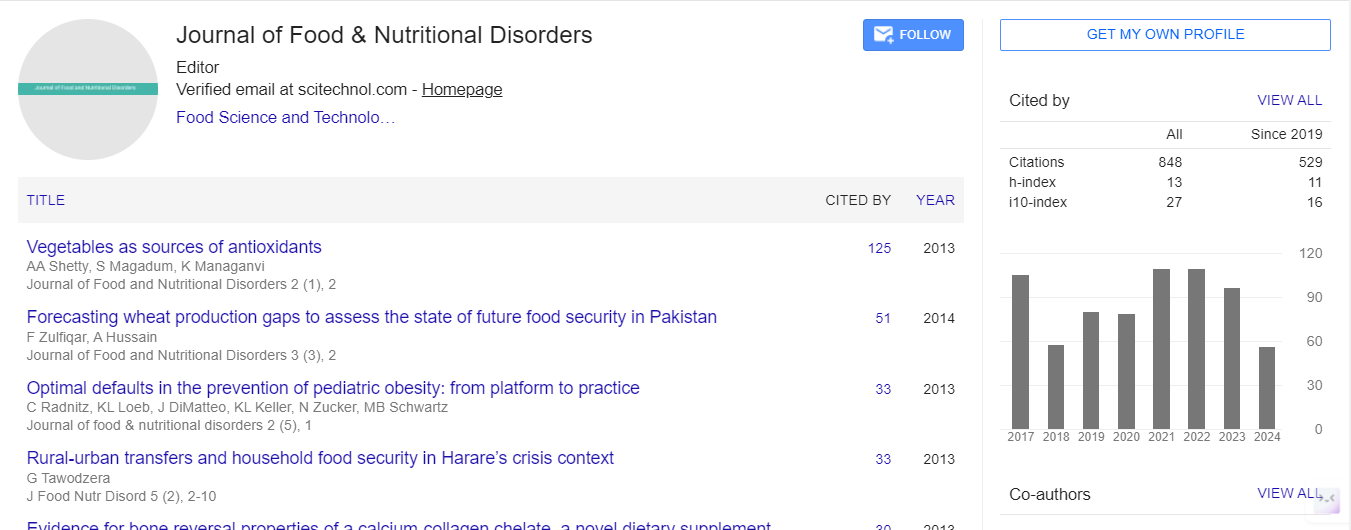
Research Article, J Food Nutr Disor Vol: 5 Issue: 5
Palm Based High-Oleic Cooking Oil and Extra Virgin Olive Oil Diets do not Affect Markers of Insulin Resistance and Glucose Tolerance in Overweight Adults
| Sin Tien Lee1,2, Phooi Tee Voon1*, Tony Kock Wai Ng3, Norhaizan Binti Esa2, Verna Kar Mun Lee4, Hazizi Abu Saad2 and Su Peng Loh2 | |
| 1Nutrition Unit, Product Development and Advisory Services Division, Malaysian Palm Oil Board, 43000 Bandar Baru Bangi, Kajang, Selangor, Malaysia | |
| 2Department of Nutrition and Dietetic, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43300 Serdang, Selangor, Malaysia | |
| 3Department of Nutrition and Dietetics, School of Health Sciences, Institute for Research, Development & Innovation, International Medical University, 57000 Bukit Jalil, Kuala Lumpur, Malaysia | |
| 4Department of Famaily Medicine, Institute for Research, Development& Innovation, International Medical University, 57000 Bukit Jalil, Kuala Lumpur, Malaysia | |
| Corresponding author : Phooi Tee Voon
Nutrition Unit, Product Development and Advisory Services Division, Malaysian Palm Oil Board, No. 6, Persiaran Institute, Bandar Baru Bangi, 43000 Kajang, Selangor, Malaysia Tel: +60387694400 Fax: +60389259446 E-mail: vptee@mpob.gov.my |
|
| Received: June 08, 2016 Accepted: August 11, 2016 Published: August 16,2016 | |
| Citation: Lee ST, Voon PT, Ng TKW, Esa NB, Lee VKM (2016) Palm Based High-Oleic Cooking Oil and Extra Virgin Olive Oil Diets do not Affect Markers of Insulin Resistance and Glucose Tolerance in Overweight Adults. J Food Nutr Disor 5:5. doi:10.4172/2324-9323.1000207 |
Abstract
Palm Based High-Oleic Cooking Oil and Extra Virgin Olive Oil Diets do not Affect Markers of Insulin Resistance and Glucose Tolerance in Overweight Adults
Background: High oleic blended cooking oil (HOBO) is an oil product of blended palm olein and canola oil with monounsaturated oleic acid as dominant fatty acid. This blend has increased the amount of oleic acid to more than 50%, with cold stability which is suitable for cooking usage in temperate countries. However, the effects of HOBO diet in human nutrition have not been studied extensively. Objective: We aimed to compare the effects of HOBO vs. oleic acid-rich extra virgin olive oil (OO) diets on markers of insulin resistance and glucose tolerance in overweight subjects. Design: A single-blind, randomized crossover study with 3 dietary interventions of 6-wk each (3-wk washout in between) was conducted in 32 overweight subjects. The 3 test fats were HOBO, OO and refined, bleached and deodorized coconut oil CO incorporated at two-thirds of 30% kcal fat containing 55% kcal from carbohydrates and 15% kcal from protein. Results: No significant differences (P>0.05) were observed in the effects of the 3 diets on markers of insulin resistance [glucose, c-peptide, insulin, fructosamine, Haemoglobin A1c (HbA1c), homeostasis model assessment of insulin resistance (HOMA-IR), homeostasis model assessment of beta cell function index (HOMA-β), and quantitative insulin sensitivity check index (QUICKI)] and glucose tolerance. Conclusions: HOBO diet did not alter markers of insulin resistance and glucose tolerance in overweight Malaysian adults compared with that of OO and CO diets.
Keywords: Glucose; Insulin; C-peptide; Insulin resistance; Glucose tolerance; High oleic; Palm olein; Olive oil; Overweight
Keywords |
|
| Glucose; Insulin; C-peptide; Insulin resistance; Glucose tolerance; High oleic; Palm olein; Olive oil; Overweight | |
List of Abbreviations |
|
| AUC: Area Under the Curve | |
| BMI: Body Mass Index | |
| CO: Refined, Bleached and Deodorized Coconut Oil | |
| CVD: Cardio Vascular Disease | |
| DM: Diabetes Mellitus | |
| HbA1c: Haemoglobin A1C | |
| HOBO: High oleic Blended Cooking Oil | |
| HOMA-IR: Homeostasis Model Assessment of Insulin Resistance | |
| HOMA-β: Homeostasis Model Assessment of Beta Cell Function Index | |
| IFG: Impaired Fasting Glucose | |
| IGT: Impaired Glucose Tolerance | |
| MUFA: Monounsaturated Fatty Acid | |
| OO: Extra Virgin Olive Oil | |
| OGTT: Oral Glucose Tolerance Test | |
| PUFA: Polyunsaturated Fatty Acid | |
| QUICKI: Quantitative Insulin Sensitivity Check Index | |
| SAFA: Saturated Fatty Acid | |
| T1DM: Type 1 Diabetes Mellitus | |
| T2DM: Type 2 Diabetes Mellitus | |
| WHO: World Health Organization | |
Introduction |
|
| Diabetes mellitus (DM) is a chronic metabolic disorder of the pancreas where it is characterized by greatly elevated blood glucose levels with disturbances in carbohydrate, fat and protein metabolism [1]. Type 2 DM (T2DM) is characterized by insulin resistance and pancreatic β cell dysfunction [2]. Insulin resistance happens when the body produces insulin but it has not been used effectively. This leads to glucose building up in the blood instead of being absorbed by the cells. Complications of DM are associated with hypoglycaemia, hypertenstion, dyslipidmia, greater cardio vascular disease (CVD) rates, greater heart attack rates, stroke, kidney disease, amputations and etc. [3-5]. | |
| The prevalence of DM has increased over the years and the continuing trend needs further attention. The recent report revised on March 2015 by the American Diabetes Association showed that 9.3% (29.1 million) of the American population have diabetes with 21 million cases diagnosed and 8.1 million cases undiagnosed. Approximately 5% of the diagnosed diabetes was type 1 DM (T1DM) while approximately 90-95% was T2DM. In addition, 85.2% of people with T2DM were overweight or obese. The World Health Organisation (WHO) has predicted that the number of DM will increase from 143 million of 1997 to about 300 million in 2025, causes mainly by dietary pattern and other lifestyle factors. | |
| Dietary fatty acids are shown to play a key role in various pathological processes involved in DM [6,7]. It was reported that the type of fat intake could affect insulin action in human body [8].Besides, studies by Wu et al. and Gillen et al. have also demonstrated that unsaturated fatty acids are able to reduce the risk of T2DM [9,10]. In addition, a meta-analysis on short-term studies by Garg revealed that high monounsaturated fat diets improve lipoprotein profiles as well as glycemic control in T2DM patients [11]. Recent studies have also reported that a monounsaturated fatty acid (MUFA) rich Mediterranean style diet is able to improve glycemic control in T2DM subjects [12-15]. Olive oil is known as the main dietary fat source in the Mediterranean diet with mainly oleic acid (55-83%) as the source of MUFA reported by Codex Alimentarius 1997. While HOBO is another MUFA rich fat alternative available in Asia that produced by blending of palm olein and canola oil. Both OO and HOBO are rich in oleic acid. However, studies on the effects of these MUFA rich fats on the risk of diabetes are insufficient [16]. | |
| T2DM is closely related to obesity. A study by Seidell calculated that around 65-75% cases of DM in Caucasian could be avoided if the whole population does not have a body mass index (BMI) of exceeding 25 kg/m2 [17]. According to an investigation carried out by Kosaka et al., it was reported that incidences of DM in ethnic groups particularly of Asian origin increased at a levels of BMI at normal range for Europeans [18]. BMI seems to have an effect on the incidence of DM. Therefore, we aim to investigate the effects of these MUFA rich diets namely i) HOBO and ii) OO on selected markers of insulin resistance and glucose tolerance in healthy overweight Malaysian adults. | |
Methods |
|
| Study design | |
| This study was conducted in Metabolic Clinic, Malaysian Palm Oil Board, Bandar Baru Bangi, Malaysia, between November 2013 and Jun 2014. Ethical approval was obtained from the Ethic Committee for Research Involving Human Subject (JKEUPM), UPM, Serdang, Malaysia with reference number of FPSK_Jun (13) 01, and the study was registered at Clinicaltrials.gov as NCT02245113. Informed consent was obtained from volunteers prior to screening selection. | |
| A randomized crossover 3x3 Latin square and single blind study design was used. The primary outcome was the change in fasting c-peptide concentration. A sample size of 30 has an 80% power to detect a difference between means of 228 with a significance p=0.01 (this was the power to detect approximately one unit standard deviation change from a mean fasting value of 552) [19]. A total of 35 subjects were recruited in this study with the allowance for dropout of 5 subjects. A total of 32 subjects completed the study and 3 subjects withdrew from the study due to personal issues (Figure 1). | |
| Figure 1: CONSORT diagram of the study. | |
| A 3-wk standardization diet on palm olein that comprised of 30% kcal from fat, 15% kcal from protein and 55% kcal from carbohydrate was provided to subjects as run in diets. At the end of the standardization period, 35 subjects were randomly stratified by a computer program to three dietary treatment based on fasting serum c-peptide levels, gender, fasting total cholesterol and menstrual cycle (for females) to evenly distribute baseline variables. The three dietary treatments were diets prepared with test fats HOBO, OO, and CO with each period lasting for 6-wk in consecutive order with a 3-wk washout in between (Figure 2). | |
| Figure 2: Flow of study. | |
| Subjects | |
| A history-health and lifestyle questionnaire and biochemical tests were used to assess the health status of all volunteers. Biochemical tests which included hematology e.g. ferritin, red blood cell, hemoglobin, packed cell volume, mean cell volume, mean cell hemoglobin, mean cell hemoglobin concentration, platelet count, differential count e.g. neutrophill, lymphocyte, monocyte, eosinophill, basophill, atypical lymphocyte, diabetes screening for glucose level, kidney function tests e.g. urea, creatine-estimated glomerular filtration rate, calcium, inorganic phosphate, uric acid, sodium, potassium, chloride, lipid profile e.g. total cholesterol, high density lipoprotein, low density lipoprotein, triglyceride and liver function tests e.g total protein, albumin, globulin, albumin/globulin ratio, total bilirubin, alkaline phosphatase, serum glutamic oxaloacetic transamine, serum glutamic pyruvate transaminase and gamma-glutamyl transpeptidase were carried out. The premenopausal female volunteers went through pregnancy test using pregnancy test kit (Avo Diagnostics, Malaysia). Anthropometric measurements of heights and weights were recorded using a SECA height measurement meter and weighing scale, Tanita (BC-418, USA). The BMI was then calculated. Inclusion criteria for the screened subjects were overweight (BMI 23.0- 27.5 kg/m2) males and females aged 20-60, free from any disease, not on any medication or treatment associated with reduction of CVD, not pregnant or lactating (for females). Exclusion criteria were having one or more of the chronic diseases (DM, CVD, cancer or any liver/renal disease); having serum total cholesterol >7.8 mmol/L; with blood clotting problem, coagulopathy or deep vein thrombosis; hypertensive (systolic pressure >140 mmHg, diastolic pressure >90 mmHg); alcoholics; chronic smokers or having smoking history. Also, subjects having difficulty following the study guidelines were excluded. | |
| Test fats and diets | |
| HOBO is palm-based oil blended with canola oil and it is a registered trademark technology by MPOB as NOVELIN. HOBO is low in saturation with ratio of saturated fatty acid (SAFA): MUFA: polyunsaturated fatty acid (PUFA) of 0.4:1.0:0.4. HOBO was produced and supplied by Ace Edible Oil Industries Sdn. Bhd. (Klang, Selangor, Malaysia). OO was imported from Spain by a local distributor (Chemney Sdn. Bhd., Selangor, Malaysia) and CO was purchased from PGEO Edible Oils Sdn. Bhd. (Pasir Gudang, Johor, Malaysia). | |
| The test fats (HOBO, OO and CO) were incorporated into diets supplying 30%, 15% and 55% energy as fat, protein and carbohydrate, respectively at which test fats contributed two-thirds of the total dietary fat. Two sets 5-d menu were planned using Nutritionist Pro Software (AXXYA Systems LLC, Stafford, TX, USA), were alternately rotated every two weeks during the interventions. | |
| The major source of carbohydrate in the diets came from rice and noodles, while protein was sourced from fish and chicken. A caterer was assigned and the chef was only allowed to prepare the experimental diets with assistants’ help, following given detailed instructions, foods’ portion size and procedures. Subjects were provided with three meals (breakfast, lunch and dinner) daily on working weekdays. The three meals in total consist of approximately 2000 kcal energy. Subjects had their breakfast and lunch in the dining hall of Nutrition Unit, MPOB under supervision of an investigator, while dinner was packed into labeled containers for them to collect for home consumption. Test fat was given accordingly to each subject for household cooking usage during weekends/public holidays. Subjects were requested to follow a dietary guideline to prepare own meal at home. | |
| Sample collection | |
| Refer to Figure 2, fasting blood samples (for markers of insulin resistance: fasting c-peptide, glucose, insulin, HbA1c, and fructosamine), were collected from subjects on the first day (1st week) before the dietary intervention as baseline and at the end of wk-6 of each dietary period. Oral glucose tolerance tests (OGTT) were carried on wk-5 of each intervention. For OGTT, subjects ingested a solution containing 75 g of dextrose dissolved in 150 ml of drinking water after an 8 hours overnight fasting, and venous blood samples were collected at 0,15,30,60 and 120 min for markers of glucose, insulin and c-peptide. The area under the curve (AUC) for glucose, insulin and c-peptide curves were calculated using GraphPad Prism 7 across time point 0 to 120 min and a comparison was made between the three test diets. | |
| Prior to the each blood sample collection days, subjects were required to avoid caffeine/alcohol/medication/supplement intake and no strenuous exercises were allowed. Test meals were given as usual. Fasting was started at 2200 with drinking water allowed. | |
| Blood samples for markers of c-peptide, insulin and fructosamine were collected into serum tubes (BD Vacutainer® serum tubes, Franklin Lakes, NJ) and for glucose were collected into fluoride tubes (BD Vacutainer® serum tubes, Franklin Lakes, NJ), stood for approximately 30 min at room temperature before centrifugation at 1300 g for 15min (at 4°C). The upper layers of tube samples were separated and stored at -80°C freezer prior to analyses. Whole blood samples were collected into EDTA tubes for HbA1c marker. | |
| Analytical methods | |
| Triplicate portions of the 5-d rotational menus diets were collected for proximate analysis. Fat content was analyzed using Soxhlet method. Protein content was determined using Kjeldahl method while carbohydrate content was calculated by difference using the 494-Atwater factors of 4 kcal/g protein, 9 kcal/g fat, and 4 kcal/g carbohydrate (Table 1). Fatty acid composition was analyzed using Perkin Elmer Autosystem Gas Chromatography (SP-2560 column, 100 mm × 0.23 mm × 0.2 mm, Agilent Technologies; flame ionization detector; helium as carrier gas at 40psi; injector temperature at 250°C; oven temperature at 240°C). | |
| Table 1: Dietary intake of the 32 subjects participated in the study. | |
| The markers of insulin and c-peptide were determined by ADVIA Centaur XP Immunoassay System (SIEMENS, Erlangen, Federal Republic of Germany); glucose and fructosamine by ADVIA 2400 Chemistry System (SIEMENS, Erlangen, Federal Republic of Germany); and HbA1c by Automatic Glycohemoglobin Analyzer Adams HbA1c HA-8180 V (ARKRAY, Inc., Kyoto, Japan). Insulin resistance was calculated by HOMA-IR using the formula of: HOMAIR= fasting glucose (mmol/L)×fasting insulin (mU/L)/22.5 [20]. HOMA-β was determined by the formula: HOMA-β=20×fasting insulin (mU/L) /[fasting glucose (mmol/L)-3.5] % [21]. QUICKI was determined using the formula: QUICKI = 1/ [log fasting insulin (μU/ mL) + log fasting glucose (mg/dL)] [22]. | |
| Statistical analyses | |
| Data was analyzed using repeated-measured ANOVA, through the Bonferroni post hoc analysis using PASW Statistic 20 software (SPSS Inc, USA). Shapiro-Wilk’s normality test using GraphPad Prism 7 to check the normal distribution of data. Homogeneity of variance test was run through all the data. Baseline values were expressed as means ± SDs and the other data were expressed as means (95% LCI, 95% UCL) and logarithmic transformed to geometric means when appropriate. Significant difference was determined at P<0.05. | |
Results |
|
| The dietary intake and fatty acid contents of the three test diets were shown in Table 1. OO diet contained the highest level of oleic acid content (18.3 ± 3.0 %en). The SAFAs content is the lowest among the other two test diets with mainly palmitic acid (6.2 ± 0.8 %en). HOBO diet contained 11.8 ± 3.2 %en oleic acid, 4.3 ± 1.2 %en linoleic acid and 6.0 ± 1.1 %en of palmitic acid. The percentage difference between the total MUFA and PUFA content between these two diets were 6.5 %en and 1.2 %en. CO diet as a positive control in this study contained mainly 23.0 ± 4.6 %en SAFA [lauric (11.4 ± 2.5 %en) and myristic acids (5.0 ± 1.1 %en)], 3.9 ± 2.3 %en oleic acid and 2.0 ± 0.8 %en of linoleic acid. This study did not aim to differentiate the effects of individual fatty acids between the three test fats therefore the PUFA content was not standardised across the diets. | |
| Table 2 shows baseline characteristics of the 32 study subjects. Subjects were all of Malaysian origin, on average age of 30 y with BMI 25.5 kg/m2. The average fasting glucose, insulin, c-peptide, fructosamine, and HbA1c were 5.26 ± 5.0 mmol/L, 10.60 ± 5.81 mU/L, 1.51 ± 0.55 μg/L, 235.53 ± 39.56 μmol/L, and 5.39 ± 0.30 %, respectively, that fell within the normal range. All subjects had no metabolic syndrome, and were predicted to have a low 10- year coronary heart disease risk of 1.5 ± 0.7 % assessed using the Framingham risk score [23]. | |
| Table 2: Baseline characteristics of the 32 study subjects. | |
| Table 3 shows fasting concentrations of insulin resistance markers for the 32 subjects, and reading fell within normal persons’ range. No significant differences were observed for all the markers tested between the OO, HOBO, and CO diets. | |
| Table 3: Fasting concentrations of markers of insulin resistance for the 32 subjects participated in study. | |
| Figures 3, 4, and 5 show OGTT postprandial readings (time points of 0,15,30,60, and 120 min) for glucose, insulin and c-peptide, respectively. Glucose level was found to elevate after 15 min and peaked at 30 min. Glucose level started to drop after 30 min and was back to normal at 120 min in all three groups. The AUC for glucose showed no significant differences between the three test diets groups. Plasma insulin and c-peptide levels were found to elevate after 15min for all three test groups. HOBO and OO demonstrated similar postprandial pattern in which plasma insulin and c-peptide started to drop at 60 min. The CO group showed faster plasma insulin clearance that started at 30 min meanwhile plasma c-peptide for this group of subjects only started to drop at 60 min. However, no significant changes were found in AUC for plasma insulin and c-peptide for all three groups. | |
| Figure 3: A) OGTT postprandial readings (time points of 0, 15, 30, 60, and 120 min) for glucose and (B) AUC for OO, HOBO, and CO diets. | |
| Figure 4: A) OGTT postprandial readings (time points of 0, 15, 30, 60, and 120 min) for insulin and (B) AUC for OO, HOBO, and CO diets. | |
| Figure 5: A) OGTT postprandial readings (time points of 0,15,30,60, and 120 min) for c-peptide and (B) AUC for OO, HOBO and CO diets. | |
Discussion |
|
| HOBO, a palm based high oleic acid blended oil diet has no reported study to date on its effect on insulin resistance and glucose tolerance. In this study, we compared the effects of HOBO with OO that is commonly used in many countries for culinary purposes and both cooking oils contained oleic acid as the dominant fatty acid on insulin resistance and glucose tolerance in overweight adults. When compared to CO diet, we found that both HOBO and OO diets do not show significant difference on the effects on fasting glucose, c-peptide, insulin, fructosamine, HOMA-IR, HOMA-B, QUICKI and different time points of glucose, c-peptide and insulin levels after a 75 g OGTT. | |
| Overweight healthy subjects were recruited due to the reason that overweight adults possess greater risk for DM compared to normal adults in the Asian population, and effects of the three test fats diets on insulin resistance and glucose tolerance of the recruited subjects would be more differentiated. Scientific evidence suggests that the Asian population have different associations between BMI and health risks than the European population. However, there is no uniform threshold for BMI to the Asian population who are generally overweight and obese. An investigation by WHO Expert Consultation concluded that Asian people with a risk of DM had lower BMI than the existing WHO cut-off (25 kg/m2), where the cut off point for observed risk varies from 22 kg/m2 to 25 kg/m2 in different Asian populations (observed countries were China, Hong Kong, Indonesia, Japan, Singapore, and Thailand) [24]. While Chiu et al. suggested a lower BMI cut-off value which is 24 kg/m2 for South Asians [25]. This study screened and selected subjects with BMI 23.0-27.5 kg/m2 that were considered overweight. | |
| DM is diagnosed based on glucose levels either fasting glucose (threshold ≥ 7.0 mmol/L) or 2-h glucose (threshold ≥ 11.1 mmol/L) after a 75 g OGTT (26). In terms of fasting glucose, the overweight subjects in our study were found in the normal range (5.23-5.26 mmol/L). The 2-h glucose reading after a 75 g OGTT also fell in the normal range (4.80- 5.01 mmol/L). Although the subjects’ BMI readings were overweight (25.5 ± 1.7 kg/m2), the risk of DM was not being demonstrated in terms of fasting and 2-h glucose levels. Therefore, the three test diets did not affect insulin resistance and impaired glucose tolerance in this 3x6 weeks study despite the differences in terms of SAFA in coconut oil as well as MUFAs levels in both OO and HOBO diets. | |
| Prediabetes defines an individual with impaired fasting glucose (IFG) and/or impaired glucose tolerance (IGT). Prediabetes individuals possess glucose levels that do not meet the criteria for diabetes but are too high to be considered normal. The FPG levels of 5.6- 6.9 mmol/L for IFG and 2-h PG OGTT values of 7.8-11.0 mmol/L for IGT, were defined [26]. WHO defines the threshold for IFG at 6.1 mmol/L [27]. According to the latest documentations, an International Expert Committee added the A1C with threshold ≥ 6.5% as the third criteria to diagnose DM [28], while the American Diabetes Association considers an A1C range of 5.7-6.4% to identify individuals with prediabetes referring to previous studies [29-31]. Insulin is a hormone produced by the pancreas to control blood sugar and fasting insulin concentrations have been used as an index of insulin resistance [32]. C-peptide is a breakdown product when the insulin hormone is produced and released. C-peptide is more stable than insulin and hence it was used as primary outcome for this study [33]. In this study, measured levels of fasting glucose, 2-h glucose, HbA1c, insulin and c-peptide of subjects on all the three test fat diets were considered normal and no significant difference were found between the three test groups. | |
| Both HOMA and QUICKI have widely been validated and applied for quantifying insulin resistance and β-cell function [34]. A metaanalysis by Song et al. further investigated its effects in relation to diabetes risk in ethnically diverse populations and stated that HOMAIR is a robust surrogate marker compared with fasting glucose, insulin, or HOMA-B in each of four ethnic groups of American women with different diabetes risk factor profiles [35]. They concluded that high HOMA-IR and low HOMA-B were independently and consistently associated with an increased diabetes risk in a multiethnic cohort of U.S. postmenopausal women. HOMA-IR, HOMA-B and QUICKI of this study’s subjects fall into normal glucose tolerance range which was comparable to those of Borai et al. reported values of 117 (101- 134), 2.0 (1.7-2.3), and 0.353 (0.343-0.364) for HOMA-IR, HOMA-B, and QUIKI, respectively for a total number of 15 subjects within 20-65 years age range [36]. The subjects in this study were found to be healthy with normal baseline characteristics although they are overweight. The subjects are categorized as non-glucose tolerance group based on their HOMA and QUICKI measurements and no significant differences were observed between the three test fats. | |
| A recent subgroup analysis by Salas-Salvadó et al. concluded that Mediterranean diet enriched with extra virgin olive oil may reduce diabetes risk among persons with high cardiovascular risk [37]. In another study, Violi et al. examined on healthy subjects and results show extra virgin olive oil improves post-prandial glucose [38]. However, human trials on coconut oil diet’s effect on insulin resistance are limited. Recently, a mice study by Deol et al. found that a moderately high fat diet (40% kcal of total fat) of coconut oil did not induce elevated fasting blood glucose and glucose intolerance at week 20 [39]. Study in diabetic rats by Kochikuzhyil et al. suggested that the presence of lauric acid in coconut oil may protect against diabetes induced dyslipidemia [40]. A 14 y follow up study on female subjects by Salmeron et al. found that the intake of saturated or monounsaturated fatty acids were not significantly associated with the risk of DM [41]. The outcomes of our study are in line with the recent review by Morio et al. who reported there is no evidence to show that dietary saturated fatty acids from varied food sources affect the risk of insulin resistance or T2DM [42]. | |
| This study has a limitation of unstandardized linoleic acid (C18:2) content across the diets as we aim to investigate selected test fats that are used commercially for household cooking purposes. The amount of fat incorporated into the diet and the short duration of the intervention may contribute to the null effects of the test diets. It was found that the quantity of fat intake leads to obesity and insulin resistance instead of the fat’s quality (saturated or monounsaturated) [42-44]. In this study, the three test diets were standardized at ~30% kcal fat that demonstrated no effects on insulin resistance and glucose tolerance. Although no significant was found on the effects of HOBO and OO on the markers tested, however, we are the first to investigate the effects of HOBO vs OO using CO as a SAFA rich control to provide preliminary health data in healthy overweight adults that contributed to the novelty of this study. | |
Conclusion |
|
| In conclusion, approximately 2000 kcal daily test diets with 30% kcal fat (test fat incorporated two-third), 15% kcal protein and 55% kcal carbohydrate provided by the three test fat diets do not impair insulin resistance and glucose intolerance after a 6-wk intervention. The HOBO diet was found to be comparable to the OO diet in perspective of insulin resistance and protects against DM in overweight Malaysian. The CO diet also show similar trend compared to the HOBO and OO diets. Therefore, we suggested that the MUFA rich OO and HOBO diets will not adversely affect insulin resistance and glucose tolerance in healthy overweight subjects if the amount of fat consumed was kept at ~30% kcal. | |
Acknowledgment |
|
| The study grant was supported by Malaysian Palm Oil Board. We would like to express our great appreciation to the Director General of Malaysian Palm Oil Board, Datuk Dr. Choo Yuen May for her approval to publish the finding. Also, we would like to acknowledge the laboratory technicians and staffs from the Nutrition Laboratory of Malaysian Palm Oil Board for their assistance in conducting the study. | |
| The authors’ responsibilities were - STL, PTV, TKWN, and NME: conceived and designed the study, coordinated to the running of the study, samples analysis and writing of manuscript; VLKM: advised on subjects screening, involved in sample collection and reviewed the manuscript; HAS and SPL: contributed to the editing of manuscript and advisory throughout the study. | |
| STL is a postgraduate student of Faculty of Medicine and Health Sciences, University Putra Malaysia. PTV is a research officer in the Nutrition Unit of the Malaysian Palm Oil Board, which is a fully government research institution. TKWN was the former head of the Cardiovascular, Diabetes, and Nutrition Research Centre, Institute for Medical Research, Kuala Lumpur, Malaysia, and is currently an associate professor in the Department of Nutrition and Dietetics, International Medical University. NME is an associate professor and she is the head of the Department of Nutrition and Dietetics, University Putra Malaysia. HAS and SPL are associate professors in the Department of Nutrition and Dietetics, University Putra Malaysia. VLKM is a family physician and the head of Department of International Medical University Medical Clinic; she is also a clinical associate professor in the Department of Family Medicine. | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi