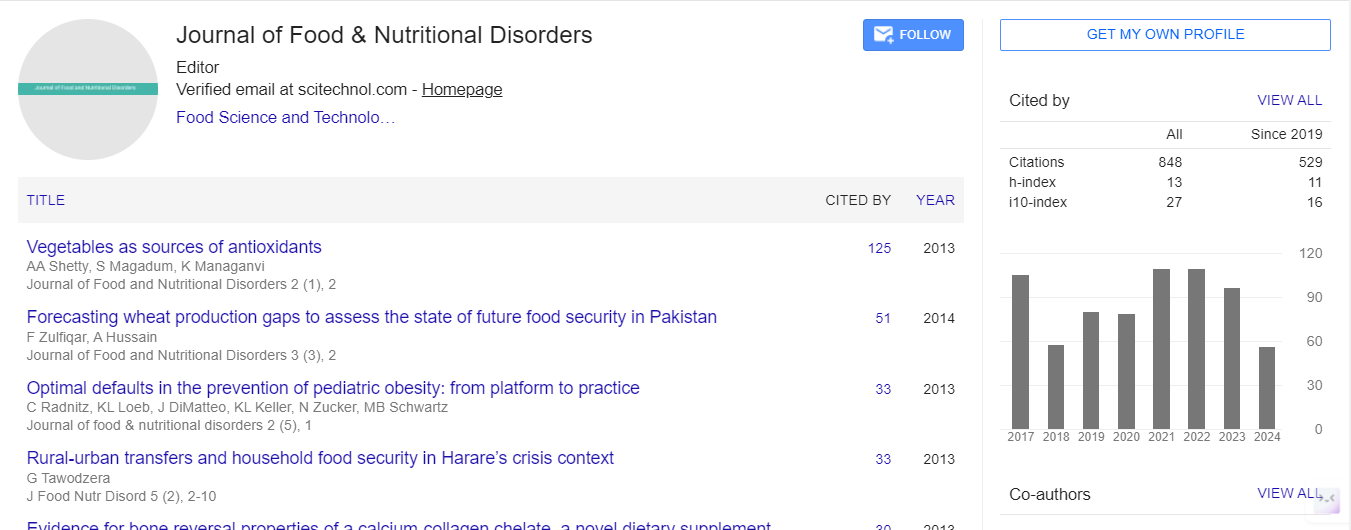
Research Article, J Food Nutr Disor Vol: 5 Issue: 1
Use of bile acids as a selection strategy for lactobacillus strains with probiotic potential
| Reyes-Nava LA1, Garduño-Siciliano L1, Estrada-de los Santos P1, Hernández-Sánchez H1, A Arauz J2, Muriel P3 and Rivera-Espinoza Y1* | |
| 1Escuela Nacional de Ciencias Biológicas, Instituto Politécnico Nacional, Av. Wilfrido Massieu esq. Cda. Manuel L. Stampa, C.P. 07738, México, DF, México | |
| 2Departamento de Farmacología, Escuela de Medicina, Universidad Autónoma de Baja California, Mexicali, Baja California. México | |
| 3Departamento de Farmacologia, Centro de Investigación y de Estudios Avanzados del IPN. México, DF, México | |
| Corresponding author : Rivera-Espinoza Y Escuela Nacional de Ciencias Biológicas, Instituto Politécnico Nacional, Av. Wilfrido Massieu esq. Cda. Manuel L. Stampa , C.P. 07738, México, D.F., México Tel: 5729-6000, ext 62464 E-mail: espinoza4@hotmail.com |
|
| Received: September 10, 2015 Accepted: December 26, 2015 Published: January 01, 2016 | |
| Citation: Reyes-Nava LA, Garduño-Siciliano L, Santos ELP, Hernández-Sánchez H, A Arauz J, et al. (2016) Use of bile acids as a selection strategy for lactobacillus strains with probiotic potential. J Food Nutr Disor 5:1. doi:10.4172/2324-9323.1000187 |
Abstract
Use of bile acids as a selection strategy for lactobacillus strains with probiotic potential
An in vitro model that simulates some gastrointestinal (GIT) tract conditions was used to select gram-positive and catalase-negative bacilli. Then, their capacity to dissociate bile salts conjugated to taurine and glycine was qualitatively and quantitatively evaluated. The in vivo experiments were performed on male Wistar rats. Lactobacillus strains were administered orally using an intubation needle, at a daily dose of 1x109 CFU/mL for 21 days. A high-cholesterol diet was administered from day 14 to 21. Total cholesterol, LDL-cholesterol, HDL-cholesterol, lipid peroxidation, and liver enzymes levels were measured.
Keywords: Lactobacillus brevis; Liver damage; Cholesterol; Agave sap
Keywords |
|
| Lactobacillus brevis; Liver damage; Cholesterol; Agave sap | |
Introduction |
|
| The fluid from the maguey cactus adult plants (agave sap) (Agave americana, A. atrovirens, A. ferox, A. mapisaga, A. salmiana) collected in the stage preceding the flowering is appreciated as a sweet and refreshing beverage in the Mexican culture. In the past, it was found to possess some health-promoting properties, such as appetite stimulant, diuretic, and antidiarrheal, among others [1,2]. The agave sap is rich in fructans [3]. The microbial diversity of the agave sap has been extensively studied [4], identifying mainly yeasts and lactic acid bacteria (LAB). However, several lactobacillus strains catalyze bile salts deconjugation reactions via the bile salt hydrolase enzymes (BSH; EC 3.5.1.24) [5]. Bacterial strains with BSH activity are often found in the gastrointestinal tract (GIT) or in the feces from mammals, which are natural environments rich in conjugated and unconjugated bile acids. Recently, BSH has been found in microorganisms isolated from kefir grains, pickles and hot water springs [6]. With the exception of two strains of Bacteroides, all BSH-positive bacteria are gram-positive. | |
| BSH is a group of enzymes that hydrolyze an amide bond between the C-24 position of the steroid moiety and an amino acid, either glycine or taurine [7]. It acts upon conjugated bile acids such as taurocholic acid (TCA), taurodeoxycholic acid (TDCA), taurochenodeoxycholic acid (TCDA), glycocholic acid (GCA), glycodeoxycholic acid (GDCA), and glycochenodeoxycholic acid (GCDCA) [8]. Some reports indicate that certain strains of lactobacilli are able to hydrolyze only the glycine-conjugated but not the taurine-conjugated bile acids [9]. In contrast, certain strains of lactobacilli are able to hydrolyze the taurine-conjugated but not the glycine-conjugated bile acids [10]. Moreover, it has been reported that no existing BSH-active strain of lactobacilli is able to deconjugate either GCA or TCA [8]. The functions of BSH are not clearly understood. However, the lactobacilli strains that possess BSH activity are preferred over the BSH-negative lactobacilli as selection criteria for probiotic strains with cholesterol lowering properties [11]. One of the hypotheses to explain their cholesterol-lowering effect is related to the deconjugation of bile acid salts. Most of the excreted conjugated bile salts (97%) are reabsorbed from the small intestine and returned to the liver through the hepatic portal circulation [12]. When they are deconjugated, the resulting free bile acids (or deconjugated bile acids) are excreted via the feces [13]. Plate assays [14], in which the deconjugation products formed on bile salt-MRS agar plates are assayed by thin layer chromatography, have been widely used to screen for the bile salt hydrolase-active form of Lactobacillus spp. Given the importance of exploring new methods to reduce serum cholesterol, this study focuses on the selection of BSH-positive (BSH+) lactobacilli from the maguey cactus sap, referred to as sap from here onwards. Using an in vitro model that simulates the gastrointestinal tract conditions, we can evaluate their capacity to dissociate bile salts conjugated to taurine and glycine and test their anti-hypercholesterolemic effect in vivo. | |
Materials and Methods |
|
| Strains | |
| For this study, Lactobacillus acidophilus LA-5 was obtained from the Christian Hansen collection, Lactobacillus casei, from a fermented product. E. coli (ATCC 160211) obtained from the Universidad Autónoma Metropolitana in Xochimilco, was used as reference. | |
| Isolation of bacilli | |
| Fresh sap samples were obtained three different geographic regions in Mexico: Huitzilac in the State of Morelos (19°02’N, 99°160’W) with an altitude of 2550 m; Jocotitlan in the State of Mexico (19°44’N, 99°45’W) with an altitude of 3900 m, and Singuilucan in the State of Hidalgo (19°59’N, 98°57’W) with an altitude of 2525 m. The brix of the samples from the three different regions was 11.5 ± 0.3 %, and the pH was 4.2, ± 0.2. A volume of 40 mL of sap from each region was collected in sterile falcon tubes, cooled with ice pellets, and transported to the laboratory. Then, the samples were concentrated by centrifugation at 10 000 rpm for 15 min. Cell pellet (1×107 CFU/mL) was placed under the simulated gastrointestinal condition according to the method developed by Vizoso [15], with some modifications [16-18]. Briefly, the cell pellet was incubated for 5 min at 37°C in 20 mL of a sterile electrolytic solution consisting of 0.01% (w/v) lysozyme (oral cavity) in 60 mM of phosphate-buffered saline (PBS), pH 6.2. Then, it was centrifuged again at 8000 rpm for 10 min at 4°C to recover the viable cells. The recovered cell pellet was incubated for 90 min at 37°C in 20 mL of a solution containing 0.05 g/L of NaCl (pH 2.0) and 3 g/L of pepsin (artificial gastric juice) and centrifuged. The resulting cell pellet was incubated at 37°C for 150 min in 20 mL of PBS (pH 6.8) supplemented with 0.5% of oxgall and 1 g/L of pancreatin (duodenal secretion). At each step of the experiment, the samples were kept under constant stirring at 180 rpm to reproduce the peristaltic movements, and the viability of the CFU was monitored through a series of dilutions in MRS agar, considering the initial count (1×107 CFU/mL) as the 100% value. The isolation of lactobacilli with BSH activity was made before and after the gastrointestinal simulation, by spreading 10 μL aliquots on MRS agar plates containing 0.5% of sodium glycocholate and 0.37 g/L of CaCl2 and incubating at 37°C for 72 h [14,19]. Only the gram-positive, catalase-negative bacilli showing precipitated halos were selected. | |
| Evaluation of the bile salt hydrolase activity | |
| Isolates were inoculated by streaking onto previously prepared MRS agar plates containing 0.5% w/v of sodium bile salts (glycocholate, GC; glycodeoxycholate, GDC; taurocholate, TC; and taurodeoxycholate, TDC) and 0.37 g/L of CaCl2. The inoculated plates were incubated for 72 h at 37°C under anaerobic conditions (Forma Scientific anaerobic chamber), according to the method described by Dashkevicz and Feighner [14] but with some modifications. The lysates that presented the largest hydrolysis halo diameter for each bile salt were selected to form the mix M1. To confirm that the precipitate was the product of the deconjugation of the bile salts, the zones on the agar plate containing the precipitate halo were cut using a penknife, and then mixed with 5 mL of water and heated to 90°C. Samples were cooled at room temperature for 10 min and the pH was adjusted to 1 using concentrated HCl to facilitate bile salts extraction [20]. Next, the samples were mixed with ethyl acetate in a 3:1 ratio and left untouched for 20 min to allow a phase separation. A 3-μL aliquot of the organic phase was placed in silica gel plates (8 cm x 13 cm, Merck, Darmstadt, Germany) to observe the products of bile salt deconjugation (namely, cholic acid and deoxycholic acid). The following bile salts were used as standards: sodium glycocholate, sodium glycodeoxycholate, sodium taurocholate, sodium taurodeoxycholate. Cholic and deoxycholic acids (Sigma Aldrich) diluted at 5% in ethyl acetate were used as controls for the deconjugated products [21]. The mobile phase contained isoamyl acetate: propionic acid:n-propanol:water mixed in a 40:30:20:10 (% v/v) ratio, respectively. Quantitative evaluation of bile salt hydrolase activity was analyzed by measuring the amount of amino acids released from the deconjugated bile salts produced by the isolates. We used the ninhydrin method, as described by Tanaka et al. [21], One unit of bile salt hydrolase was defined as the amount of enzyme releasing 1 μmol of amino acid per substrate, per minute. The protein concentration was determined by the Bradford method [22], using bovine serum albumin (Sigma) as the standard. The lysates showing the best BSH specific activity were selected. | |
| Identification of strains | |
| The cultures of bacilli were characterized by substrate fermentation (API 50 CHL systems, bioMérieux SA, France). For the genotyping, bacterial DNA was isolated using the Wizard Genomic DNA purification kit (Promega). The 16S rRNA gene sequences (c.a. 1,500 bp) were amplified with the universal fD1/rD1 primers [23] using a Maxygene Gradient thermocycler (Axygen), according to the protocol described by Estrada-de los Santos et al. [24]. The amplified 16S rRNA genes were purified with the High Pure PCR Product Purification Kit (Roche) and sequenced at Macrogen (www.macrogen. com) using the fD1/rD1 primers. The sequences were analyzed using the CAP3 Sequence Assembly Program (doua.prabi.fr/software/ cap3). Multiple alignments were performed with the Muscle 3.57 software [25], and a phylogenetic analysis was performed with the maximum likelihood (ML) setting using the PhyML program [26]. The among-site variation rate was modeled on a gamma distribution with four rate categories [27], each category being represented by its mean according to the GTR+G model. The ML tree searches were initiated from a Bio NJ seed tree, retaining the best trees among those found with Nearest Neighbor Interchange setting. The ML topologies robustness was evaluated using a Shimodaira–Hasegawa (SH)-like test [28]. The ML trees were visualized using the MEGA version 6 program [29]. | |
| Sensitivity to antibiotics | |
| To evaluate the sensitivity of the isolates to different antibiotics, bacteria were massively inoculated with a sterile cotton-tipped swab, left to dry for 15 min and then multidiscs (BIO-RAD gram-positive II) were set on the plates. The multidiscs contained: cephalothin (30 μg), ampicillin (10 μg), cefotaxime (30 μg), ceftazidime (30 μg), cefuroxime (30 μg), dicloxacillin (1 μg), erythromycin (15 μg), gentamicin (10 μg), pefloxacin (5 μg), penicillin (10 U), tetracycline (30 μg), and trimethoprim-sulfamethoxazole (25 μg). The plates were incubated at 37°C for 18-24 h. The results were expressed according to the following criteria: no halo (0 mm) meaning resistant, and presence of a halo (> 0 mm) meaning sensitive. Two known lactobacilli strains were used as reference (L. casei and L. acidophilus). | |
| In vivo study of Lactobacillus brevis strains | |
| To evaluate the activity of the L. brevis strains on cholesterol (strains Lb5H-PTA-120748, Lb2H-PTA-120749, Lb3H-PTA-120750, Lb9HPTA- 120751, Lb13H-PTA-120752) and hepatic enzymes levels (Lb9H-PTA-120751), each strain was grown to a 1x109 CFU/mL concentration. The strains (St), diluted in water for the cholesterol experiment and in milk for the liver experiment, were administered daily [30]. The experiments were performed on male Wistar rats, weighing approximately 250 ± 20 g. The animals were maintained on a standard rat chow diet with free access to drinking water. Eight animals were housed per polycarbonate cage, under controlled conditions at a temperature of 22 ± 2°C with 50-60% relative humidity under a 12:12 h light-dark cycle. The study protocols were complied with the institutional guidelines and the official Mexican regulations [31]. | |
| Cholesterol levels evaluation: The rats were divided into four groups of six. In the control diet group, the rats had access to a standard control diet (CD) (Laboratory chow 5001 Lab rodent diet, PMI Nutrition International, Inc., Bienwood, MO, USA) and water ad libitum and received 1 ml of water using a gavage needle, daily. For the high-cholesterol diet group (HLD), a specific hypercholesterolemic diet rich in cholesterol was prepared according to Garduño et al. 1997 [32]. The animals of the microorganisms (MO) groups were administered each strain of L. brevis at a dose of 1×109 CFU/mL once a day orally using an intubation needle for the duration of the experiment. Then, a high cholesterol diet was provided from days 14 to 21. After 21 days, each animal was fasted for 12-15 h. After fasting, blood samples were obtained and centrifuged for 15 min at 12000 rpm. The serum was obtained and the total cholesterol was measured using an automated Selectra II Vitalab Wiener Lab analyzer [33]. The low-cholesterol density lipoprotein (LDL) levels were calculated using the following equation: LDL= Total cholesterol-high density lipoprotein (HDL)-(Triglycerides x 0.45) [34]. LDL-cholesterol and HDL-cholesterol were determined using an Abbot VP Bichromatic Analyzer. All data are expressed as the means ± standard deviation (SD) and were analyzed statistically using the GraphPad Prism® software. The parameters with a P value of less than 0.5 were considered significant. | |
| Hepatic enzymes activity evaluation: The rats were divided into four groups of eight, and specifically treated as follows: Group I (Control): L. brevis vehicle (1 mL of milk, oral gavage) once daily for 14 days; Group II (hepatic damage): the animals were administered a single dose of lipopolysaccharide (LPS) (50 μ/kg, i.p) (from Escherichia coli 026:B6, Sigma) + D-galactosamine (500 mg/Kg, i.p.) (sigma G-00500, D-GalN) intraperitoneally on day 14 (Abdel-Salam and Sayed, 2012), and received water once daily by gavage; Group III (hepatic damage + MO): the rats were treated as group II but were also administered a daily oral dose (1×109 CFU/ mL/rat) of MO (L. brevis Lb9H-PTA-120751) by gavage; Group IV (MO): the rats were administered MO (1×109 CFU/ mL/rat) once daily by gavage. The animals were sacrificed by exsanguination under light anesthesia, 72 h after the intraperitoneal administration of LPS+D-GalN. Blood samples were collected by cardiac puncture, and the liver was rapidly removed. The serum was obtained to determine liver damage by measuring the enzymatic activities of alanine aminotransferase (ALT) [35], alkaline phosphatase (ALP) [36], and γ-glutamyl transpeptidase (γ-GTP), as previously described [37]. The lipid peroxidation levels were determined from liver homogenates by the quantification of malondialdehyde (MDA) formation using the thiobarbituric acid method [38]. | |
| Statistical analyses | |
| The data were expressed as the means ± SE. Comparisons were performed using one-way ANOVA followed by a Tukey’s, Dunnett, or Bonferroni post-hoc test, using the GraphPad Prism 5 software. The differences were considered statistically significant when p ≤ 0.05. | |
Results |
|
| Isolation of bacilli | |
| After surviving the simulated GIT conditions, a bacterial population at concentrations of 7×103, 9.7×105, and 3.5×105 CFU/ ml was obtained from Huitzilac, Jocotitilán, and Singuilucan agave sap samples. From the tested agave sap samples, 12 colonies (1H, 2H, 3H, 5H, 7H, 8H, 9H, 10H, 11H, 12H, 13H, and 14H) from Huitzilac and 2 colonies (4J and 6J) from Jocotitlan were selected as short bacilli, gram-positive (+), and catalase negative (-) that were able to grow on agar MRS containing 0.5% of sodium glycocholate and 0.37 g/L of CaCl2 (Figure 1). No colonies with these characteristics were obtained from the Singuilucan agave sap. Only the 5H and 9H colonies from Huitzilac were resistant to the GIT-simulating conditions, and no colonies from the Jocotitlán and Singuilucan agave sap possessed this capability. | |
| Figure 1: Representative result of the isolation of bacilli possessing bile salt hydrolase activity using MRS-glycocholate agar medium. A. Halos of bile salt precipitated formed by bacilli on plates containing 0.5% glycocholate(GC); B. The morphological characteristics of isolated bacilli. | |
| Evaluation of the bile salt hydrolas activity | |
| The colonies selected spreaded onto petri dishes containing a 0.5% of concentration of different bile acid salts (sodium taurocholate, taurodeoxycholate, glycocholate and glycodeoxycholate) formed hydrolysis halos (ranging from 0.5 to 4 mm in diameter) with a granular precipitate around the white colonies (Figure 2A-2D). The diameter obtained for each colony in the presence of bile salts was variable (Table 1). The 5H and 9H strains showed the largest halos. The 5H strain produced a halo diameter of 4 mm when it was incubated in taurocholate, and of 1 mm in other bile salts. The 9H strain produced a halo diameter of 1 mm regardless of the bile salt. Larger halos in the presence of glycocholate (2 mm), glycodeoxycholate (3 mm), taurocholate (4 mm), and taurodeoxycholate (3 mm) were generated by the 3H, 2H, 5H, and 13H strains, respectively. When analyzing the precipitates that were produced by the selected strains on the agar MRS-bile salts plates by thin layer chromatography, the specific bile salt and the deconjugation product were identified (Figure 3A and 3B). Indeed, when glycocholate or glycodeoxycholate were used, cholate and deoxycholate were produced. Therefore, the precipitated halo and the white granular material around the strains represented the deconjugated bile salts observed in (Figure 2A-2D) and correlate with BSH activity. | |
| Figure 2: Representative result of bile salts deconjugation by lactobacilli isolated on MRS-bile agar medium. The plates were incubated anaerobically for 72 h at 37°C. A, B, C and D. Assay plates containing 0.5% glycocholate (GC), 0.5% glycodeoxycholate (GDC), 0.5% taurocholate (TC) and 0.5% taurodeoxycholate (TDC), respectively. In the assays shown, precipitation or formation of white and opaque granular shiny colonies on the agar was indicative of a BSH activity speciï¬Âcity for all bile salts tested. | |
| Figure 3: Thin-layer chromatogram of ethyl acetate-extracted agar plugs. When lactobacilli isolates were grown on MRS plates containing 0.5% GC, they produced white colonies with precipitate halos of bile salts. The agar zone with bile salt precipitate was cut for analysis. GC, glycocholate; CA, cholic acid; TDC, taurodeoxycholate; DC, deoxycholic acid; lanes 1-14, isolated strains shown in the same order as in the Table 1. | |
| Table 1: Screening of the bile salt hydrolase (BSH) activity of isolates on glycocholate (GC), glycodeoxycholate (GDC), taurocholate (TC) and taurodeoxycholate (TDC) by qualitative and quantitative methods. | |
| Lactobacilli from agave sap present different specific BSH activity depending on the bile salt studied (Table 1). The highest SBH activity was observed with the 6J strain on glycocholate and glycodeoxycholate (1970 and 1534 U/mg, respectively) and with the 4J strain on taurocholate and taurodeoxycholate (685 and 587 U/mg, respectively). These differences were significant when compared to other lactobacilli. By selecting the largest hydrolysis halos obtained with different bile salts, a mix of strains was generated to evaluate their possible hypolipidemic activity in an in vivo model. The strains selected for in vivo analysis, 3H (for glycocholate), 2H (for glycodeoxycholate), 5H (for taurocholate), and 13H (for taurodeoxycholate), were combined to form the M1 mix. The 6J and 4J strains were combined to form the mix 2 (M2). The 9H strain, isolated at the end of the GIT-simulating conditions selection process, was also included in the mix M2. | |
| Identification of strains | |
| Nine microorganisms (MOs) obtained from fresh agave sap were identified as Lactobacillus collinoides and five, as Lactobacillus brevis (Table 2). According to 16S rRNA gene sequences, isolates with the largest hydrolysis halos obtained with different bile salts (3H, 2H, 5H and 13H), the highest bile salt hydrolase activity (6J and 4J) and the culture isolated at the end of the GIT-simulating conditions selection (9H), were over 98.2%, similar to Lactobacillus brevis (ATCC 14869T (KI271266)), allowing an unambiguous identification of the strains as L. brevis. Consequently, a phylogenetic analysis was performed with the species closest to L. brevis. The results showed a robust group including the L. brevis-type strains (Figure 4). Therefore, from now onwards, the acronym Lb was added in front of the strain labels for the 2H, 3H, 5H, 9H, and 13H isolates. For example, the Lb2H label denotes a Lactobacillus brevis, colony number 2, isolated from Huitzilac. Five strains were deposited to the American Type Culture Collection (ATCC) and given the following names: Lb5HPTA- 120748, Lb2H-PTA-120749, Lb3H-PTA-120750, Lb9HPTA- 120751, and Lb13H-PTA-120752. | |
| Figure 4: Phylogenetic tree based on the alignment of the 16S rRNA gene sequences of selected species of Lactobacillus. The bar represents the number of expected substitutions per site under the GTR+G model. The NCBI database accession numbers are shown in parenthesis. The strains analyzed in this study are in bold. | |
| Table 2: Identification of lactobacilli isolates using the API 50 CH fermentation system and 16S rDNA sequence analysis of lactobacilli with better bile salt hydrolase activity. | |
| Sensitivity to antibiotics | |
| The strains of Lactobacillus brevis selected in this study were sensitive to 10 antibiotics (cephalexin, ampicillin, cefotaxime, ceftazidime, cefuroxime, erythromycin, gentamicin, penicillin, tetracycline, and trimethoprim-sulfamethoxazole). The Lb2H, Lb5H, Lb9H, and Lb13H, strains were resistant to dicloxacillin and pefloxacin. The 3H strain was resistant to ceftazidime (Table 3). | |
| Table 3: Susceptibility of lactobacilli isolates to antibiotics using the disc diffusion method. | |
| Cholesterol levels evaluation | |
| The total cholesterol (TCH) levels in rats consuming the control diet (CD) were 26.2 mmol/L, whereas in rats fed the hyperlipidemic diet (HLD), the TCH levels were 50 mmol/L (p ≤ 0.05). The M1 mix administered to the HLD-group did not prevent the increase in TCH levels. In contrast, the M2 mix significantly prevented the increase in TCH induced by the HLD (p ≤ 0.05) (Figure 5A). To dissect the effects of each one of the strains composing the M2 mix, the strains were evaluated individually. Overall, the best effect on TCH levels was produced by the Lb9H strain (100%) (Figure 5B). The serum HDLcholesterol concentration in the CD-fed animals was 7.1 mmol/L, whereas in the HLD group, high cholesterol diet administration decreased the TCH levels to 3.6 mmol/L (an approximately 50% effect, p ≤ 0.05) (Figure 6A). The HLD+M1 and HLD+M2 groups showed no significant difference with HLD group (p ≤ 0.05). Indeed, our individual strain analyses revealed that they were unable to prevent the decrease in HDL (Figure 6B). The levels of serum LDL-cholesterol in the CD group was 14.6 mmol/L, and the HLD increased the LDL levels to 39 mmol/L (a 150% increment, p ≤ 0.05) (Figure 7A). The M2 mix partially but significantly prevented this increase (p ≤ 0.05). The individual strain analyses revealed an effect of 55% when Lb9H was administered (HLD+Lb9H) (Figure 7B). | |
| Figure 5: Effects of two mixtures of Lactobacillus strains on serum total cholesterol levels in rats fed a high cholesterol diet. CD, control diet; HLD, hyperlipidemic diet; HLD+M1, hyperlipidemic diet + Lactobacillus mixture 1 (L. brevis Lb2H, Lb3H, Lb5H and Lb13H); HLD+M2, hyperlipidemic diet + Lactobacillus mixture 2 (L. brevis Lb4J, Lb6J and Lb9H). Each bar represents the mean values from 6 rats ± SE. An “a” denotes a significant difference from the CD-group (p≤ 0.05) and a “b” denotes a significant difference from the HLD-group (p≤ 0.05). B. Effects of Lactobacillus brevis Lb4J (Lb4J), L. brevis Lb6J (Lb6J) and L. brevis Lb9H (Lb9H) strains on serum total cholesterol levels in rats fed a high-cholesterol diet. CD, control diet; HLD, hyperlipidemic diet; HLD+Lb4J, hyperlipidemic diet + Lb4J; HLD+Lb6J, hyperlipidemic diet + Lb6J; HLD+Lb9H, hyperlipidemic diet + Lb9H. Each bar represents the mean values from 6 rats ± SE. An “a” denotes a significant difference from the CD-group (p≤ 0.05) and a “b” denotes a significant difference from the HLD-group (p≤ 0.05). | |
| Figure 6: Effects of two mixtures of Lactobacillus strains on high-density lipoprotein cholesterol in rats fed a high cholesterol diet. CD, control diet; HLD, hyperlipidemic diet; HLD+M1, hyperlipidemic diet + Lactobacillus mixture 1 (L. brevis Lb2H, Lb3H, Lb5H and Lb13H); HLD+M2, hyperlipidemic diet + Lactobacillus mixture 2 (L. brevis Lb4J, Lb6J and Lb9H). Each bar represents the mean values from 6 rats ± SE. An “a” denotes a significant difference from the CD group (p≤ 0.05) and a “b” denotes a significant difference from the HLD group (p≤ 0.05). B. Effects of Lactobacillus brevis Lb4J (Lb4J), L. brevis Lb6J (Lb6J) and L. brevis Lb9H (Lb9H) strains on high-density lipoprotein cholesterol in rats fed a high cholesterol diet. CD, control diet; HLD, hyperlipidemic diet; HLD+Lb4J, hyperlipidemic diet + Lb4J; HLD+Lb6J, hyperlipidemic diet + Lb6J; HLD+Lb9H, hyperlipidemic diet + Lb9H. Each bar represents the mean values from 6 rats ± S.E. The values were not significantly different between them (p ≤ 0.05). | |
| Figure 7: Effects of two mixtures of Lactobacillus strains on low-density lipoprotein cholesterol in rats fed a high cholesterol diet. CD, control diet; HLD, hyperlipidemic diet; HLD+M1, hyperlipidemic diet + Lactobacillus mixture 1 (L. brevis Lb2H, Lb3H, Lb5H and Lb13H); HLD+M2, hyperlipidemic diet + Lactobacillus mixture 2 (L. brevis Lb4J, Lb6J and Lb9H). Each bar represents the mean values from 6 rats ± SE. An “a” denotes a significant difference from the CD-group (p≤ 0.05) and a “b” denotes a significant difference from the HLD-group (p≤ 0.05). B. Effects of Lactobacillus brevis Lb4J (Lb4J), L. brevis Lb6J (Lb6J) and L. brevis Lb9H (Lb9H) strains on low-density lipoprotein cholesterol in rats fed a high cholesterol diet. CD, control diet; HLD, hyperlipidemic diet; HLD+Lb4J, hyperlipidemic diet + Lb4J; HLD+Lb6J, hyperlipidemic diet + Lb6J; HLD+Lb9H, hyperlipidemic diet + Lb9H. Each bar represents the mean values from 6 rats ± SE. An “a” denotes a significant difference from the CD group (p≤ 0.05) and a “b” denotes a significant difference from the HLD group (p < 0.05). | |
| Hepatic enzymes activity evaluation: | |
| Given the important positive effect of the Lb9H strain on the lipid profile, this strain was evaluated for safety by assessing the levels of liver-damage associated enzymes in rats to measure its protective effect against liver damage induced by LPS and D-galactosamine administration. An increase in serum ALT activity is associated with hepatocyte death, and an elevation in serum γ-GTP activity is a sign of cholestasis. Treatment with Lb9H showed a partial prevention of the ALT increase (Figure 8) and totally inhibited the (LPS+D-GalN)- induced increase in-GTP (Figure 9) and ALP serum enzyme activities (Figure 10). As shown in Figure 11, acute LPS+D-GalN intoxication increased MDA levels (an indicator of lipid peroxidation) significantly over control values. Importantly, Lb9H co-treatment was able to completely prevent the elevation of the lipid peroxidation levels. | |
| Figure 8: Effect of Lactobacillus brevis Lb9H (Lb9H) on serum alanine aminotransferase (ALT) in rats with liver damage induced by LPS+DGalN. Control, rats administered with milk; Lb9H+ (LPS+DGalN), L. brevis Lb9H+(LPS+DGalN). Each bar represents the mean values from 6 rats ± SE in triplicate. An “a” denotes a significant difference from the Control group (p ≤ 0.05) and a “b” denotes a significant difference from the LPS+DGalN group (p≤ 0.05). | |
| Figure 9: Effect of Lactobacillus brevis Lb9H (Lb9H) on gamma-glutamyl transpeptidase (γ-GTP) in rats with liver damage induced by LPS + D- galactosamine (LPS+DGalN). Control, rats administered with milk; Lb9H+ (LPS+DGalN), L. brevis Lb9H+(LPS+DGalN). Each bar represents the mean values from 6 rats ± S.E in triplicate. An “a” denotes a significant difference from the Control group (p ≤ 0.05) and a “b” denotes a significant difference from the LPS+DGalN group (p ≤ 0.05). | |
| Figure 10: Effect of Lactobacillus brevis Lb9H (Lb9H) on alkaline phosphatase (ALP) in rats with liver damage induced by LPS + D-galactosamine (LPS+DGalN). Control, rats administered with milk; Lb9H+ (LPS+DGalN), L. brevis Lb9H+ (LPS+DGalN). Each bar represents the mean values from 6 rats ± SE in triplicate. An “a” denotes a significant difference from the Control group (p ≤ 0.05) and a “b” denotes a significant difference from the LPS+DGalN group (p ≤ 0.05). | |
| Figure 11: Effect of Lactobacillus brevis Lb9H (Lb9H) on lipid peroxidation in rats with liver damage induced by LPS + D-galactosamine (LPS+DGalN). Lipid peroxidation was expressed in nmol malondialdehyde (MDA)/mg of protein. Control, rats administered with milk; Lb9H+ (LPS+DGalN), L. brevis Lb9H+(LPS+DGalN). Each bar represents the mean values from 6 rats ± SE in triplicate. An “a” denotes a significant difference from the Control group (p ≤ 0.05) and a “b” denotes a significant difference from the LPS+DGalN group (p ≤ 0.05). | |
Discussion |
|
| Two colonies (5H and 9H) endowed with the desired properties were isolated from agave sap after exposition to GITsimulating conditions. This selection results from the capacity of these strains to express various proteins and enzymes in response to stress. For example, Lactobacillus reuteri is able to produce 40 different proteins when exposed to acidic pH [39]. Regardless of the shape or texture of the halos around the selected colonies, we observed the presence of cholate- and deoxycholatecontaining agar around them. Via thin layer chromatography, we observed that for the cholic and deoxycholic acid bile salts deconjugation products, the spots were more intense than those observed with tauroconjugated (taurocholate taurodeoxycholate) and glyco-conjugated (glycocholate and glycodeoxycholate) bile salts Regardless of the shape or texture of the halos around the selected colonies, we observed the presence of cholate- and deoxycholatecontaining agar around them. Via thin layer chromatography, we observed that for the cholic and deoxycholic acid bile salts deconjugation products, the spots were more intense than those observed with tauroconjugated (taurocholate taurodeoxycholate) and glyco-conjugated (glycocholate and glycodeoxycholate) bile salts (Figure 3A and 3B). When comparing the qualitative and quantitative effects of SBH activity, we found that the size of the hydrolysis halos did not correlate with the specific activity shown by the lactobacilli strain. For example, the 3H isolate, which formed a halo of 2 mm of diameter when grown on glycocholate, presented a SBH activity of 539 U/mg, whereas the 6J isolate (1.2 mm halo) showed a 3.5 times greater SBH activity level (1970 U/mg). The 5H isolate, which formed the largest halo (4 mm), showed a specific SBH activity of 100 U/mg, which is very small compared to the 4J isolate (3 mm halo; 685 U/mg). In addition, the 9H isolate showed a specifically higher SBH activity when grown on glycocholate (1307 U/mg), which is 13 times higher than for 5H. and 3B). When comparing the qualitative and quantitative effects of SBH activity, we found that the size of the hydrolysis halos did not correlate with the specific activity shown by the lactobacilli strain. For example, the 3H isolate, which formed a halo of 2 mm of diameter when grown on glycocholate, presented a SBH activity of 539 U/mg, whereas the 6J isolate (1.2 mm halo) showed a 3.5 times greater SBH activity level (1970 U/mg). The 5H isolate, which formed the largest halo (4 mm), showed a specific SBH activity of 100 U/mg, which is very small compared to the 4J isolate (3 mm halo; 685 U/mg). In addition, the 9H isolate showed a specifically higher SBH activity when grown on glycocholate (1307 U/mg), which is 13 times higher than for 5H. In our study, the BSH activity, in all strains of lactobacilli was higher for the deconjugation of glycine-conjugated compared with taurine-conjugated bile salts. Various studies have reported that sodium glycocholate is less toxic to Lactobacillus strains than sodium taurocholate [40]. However, the viability studies with L. amylovorus indicated that glycoconjugated bile salts exerted a much higher toxicity than tauroconjugates [41]. Previous studies reported that glycine-conjugated bile salts were more efficiently deconjugated by the L. acidophilus strain than the taurineconjugated bile salts [42]. Considering the fact that the majority of human bile salts are glycine-conjugated, the BSH activity of the L. brevis strains shown in our study might be important for the deconjugation of bile salts in the human intestine [43]. The differences in the ability to hydrolyze various bile salts may result from the presence of more than one SBH enzyme in the L. brevis strains isolated from agave sap. Different classes of BSH between strains of animal and human origin have been reported. Indeed, the L. johnsonii NCC 533 strain encodes for three BSHs in their genome [44] and the L. plantarum WCFS1 encodes four [45]. However, some isolates from the same species can display variable BSH phenotypes. Indeed, the L. brevis ATCC 14869 strain exhibits BSH activity, whereas the L. brevis BCCM 7944 does not, although both are issued from human feces [7]. Regarding the identification of microorganisms, phenotypic techniques such as the API system are still considered as powerful tools capable of discriminating between Lactobacillus species. However, the use of genetic methods for the taxonomy of Lactobacillus is becoming the mainstay for reliable identification because of its high sensitivity [46]. Using these two approaches, we could confidently conclude that our isolated MOs were Lactobacillus brevis strains. All of the Lactobacillus brevis strains were resistant to dicloxacillin and pefloxacin. The former is a low-spectrum penicillin [47], and the latter is a second-generation quinolone. This result is in agreement with previous observations indicating that Lactobacilli present an intrinsic resistance to quinolones [48]. Consequently, the resistance to pefloxacin is an intrinsic characteristic of the L. brevis strains found in agave sap. The LbH3 strain also presented resistance to ceftazidime (a third-generation cephalosporin). The OMS, in 2007, classified the third- and fourth-generation cephalosporins as “critical importance antibiotics in human medicine” [48]. However, MOs with probiotic potential should not be selected on the sole basis of their ability to resist to antibiotics but also in the absence of any resistance genes to avoid transferring it to other bacteria [49]. Therefore, it is important to conduct studies evaluating whether resistance genes are present in the strains of L. brevis isolated from agave sap, which is the logical follow-up of this work. When evaluating the effect of the strains presenting the best capacity for glycine- or taurine-conjugated bile salts hydrolysis, the Lb6J and Lb4J strains, respectively, on the lipid profile, we observed a partially protective effect on the elevation of blood cholesterol. However, the Lb9H strain, isolated by the GIT-stimulating conditions offered 100% prevention on cholesterol increase. The hypolipidemic activity produced by the Lb9H strain may be because this strain was found at the end of the GIT-simulating selection. This might suggest that it survived more easily than the other strains in the GIT of the mice, therefore maintaining an appropriate concentration to exert a beneficial effect and prevent the increased levels of total and LDL cholesterol induced by the HLD. Additionally, the BSH is associated with GIT-colonizing bacteria, and may impart a selective advantage in the GIT environment [50]. Furthermore, other mechanisms that do not involve the BSH may be inhibiting the intestinal absorption of cholesterol in vivo, for example by incorporating cholesterol into the cells [51]. | |
| The effect of the Lb9H strain identified in this work was superior to the effects reported with other microorganisms. When a HLD was administered along with supplements of Bacillus polyfermenticus (3.1x106 CFU) for 6 weeks, a 14% reduction of total cholesterol was observed (although the difference was not statistically significant) and a decrease of 24% of cholesterol-LDL was obtained, p ≤ 0.05 [52]. However, it is important to note that the study of L. brevis, reported here, is preventive. Therefore, further studies, including curative ones, are needed. | |
| In another study, the cholesterol lowering effects of several strains of L. brevis isolated from turnip “tsuda kabu” and pickled turnip “tsuda kabu zuke”, respectively, were evaluated. However, the findings indicated that viable L. brevis 119-2 cells can inhibit the intestinal absorption of cholesterol in vivo by the incorporation of cholesterol into the cells [51]. An hepatic gene analysis by DNA microarray suggested that the potential mechanism for the cholesterol-lowering effect of L. brevis-2 in vivo was the inhibition of 3-hydroxy-3-methylglutaryl-CoA reductase activity by the insulin-induced gene (insig) protein, and an induction of cholesterol catabolism into bile acid by the cytochrome P450A1. Additionally, this MO decreased serum low density lipoprotein (LDL)-cholesterol by inducing an overexpression of the LDL receptor gene, in contrast to L. acidophilus ATCC 43121, which increased HDL-cholesterol levels by upregulating the ATP-binding cassette sub-family [53]. | |
| The serum markers of liver damage evaluated herein were ALT, ALP, and γ-GTP. ALT is a cytosolic enzyme of the hepatocytes, and its elevation in serum reflects an increase in hepatocyte membrane permeability and thus necrosis [54]. γ-GTP is an enzyme embedded in the hepatocyte membrane and it is considered as of the best indicators of cholestasis. In our study we found that the Lb9H strain prevented the increase of these serum enzymes. Also the Lb9H strain was able to completely prevent the elevation of lipid peroxidation. In another study using L. brevis SBC-8803, this strain ameliorated fatty liver ethanol-induced liver injury by suppressing the upregulation of TNF-α, SREBP-1, and SREBP-2 in the liver [55]. This could explain the beneficial effect associated with the prevention of liver damage, however, other mechanisms could be involved. | |
Conclusion |
|
| From our research, we could conclude that in the agave sap, a number of beneficial lactobacilli capable of dissociating conjugated bile salts exist. In addition, these MOs positively modulated the blood lipid profile resulting from a HLD. Specifically, for one strain (Lb9HPTA- 120751), the potential to prevent cardiovascular and liver diseases is promising. Our results are under evaluation to obtain a patent (Mx/a/2014/012569). | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi