Research Article, J Sleep Disor Vol: 8 Issue: -1
Treatment of Primary and Secondary Sleep RelatedBruxism with the Luco Hybrid OSA Appliance
Ken Luco*
Luco Hybrid OSA Appliance Inc, University of Alberta, Kingston, Canada
*Corresponding Author: Ken Luco
DDS, President at Luco Hybrid OSA Appliance Inc, 1419 Butternut Creek Road, Canada
Tel: 613 888 6019
Fax: 613 544 0885
E-mail: drluco@sympatico.ca
Received: December 03, 2018 Accepted: December 21, 2018 Published: December 28, 2018
Citation: Luco K (2018) Treatment of Primary and Secondary Sleep Related Bruxism with the Luco Hybrid OSA Appliance®. J Sleep Disor: Treat Care 7:4. doi: 10.4172/2325-9639.1000223
Abstract
Sleep-related bruxism is classified as a repetitive jaw-muscle activity characterized by clenching or grinding and/or bracing or thrusting of the mandible during sleep. It has been shown in previous studies to occur in both a primary form as well as a secondary form. The Luco Hybrid OSA Appliance�?? has been shown to be a very effective treatment of primary sleep-related bruxism as well as controlling the hyperstimulation of the trigeminal cardiac reflex that accompanies sleeps related-bruxism. The aim of this study is to determine if this device is effective in the treatment of secondary sleep relatedbruxism resulting from sleep disordered breathing as well as from administration of selective serotonin reuptake inhibitor (SSRI) type medications. It was found that the Luco Hybrid OSA Appliance�?? was effective in the treatment of primary and both forms of secondary sleep-related bruxism however the SSRI group took significantly longer to respond (8 weeks vs. 2-3 weeks) to treatment.
Keywords: Sleep-related bruxism; Sleep disordered breathing; Luco Hybrid OSA Appliance; Trigeminal cardiac reflex; Trigemino-cardiac reflex; HTR2A polymorphism; SSRI selective serotonin re-uptake inhibitors
Abbreviations
AHI: Apnea-Hypopnea Index; BRIN: Sleep Bruxism Index or hourly rate of sleep bruxism events; HSS: Home Sleep Study; LHOA: Luco Hybrid OSA Appliance®; MAD: Mandibular Advancement Appliance; SRB: Sleep-Related Bruxism; SDB: Sleep Disordered Breathing; SB1: Primary Sleep-Related Bruxism; SB2A: Sleep-Related Bruxism due to Sleep Disordered Breathing; SB2B: Sleep-Related Bruxism due to Selective Serotonin Re-Uptake Inhibitors; SSRI: Selective Serotonin Re-Uptake Inhibitors; TCR: Trigeminal Cardiac Reflex Or Trigemino-Cardiac Reflex; TMJ: Temporomandibular Joint; TMD: Temporomandibular Dysfunction
Introduction
Sleep-related bruxism (SRB) is classified by the American Academy of Sleep Medicine (AASM) in the International Classification of Sleep Disorders (3rd Edition) (ICSD) as a sleep related movement disorder characterized by clenching or grinding and/or bracing or thrusting of the mandible during sleep [1,2]. Although SRB primarily occurs in a primary form, recent studies have linked SRB to sleep disordered breathing (SDB) [3], selective serotonin re-uptake inhibitor (SSRI) medications [4] as well as genetic mutations including the HTR2A [5,6] and DDR3 polymorphisms [7] (affecting both the serotonergic and dopaminergic systems of the central nervous system). In a recent paper by the author, it was shown that primary SRB, SRB occurring with SDB, and SRB occurring due to SSRI administration, all stimulate SRB similarly with a hyper-stimulation of the trigeminal cardiac reflex (TCR) [8]. In 2016, the FDA cleared the Luco Hybrid OSA Appliance® (LHOA) as the first treatment of SRB (K160477) [9]. The LHOA has been shown to reduce the bruxism index (BRIN) as well as the TCR hyper-stimulation to normal levels in SRB [10]. The purpose of this study was to determine the effectiveness of the LHOA in reducing the BRIN and TCR hyper-stimulation for SRB due to SDB and SRB due to SSRI administration.
The LHOA is classified as a mandibular advancement appliance (MAD) as it repositions the mandible in a forward positioning (Figure 1). It places the bite into a forward positioning over the maxillary cuspids activating the periodontal masseter reflex, a masticatory protective reflex, limiting the masseter and temporalis muscles to normal bite forces. These two powerful muscles are the primary muscles involved in sleep bruxism and have been previously shown to exert forces far greater than normal chewing.
Studies have shown that conventional mandibular advancement appliances can result in temporomandibular dysfunction (TMD) (specifically crepitation of the TMJ) within one to three months of use in patients suffering from both SDB and SRB [11]. With SRB inactivating the masseter and temporalis inhibitory reflexes, the masseter and temporalis muscles are allowed to contract with 100% of their capacity, which can overload the teeth, supporting periodontium and the TMJ [12]. The FDA lists TMD as a known risk in their guidance documents for mandibular advancement appliances. The LHOA does not carry this risk with its activation of the periodontal masseter reflex. In 2016, the FDA cleared the LHOA as the first treatment of sleep bruxism and to aid in treatment of tension and migraine type headaches in adults [13].
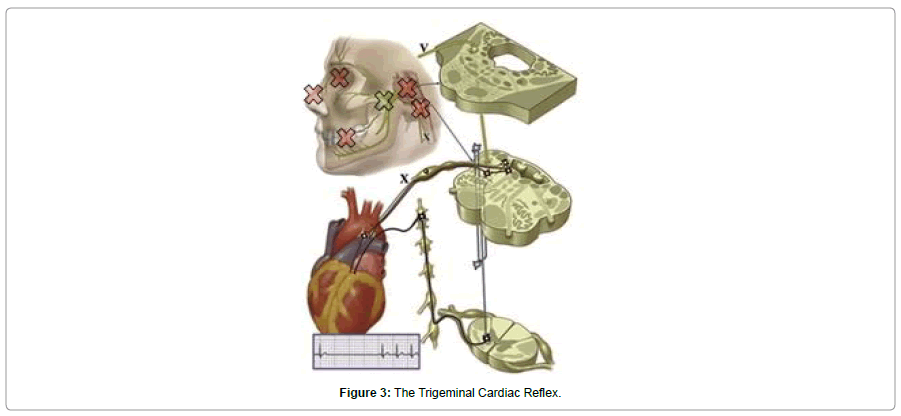
The trigeminal cardiac reflex
The TCR [14] was first discovered in humans in 1999 by Stefanovska [15]. It is characterized typically as a rapidly occurring significant bradycardia, hypotension, apnea, decreased cranial pressure and altered gastric motility in response to stimulation of one of the many branches of the trigeminal nerve [16]. The TCR may be stimulated along the trigeminal nerve either externally or internally, by electrical, mechanical or chemical stimulation and actually a represents a collection of cranial reflexes. A recent classification of the TCR proposed six different presentations of the TCR that make up the collective TCR reflex [17]. The skin around the nasal mucosa is the first, and has been traditionally been labelled the “Naso-cardiac” or “Diver’s Reflex [14]” as placing the face into cold water can elicit the response. The “Oculo-cardiac Reflex [18]”, the second type, is elicited by external pressure inward on the orbit of the eyes. The “Maxillo- Mandibular Reflex [19]” results due to stimulation either of the V2 and/or V3 branches of the trigeminal nerve, externally. The” Gasserian Ganglion Reflex [20]” occurs with direct stimulation of the Gasserion ganglion. The “Central Reflex [21]” results from direct stimulation of the brainstem branches of the trigeminal nerve in the pons in the region of the trigeminal sensory nucleus and the last, the “Brainstem Reflex [22]”, results from direct stimulation of the brainstem below the trigeminal sensory nucleus region.
Of significance, the Gasserian Ganglion Reflex [17] is the only reflex of the six that has a converse effect on the TCR. Tachycardia, hypertension, hyperpnea [23], increased cranial pressure [24] and increased gastric activity (often manifesting as acid reflux [25] (GERD)) occurs with stimulation of the TCR In this region. Both sleep bruxism and restless leg syndrome have been shown to affect the TCR at the level of the Gasserion Ganglion [26].
Studies have also shown that heart rate changes are the most significant measurement of TCR stimulation [23] and it is medically accepted that there must be a change (increase or decrease) of 20% or greater in heart rate before a diagnosis of TCR stimulation may be made [27]. In SRB, the increase in heart rate that occurs is routinely in the order of 50-90%+, indicating SRB is hyper-stimulating the TCR, well above the minimum 20%.
The study design
The purpose of this study was to determine if the LHOA device is effective in the treatment of secondary SRB and associated hyperstimulation of the TCR. With recent publications demonstrating SSRI stimulation/exacerbation of SRB as well as synchronicity of SRB with SDB events, both of these secondary SRB groups were included in the study.
Subject selection
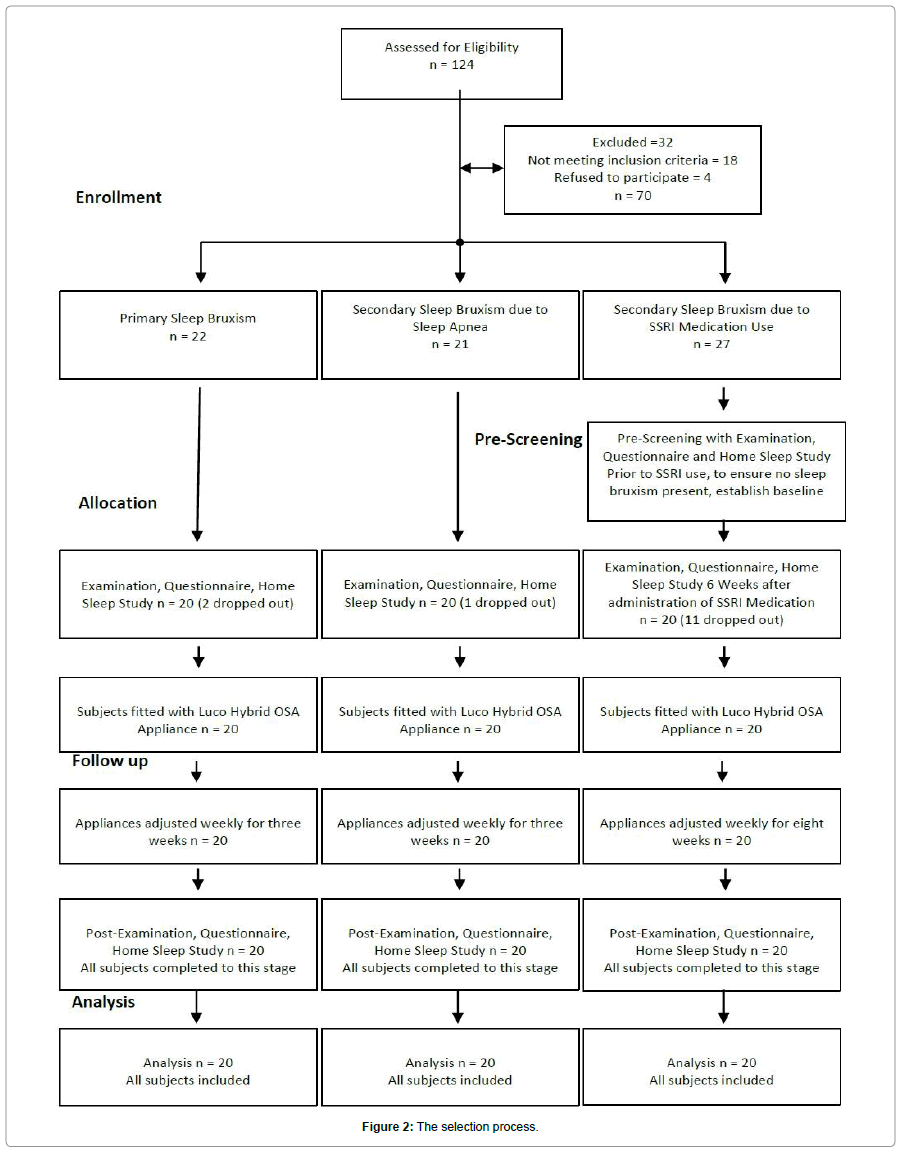
The subjects for each group were selected from a pool of onehundred eighty-four patients referred to our clinic for management of chronic moderate to severe facial/TMJ pain. All completed a detailed pain questionnaire, were examined and broken into one of the three groups. Figure 2 diagrams the selection process used. For the primary (SB1) and secondary due to sleep disordered breathing (SB2A) groups, a diagnosis of probable sleep bruxism (based upon reported symptoms as well as clinical findings) was necessary to be included (Figures 3 and 4). A total of 20 subjects for each group were eventually selected. For the third group, secondary sleep bruxism due to SSRI use, the subjects were selected based upon the criteria of no history of SSRI use, and no diagnosis of sleep bruxism (based upon reported history and clinical findings) but were previously diagnosed with clinical depression. 31 subjects were subsequently were placed on SSRI type medications for a period of 6 weeks, after which they all developed symptoms of SRB. 20 subjects who met these criteria were included in this group. For all three groups, there were 10 male and 10 female subjects (Figure 2). The SSRI medications studied included sertraline (n=7), fluoxetine (n=6), paroxetine (n=4) and citalopram (n=3). Of the 31 subjects, 11 dropped out leaving 20 in the study. All subjects provided informed consent for the study.
The reported historical elements considered significant were bruxism during sleep, witnessed by a spouse or by the patient awakening, aware of bruxism. Hypersensitivity of the teeth to temperatures and a history of temporal, frontal or occipital headaches were a requirement. Significant clinical findings included restricted mandibular range of motion, pain on palpation of the masseter, temporalis and posterior digastric muscle groups, pain on palpation of the TMJ and/or the presence of mandibular tori, linea alba (buccal) lesions. Scalloping of the lateral borders of the tongue and a mandibular tremor were also considered significant. The SB1 and SB2A groups tested positive for most or all of these clinical signs as well as providing a positive history. The SB2B group demonstrated none of these clinical signs and initially had a negative history for sleep bruxism at the onset.
All three groups underwent polysomnographic sleep recordings with EMG and audio at the onset, as a baseline as well as to screen for pre-existing primary sleep bruxism. The SB1 and SB2A groups all resulted in a positive polysomnographic diagnosis of SRB. The SB2CB group’s initial polysomnograph tested negative for SRB. The SB2B group were started and titrated on an SSRI class medication over a 6 week period. After this time, a second pain history, examination and HSS were completed. All tested positive in the HSS for SRB in the post-SSRI polysomnogram.
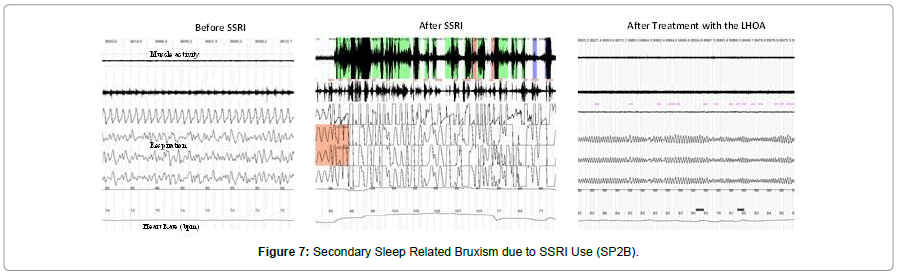
All three groups were fitted with a LHOA and titrated over a 3 week period (weekly appointments) maintaining a 75% forward positioning for all subjects. After this, a pain history, an examination, and post-treatment HSS were completed for the SP1 and SP2A groups. For the PS2B (SSRI) group, it was necessary to extend the treatment an additional 5 weeks (to 8 weeks) due to incomplete response and the need for additional titration. Figure 5 demonstrates a sleep tracing of secondary sleep related bruxism due to SSRI use. The first tracing is the baseline tracing and the second is 6 weeks after SSRI use in the same patient. The events are very similar in appearance to primary sleep related bruxism. They are not associated with sleep disordered breathing and show a rapid increase in heart rate typical of TCR activation seen in SRB, from 64 bpm to 108 bpm (a 68.8% increase). The Right Tracing is after treatment with the Luco Hybrid OSA Appliance. At 8 weeks, a third post treatment history, examination and HSS were eventually completed on the SR2B group.
Results
SB1 group: Primary SRB
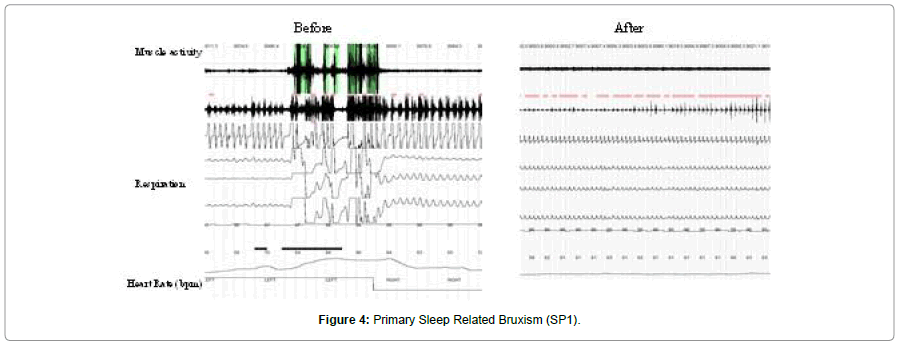
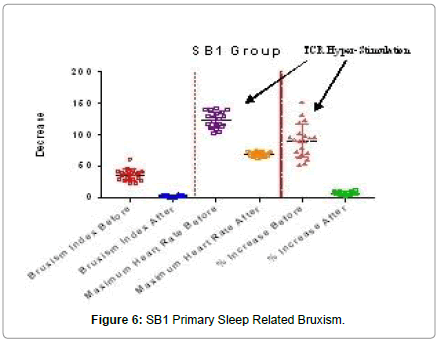
All subjects in the SB1 (primary SRB) group demonstrated a mandibular tremor, with is characteristic of SB [28]. Mean BRIN was 34.1 with a range of 26.7 to 61.5. Mean maximum heart rate was 123.5 bpm with a range of 101.8 to 141.8 bpm. The mean % increase in heart rate was 90.6% with a range of 52.4 to 151.8%. This increase of 90.6% in mean maximum heart rate far exceeds the medically accepted increase/decrease of 20% for a positive diagnosis of TCR stimulation. The mean BRIN of 31.4/hr indicated the presence of severe SRB (Table 1). Figure 6 demonstrates significant sleep related bruxism before and normal after treatment with a Luco Hybrid OSA Appliance®. A significant increase in mean maximum heart rate as well as in mean % increase in heart rate due to sleep related bruxism events can also be seen. After treatment with the Luco Hybrid OSA Appliance®, mean maximum heart rate and mean % increase were significantly reduced, into the normal range. The mean % increase in heart rate before of 90.6% was well above the 20% threshold for TCR hyperstimulation. After treatment with the Luco Hybrid OSA Appliance, the % increase was 6.8% indicating no stimulation of the TCR.
| Bruxism Index | Maximum Heart Rate | % Increase Heart Rater | ||||||
|---|---|---|---|---|---|---|---|---|
| Gender | Age | Before | After | Before | After | Before | After | |
| 1 | M | 30 y 3 m | 29.3 | 2.2 | 108.6 | 66.3 | 73.8 | 6.8 |
| 2 | M | 24 y 7 m | 41.8 | 2.4 | 116.1 | 71.5 | 69.7 | 7 |
| 3 | F | 31 y 5 m | 31.6 | 2.9 | 129.6 | 64.8 | 123.8 | 11.5 |
| 4 | F | 33 y 5 m | 38.1 | 3 | 141.8 | 66.7 | 131.7 | 11 |
| 5 | F | 24 y 6 m | 22.8 | 2.4 | 139.8 | 72.5 | 95.5 | 4.6 |
| 6 | M | 29 y 7 m | 26.7 | 1 | 101.8 | 66.3 | 52.4 | 5.7 |
| 7 | F | 35 y 8 m | 31.4 | 1.9 | 112.5 | 69.3 | 63.8 | 4.1 |
| 8 | M | 33 y 3 m | 26.9 | 2.4 | 117.5 | 64.1 | 96.2 | 10.3 |
| 9 | M | 46 y 2 m | 28.7 | 1.7 | 135.6 | 73.5 | 90.4 | 5 |
| 10 | F | 35 y 8 m | 39.1 | 3.3 | 129.5 | 70.1 | 95 | 6.2 |
| 11 | F | 33 y 0 m | 45.5 | 4 | 111.3 | 74.8 | 54.4 | 5.4 |
| 12 | F | 27 y 1 m | 39.1 | 3.1 | 104.5 | 70.1 | 71.9 | 5.1 |
| 13 | M | 27 y 9 m | 22.7 | 3.6 | 116.7 | 64.8 | 90.4 | 6.6 |
| 14 | M | 38 y 5 m | 37.1 | 4.1 | 128.8 | 69.9 | 92.8 | 5.4 |
| 15 | M | 42 y 3 m | 39.4 | 3.1 | 134.7 | 70.2 | 97.5 | 4.3 |
| 16 | F | 24 y 5 m | 41 | 3.3 | 115.9 | 74 | 60.1 | 2.8 |
| 17 | F | 31 y 6 m | 44.4 | 3.8 | 140 | 62.1 | 151.8 | 13.1 |
| 18 | M | 44 y 6 m | 61.5 | 4.8 | 139.1 | 68.7 | 119.4 | 8.9 |
| 19 | M | 37 y 5 m | 33.7 | 2.7 | 124.7 | 66.2 | 102.1 | 7.6 |
| 20 | F | 32 y 3 m | 35 | 2.1 | 121 | 70 | 79.5 | 4 |
| Mean | 33 y 2 m | 34.1 | 2.9 | 123.5 | 68.8 | 90.6 | 6.8 | |
| Range | 26.7 to 61.5 | 1.0 to 4.8 | 101.8 to 141.8 | 62.1 to 74.8 | 52.4 to 151.8 | 2.8 to 13.1 | ||
Table 1: SB1 Primary Sleep Bruxism Group.
After treatment with a LHOA, mean BRIN significantly decreased from 34.1 to 2.9 (91.57% reduction, p<0.0001) indicating the device successfully treated the sleep bruxism. The mean maximum heart rate was significantly reduced from 123.5 bpm to 68.8 bpm (44.3% decrease, p<0.0001), indicating that the TCR was no longer being hyper-stimulated (Figure 3).
SB2A group: Secondary SRB due to sleep disordered breathing
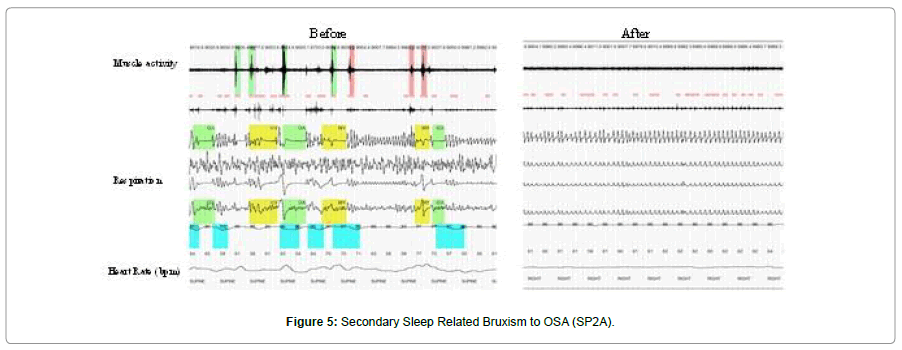
All subjects in the SB2A group also demonstrated a mandibular tremor, with is characteristic of SB. Mean maximum heart rate was 125.0 bpm with a range of 105.7 to 141.8 bpm. The mean % increase in heart rate was 101.3% with a range of 72.7 to 140.6%. This mean increase of 101.3% in heart rate far exceeds the medically accepted increase/decrease of 20% for a positive diagnosis of TCR stimulation. The mean AHI was 25.5 with a range of 19.0 to 33.0 indicating sleep disordered breathing. The mean BRIN of 33.1 indicated the presence of severe SRB (Table 2). Figure 7 of the SB2A group demonstrates a significant increase in mean maximum heart rate as well as in mean % increase in heart rate due to sleep disordered breathing events. The AHI score indicates moderate OSA before and normal after treatment. After treatment with the Luco Hybrid OSA Appliance, mean maximum heart rate and mean % increase were significantly reduced, into the normal range. The mean % increase in heart before treatment of 101.3% was well above the 20% threshold for TCR hyperstimulation. After treatment with the Luco Hybrid OSA Appliance, the % increase was 6.8 indicating no further stimulation of the TCR.
| Bruxism Index | Maximum Heart Rate | % Increase Heart Rate | AHI | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Gender | Age | Before | After | Before | After | Before | After | Before | After | |
| 1 | M | 49 y 6 m | 41.1 | 4.1 | 114.1 | 66.3 | 89.9 | 6.8 | 26.3 | 4 |
| 2 | M | 41 y 5 m | 38.4 | 1.1 | 124.5 | 71.5 | 124.7 | 7 | 22.7 | 3.9 |
| 3 | F | 42 y 1 m | 36.8 | 4.5 | 114.7 | 64.8 | 96.4 | 11.5 | 29.1 | 3.4 |
| 4 | F | 33 y 2 m | 31.8 | 2 | 132.6 | 66.7 | 136.4 | 11 | 30.1 | 3.7 |
| 5 | F | 40 y 1 m | 34.1 | 3.3 | 133.9 | 72.5 | 101.7 | 4.6 | 32.7 | 2.8 |
| 6 | M | 30 y 7 m | 29.7 | 4.2 | 111.2 | 66.3 | 86.3 | 5.7 | 24.9 | 4.8 |
| 7 | F | 26 y 4 m | 32.5 | 2 | 105.7 | 69.3 | 72.7 | 4.1 | 33 | 4 |
| 8 | M | 24 y 1 m | 26.5 | 4.8 | 124.8 | 64.1 | 97.8 | 10.3 | 23.1 | 2 |
| 9 | M | 30 y 11 m | 22.8 | 3.1 | 126.1 | 73.5 | 94.6 | 5 | 21.9 | 1.7 |
| 10 | F | 50 y 5 m | 36.7 | 1.7 | 127.6 | 70.1 | 113.7 | 6.2 | 21.4 | 1.8 |
| 11 | F | 34 y 11 m | 40.1 | 0.1 | 122.2 | 74.8 | 84.3 | 5.4 | 26.8 | 2.1 |
| 12 | F | 28 y 1 m | 29.7 | 0.7 | 115.4 | 70.1 | 86.7 | 5.1 | 25.3 | 3 |
| 13 | M | 48 y 4 m | 22.4 | 1.7 | 124.8 | 64.8 | 85.4 | 6.6 | 20.7 | 2.8 |
| 14 | M | 24 y 10 m | 32.9 | 2.9 | 123.8 | 69.9 | 86.7 | 5.4 | 19 | 2.4 |
| 15 | M | 37 y 2 m | 38.9 | 4.1 | 129.3 | 70.2 | 118 | 4.3 | 30.4 | 1.7 |
| 16 | F | 25 y 10 m | 29.1 | 3.1 | 141.8 | 74 | 131.7 | 2.8 | 27.7 | 3.4 |
| 17 | F | 43 y 0 m | 33.3 | 3.4 | 134.7 | 62.1 | 100.1 | 13.1 | 21.5 | 4.9 |
| 18 | M | 40 y 7 m | 37.4 | 4.8 | 138.6 | 68.7 | 140.6 | 8.9 | 26.1 | 4.1 |
| 19 | M | 43 y 7 m | 31.9 | 2.9 | 132.2 | 66.2 | 98.2 | 7.6 | 27.6 | 1.7 |
| 20 | F | 23 y 2 m | 36.8 | 1.8 | 121.7 | 70.2 | 80.8 | 4 | 19.3 | 1 |
| Mean | 35 y 11 m | 33.1 | 2.8 | 125 | 68.8 | 101.3 | 6.8 | 25.5 | 3 | |
| Range | 22.4 to 40.1 | 0.1 to 4.8 | 105.7 to 141.8 | 64.8 to 73.5 | 72.7 to 140.6 | 2.8 to 13.1 | 19.0 to 33.0 | 1.0 to 4.9 | ||
Table 2: SB2A Secondary Sleep Bruxism due to Sleep Disordered Breathing Group.
After treatment with a LHOA, there was a significant reduction in mean BRIN decreased from 33.1/hr to 2.8/hr (91.5% reduction, p<0.0001) indicating the device successfully treated the sleep bruxism. The mean maximum heart rate was significantly reduced from 101.3 bpm to 68.8 bpm (32.1% reduction (p<0.0001). This strongly suggests that the TCR was no longer being hyperstimulated (Figure 4).
SB2B group: Secondary SRB due to SSRI administration
This group of patients was tested on three occasions: before the administration of an SSRI type medication, 6 weeks after administering an SSRI medication, and 8 weeks after therapy with a LHOA. Mean BRIN at onset was 2.1 (range of 0.2 to 4.7), after SSRI was 29.1 (range of 18.6 to 59.7, a significant increase of 1285.7% (p<0.0001). Maximum heart rate at onset was 68.2 bpm (range of 60.1 to 74.3 bpm), after SSRI was 127.0 bpm (range of 99.7 to 138.7 bpm, a significant increase of 86.2% (p<0.0001). The percentage increase of heart rate at onset was 5.4% (range of 2.2 to 10.4%), after SSRI was 90.3% (range of 51.8 to 117.7 bpm), an increase of 1572.2% (Table 3).
| Bruxism Index | Maximum Heart Rate | % Increase in Heart Rate | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Gender | Age | Before SSRI | After SSRI | After Treatment | Before SSRI | After SSRI | After Treatment | Before SSRI | After SSRI | After Treatment | |
| 1 | M | 37 y 6 m | 3 | 38.6 | 8.1 | 60.1 | 124 | 59.2 | 3.8 | 113.4 | 3.5 |
| 2 | F | 30 y 4 m | 2.1 | 59.7 | 5.2 | 68.7 | 119 | 66.8 | 3.5 | 78.1 | 4.7 |
| 3 | F | 22 y 3 m | 0.7 | 28.4 | 3.9 | 66.6 | 99.7 | 67.4 | 2.9 | 51.8 | 4.3 |
| 4 | M | 21 y 4 m | 0.2 | 22.6 | 6.7 | 70.1 | 114.7 | 69.4 | 2.9 | 66 | 0.9 |
| 5 | M | 19 y 10 m | 1.1 | 19.8 | 11.2 | 65.4 | 135.4 | 62.3 | 9.2 | 117.7 | 0.8 |
| 6 | F | 33 y 4 m | 2.6 | 31.4 | 3.8 | 64.8 | 128.4 | 66.8 | 2.2 | 96.6 | 2.6 |
| 7 | M | 43 y 9 m | 1.7 | 42.4 | 5.1 | 72.2 | 134.4 | 69.8 | 6 | 92.8 | 2.5 |
| 8 | F | 42 y 0 m | 3.5 | 39.7 | 5.9 | 74.3 | 113.8 | 73.5 | 4.5 | 58.1 | 3.8 |
| 9 | F | 30 y 7 m | 3.1 | 16.8 | 6.9 | 70.3 | 127.1 | 70.1 | 2.2 | 82.1 | 2.6 |
| 10 | M | 38 y 5 m | 1.7 | 22.6 | 11.1 | 68.9 | 131.8 | 68.5 | 10.4 | 106.6 | 10.1 |
| 11 | M | 56 y1 1m | 1.5 | 28.7 | 9.3 | 61.8 | 122.5 | 66.6 | 8.2 | 104.8 | 13.5 |
| 12 | M | 44 y 0 m | 2.7 | 23.4 | 8.1 | 69.8 | 131.8 | 71 | 7.7 | 88.8 | 2.6 |
| 13 | F | 47 y 5 m | 2.8 | 29.1 | 6.8 | 66.8 | 136.5 | 66.8 | 5.7 | 106.5 | 1.5 |
| 14 | F | 50 y 8 m | 3.8 | 31.5 | 6.6 | 70.1 | 128.9 | 69.2 | 3.5 | 87.6 | 1.5 |
| 15 | M | 48 y 9 m | 4.7 | 33.7 | 7.2 | 69.1 | 144.8 | 69.9 | 6.1 | 112.6 | 2.8 |
| 16 | M | 28 y 10 m | 1 | 24.6 | 5.8 | 65.8 | 116.8 | 67.8 | 4.6 | 74.6 | 3.4 |
| 17 | F | 35 y 1 m | 1.9 | 19.5 | 11.8 | 69.7 | 129.8 | 68.3 | 10.3 | 90.6 | 0.6 |
| 18 | F | 35 y 9 m | 0.1 | 28.6 | 9.1 | 72.5 | 138.7 | 72.2 | 4.9 | 92.6 | 2.4 |
| 19 | M | 46 y 9 m | 1.4 | 18.6 | 10.9 | 69.9 | 132.5 | 71.8 | 3.7 | 88.7 | 2.7 |
| 20 | F | 53 y 2 m | 1.8 | 22.8 | 6.3 | 66.8 | 129.7 | 69.8 | 4.7 | 96.5 | 6.2 |
| Mean | 38 y 2 m | 2.1 | 29.1 | 7.5 | 68.2 | 127 | 68.4 | 5.4 | 90.3 | 3.7 | |
| Range | 0.1 to 4.7 | 16.8 to 59.7 | 3.8 to 11.8 | 60.1 to 74.3 | 99.7 to 138.7 | 59.2 to 73.5 | 2.2 to 10.4 | 51.8 to 117.7 | 0.8 to 13.5 | ||
Table 3: SB2B Secondary Sleep Bruxism due to SSRI Medication Use Group.
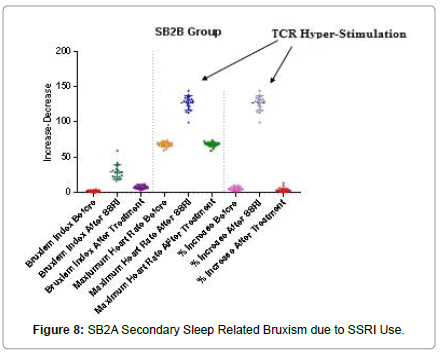
After treatment with the LHOA, a significant reduction in the mean BRIN of 74.2% (p<0.0001) was seen with a mean BRIN after SSRI use of 29.1 (range of 18.6 to 59.1) reduced to 4.9 (range of 3.8 to 11.2) following treatment. Mean maximum heart rate was significantly reduced after treatment from 127 bpm (range of 99.7 to 138.7) to 68.4 bpm (range of 59.2 to 71.8 bpm), a significant reduction of 46.1% (p<0.0001). The mean % increase in heart rate was significantly reduced (p<0.0001) after treatment with the LHOA by 95.9% with a mean % increase after the SSRI of 90.3% range of 51.8% to 117.7% to a mean % increase after treatment of 3.7% (range of 0.8 to 13.5%) (p<0.0001). Figure 8 demonstrates the significant increase in mean bruxism index, mean maximum heart rate and mean % increase seen after 6 weeks use of the SSRI. After treatment for 8 weeks with the Luco Hybrid OSA Appliance®, there was a significant reduction in mean bruxism index, mean maximum heart rate and mean % increase in heart rate, similar to the levels of the pre-SSRI measurements. The mean % increase in heart rate after SSRI use of 90.3% was well above the 20% threshold of TCR hyperstimulation. After treatment with the Luco Hybrid OSA Appliance®, the % increase was 3.7%, demonstrating no activation of the TCR.
Of significance, it took significantly longer to treat the SSRI group. The medication’s effect of increasing serotonin levels in the central nervous system and the HTR2A polymorphism common to SRB patients is likely underlying the resistance to treatment by hyperstimulation of the SRB. At 8 weeks however, all SSRI patients significantly responded, compared with 3 weeks for the other two groups.
Analysis
Analysis using the paired t test (two-tailed) was performed (using GraphPad Prism version 7.00 for Windows, GraphPad Software, La Jolla California USA, www.graphpad.com), for statistical comparison of before and after states.
For the SB1 and SB2A groups, % increase was calculated using the following formula:
% Decrease=Pre-Treatment Value/Post-Treatment Value–Post Treatment Value×100
For the SB2B group, % increase was calculated using the following formula:
% Increase=Post-SSRI Value–Initial Value/Initial Value×100
% Decrease=Post-SSRI Value–Post-Treatment Value/Post-SSRI Value×100
Discussion
SB1 group
The SB1 group responded as expected. There was a significant reduction (p<0.0001) in all parameters. Mean BRIN was significantly reduced by 91.5% (p<0.0001) and mean maximum heart rate by 44.3% (p<0.0001). The % change in heart rate was significantly reduced by 92.5% (p<0.0001). The LHOA was demonstrated to be effective in the treatment of primary SRB and in stabilizing the hyperstimulation of the TCR (7.5% P<0.0001) less than 20%. The results confirmed the previous published study 10. SSRI type medications prevent the resorption of serotonin in the central nervous system. With the HTR2A polymorphism known to affect SRB patients, this suppresses the masseter inhibitory reflex and activates central pattern generators responsible for the movements and forces seen in sleep bruxism. The HTR2A gene produces the 2A serotonin receptor and the polymorphism produces an excess of receptors rendering the system to be hypersensitive to serotonin. With the SSRI increasing serotonin levels and the HTR2A producing an excess of receptors, the inhibitory control centers of the masseter inhibitory reflex, located in the mesencephalic nucleus of the pons, are inactivated [29].
SB2A group
OSA and SRB have been demonstrated to have a relationship in studies [3]. This secondary form of SRB has been shown to be reduced with treatment of the OSA and this was confirmed in this study. All parameters improved significantly (p<0.0001). Mean BRIN was significantly reduced by 91.5% (p<0.0001), maximum heart rate was significantly reduced by 45.0% (p<0.0001), mean % increase in heart rate was significantly reduced by 93.3% and mean AHI was reduced by 88.34% (P<0.0001). Treatment with the LHOA was effective in treatment of the OSA, SRB and to stabilize the TCR stimulation under 20% (6.7%, p<0.0001). The LHOA is also FDA cleared for the treatment of sleep apnea and snoring in adults [30]. None of the subjects in the study developed TMJ symptoms or crepitation of their TMJ or were forced to drop out of the study due to TMJ complications.
PS2B group
The findings in this group were consistent with previous studies that SSRI type medications can initiate or exacerbate existing SRB [30]. Before SSRI use, all parameters fell within the normal range. 6 weeks after SSRI use, there was a significant increase in all parameters compared with the pre-SSRI values. The SSRI’s that were used by subjects in the study included sertraline, fluoxetine, citalopram and fluvoxamine. All subjects were titrated to therapeutic levels by their medical doctors.
Mean BRIN was significantly increased with SSRI use from 2.1 to 29.1 (p<0.0001). This was consistent with previous studies. Mean BRIN was not significantly different pre SSRI and after treatment with the LHOA (P<0.0001) demonstrating the LHOA was effective in reducing the mean BRIN to normal levels, even in the presence of SSRI use.
Mean maximum heart rate increased with SSRI use from 68.2 bpm to 127 bmp (86.2%, p<0.0001). This clearly demonstrated hyperstimulation of the TCR with SSRI use in excess of 20%. Treatment with the LHOA significantly decreased mean maximum heart rate by 31.9% (p<0.0001), from 127 bpm to 68.4 bpm indicating that treatment with the LHOA effectively stabilized the TCR and returned the values to pre SSRI use (68.2). There was no significant difference in pre-SSRI and post-LHOA values for all parameters studied. This group proved to be much more challenging in treating that the other two groups requiring a mean of 6 additional titration appointments compared to 2 for the SP1 and SP2A groups.
Conclusions
Treatment of SRB with the LHOA, whether primary or secondary due to SDB or use of SSRI type medications, was effective and significantly reduced the maximum heart rate, BRIN and % increase in heart rate to normal values. Treatment of the PS2B group was more complex and prolonged than the other groups, confounded by the SSRI medications. Even so, the LHOA was effective in treating the SRB.
SRB has been shown in many studies to cause significant increases in maximum heart rate, increases in both intra-cranial and in blood pressure, and an increase in gastric hypermotility (resulting in GERD). Treatment with the LHOA reduces these effects significantly, potentially reducing the cardiac risks associated with the TCR. Further investigation on a larger scale is definitely warranted as this non-pharmaceutical approach could provide a safe and well tolerated adjunct in cardiovascular disease therapy.
References
- American Academy of Sleep Medicine (2014) International classification of sleep disorders.
- Carra MC (2018) Sleep-related bruxism. Curr Sleep Med Reports 4: 28-38.
- Winck M, Drummond M, Viana P, Pinho JC, Winck JC (2017) Sleep bruxism associated with obstructive sleep apnoea syndrome - A pilot study using a new portable device. Rev Port Pneumol 23: 22-26.
- Melo G, Dutra KL, Filho RR, Ortega AOL, Porporatti AL, et al. (2018) Association between psychotropic medications and presence of sleep bruxism: A systematic review. J Oral Rehabil 45: 545-554.
- Unschuld PG, Ising M, Erhardt A, Lucae S, Kloiber S, et al. (2007) Polymorphisms in the serotonin receptor gene HTR2A are associated with quantitative traits in panic disorder. Am J Med Genet B Neuropsychiatr Genet 144B: 424-429.
- Piatto VB, Carvalho TB, De Marchi NS, Molina FD, Maniglia JV (2011) Polymorphisms in the 5-HTR2A gene related to obstructive sleep apnea syndrome. Braz J Otorhinolaryngol 77: 348-355.
- Oporto GH, Bornhardt T, Iturriaga V, Salazar LA (2018) Single nucleotide polymorphisms in genes of dopaminergic pathways are associated with bruxism. Clin Oral Investig 22: 331-337.
- Luco K (2018) A study comparing primary and secondary sleep bruxism and their effect on the trigeminal cardiac reflex. J Sleep Disor Treat Care 7.
- Luco K (2016) Publishing health services. Department of Health & Human Services.
- Luco K (2018) Short-term effects of the luco hybrid obstructive sleep apnea appliance in adults with sleep bruxism: A Quasi-experimental study. J Sleep Disord Treat Care 7.
- Duran-Cantolla J, Crovetto-Martínez R, Alkhraisat MH, Crovetto M, Municio A, et al. (2015) Efficacy of mandibular advancement device in the treatment of obstructive sleep apnea syndrome: A randomized controlled crossover clinical trial. Med Oral Patol Oral Cir Bucal 20: e605-e615.
- Ohmori H, Kirimoto H, Ono T (2012) Comparison of the physiological properties of human periodontal-masseteric reflex evoked by incisor and canine stimulation. Front Physiol 3: 233.
- US Food and Drug Administration (2018) Industry (Medical devices).
- Panneton WM (2013) The mammalian diving response: An enigmatic reflex to preserve life? Physiology (Bethesda) 28: 284-297.
- Stefanovska A, Bracic M, Kvernmo HD (1999) Wavelet analysis of oscillations in the peripheral blood circulation measured by laser doppler technique. IEEE Trans Biomed Eng 46: 1230-1239.
- TeachMeAnatomy (2018) The Trigeminal Nerve (CN V).
- Meuwly C, Golanov E, Chowdhury T, Erne P, Schaller B (2015) Trigeminal cardiac reflex: new thinking model about the definition based on a literature review. Medicine (Baltimore) 94: e484.
- Dunville LM, Kramer J (2018) Oculocardiac reflex. StatPearls Publishing, Florida, United States.
- Walker HK (1990) Cranial nerve V: the trigeminal nerve. The history, physical, and laboratory examinations, (3rd edtn), Boston: Butterworths, Massachusetts, United States.
- Luco K (2018) The relationship of the trigemino-cardiac reflex to sleep bruxism. Journal of Neurology and Brain Disorders 1.
- Duan HZ, Zhang Y, Li L, Zhang JY, Yi ZQ, et al. (2017) Trigemino-cardiac reflex in skull base surgery. Beijing Da Xue Xue Bao 49: 164-168.
- Mizumori T, Sumiya M, Kobayashi Y, Inano S, Yatani H (2013) Prediction of sleep bruxism events by increased heart rate. Int J Prosthodont 26: 239-243.
- Nashed A, Lanfranchi P, Rompre P, Carra MC, Mayer P, et al. (2012) Sleep bruxism is associated with a rise in arterial blood pressure. Sleep 35: 529-536.
- Castroflorio T, Bargellini A, Rossini G, Cugliari G, Deregibus A (2017) Sleep bruxism and related risk factors in adults: A systematic literature review. Arch Oral Biol 83: 25-32.
- Winter AC, Berger K, Glynn RJ, Buring JE, Gaziano JM, et al. (2013) Vascular risk factors, cardiovascular disease, and restless legs syndrome in men. Am J Med 126: 220-227.
- Meuwly C, Chowdhury T, Sandu N, Golanov E, Erne P, et al. (2017) Definition and diagnosis of the trigeminocardiac reflex: a grounded theory approach for an update. Front Neurol 8: 533.
- Laine CM, Yavuz SU, D'Amico JM, Gorassini MA, Turker KS, et al. (2015) Jaw tremor as a physiological biomarker of bruxism. Clin Neurophysiol 126: 1746-1753.
- Luco K (2017) How sleep bruxism and tension headaches affect the masseter inhibitory reflex. J Sleep Disord Treat Care 6.
- US Food and Drug Administration (2019) 510(k) Premarket notification.
- Rajan R, Sun YM (2017) Reevaluating antidepressant selection in patients with bruxism and temporomandibular joint disorder. J Psychiatr Pract 23: 173-179.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi