. Sci Rep 5: 11398.
- Eghtedari M, Liopo AV, Copland JA, Oraevsky AA, Motamedi M (2009) Engineering of hetero-functional gold nanorods for the in vivo molecular targeting of breast cancer cells. Nano Lett 9: 287-91.
- Chen T, Shukoor MI, Chen Y, Yuan Q, Zhu Z, et al. (2011) Aptamer-conjugated nanomaterials for bioanalysis and biotechnology applications. Nanoscale 3: 546-556.
- Kreibig U, Vollmer M (1995) Optical properties of metal clusters. Springer, Heidelberg, Germany.
- Jain PK, Lee KS, El-Sayed IH, El-Sayed MA (2006) Calculated absorption and scattering properties of gold nanoparticles of different size, shape, and composition: applications in biological imaging and biomedicine. J Phys Chem B 110: 7238-7248.
- Li PC, Wang CR, Shieh DB, Wei CW, Liao CK, et al. (2008) In vivo photoacoustic molecular imaging with simultaneous multiple selective targeting using antibody-conjugated gold nanorods. Opt Express 16: 18605-18615.
- Shah AJ, Alles EJ, Box C, Eccles SA, Robinson SP, et al. (2014) Non-invasive molecular profiling of cancer using photoacoustic imaging of functionalized gold nanorods. SPIE BiOS, San Francisco, California, United States.
- Huang X, El-Sayed IH, Qian W, El-Sayed MA (2006) Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J Am Chem Soc 128: 2115-2120.
- Nikoobakht B, El-Sayed MA (2003) Preparation and growth mechanism of gold nanorods (NRs) using seed-mediated growth method. Chem Mater 15: 1957-1962.
- Gou L, Murphy CJ (2005) Fine-tuning the shape of gold nanorods. Chem Mater 17: 3668-3672.
- Pérezâ€ÂJuste J, Lizâ€ÂMarzán LM, Carnie S, Chan DY, Mulvaney P (2004) Electricâ€Âfieldâ€Âdirected growth of gold nanorods in aqueous surfactant solutions. Adv Funct Mater 14: 571-579.
- Sau TK, Murphy CJ (2004) Seeded high yield synthesis of short Au nanorods in aqueous solution. Langmuir 20: 6414-6420.
- Yong KT, Sahoo Y, Swihart MT, Schneeberger PM, Prasad PN (2008) Templated synthesis of gold nanorods (NRs): the effects of cosurfactants and electrolytes on the shape and optical properties. Top Catal 47: 49-60.
- Bullen C, Zijlstra P, Bakker E, Gu M, Raston C (2011) Chemical kinetics of gold nanorod growth in aqueous CTAB solutions. Cryst Growth Des 11: 3375-3380.
- Wang Y, Long S, Vdovic S, Wang X (2012) Fine tuning of the longitudinal plasmon resonance of gold nanorods by depleting gold precursor. Chem Mater 25: 1372-1376.
- Carattino A, Khatua S, Orrit M (2016) In situ tuning of gold nanorod plasmon through oxidative cyanide etching. Phys Chem Chem Phys 18: 15619-15624.
- Tsung CK, Kou X, Shi Q, Zhang J, Yeung MH, et al. (2006) Shortening of single-crystalline gold nanorods by mild oxidation. J Am Chem Soc 128: 5352-5353.
- Ni W, Kou X, Yang Z, Wang J (2008) Tailoring longitudinal surface plasmon wavelengths, scattering and absorption cross sections of gold nanorods. ACS Nano 2: 677-686.
- Link S, Burda C, Nikoobakht B, El-Sayed MA (2000) Laser-induced shape changes of colloidal gold nanorods using femtosecond and nanosecond laser pulses. J Phys Chem B 104: 6152-6163.
- Zijlstra P, Chon JW, Gu M (2009) White light scattering spectroscopy and electron microscopy of laser induced melting in single gold nanorods. Phys Chem Chem Phys 11: 5915-5921.
- Chang SS, Shih CW, Chen CD, Lai WC, Wang CC (1999) The shape transition of gold nanorods. Langmuir 15: 701-709.
- Chen YS, Frey W, Kim S, Homan K, Kruizinga P, et al. (2010) Enhanced thermal stability of silica-coated gold nanorods for photoacoustic imaging and image-guided therapy. Opt Express 18: 8867-8878.
- Zhu J (2005) Shape dependent full width at half maximum of the absorption band in gold nanorods. Phys Lett A 339: 466-471.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi