Review Article, J Clin Nutr Metab Vol: 1 Issue: 2
Methionine: The One Carbon Metabolic Cycle and its Relation to Pathogenesis
Hector B Crespo-Bujosa1* and Michael J Gonzalez2
1EDP University of Puerto Rico, Hato Rey, Puerto Rico
2University of Puerto Rico, San Juan, Puerto Rico
*Corresponding Author : Hector B Crespo-Bujosa
EDP University of Puerto Rico, Hato Rey, Puerto Rico
Tel: 1 (855) 999 3378
E-mail: hector.crespo@edpuniversity.edu
Received: September 12, 2017 Accepted: September 15, 2017 Published: September 22, 2017
Citation: Crespo-Bujosa HB, Gonzalez MJ (2017) Methionine: The One Carbon Metabolic Cycle and its Relation to Pathogenesis. J Clin Nutr Metab 1:2.
Abstract
Methionine (Met) is a sulfur-containing amino acid that has a methyl group attached to its sulfur. It is the precursor of the sulfur-containing amino acids homocysteine (Hcy), cysteine and taurine. Met metabolism or one-carbon metabolic cycle requires folate, B6 and B12 in order to function adequately. Deficient nutritional intake of these vitamins affects this process paving the way for pathogenesis. A review of literature exposes how different conditions (neurological, physical and psychological/psychiatric) have as a common denominator of the energetic and metabolic problems. In addition it is shown that Metabolic Correction is a key factor in addressing these chronic conditions.
Keywords: Methionine; One-carbon metabolism; Amino acid; Pathogenesis
Introduction
Methionine: Metabolism, pathogenesis, nutrition and supplementation
All illness has both psychological and physical dimensions Butler and Zeman [1].
This statement, although simple, wisely exposes the profound interrelation between abstract and concrete processes essential in human health and disease. It is an invitation to analyze the manner in which healthcare professionals, through their specialties, are treating many conditions. In addition, it opens the door to take a deeper look into what illness is by taking into account the energetic and metabolic implications of nutritional intake in health and disease. This would include comprehending aspects as specific as the Methionine (Met) metabolism which is the focus of this article.
In order to understand this process it is important to state that human metabolism and structure require various elements to be built on and function properly. In this matter amino acids and proteins become of vital significance. According to Mosby [2], an amino acid (AA) is an organic chemical compound composed of one or more basic amino groups and one or more acidic carboxyl groups. They are the basic constituents of proteins Youngson [3]. The 8 essential AA are isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine, and although there are more than 100 AA, 20 of them are building blocks of proteins Mosby [2]. On the other hand, proteins, as defined by Mosby [2], are any large group of naturally occurring complex organic nitrogenous compound. Each is formed of large combinations of amino acids of fifty (50) or more usually, and contain carbon, hydrogen, nitrogen, oxygen, and in some cases sulfur, phosphorus, iron, iodine, or other essential constituents of living cells. In this case, sulfur is key to the processes that involve Met among other AAs. As it has been shown, there is a clear basis to explore the internal mechanisms that organize the generation of energy, structures and functions, as well as how it can dysfunction and produce illness. This is the case of amino acid Methionine.
Methionine and the (One carbon) metabolic cycle
In order to address the Met metabolic cycle, it is essential to define what metabolism is. Metabolism, as defined in Mosby [2], comes from the Greek “metabole” meaning change and “ismos” or process, and refers to the sum of all the chemical processes that take place in living organisms that result in growth, energy generation, elimination of wastes, and other body functions as they relate to the distribution of nutrients in the blood after digestion. This process, although invisible to the naked eye, is essential for life itself. In addition to this mechanism, or, as part of is synthesis of chemicals has to take place. Synthesis means “putting together”; to synthesize refers to “form by building”, as in the formation of complex chemical compounds, for example in the case of proteins which are formed from simpler units of amino acids Mosby [2]. Therefore, these processes become the cornerstone of the Met metabolic cycle.
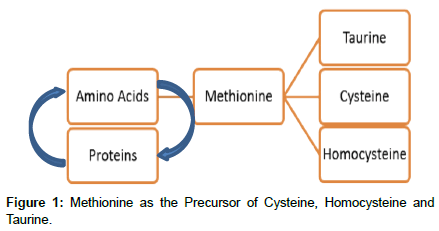
Methionine is (besides cysteine) one of the two (2) sulfur containing proteinogenic amino acids and is essential for life and exists in two (2) isomers, L- and D-methionine, of which the L-form predominates in nature Willke et al. [4]. According to Seth et al. [5] Met has a methyl (−CH3) group attached to its sulfur and is the precursor of the sulfur-containing amino acids homocysteine (Hcy), cysteine, and taurine (Figure 1).
Figure 1 shows the interrelation between AA and proteins in which the first is essential in the production of proteins at the time that proteins are necessary for AA production. Met shown as the precursor of Taurine, Cysteine and Homo cysteine.
According to Bassett et al. [6] one-carbon metabolism comprises a complex network of biochemical pathways that involve interactions between several B vitamins, Homocysteine and methionine. Homocysteine, naturally occurring Kaur and Sekhon [7] sulfur-containing amino acid [8,7] is of significant importance in cellular metabolism Krause et al. [9]. Hcy is the only direct precursor for l-methionine synthesis through a reaction that requires vitamin B12, representing a connection with “one-carbon” unit’s metabolism Manolescu et al. [10] and a derivate from the metabolism of methionine, an essential amino acid, and is metabolized by one of two pathways: re-methylation or transsulfuration [2]. The methionine cycle is completed by the re-methylation of homocysteine Finkelstein [11].
In addition McBean [12] explains that sulfur amino acid metabolism is of critical importance in mammalian cells and is linked to provision of methyl groups for a great number of biochemical processes. Met is the starting material for numerous other biochemical molecules and is converted via the enzyme methionine adenosyltransferase (MAT) to Sadenosylmethionine (SAMe), the most important methyl donor in the body Miller [13]. More specifically, dietary Met is activated by conversion to SAMe in an ATP-dependent reaction catalyzed by methionine MAT and through action of methyltransferase, produces S-adenosylhomocysteine (SAH), followed by Hcy McBean [12]. Finally, the Met cycle is completed by the re-methylation of Hcy Finkelstein [11] where it can be either re-methylated back to methionine using a methyl group provided by methyl tetrahydrofolate (MTHF), or irreversibly converted into cysteine via McBean [12]. The canonic role of the transsulfuration pathway is to clear Hcy, a toxic intermediate in the Met cycle, and to divert it to cysteine synthesis Kabil et al. [14].
Nutrition, Methionine metabolism and pathogenesis
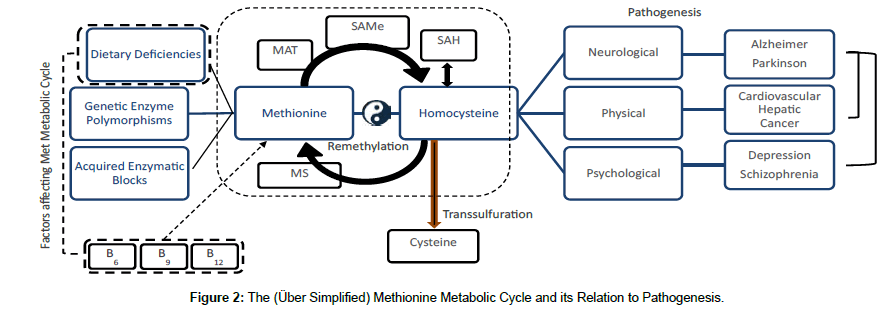
Pathogenesis is the mechanism involved in the development of disease Youngson [3]. In order to understand it there must be an integrative view of the many factors that play a key role which includes nutrition. Chronic nutritional deficiencies or lack of supplements such as folate, vitamin B6 and B12, Omega-3 and minerals may disturb normal one-carbon metabolism or methionine cycle Seth et al. [5]. Optimal Met metabolism is vital for numerous biochemical processes in the body; however, dietary deficiencies, genetic enzyme polymorphisms, and acquired enzymatic blocks can negatively influence the normal metabolism of this essential amino acid Miller [13]. The methionine metabolic cycle is strongly regulated by the supply of essential B-vitamins Halsted [15]. Disruption of this cycle leads to hyperhomocysteinemia (HHcy), and is also associated with genetic abnormality, epigenetic alterations and also with DNA hypomethylation that cause the cell damage and neuronal injury in the brain through the oxidative stress Seth et al. [5]. Abnormalities of re-methylation or transsulfuration pathways lead to HHcy and have been observed in approximately 5% of the general population and are associated with an increase for disorders such as autoimmune disorders, vascular and neurodegenerative diseases, birth defects, diabetes, renal disease, osteoporosis, neuropsychiatric disorders, and cancer Brustolin et al. [8] (Figure 2).
Figure 2 shows the essentials of Met metabolism in which dietary deficiencies, in particular of folate, B6 and B12, in isolation or addition to genetic enzyme polymorphisms and acquired enzymatic blocks hinder the efficiency of the cycle. Hcy increases its levels giving way to pathogenesis of neurological, physical and psychological conditions. This shows the importance of homeostasis of Met Cycle and adequate nutritional intake to keep the internal balance. On the other hand, it is important to point out that there are comorbidities between neurological and psychological conditions.
Methionine metabolic cycle and pathogenesis
There are many internal and external factors that contribute to pathogenesis of physical, neurocognitive and psychological conditions. As stated above, Met metabolic cycle defects may arise from different factors including nutritional or dietary deficiencies. Among the many conditions that are associated to this mechanism and can affect human wellbeing are:
1. Stress
2. Mental Illness
3. Neurocognitive Illness
4. Hyperhomicysteinemia, atherosclerosis and cardiovascular disease
5. Cancer
6. Liver illness
7. DNA and Epigenetics aspects of pathogenesis
8. Stress and mental illness
Although stress is not a disease by itself, continuous exposure to stressful stimuli has been directly related to the onset, progression or outcome of many pathological processes Munhoz et al. [16]. Some of the detrimental actions of stress hormones in the brain may be mediated via the release of secondary mediators Munhoz et al. [16].
The response of an organism to stress includes both physical and behavioral adaptations, as, for example, the “fight or flight” reaction Munhoz et al. [16]. During stressful situations including physical or emotional overwork, infection, injury, fad or poor dieting, excessive use of alcohol and/or drugs and intense sports workouts, the nervous system can become depleted of B vitamins Presser [17]. On the other hand Ramsey and Muskin [18] pointed out that there is an increasing amount of literature that associates dietary choices to brain health and the risk of psychiatric illness pointing to the fact that vitamin deficiencies can affect psychiatric patients in the following ways:
1. Deficiencies may play a causative role in mental illness and exacerbate symptoms
2. Psychiatric symptoms can result in poor nutrition
3. Vitamin insufficiency (defined as subclinical deficiency) may compromise patient recovery.
However, this can produce significant consequences at the time that it is an essential part of a complex network of factors. In this case Spillmann and Fava, and Alpert and Mischoulon cited in Mischoulon and Fava [19] indicate that SAMe has been identified as a major methyl donor in the brain that is involved in the pathways for synthesis of hormones, neurotransmitters, nucleic acids, proteins, and phospholipids. SAMe is required for the synthesis of norepinephrine, dopamine, and serotonin, and also plays a role in other intracellular metabolic pathways. They explain that SAMe is synthesized from the amino acid L-methionine through a metabolic pathway (onecarbon cycle), which relies, in part, on adequate concentrations of the vitamins folate and B12. Deficiencies of both of these vitamins have also been linked to depression [19]. It has been postulated that exogenous addition of SAMe may result in increased synthesis of these neurotransmitters, which may account for its antidepressant effect Mischoulon and Fava [19]. This shows the importance of metabolism in psychological and psychiatric illness.
Neurocognitive illness
Antioxidants are well known for their neuroprotective properties against reactive oxygen species in cortical neurons and auditory cells, and L-carnitine and D-methionine are among agents that provide such protection
Wu et al. [20]. In contrast, attention has also been given to the link between high concentrations of Hcy in plasma and damage of neural cells, that lead to several mechanisms of neurotoxicity of the Hcy, such as the generation of reactive oxygen intermediates, pro-thrombotic effects, oxidative stress, the formation of homocysteine derivatives, accumulation of the β-amyloid and the activation of apoptosis among others Krause et al. [9]. Krause and Müller [21] indicate that apart from Alzheimer’s disease’s AD’s distinct pathological markers, its neurodegenerative conditions are characterized by chronic neuro inflammatory processes that are not exclusive to the condition. These could be associated to imbalances in internal homeostasis. In fact there are mechanisms of neurotoxicity caused by the Hcy in Alzheimer disease, cardiovascular illnesses, Dementia and Parkinson´s disease Krause et al. [9]. In contrast, on a research conducted by Wu et al. [20] they identified that the antioxidants L-carnitine and D-methionine modulate cortical electrical spike activity primarily through gama aninobutyric acid GABAA receptor activation, suggesting the involvement of GABA ergic mechanisms that possibly contribute to the protective actions of these compounds.
Hyperhomocysteinemia, atherosclerosis and cardiovascular disease
Among the many factors associated to pathogenesis and the Met metabolic cycle, Hyperhomocysteinemia (HHcy) has been taken into account as smoking, dyslipidemia, hypertension, and obesity as an independent risk factor for cardiovascular disease Miller and Kelly [22]. However, mild to moderate elevations in Hcy levels were reported recently to be risk factors for atherosclerosis and thrombosis Yakut et al. [23]. Hcy provides a suitable background for atherosclerosis by impairing the mechanisms of coagulationfibrinolysis, endothelial functions and proliferation of smooth muscle cells Yakut et al. [23]. Atherosclerosis is a neuropathological process characterized by irregularly distributed yellow fatty plaques in large and medium-sized arteries Zillmer et al. [24]. It is considered to be a pathophysiologic process that leads to several diseases like coronary artery disease, cerebrovascular disease, carotid artery disease and peripheral artery disease Yakut et al. [23]. In addition to its possible role in cardiovascular disease, increased homocysteine levels have been associated in a variety of pathologies including neural tube defects, pre-menstrual syndrome, spontaneous abortion, placental abruption, osteoporosis, renal failure, diabetic microangiopathy, and neuropsychiatric disorders Miller and Kelly [22].
Cancer
Cancer is a multistage process that frequently involves ‘‘alterations” or ‘‘changes” in the transcriptional activity of genes associated with many critical cellular processes for tumor development including proliferation, senescence, inflammation, and metastasis, among others Franco et al. [25]. Transformation of a normal cell to a malignant one requires phenotypic changes often associated with each of the initiation, promotion and progression phases of the carcinogenic process [25].
According to Locasale [26] one-carbon metabolism involving the folate and methionine cycles integrates nutritional status from amino acids, glucose and vitamins, and produces diverse outputs, such as the biosynthesis of lipids, nucleotides and proteins, the maintenance of redox status and the substrates for methylation reactions. In addition, the author states that even though this process was considered as one of “general maintenance”, it has been recently demonstrated that this pathway has an additional complexity, and the genetic and functional evidence suggests that hyper activation of this pathway is a driver of oncogenesis and establishes a link to cellular epigenetic status.
On the other hand Xiao et al. [27] indicate that folate is critically involved in synthesis, repair, and methylation of DNA, while nutrients in the one-carbon metabolism (methionine metabolic cycle) pathway may be involved in carcinogenesis. A meta-analysis performed by Wu et al. [28] suggested that serum pyridoxal 5’-phosphate (PLP, active form of vitamin B6) levels and dietary methionine intake could be significantly associated with reduced risk of breast cancer, particularly in post-menopausal women; however the findings require further confirmation by a well-conducted randomized trial.
Liver disease
According to Halsted [15] there is convincing evidence which associates aberrant B-vitamin (folate, B6 and B12) dependent hepatic Met metabolism to the pathogenesis of alcoholic liver disease (ALD). The deficiency of these vitamins in addition to chronic alcohol exposure triggers an aberrant Met metabolism in the pathogenesis of ALD Perez et al. [29]. In addition Methionine adenosyltransferases MAT I and MAT III (encoded byMat1a) catalyze S-adenosylmethionine synthesis in normal liver Presser [17]. According to Lu and Mato [30], at present most of the hepatic diseases studied, including cirrhosis, hepatocellular carcinoma or acute liver injury, concur with a reduction in SAMe or S-Adenosyl methionine (AdoMet) concentrations due to a decrease in Mat1a expression and the concomitant increase in that of Mat2a and Mat2b. On the other hand Folate (B9) has been identified as a dietary transmethylation donor for the production of SAMe, which is the substrate for all methyltransferases that regulate gene expressions in pathways of liver injury, and is also a regulator of the transsulfuration pathway that is essential to produce glutathione (GSH), which is the principal antioxidant for defense against oxidative liver injury [15]. While Vitamin B12 regulates trans methylation reactions for SAMe production, B6 regulates transsulfuration reactions for GSH production [15].
DNA and epigenetics
Epigenetics refers to altered levels of a gene’s transcriptional activity without directly affecting its primary DNA nucleotide sequence Franco [25]. According to Stenvinkel et al. [31] epigenetics can be understood as the properties of the genome that are not explained by the primary DNA sequence, but the result of modifications of DNA and/or associated proteins due to processes such as acetylation, methylation, ubiquitination, sumoylation and phosphorylation. Epigenetics appears to go against the traditional principles of genetics where early 20th century data supports the Darwinian Theory that genes are the basis of phenotype, and any change in phenotype is due to alterations in DNA sequence Horsburgh et al. [32]. On the other hand Epigenetics may be particularly important for patients suffering from uraemic toxicity and due to the elevated prevalence of inflammation and HHcy in combination with a high cardiovascular mortality rate in chronic kidney disease patients, this population may be particularly well suited for studies associating phenotype with epigenetic alterations and clinical outcome [31].
DNA methylation is the most widely studied mechanism of epigenetic modifications intended to regulate gene expression Yasmin et al. [33]. According to Tomizawa et al. [34] DNA methylation is one of the essential factors in the control of gene expression, and alterations of this pattern has been linked to various neurological, behavioral and neurocognitive dysfunctions. They indicate that in recent studies have pointed out the importance of epigenetics in brain development and functions including learning and memory. Nutrients related to one-carbon metabolism are known to play important roles in the maintenance of genomic DNA methylation. In adition Hcy is remethylated and converted to Met by 5-methyltetrahydrofolatehomocysteine methyltransferase (MTR), which requires folate and vitamin B12 or betaine-homocysteine methyltransferase (BHMT) as a methyl donor Shiraki et al. [35].
In relation to nutritional intake, Folate is critically involved in synthesis, repair, and methylation of DNA Xiao et al. [27]. Embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) have a limitless ability to replicate; they are pluripotent and can give rise to all cell types Shiraki et al. [35]. Studies suggest that human ESCs/ iPSCs require high amounts of Met and express high levels of enzymes involved in Met metabolism. Met deprivation results in a rapid decrease in intracellular SAMe, triggering the activation of p53-p38 signaling, reducing NANOG expression, and poising human iPSC/ ESCs for differentiation, follow by potentiated differentiation into all three germ layers; when exposed to prolonged Met deprivation, the cells undergo apoptosis Shiraki et al. [35].
Nutrition, supplementation and metabolic correction in treating methionine metabolism defects
As it has been exposed through a brief exposition of Met metabolic cycle defects and its relation to pathogenesis, it becomes obvious that there is a nutritional and energetic problem that is at the epicenter of illness. At this point the concept of Metabolic Correction (MC) introduced by Dr. Michael Gonzalez and Dr. Jorge Miranda-Massari becomes a key factor in addressing illness from an energetic standpoint. MC is a functional term introduced by Drs. Gonzalez and Miranda-Massari in 2011 to explain the mechanism of how nutrients are capable of correcting biochemical disruptions that promote the disease state Miranda-Massari, Gonzalez, Jiménez et al. [36] cited in Gonzalez and Miranda-Massari, 2012. According to Miranda-Massari et al. [37] MC is: a) preventive, b) coordinated, c) evidence-based, d) optimizes physicians treatment decisions making process, e) proactive and participatory, engaging patients in lifestyle choices, and f) active health maintenance to compensate for genetic susceptibilities. According to Gonzalez and Miranda-Massari [36] MC provides the biochemical elucidation of the utilization of nutrients for both preventive and therapeutic purposes against disease. They add that it is a functional biochemical/physiological concept that explain how improvements in cellular biochemistry help the body achieve metabolic or physiological optimization, and that impaired or incomplete cellular biochemical reactions are amended with MC.
In the case of the conditions exposed, vitamins have been shown to be of vital importance to Met metabolism, homeostasis and adequate functioning of many systems in the human body. The literature points out to B vitamins as cofactors of significant importance. The B group of vitamins, also known as B vitamins are coenzymes without which many body enzymes cannot function appropriately [3,38]. According to Presser [17], B-Complex is known as the energy vitamins and stress fighters because they are intimately involved in cellular energy metabolism, specifically the glycolytic, Krebs cycle and pentose pathways. As stated by Mozaffarian [39], several epidemiological studies have exposed that blood concentrations of vitamins B6, B12, and folic acid are linked to people’s performance on tests of memory and abstract thinking. B vitamins act as cofactors in the conversion of carbohydrates into glucose which the body burns to produce energy, and are necessary for normal functioning of the nervous system and could be the single most important factor for the health of the nerves [17]. The B-Complex vitamins are also essential for healthy hair, skin and nails Presser [17].
On the other hand folate and vitamin B12 deficiency can produce a megaloblastic anemia and demyelination in the spinal cord and brain, causing peripheral neuropathy, myelopathy, gait disturbance, incontinence, visual impairment, and neuropsychiatric syndromes Scharre [40]. However, administration of vitamin B12 may reverse or stop the progression of the neurological symptoms and dementia Scharre [40]. Folic acid supplements are valuable in preventing neural tube defects [40]. These vitamins have been pointed out by the literature as key elements for proper brain functioning and therefore adaptive social behaviour.
In a research conducted by Zhong et al. [41] they found that increasing maternal consumption of methionine as DL-methionine (DLM) or DL-2-hydroxy-4-methylthiobutanoic acid (HMTBA), promoted neonatal intestinal growth by increasing morphological development or up-regulating expression of genes responsible for nutrient metabolism. They also observed that increasing maternal consumption of HMTBA promoted neonatal intestinal antioxidant capacity without compromising maternal energy homeostasis during early lactation. In the case of HHcy treatment, in addition to the inclusion of foods that are high in folate and vitamins B6 and B12 to the diet, supplementation of folate, vitamins B6 and B12 to reduce homocysteine levels, and patient education about lifestyle changes such as stress reduction, regular exercise and smoking cessation Mannella and Smith [42]. In the following table (Table 1) the sources of folate and vitamins B6 and B12 are identified according to Mozaffarian [23]. It is important to point out that the quantities of intake exposed on the table are typical and persons with illness would probably require higher doses.
| Vitamin | Foods |
|---|---|
| Vitamin B6 | |
| (Pyridoxal, Pyridoxine, Pyridoxamine) • Aids in lowering homocysteine levels • Helps convert tryptophan to niacin and serotonin, a neurotransmitter that plays key roles in sleep, appetite, and moods • Helps make red blood cells • Influences cognitive abilities and immune function |
Meat, fish, poultry, legumes, tofu and other soy products, potatoes, non-citrus fruits such as bananas and watermelons |
| Folic Acid / Vitamin B9 | |
| (Folate, Folacin) • Vital for new cell creation • Helps prevent brain and spinal birth defects when taken early in pregnancy; should be taken regularly by all women of childbearing age • Can lower levels of Hcy • May reduce risk for colon cancer • Offsets breast cancer risk among women who consume alcohol |
Fortified grains and cereals, asparagus, okra, spinach, turnip greens, broccoli, legumes such as black-eyed peas and chickpeas, orange juice, tomato juice |
| Vitamin B12 | |
| (cobalamin) • Aids in lowering homocysteine levels • Assists in making new cells and breaking down some fatty acids and amino acids • Protects nerve cells and encourages their normal growth • Helps make red blood cells |
Meat, poultry, fish, milk, cheese, eggs, fortified cereals, fortified soy milk |
Table 1: Vitamins B and Sources (adapted from Presser and Mozaffarian).
Conclusion
Diverse health fields analyze illness from a specialized perspective which sometimes could be incomplete due to the specificity of its nature. However, an integrative model of health has proven to be more adequate because it matches the multidimensional essence of human beings. In addition, it is imperative that health professionals take one step back in order to better observe the whole picture of what the illness process encompasses before taking the step forward in implementing a treatment. By understanding the subjacent factors illness can be better understood and addressed. In fact, this is a deeper clinical exercise in terms of finding the genesis of the condition instead of treating symptoms. When clinical analysis takes place, symptoms are to be identified as something more than criteria for diagnosis. They should be seen at the by-product of a metabolic and energetic deficit. In this case, Metabolic Correction becomes essential in treating illness and correcting deficits.
In my reflection about this interesting topic I can conclude that:
1. Pathogenesis goes beyond evident signs and symptoms.
2. In order to help individuals heal and improve their quality of life Naturopathy professionals must seek the cause (Tolle Causam) and promote the body’s healing power (Vis Medicatrix Naturae).
3. The beginning treatment will probably require significant supplementation while the system regains homeostasis and the person learns that healthy habits and a proper nutrition that will serve as the basis for gaining and maintaining health.
4. B vitamins are necessary for normal functioning of the nervous system and could be the single most important factor for the health of the nerves [26].
5. Sub-optimal levels of B vitamins are important factors in Methionine Metabolism defect and are associated with pathogenesis.
6. Nutritional intake and supplementation of Folate, Vitamin B6, B12 and Omega-3 is important for Met and Hcy cycles homeostasis and prevent several neurocognitive, physical and psychological conditions.
7. In the end, it’s all about energy… and it should be provided through nutrition and supplementation in order to achieve homeostasis, wellbeing and health.
References
- Butler C, Zeman AZ (2005) Neurological syndromes which can be mistaken for psychiatric conditions. J Neurol Neurosurg Psychiatry 1: 31-38.
- Mosby’s dictionary of medicine, nursing, and health professions(2009) Missouri, USA
- Youngson RM (2006) Collins dictionary of human biology. Human biology defined and explained. London: Harper Collins Publishers, UK.
- Willke T (2014) Methionine production—a critical review. Appl Microbiol Biotechnol 98: 9893-9914.
- Seth R, Agrawal A, Singh GPI, Dubey GP (2015) Hyperhomocysteinemia as risk factor for depression: A review. Pharmaceutical and Biological Evaluations 2: 133-141.
- Bassett JK, Hodge AM, English DR, Baglietto L, et al. (2012) Dietary intake of B vitamins and methionine and risk of lung cancer. Eur J Clin Nutr 66: 182-187.
- Kaur R, Sekhon BS (2013) Hyperhomocysteinemia: An overview. Ann Pharmacother 4: 1-11.
- Brustolin S, Giugliani R, Félix TM (2010) Genetics of homocysteine metabolism and associated disorders. Braz J Med Biol Res 43: 1-7.
- Krause M, Jiménez Reséndiz SP, Morgado Vázquez JS (2009) La homocisteína: un aminoácido neurotóxico. REB 28: 3-8.
- Manolescu BN, Oprea E, Farcasanu IC, Berteanu M and Cercasov C (2010) Homocysteine and vitamin therapy in stroke prevention and treatment: a review. Acta Biochim Pol 57: 467-477.
- Finkelsteinn JD (2003) Methionine metabolism in liver diseases. Am J Clin Nutr 77: 1094-1095
- McBean GJ (2012) The transsulfuration pathway: a source of cysteine for glutathione in astrocytes. Amino Acids 42: 199-205.
- Miller AL (2003) The Methionine-Homocysteine cycle and its effects on cognitive diseases. Altern Med Rev 8: 7-19.
- Kabil O, Vitvitsky V, Xie P, Banerjee R (2011) The quantitative significance of the transsulfuration enzymes for H2S production in murine tissues. Antioxid Redox Signal 15: 363-372.
- Halsted CH (2003) B-Vitamin dependent methionine metabolism and alcoholic liver disease. Clin Chem Lab Med 51: 457-465.
- Munhoz CD, García-Bueno B, Madrigal JL, Lepsch LB, Scavone C, et al. (2008) Stress-induced neuroinflammation: mechanisms and new pharmacological targets. Braz J Med Biol Res 4: 1037-1046.
- Presser A (2009) Smart Supplementation. B-Vitamins. Literature Education Series on Dietary Supplements.
- Ramsey D, Muskin PR (2013) Vitamin deficiencies and mental health: How are they linked? Identifying and correcting deficiencies can improve brain metabolism and psychopathology. Curr Psychiatry 12: 37-44.
- Mischoulon D, Fava M (2002) Role of S-adenosyl-L-methionine in the treatment of depression: a review of the evidence. Am J Clin Nutr 76: 1158-61.
- Wu C, Gopal KV, Moore EJ, Gross GW (2014). Antioxidants L-carnitine and D-methionine modulate neuronal activity through GABAergic inhibition. J Neural Transm 121: 683-693.
- Krause DL, Müller N (2010) Neuroinflammation, microglia and implications for anti-inflammatory treatment in Alzheimer’s disease. Int J Alzheimers Dis 2010: 732806
- Miller AL, Kelly GS (1996) Methionine and Homocysteine metabolism and the nutritional prevention of certain birth defects and complications of pregnancy. Altern Med Rev 1: 220-235.
- Yakut M, Ayyıldız O, Batum S, AlanS (2011)The relationship between hyperhomocysteinemia, haemostatic factors and acute coronary syndrome in southeastern turkey: a prospective, comparative study. Int J Clin Med 2: 272-277.
- Zillmer EA, Spiers MV, Culbertson WC (2008) Principles of neuropsychology, USA.
- Franco R, Schoneveld O, Georgakilas AG, Panayiotidis MI (2008) Oxidative stress, DNA methylation and carcinogenesis. Cancer Lett 266: 6-11.
- Locasale JW (2013) Serine, glycine and one-carbon units: cancer metabolism in full circle. Nat Rev Cancer 13: 572-583.
- Xiao Q, Freedman ND, Ren J, Hollenbeck AR, Abnet CC, et al. (2014) Intakes of folate, methionine, vitamin B6, and vitamin B12 with risk of esophageal and gastric cancer in a large cohort study. Br J Cancer 110: 1328-1333.
- Wu W, Kang S, Zhang D (2013).Association of vitamin B6, vitamin B12 and methionine with risk of breast cancer: a dose–response meta-analysis.Br J Cancer 109: 1926-1944.
- Pérez C, Pérez-Zúñiga FJ, Garrido F, Reytor E, Portillo F, et al. (2016) The oncogene PDRG1 is an interaction target of Methionine Adenosyltransferases. PLoS ONE 11: e0161672.
- Lu SC, Mato JM (2012) S-adenosylmethionine in liver health, injury, and cancer. Physiol Rev 92: 1515-1542.
- Stenvinkel P, Karimi M, Johansson S, Axelsson J, Suliman M, et al. (2007) Impact of inflammation on epigenetic DNA methylation– a novel risk factor for cardiovascular disease? J Intern Med 261: 488-499.
- Horsburgh S, Robson-Ansley P, Adams R, Smith C (2015) Exercise and inflammation-related epigenetic modifications: focus on DNA methylation. Exerc Immunol Rev 21: 26-41.
- Yasmin R, Siraj S, Hassan A, Khan AR, Abbasi R, et al. (2015) Epigenetic regulation of inflammatory cytokines and associated genes in human malignancies Mediators Inflamm 2015: 201703.
- Tomizawa H, Matsuzawa D, Ishii D, Matsuda S, Kawai K, et al. (2015) Methyl-donor deficiency in adolescence affects memory and epigenetic status in the mouse hippocampus. Genes, Brain and Behav 14: 301-309.
- Shiraki N, Shiraki Y, Tsuyama T, Obata F, Miura M, et al. (2014) Methionine metabolism regulates maintenance and differentiation of human pluripotent stem cells. Cell Metab 19: 780-794.
- González MJ, Miranda-Massari JR (2012) metabolic correction: A functional explanation of orthomolecular medicine. J Orthomol Med 27: 1-8.
- Miranda-Massari JR, González MJ, Duconge J, Berdiel MJ, Alfaro I, et al. (2016) metabolic correction therapy: Changing the healthcare management paradigm.
- Youngson RM (2005) Collins dictionary of medicine. Medicine defined and explained. London, USA.
- Mozaffarian D (2012) The truth about vitamins and minerals. Choosing the nutrients you need to stay healthy. Boston, USA.
- Scharre DW (2007) Infectious, Inflammatory, and Demyelinating Disorders. The Guilford Press, USA.
- Zhong H, Li H, Liu G, Wan H, Mercier Y, et al. (2016) Increased maternal consumption of methionine as its hydroxyl analog promoted neonatal intestinal growth without compromising maternal energy homeostasis. J Anim Sci Biotechnol 7: 46.
- Mannella H, Smith N (2016) Hyperhomocysteinemia, Cinahl Information Systems. Quick Lesson, USA.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi