Research Article, J Pharm Sci Emerg Drugs Vol: 8 Issue: 2
Development of Topical Oral Anti-Inflammatory Gel Containing Zanthoxylum limonella seed Extracted: A Preclinical Study
Aroonsri Priprem1*, Sucharat Limsitthichaikoon2, Kouichi Nakagawa3 and Vassana Netweera41Department of Pharmaceutical Technology, Khon Kaen University, Khon Kaen, 40002, Thailand
2Department of Pharmaceutical Technology, College of Pharmacy, Rangsit University, Pathum Thani, 12000, Thailand
3Division of Regional Innovations, Graduate School of Health Sciences, Hirosaki University, 66-1 Hon-cho, Hirosaki 036-8564, Japan
4School of Health Science, Mae Fah Luang University, Chiang Rai, 57100, Thailand
*Corresponding Author : Aroonsri Priprem
Department of Pharmaceutical
Technology, Faculty of Pharmaceutical Science, Khon KaenUniversity, Khon Kaen,
40002, Thailand
Tel: 66804072546
E-mail: aroonsri@kku.ac.th
Received date: May 08, 2020; Accepted date: May 21, 2020; Published date: May 28, 2020
Citation: Priprem A, Limsitthichaikoon S, Nakagawa K, Netweera V (2020) Development of Topical Oral Anti-Inflammatory Gel Containing Zanthoxylum limonella seed Extracted: A Preclinical Study. J Pharm Sci Emerg Drugs 8:2. doi: 10.37532/jpsed.2019.8(2).135
Abstract
Background: This study aims to investigate the stability of Zanthoxylum limonella Alston. seed (ZL) at various storage temperatures and anti-inflammatory activity of the topical oral gel (ZG) in a preclinical study.
Methods: The seed was stored in 2 different conditions: accelerated condition (50 °C, 70 °C and 90 °C) over 96 h and long-term condition (4 °C, 25 °C and 50 °C) over 6 months. Anti-inflammatory activity of the topical oral in rats was observed by using croton oil ear edema and oral punch wound healing.
Results: High storage temperature decreased the total phenolic content of ZL extract which the rate of degradation followed zero-order kinetic. In the long term condition, ZL stored at 50°C generated free radical while at 4 °C and 25 °C found the non-significant difference from the initial. The rate of release and permeation of phenolic content detected in receptor medium of the ZL into a mucoadhesive gel was 0.11 and 0.47 µg/cm2/h with no lag time indicated the fast release and permeation. The anti-inflammatory study suggests an effective dose of 10% of ZG which accelerated the wound size and reduced erythema compared to blank gel (p<0.001) and similar to FG (p=0.8) since the third day of treatment. The anti-inflammatory and wound healing activity of the ZG demonstrated re-epithelialized and wound closure, as supported by results of histopathology.
Conclusion: The topical oral gel contained the Zanthoxylum limonella seed extracts in the preclinical of rats was proved usefulness as an alternative topical oral product.
Keywords: Zanthoxylum limonella, Antioxidant, Anti-inflammation, Topical oral gel, EPR, free radical
Introduction
Certain types of inflammatory tissue injury are mediated by reactive oxygen metabolites [1]. Reactive oxygen species (ROS) may mediate the inflammation and tissue dysfunction. Injuries of oral mucosa, caused from the tooth, hard objects and/or fixed orthodontic appliances, can injure oral mucosa and affect oral routine function by introducing pain, swelling and inflammation, followed by a reduction in saliva secretion which may cause mouth dryness, gingivitis and subsequently microbial infection [2-4]. Topical application of antiinflammatory treatment is preferable, however, needs to overcome the complex mucosal permeability phenomenon influenced by mucosal structure, saliva secretion and the nature of the penetrants [5]. Mucoadhesive polymers used in the topical product are aimed to prolong the contact time of the residence time of the dosage form at the target of the action [6].
Zanthoxylum limonella Alston seed is widely distributed in the north part of Thailand. It has been traditionally used as the food ingredient and folk medicine various purpose, including stomach ache, toothache, intestinal worms, rheumatism, scabies, snake bites, fever and cholera [7]. Itthipanichpong and his colleagues found 33 chemical components in the essential oil from fruit (seed), which evaluated by using GC/MS analysis [8]. The main components are volatile oils and phenolic compounds including Limonene (31.09%), terpin-4-ol (13.94%) and sabinene (9.13%). The components can be useful as a topical [9] application as antioxidant agents [7,10]. However, antioxidant activity related anti-inflammatory tissue action has not been fully understood, yet.
Stable free radicals can be detected by electron paramagnetic resonance (EPR). Stable free radicals in pepper seeds were measured by using EPR [11], which detects the unpaired electron spin of the resonance phenomena. The resonance of unpaired electron in sesame seeds under the applied magnetic field was also measured [12]. The stable radicals can be produced by the antioxidant activities during the storage of the seeds. Thus, stable free radicals of Zanthoxylum limonella fruit can be measured by EPR. Furthermore, free radicals represent the stability of the main components (limonene, phenols, etc.), which is beneficial to verify the appropriate condition of fruit due to gain the most potential of the main components. However, the detail relation between total phenolic contents and the free radical are not known.
In this paper, we investigated the stability and antioxidant activity of Zanthoxylum limonella seed in order to be developed a mucoadhesive topical oral gel. Stable free radical in the seed was measured by EPR. Anti-inflammatory activity of the Zanthoxylum limonella gel in a preclinical of a buccal wound of rats was also investigated which inhibit tissue inflammation. In addition, we discussed the results about the wound sizes reduction.
Materials and Methods
Plant sample
Seeds of Zanthoxylum limonella Alston (so-called the seeds) were harvested during November, December 2015 and January 2016 in the North of Thailand.
Sample preparation for stability testing
These seeds were divided into 2 groups and kept in different conditions. The first group was stored in accelerated temperature at 50, 70 and 90 °C for 96 h. At each predetermined time, triplicate samples of each group were analyzed and averaged for determination of the percentage of total phenolic content remaining in the samples. The sampling period of ZL sample stored at 50 °C were randomly sampling in every 24 h, as collected at time 0, 24, 48, 72, and 96 h, at 70 °C were randomly sampling in every 12 h, as collected at time 0, 12, 24 and 48 h and at 90°C were randomly sampling in every 3 h, as collected at time 0, 3, 6, 9, and 12 h. All samples of the first group were extracted with absolute methanol (methanolic extracts) and proceeded to analyze total phenolic content and DPPH assay. Then the percentage remaining of total phenolic at an initial time of each temperature was plotted with the time (month).
The second group was stored in normal condition as 4, 25, and 50 °C in a closed container containing silica gel for humidity control. Nine tubes of the samples from each batch (approximately 100 tubes per batch, 6 batches in total) were randomly sampled and stored at 4, 25, and 50°C follow ICH Q1A(R2) guideline [13] for 6 months. The samples were randomly sampling ever months to analyze free radical content with EPR assay.
Total phenolic contents
Folin-Ciocalteu’s reagent (VWR International, France) and sodium carbonate anhydrous (Fluka-Garantie, Switzerland) were used to oxidize and neutralize the extracts, respectively. The blue color mixture was measured at 765 nm using a UV spectrophotometer (Shimadzu, Japan) after 15 min incubation at 45 °C [14]. The results were compared with the gallic acid, the standard for total phenolic quantification, and expressed as microgram of gallic acid equivalent per milliliter (μg GAE/mL).
EPR Measurement
A JEOL RE-3X X-band (9 GHz) EPR spectrometer (JEOL Inc. Tokyo, Japan) was used to measure stable radical in ZL seed samples. Typical spectrometer settings were as follows: magnetic field, 337.0 mT; sweep width,5.0 mT; sweep time, 4 min; modulation frequency, 100 kHz; magnetic field modulation, 0.32 mT; amplitude,500; time constant, 1s; microwave power,5 mW. All measurements were performed at ambient temperature. The details of EPR are also described previously [10-12].
Control, 1-, 3- and 6-month sampling were selected and inserted into an EPR tube for the measurement. The position of a tube was put in the center of the microwave cavity. After EPR measurements, ZL seeds were weighed by using an analytical balance.
Gel preparation
A blank gel (BG), consisting of 1% each of sodium polyacrylate and carbomer 934P (GMP, Thailand) in isotonic phosphate buffer saline (pH 7.4), was prepared. The ZL extract, lidocaine (Sigma-Aldrich, U.S.A.) and fluocinolone acetonide (Sigma-Aldrich, U.S.A., U.S.A.) were formed by thoroughly mixing with BG in order to compare each ingredient in the same gel base [14,15]. The gels were packed in sealed collapsible tubes with minimum air and autoclaved (121°C, 1 bar, 15 min) for sterilization.
Physicochemical properties, including color, pH, and texture analysis of the ZG were evaluated. Firmness, compressibility, adhesiveness and cohesiveness of ZG, measured using a texture analyzer (TA.XT plus, Stable Microsystem, U.K.) with a half inch probe compressed at a rate of 6 mmsec-1 into a 60 g sample in a 3-cm diameter glass container, were analyzed by a texture profile of a double compression (90-sec) with a 15-sec rest [16].
In vitro release and permeation of ZG
In vitro release was conducted using Franz diffusion cells (Crown Glass, U.S.A.) assembled by mounting cellulose acetate membrane pore size 0.45 μm (Whatman®, England) as the barrier between 5 ml of artificial saliva solution pH 7.28 as the receptor medium (600 rpm, 37 °C), and ZG as the donor. Receptor medium was taken for total phenolic content determinations.
In vitro permeation test was conducted using Franz diffusion cells (Crown Glass, U.S.A.) assembled by mounting excised porcine esophagus as the barrier [17,18] between 5 ml of 1% albumin solution as the receptor medium (600 rpm, 37 °C), and ZG as the donor. Receptor medium was taken for total phenolic content determinations.
Animal study
Adult male Wistar rats (weighing 250-350 g, 60 in total) were obtained from the National Laboratory Animal Center, Mahidol University, Salaya, Nakhonpathom, Thailand, and the animal handling was under the supervision of the certified veterinarian of the Northeast Laboratory Animal Center, Khon Kaen University, Thailand. The study protocol has been reviewed and approved by the Animal Ethics Committee of Khon Kaen University, based on the Ethic of Animal Experimentation of the National Research Council of Thailand (NELAC 23/2557) [19]. The animals were housed under natural conditions (22 ± 3 °C, 50 ± 5%RH, 12h/12h light-dark cycle) for 1 week prior to experimentation. They had free access to a rodent diet and clean water and were fed and provided filtered water ad libitum. Experiments were carried out on groups of 6 animals with free access to food and water in a Heating Ventilating and Air Conditioning (HVAC) system (Northeast Laboratory Animal Center, 2010).
Anti-inflammatory by ear edema induction
Rats were anesthetized by intraperitoneal injection of pentobarbital sodium (Nembutal, Ceva Corporate, France). Inflammation of the posterior edge of the left ear of each rat was induced by applying 0.5 mg/cm2 of croton oil (diluted in acetone). Right ears of the rats were treated with the same volume of acetone for blank comparisons. Immediately after the inducement, predetermined topical preparations, i.e. 1% and 10% ZG, 2% LG, 0.1% FG, and BG, were topically applied to the assigned ears of the rats. Thicknesses of both ears of each rat were measured at predetermined time intervals using a digital vernier caliper (Mitutoya, Japan). Anti-inflammatory activity was calculated from ear edema inhibition based on thicknesses of the right ear of each rat, compared to an equation, as follows:

where ΔTblank=difference of ear thickness between after and before treated with blank, Tpre-treated=ear thickness of the same rat at the same site before the treatment (mm) and ΔTsample=difference of ear thickness between after and before treated with samples.
Oral wounds
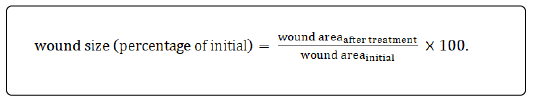
Oral wound induction and tissue sampling were conducted as previously described [20]. In brief, anesthetized rats (by intraperitoneal injections of Nembutal) were subjected to a 5-mm oral biopsy punch onto the left buccal cavity by one person, pre-trained for this practice. Treatment groups (n=5 each) were randomly assigned as follows: negative control (no wound and no treatment), BG, positive control; 0.1% FG and 2% LG and 10% ZG. All treatments were applied to the wounds twice daily as a total volume of 0.1 ml using an automatic pipette. The degree of erythema was double-blindly assessed by scoring the inflammation level of each wound in reference to the adjacent normal mucosa as follows: non-inflamed (the same color), mild (slightly red), moderate (medium red) and severe (red) and estimated as % of initial [21]. Wound sizes were measured as the areas illustrated in the photographs taken by fixed conditions (light and distance) using a digital camera with a USB probe and LED light (Andonstar Professional Electronic, China) and scale to compare with that of the initial, as follows:
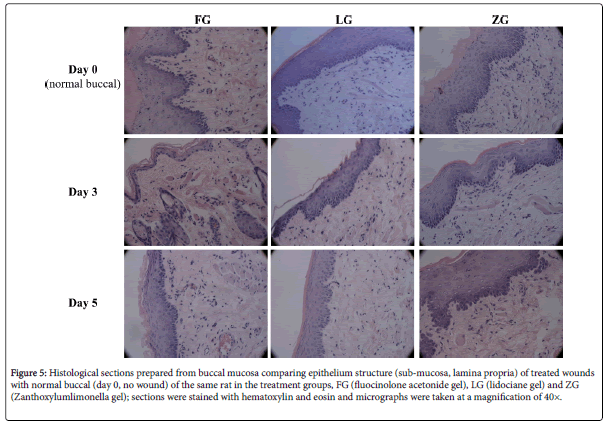
Histological evaluation
Rats were sacrificed after day 3 or 5, the wound area and the normal area of the same rat were excised, hematoxylin and eosin stained (H&E, Sigma-Aldrich, U.S.A.) and cross-sectioned for about 4-mm thick tissue samples. Each tissue sample was randomly selected for 3 zones for photographs by using Zeiss Axiovert 25 Light Field microscope (Axiocam, Germany).
Results
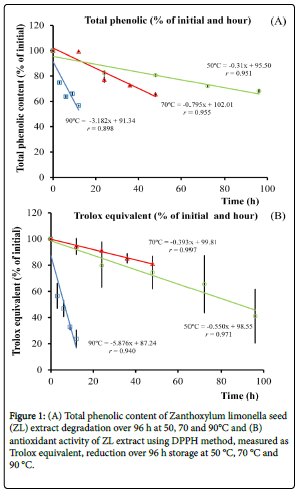
The percentage of total phenolic remaining of ZL extract (percentage of initial) stored at three different temperatures (50 °C, 70 °C and 90 °C) over 96 h shown in Figure 1A. The total phenolic contents of ZL extracts were decreased during storage at high temperature which the percentage of total phenolic remaining of ZL reduced when stored at 90 °C which significantly faster than those of 50 and 70 °C (p<0.001). The degradation rate (slope) of ZL extracts storage at 50, 70 and 90 °C were 0.31, 0.79 and 3.2 %·h-1, respectively. In addition to total phenolic content, DPPH assay of antioxidant activity of ZL extract storage at 50, 70 and 90 °C was carried out and calculated to Trolox equivalent as shown in Figure 1B. The reduction of antioxidant activity was observed which storage in 90 °C significantly reduced antioxidant activity faster than those of 50 and 70 °C storage temperature (p<0.001). The reduction rate (slope) of ZL extract storage at 50, 70 and 90 °C were 0.39, 0.55 and 5.88%·h-1, respectively. Both total phenolic and antioxidant activity affected by the temperature which at 90°C showed the fastest rate of degradation within 15 h.
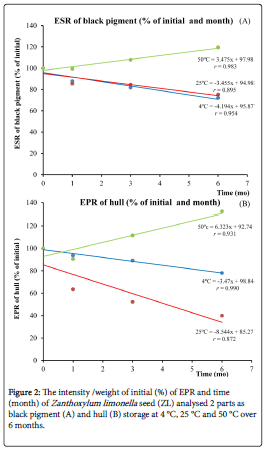
We also measure the organic radical in the seeds at three different temperatures. The signal (radical)is observed at 337 mTwhich corresponds to the organic radical in the ZL seed [9](Shown in Supplementary Figure 1).The intensity/weight of initial (%) observed by EPR and time (month) of black pigment and hull of ZL seed, shown in Figure 2, showed that storage condition at 50 °C detected the intensity of free radical greater than at 4 °C and 25 °C in both samples; black pigment and hull. The EPR results of pigments in the seeds show that the organic radical increase at 90 °C (Figure 2A) is directly relatedto the total phenolic contents and Trolox reduction (Figures 1A and 1B).
The blank mucoadhesive oral gel (BG) was prepared as a slightly viscous clear gel which was well mixed to incorporate ZL extract solution, lidocaine, and fluocinolone acetonide powder to form the sample gels. BG, FG, and LG were odorless and tasteless while ZG contained the fresh smell of the extracted. The physicochemical properties of ZG were shown in Table 1. The pH of ZL extracts was increased after incorporated into the gel base with slightly viscous clear gel.
| Parameters | ZL (extract solution) | ZG (gel) |
|---|---|---|
| Physicochemical Properties | ||
| -color | clear yellow solution | slightly viscous clear gel |
| -pH | 3.4-3.8 | 7.4-7.6 |
| Total phenolic content | ||
| (mg GAE*/g, n=6) | 0.33 ± 0.02 | - |
| Texture analysis (n=5) | ||
| firmness (N) | - | 0.10 ± 0.00 |
| compressibility (N × sec) | - | 0.19 ± 0.02 |
| adhesiveness (N × sec) | - | 0.06 ± 0.00 |
| cohesiveness (N × sec) | - | 0.17 ± 0.00 |
| Texture analysis (n=5) | - | 0.10 ± 0.00 |
Table 1: Characteristics of ZL, Zanthoxylum limonella Alston. seed extract and ZG, Zanthoxylum limonella Alston. seed extract in the gel base.
Remark: * as gallic acid equivalent
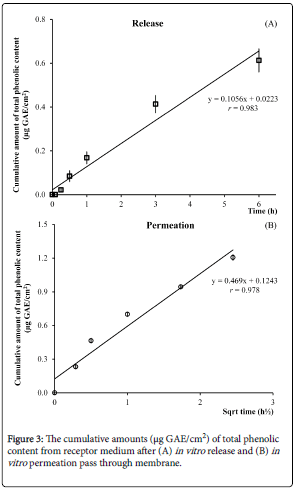
In vitro release and permeation studies of ZG indicated as the percentage cumulative total phenolic permeated through the membrane and time (h), shown in Figure 3 and the release rate was 0.156 μg/cm2/h with no lag time. The permeation of total phenolic of ZG was followed Higuchi model and permeation rate was 0.469 μg/cm2/h with no lag time.
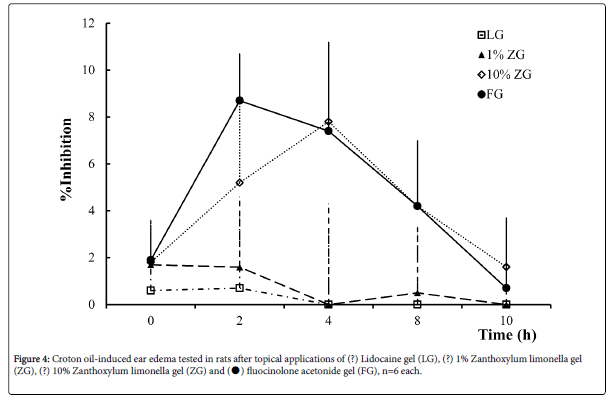
Figure 4 demonstrates percentage inhibition of rats’ ear edema of initial and time (h) after treated with all gel samples. Treated with FG gave the peak of anti-inflammatory activity (about 9% inhibition) within 2 h after application, then the activity began to decline, which remained the inflammatory inhibition for 8 h. Treated with 10%ZG showed inflammation inhibit activity started by accelerated since 2-4 h and declined which prolonged inhibition activity for 8 h while 1% ZG and LG showed no inhibition activity comparing to the BG. Figure 4: Croton oil-induced ear edema tested in rats after topical applications of (?) Lidocaine gel (LG), (?) 1% Zanthoxylum limonella gel (ZG), (?) 10% Zanthoxylum limonella gel (ZG) and (●) fluocinolone acetonide gel (FG), n=6 each. The comparison
The comparison percentage of redness (inflammation) and wound size reduction of the buccal wounds (% of the baseline) from day3 and day 5 after treated with BG, FG, and ZG were shown in Table 2. All buccal wounds of rats treated with all samples exhibited the inflammation and wound size reduction after 3 days of the treatments. Treated with ZG and FG had the most profounding effect on erythema score (62.67% and 65.50% of initial) in day 3 and (71.46% and 67.42% of initial) in day 5 compared to LG and BG (about 50% reduction). Moreover, ZG and FG reduced the wound size by 71.29% and 72.82% in 3 days and 93.90 and 85.31% in day 5, respectively.
| Treatments | Day 3 | Day 5 | ||
|---|---|---|---|---|
| Redness reduction (%) | Wound size reduction (%) | Redness reduction (%) | Wound size reduction (%) | |
| BG | 42.93 | 48.37 | 50.06 | 74.27 |
| LG | 46.64 | 48.9 | 56.81 | 73.78 |
| ZG | 62.67* | 71.29* | 71.46* | 93.90* |
| FG | 65.50* | 72.82* | 67.42* | 85.31* |
Table 2: The results comparison percentage of redness (inflammation) and wound size reduction of the baseline from day 3 and day 5 after the treatments of the gel samples blank gel (BG), lidocaine gel (LG), Zanthoxylum limonella Alston. seed extract incorporated in the gel base (ZG) or fluocinolone acetonide gel (FG) groups.
Cross-sections of surface regions of the treated wounds were histologically compared with the normal buccal tissues in Figure 5, in comparison of FG, BG, and ZG treated of normalbuccal, treated for 3 days and 5 days. Layers of non-keratinized stratified squamous epithelia with nucleated basal cells interfaced with the lamina propria were well defined in wound samples treated by BG, LG, and ZG as similar as the normal buccal. The FG-treated wound sample consists of thin superficial and intermediate layers with very few cuboidal nucleated epithelial cells which differ from the normal sample.
Figure 5: Histological sections prepared from buccal mucosa comparing epithelium structure (sub-mucosa, lamina propria) of treated wounds with normal buccal (day 0, no wound) of the same rat in the treatment groups, FG (fluocinolone acetonide gel), LG (lidociane gel) and ZG (Zanthoxylumlimonella gel); sections were stained with hematoxylin and eosin and micrographs were taken at a magnification of 40×.
Discussion
The percentage remaining of the total phenolic content and the rate of degradation of ZL extract were rapidly decreased while the high temperature which stored at 90 °C was declined faster than stored at 50 and 70 °C. In long-term condition, the free radical scavenging detected by EPR also revealed that stored at 50 °C affected the free radical scavenging. The normal condition of storage temperatures such as 4 and 25 °C could preserve the phenolic content and/or prevent initiating of the free radical reaction. Antioxidant activity of ZL showed the relationship between the phenolic content, free radical scavenging, and high temperature which similar to other spices stability [22].
The pH of all gel samples was prepared as similar to the pH of normal oral, 7.4 with slightly viscous and easier to apply onto the membrane. In vitro release and permeation studies of ZG demonstrated that total phenolic was easily released and penetrated pass through the membrane.
The main irritant compounds of croton oil are 12-otetracanoilphorbol- 13-acetate and other phorbol esters [23]. The croton oil dilutes with acetone in order to rapidly vaporize and lefts croton oil to activate a cascade pathway of arachidonic acid production. Irritants stimulate the release of inflammatory mediators such as leukotrienes, histamine, prostaglandins, and serotonin by cyclooxygenase (COX) and 5-lipoxygenase (5-LOX) enzymes which COX and 5-LOX can be inhibited anti-inflammatory such as corticosteroids [24]. Applying croton oil to rat ears resulted in signs of inflammation, i.e. redness, swelling, which can be observed within 5 min and remained for 2-4 h.
Topical ZG at concentration 10% began to exhibit some antiinflammatory activity cover from 2-4 h and gave the peak of inflammation reduction at 4 h which showed a nonsignificant difference to FG at 4 h. Moreover, 10% ZG, ear edema was inhibited and prolonged till 8 h at which the extent of activity was similar to FG group. This suggests that a single topical application of ZG to the skin could inhibit inflammation caused by an irritant used in croton oilinduced ear edema model. It was anticipated that an effective dose of ZL extracts (10%) should be higher than corticosteroids. This study showed that the extracted of ZL was showed less potent than fluocinolone acetonide by the dose and onset of action.
Oral wound healing was assessed in this study as a reduction of erythema, i.e. anti-inflammation, and reduction of wound size, i.e. wound healing in which re-epithelization occurs as the cells migrate to fill the wound gaps [25,26]. Normally, the wound healing process starts just a few minutes after wounding, but its success depends on a number of complex chemotactic and enzymatic activities as well as reepithelization usually detectable within one week. Anti-inflammatory and wound area of buccal of rats was reduced observed since the first day. Treated with ZG and FG reduced both redness and wound size significant difference than treated BG and LG (all, p<0.001). Moreover, the ZG also reduced wound sizes of rats significantly better than the used of LG and BG at day 3 and showed no significant to FG (p=0.78). Lidocaine is an anesthetic agent and showed mild anti-inflammatory [27]. However, the anti-inflammatory activity and wound size reduction of the rats treated with LG showed no significant difference from the BG group which may because of the self-healing. FG reduced the erythema scores by half since day 3 and, but it took 5 days to reduce wound sizes to about 72.82% of initial. The re-epithelization generally follows and overlaps with the inflammatory phase [28] and it was surprising that wound healing action of FG was not well manifested and revealed by histological disorientation of the epithelium cells in the cross-section sample of the buccal wound which was excised at day 3 but recovery at day 5 suggesting that fluocinolone have both anti-inflammatory and wound healing effects. ZG reduced the erythema scores by half since day 3, but it took 5 days to reduce wound sizes to about 72.82% of initial, suggesting anti-inflammatory but poor wound healing activity.
Conclusion
Zanthoxylum limonella Alston. seed extracts incorporated in a mucoadhesive gel (at a neutral pH) is able to stabilize the total phenolic contents and be topically delivered for oral anti-inflammatory. In vivo testing in ear edema and buccal wounds of rats indicates antiinflammatory activities at 10% ZG. Thus, lead to confirm antiinflammatory activity by the Zanthoxylum limonella Alston. seed extracts in the topical oral gel and could be further studied in human clinical trials.
Acknowledgments
This work receives a scholarship under the Post-doctoral Program from Research Affairs and Graduate School, Khon Kaen University (58226). Mr. Apichet Pomjareet is thanked for assisting with the total phenolic assay and EPR measurement. Ms. Nanthiya Wongsaengta and Ms. Bhattaranit Khampanjiraroch for general assistance. The authors declare that they have no competing interests.
References
- Conner EM, Grisham MB (1996)Inflammation, free radicals,and antioxidants. J Nutr 12: 274-277
- Hagg U, Kaveewatcharanont P, Samarnayake YH, Samarnayake LP (2004) The effect of fixed orthodontic appliances on the oral carriage of Candida species and Enterobacteriaceae. Eur J Orthod 26: 623-629
- Casaroto AR, Lara VS (2010) Phytomedicines for Candida-associated denture stomatitis. Fitoterapia 81: 323-328
- Pahwa N, Kumar A, Gupta S (2011) Short term clinical effectiveness of a 0.07% cetylpyridinium chloride mouth rinse in patients undergoing fixed orthodontic appliance treatment. The Saudi Dent J 23: 135-141
- Squier CA (1991) The permeability of oral mucosa. Crit Rev Oral Biol Med2: 13-32
- Verma N, Chattopadhyay P (2011) Polymeric platform for mucoadhesive buccal drug delivery system: a review. Int J Curr Pharm Res 3:3-8
- Tangjitjaroenkun J, Supabphol R, Chavasiri W (2012) Antioxidant effect of Zanthoxylum limonella Alston. J Med Plants Res 6: 1407-1414
- Itthipanichpong C, Ruangrungsi N, Pattanaautsahakit C (2002) Chemical compositions and pharmacological effects of essential oil from the fruit of Zanthoxylum limonella. J Med Assoc Thai 85: S344-354
- U.S. Food and Drug Administration (FDA). Guidance for Industry: Bioanalytical Method Validation. In: Services DoHaH, editor. Rockville, MD: U.S. Food and Drug Administration (FDA); 2001.
- Nakagawa K, Promjareet A, Priprem A, Netweera V, Hara H (2016) Investigation of scavenging activities and distribution of paramagnetic species in Zanthoxylum limonella seeds. Free Radic Res 50: 1432-1440
- Nakagawa K, Epel B (2014) Locations of radical species in black pepper seeds investigated by CW EPR and 9GHz EPR imaging. Spectrochim Acta A Mol Biomol Spectrosc 131:342-346
- Nakagawa K, Hara H (2015) Investigation of radical locations in various sesame seeds by CW EPR and 9-GHz EPR imaging. Free Radic Res 49: 1-6
- ICH guideline. Guidance for Industry Q1A(R2) Stability Testing of New Drug Substances and Products. Switzerland: The International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH); 2003.
- Singleton VL, Rossi JA (1965) Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic 16: 144-158
- Nachajski MJ, Zgoda MM (2010) The influence of polymeric excipients on the process of pharmaceutical availability of therapeutic agents from a model drug form. Part I. In formulations with controlled disintegration and release time. Polim Med 40: 65-77.
- Friedman HH, Whitney JE, and Szczesniak AS (1963) The Texturometer-A New Instrument for Objective Texture Measurement. J Food Sci 28: 390-396
- Diaz-del CI, Jacques Y, Pizzolato GP, Guy RH, Falson F (2005) Comparison of the lipid composition of porcine buccal and esophageal permeability barriers. Arch Oral Biol 50: 981-987
- Sudhakar Y, Kuotsu K, Bandyopadhyay AK (2006) Buccal bioadhesive drug delivery--a promising option for orally less efficient drugs. J Control Release 114: 15–40
- Garber JC (2011) Washington DC Guide for the Care and Use of Laboratory Animals 8th ed. The National Academies Press
- Cavalcante GM, dePaula RJS, deSouza LP, Sousa FB, Mota MRL, et al. (2011) Experimental model of traumatic ulcer in the cheek mucosa of rats. Acta Cir Bras 26: 227-234
- Gallin JI, Snyderman R, Fearon DT, Haynes BF, Nathan C (1999) Philadelphia Inflammation: Basic Principles and Clinical Correlates. 3rd ed.
- Settharaksa S, Jongjareonrak A, Hmadhlu P, Chansuwan W, Siripongvutikorn S (2012) Flavonoid, phenolic contents and antioxidant properties of Thai hot curry paste extract and its ingredients as affected of pH, solvent types and high temperature. Int Foo
- Garbacki N, Gloaguen V, Damas J, Hoffmann L, Tits M, et al. (2000) Inhibition of croton oil-induced oedema in mice ear skin by capsular polysaccharides from cyanobacteria. Naunyn Schmiedebergs Arch Pharmacol 361: 460-464
- Charlier C, Michaux C (2003) Dual inhibition of cyclooxygenase-2 (COX-2) and 5-lipoxygenase (5-LOX) as a new strategy to provide safer non-steroidal anti-inflammatory drugs. Eur J Med Chem 38: 645–59
- Tomikawa K, Yamamoto T, Shiomi N, Shimoe M, Hongo S, et al. (2002) Smad2 decelerates re-epithelialization during gingival wound healing. J Dent Res 91: 764-770
- Larjava H, Koivisto L, Hakkinen L, Heino J (2011) Epithelial integrins with special reference to oral epithelia. J Dent Res 90: 1367-1376
- Pochapski MT, Neto JL, Jassen JL, Farago PV, Santos FA (2012) Effect of Lidocaine- and Prilocaine-Based Topical Anesthetics on the Inflammatory Exudates in Subcutaneous Tissue of Rats. Anesth Prog 59: 57–61
- Guo S, DiPietro LA (2010) Factors affecting wound healing. J Dent Res 89: 219-229
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi