Research Article, J Pharm Sci Emerg Drugs Vol: 6 Issue: 1
Synthesis and Quality Control of 68Ga-PSMA PET/CT Tracer used in Prostate Cancer Imaging and Comparison with 18F-Fluorocholine as a Reference Point
Marcin Szydlo*, Dawid Pogoda, Tomasz Kowalski, Mateusz Pocięgiel, Michał Jadwiński and Andrea d’Amico
Department of PET Diagnostic, Maria Skłodowska-Curie Memorial Cancer Center, Institute of Oncology, Gliwice, Poland
*Corresponding Author : Marcin Szydlo
Department of PET Diagnostic, Faculty of Diagnostic, Maria Sklodowska Curie Memorial Cancer Center, Institute of Oncology, 44-101 Gliwice, Poland
Tel: +48-32-278-9912
Fax: +48-32-278-8543
E-mail: Marcin.Szydlo@io.gliwice.pl
Received: February 16, 2018 Accepted: March 08, 2018 Published: March 15, 2018
Citation: Szydlo M, Pogoda D, Kowalski T, Pocięgiel M, Jadwiński M, et al. (2018) Synthesis and Quality Control of 68Ga-PSMA PET/CT Tracer used in Prostate Cancer Imaging and Comparison with 18F-Fluorocholine as a Reference Point. J Pharm Sci Emerg Drugs 6:1. doi:10.4172/2380-9477.1000126
Abstract
The clinical usefulness of choline-based PET/CT tracers (11C-Choline and 18F-Fluorocholine) in prostate cancer recurrence imaging is well established. However, choline uptake is not increased in a considerable number of patients. In 2012 was proposed the new prostate-specific PET radio ligand PSMA 68Ga-labeled, with superior sensibility and specificity than choline-based tracers. This work has as aim to describe the procedures for labeling PSMA ligands and its quality control procedures as a medicinal product and its comparison with 18F-fluorocholine synthesis and quality control procedures. Both the manual and automated synthesis methods for 68Ga-PSMA were taken into account and set of a quality control methods based mainly on chromatographic and spectrometric methods used to determine chemical, radiochemical and radionuclide purity. PSMA synthesis time, dose of radiation to operators and quality control protocols were compared to choline. Finally, some images of PET performed with 18F-Fluorocholine and 68Ga-PSMA in the same patients were showed.
Keywords: Radiopharmacy; Choline; PSMA; PET/CT; Prostate; Cancer; Synthesis; Quality
Introduction
Prostate cancer is one of the more frequent malignancies in developed countries, with high rates of morbidity and mortality [1]. After radical treatment, the rise of serum prostate-specific antigen (PSA) is the most important marker of recurrence. Imaging of recurrence is traditionally based on bone scan, ultrasonography and computed tomography.
In the last years, positron emission imaging (PET) with choline radiolabeled with carbon-11 or fluorine-18 was demonstrated to be able to localize recurrence site in patients with PSA level higher than 2 ng/ml [2]. Recently, a new PET radiopharmaceutical was proposed for early localization of prostate cancer relapses: prostate-specific membrane antigen ligand 68Ga-HBED-CC-PSMA (68Ga-PSMA), with diagnostic performance markedly superior to 18F-fluorocholine PET [3].
Uptake of 68Ga-PSMA reflects increased density of N-acetyl-Laspartyl- L-glutamate peptidase 1 (NAALAD1) [4]. For this reason an increasing number of PET laboratories are switching from choline to PSMA imaging for clinical purposes.
This work has as aim to describe the procedure for labeling PSMA ligands and its quality control as a medicinal product and then to compare results of 68Ga-PSMA and 18F-fluorocholine.
Finally, a pictorially essay of some compared 18F-fluorocholine and 68Ga-PSMA PET images in a series of patients from our site was given.
Materials and Methods
The production of PSMA peptide labeled with 68Ga gallium is done with the use of the Eckert-Ziegler pharmacy approved 68Ge- 68Ga generator with and hydrochloric acid 0.1 M as an eluent. The labeling kit contains 30 μg PSMA peptide, 45 mg sodium acetate, 25mg ascorbic acid, ethanol (99,8% and 50%), water for injection, sterile filter 0,22 μm (Millipore), 0,9% NaCl and C18 SPE cartridge (Waters). The Scintomics GRP automated synthesis module is used for automated synthesis and the manual preparation is carried in a shielded hot cell. All radioactivity measurements are proceeding with use of dose calibrator. All materials and methods used during quality control are described in Quality control methods section.
Manual synthesis of 68Ga-PSMA
A 10 ml vial containing PSMA peptide, sodium acetate and ascorbic acid (buffer pH like in Eder et al [6]) is heated in a heating block at 80ºC. 68Ge/68Ga generator, connected directly to the synthesis vial, is eluted with 5 ml 0,1M HCl. Heating is continued for at least 10 min. After synthesis, the reaction mixture is pushed through the C18 cartridge (preconditioned with sequence of 2 ml of 99,8% ethanol, 10 ml of water and dried with 10 ml of air) to trap labeled peptide. After trapping of synthesis final product the C18 cartridge is connected to the waste vial and rinsed with 5 ml of water for injection. The final product is eluted from C18 cartridge with 0,5 ml 50% ethanol and 10 ml 0.9% saline to a 30 ml sterile vial through the sterile filter 0,22 μm. At the end of synthesis the activity product, waste vial and C18 cartridge is measured in dose calibrator.
Automated synthesis of 68Ga-PSMA
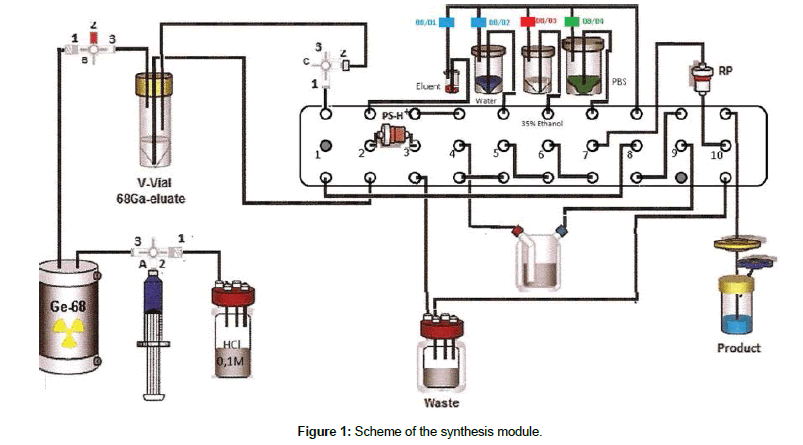
The preparation of the synthesis module is done by preparing and connecting the reagents to the appropriate tubing’s and by filling the reactor with the 20 μl of PSMA peptide solution in water at the concentration of 1 mg/ml and 2 ml of 1,5 M HEPES buffer solution as it is shown on Figure 1. After the initial check of the Scintomics GRP module software the preparations are complete.
The production process progress in two steps similarly to René Martin et.al. [5]. Firstly the 68Ga gallium is eluted from the generator with the use of 4ml 0,1M HCl to the V-vial. The second step comes after the elution, when the operator of the synthesis module starts the labeling process. The whole script runs for approximately 20 minutes and the temperature of heating block is set to 125ºC to ensure that the labeling runs in around 100ºC. After the start of labeling the 68Ga gallium chloride is transferred to the PSH+ cartridge to remove the excess of the hydrochloric acid and then eluted to the reactor with 2 ml of acidified 5,0 M NaCl. The labeling process takes around 10 minutes and then the purification part of the script begins. First the reaction mixture is send through the C18 cartridge to the waste bottle. C18 cartridge is preconditioned with 2 ml of 99,8% ethanol and 10 ml of water and dried with air at the end. The 68Ga gallium labeled PSMA peptide is adsorbed by the cartridge and the eluent and HEPES buffer (2-[4-(2-hydroxyethyl) piperazin-1-yl] ethane sulfonic acid) present in the reaction mixture are removed. Then the C18 cartridge is flushed with water to ensure removal of HEPES buffer and other water soluble impurities. When the purification step is complete the product is removed from the C18 cartridge by flushing it with 2 ml of 35% ethanol solution and transferred through the 0,22 μm Cathivex- Gv (Millipore) filter to the product vial. After the removal of the 68Ga gallium labeled PSMA, the C18 cartridge is further flushed with 20 ml of phosphate buffered saline solution (PBS) to ensure the proper concentration and a pH level of the obtained product.
Quality control methods
Quality control methods are in most based on European Pharmacopoeia (PhEur) monograph describing Gallium (68Ga) Edotreotide for Injection [6], despite liquid chromatography method, which is based on Eder and colleagues [7].
Gamma ray spectroscopy: This examination is about measurement of the energy of the detected gamma quantum. The gamma ray spectroscopy is proceed with use of multichannel analyzer with 3” x 3” NaI detector All of the positron emitting isotopes are characterized by energy of the photon, which is emitted during positron annihilation with electron, and it is expected to be 511keV [8]. This test is one of the nuclide identification tests.
Half-life determination: Determination of the half-life is the second test that the purpose is to define the isotope. Half-life is obtained with use of dose calibrator for the activity measurements. Each activity measurement lasts 20 minutes and is repeated every minute. The expected half-life of Gallium-68 is 67.6 minutes [9] and can be calculated using following equation [10]:
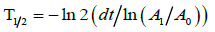
Where: dt-time difference
A1-ending activity
A0-starting activity
These two tests mentioned above uniquely identify the nuclide as Gallium-68.
Determination of residual HEPES buffer: According to PhEur monograph [6] HEPES content is determined by thin-layer chromatography (TLC). It is needed to prepare reference solution of HEPES at concentration of 20 μg/ml. Applicant 5 μl of each, reference and test solution (the test solution is the [68Ga]-PSMA final product sample), on TLC silica gel F254 plate with an aluminium backing (3 cm x 8 cm) as a two separate spots and develop on a path over 2/3 of the plate with use of water: acetonitrile (25: 75 V/V) solution as a mobile phase. Expose to iodine vapor for four minutes after drying. Spot corresponding to the test solution should be not more intense than the spot corresponding to the reference solution (less than 20μg/ ml of HEPES in test solution).
Residual solvents: The only organic solvent used during [68Ga]-PSMA labeling is an ethanol. In Pharmacopoeia there are three method described to determine the amount of ethanol in a pharmaceutical product: distillation with a vertical condenser (A) and gas chromatography (GC) with a head-space (B) a without headspace (C) [11]. In this study gas chromatography (C) is used in accord with the pharmacopoeial method [12]. Any calibrated analytical GC method adapted to the method mentioned above should be suitable for this analysis. In our apparatus are used the same settings as for FDG analysis i.e. volume of sample injection is 1,0 μl; split ratio 5,0:1 at a total flow of 16,2 ml/min with helium as a carrier gas. Used column is HP-Inn wax (30 m x 0,32 mm; film: polyethylene glycol 0,50 μm) in column heater settled up to 50ºC. Detection is carried out on an FID detector (Flame Ionization Detector).
Bacterial endotoxin purity: The bacterial endotoxin test is performed using Endosafe®-PTS™ (Charles River) apparatus and the kinetic chromogenic LAL-test method applied, the same as during [11C]-choline quality control [13].
Radionuclide purity: Gamma spectroscopy of the final product sample also is carried out in the same way as in paper mentioned above [13-18] in a Canberra-Packard gamma spectrometer equipped with a high-purity germanium (HPGe) detector. The spectra acquisition is performer two days after synthesis. Acquisition is proceeded for 180 minutes to obtain high a signal-to-noise ratio. The sample volume should be at least 1 ml. Spectrum is analyzed using Genie 2000 software.
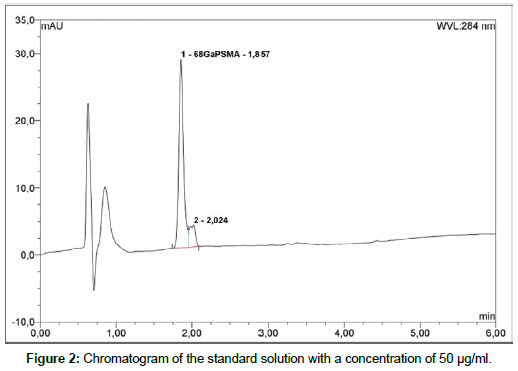
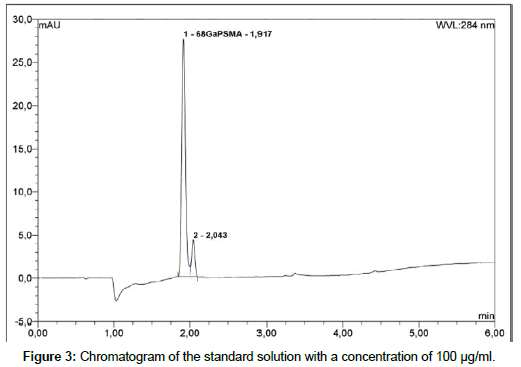
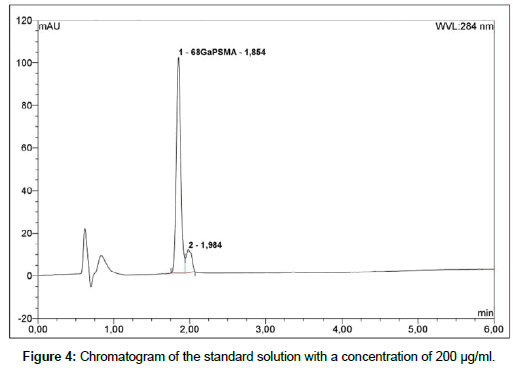
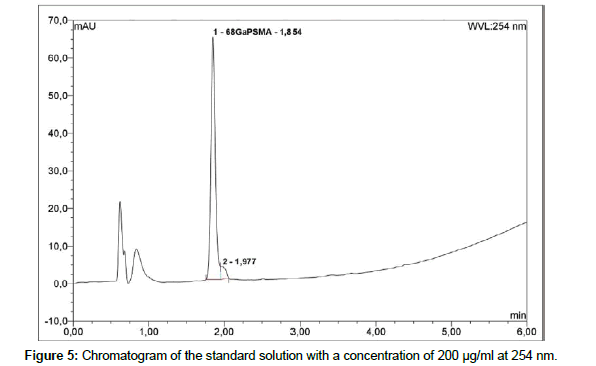
Liquid chromatography: To determine radiochemical and chemical purity of 68Ga-PSMA a reversed-Phase HPLC with Chromolith Performance RP-18e column; 100 x 4.6 mm (Merck) is used. The HPLC system is Thermo Fisher Dionex Ultimate 3000 equipped with on-line degasser, UV and radiometric (Bioscan B-FC- 1000) detectors. Measurement is carried out in linear A-B gradient (0 % B to 100 % B) at flow rate 4 ml/min. Solvent A consist of 0,1 % aqueous trifluoroacetic acid (TFA) and solvent B is 0,1 % TFA in acetonitrile [19]. UV absorbance is measured at wavelength 254.0 nm and 284.0 nm. The injection volume is 50 μl. All of obtained chromatograms were compared with natGaPSMA standard solution. To find the best concentration of the standard solution, 1 mg of natGaPSMA-11 (ABX, Radeberg, Germany) was dissolved in 1 ml (1:1 v/v CH3CN/H2O) and from this stock the solutions with following concentrations were prepared: 50 μg/ml, 100 μg/ml and 200 μg/ml. Three standard solutions with concentrations of 50 μg/ ml (Figure 2); 100 μg/ml (Figure 3) and 200 μg/ml (Figure 4) were analyzed during the study with an UV detector at 284 nm. As it is seen on the recorded chromatograms, the best results were given from solution containing 200 μg/ml of natGaPSMA. Therefore, it was decided to record one more chromatogram with use of the 200 μg/ml standard solution at 254 nm (Figure 5) [7]. The better results were obtained during the measurements at a wavelength of 284 nm and that is why it was decided to measure the 68Ga-labeled PSMA-11 peptide at this wavelength.
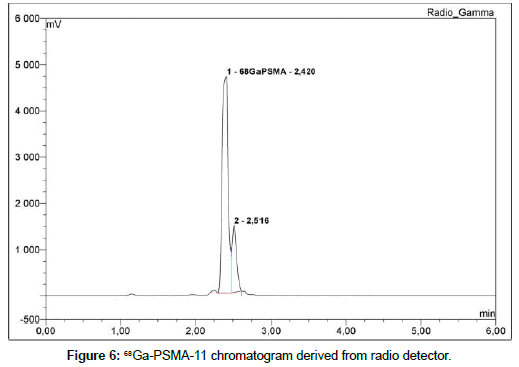
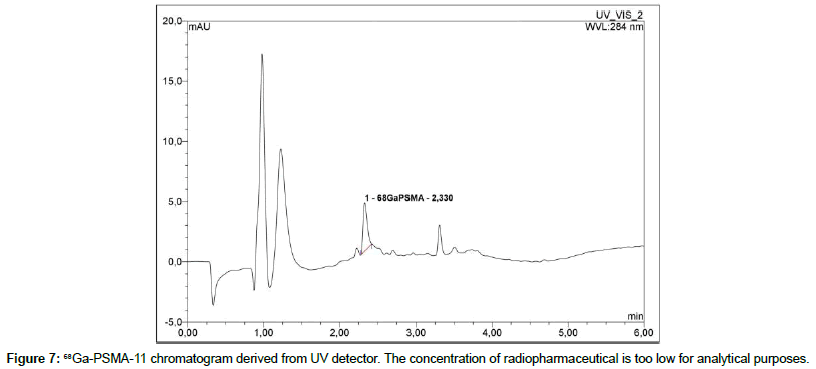
68Ga-labeled PSMA peptide is analyzed with an UV detector at 284 nm and a radiometric detector. During the tests it was found that the concentration of 68Ga-PSMA in the sample diluted with a saline is below the detection limit of radiometric detector (synthesis product is diluted with saline to provide isotonicity). Therefore, for the further analysis the undiluted product is used. There are two signals that can be observed in the obtained chromatograms in a ratio of approximately 82%/12%. The more intense signal comes from 68Ga- PSMA-11, the second signal comes from its less thermodynamically stable diastereomer and does not affect the test result (Figure 6). These signals can be observed both on the UV detector and the radiometric detector. However, the concentration of 68Ga-PSMA-11 in the test sample is too low to obtain a reliable result on the UV detector (Figure 7).
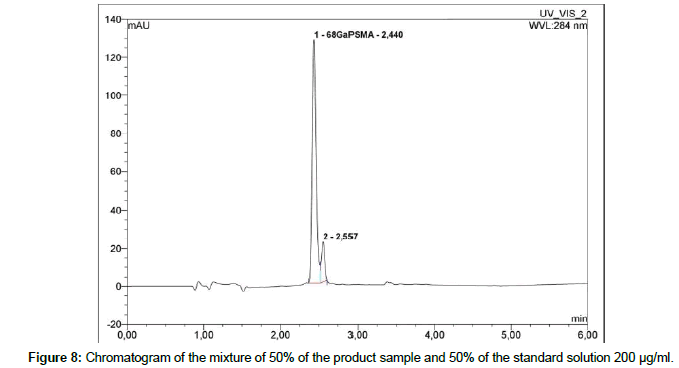
To confirm the identity of the compound, the mixture of 50% of the product sample and 50% of a standard solution of 200 μg/ml were measured (Figure 8).
Results
The activity of the 68Ga gallium labeled PSMA peptide depends on the activity eluted from the 68Ge-68Ga generator. For the 40 mCi of eluate the Scintomics GRP automated synthesis module can produce around 23 - 25 mCi of product.
The activity of the product obtained by automated method of labeling is a little lower than analogical product obtained with the use of manual method because of the more complicated purification (necessity of complete removal of the HEPES buffer). But otherwise it is still comparable to the manual method. On the other hand because of the automatization of the process the human error factor is minimized and the dosages of radiation received by synthesis operators are lower, which means the process is safer.
Short pictorial essay
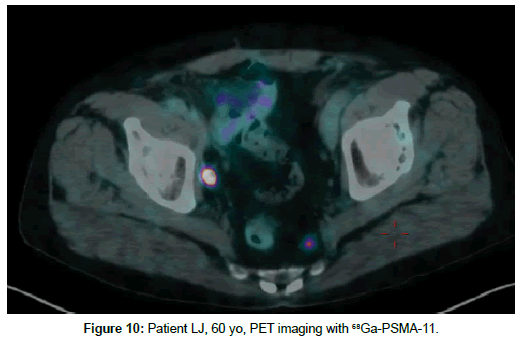
Patient LJ, 60 yo, prostate cancer treated with prostatectomy in 2013. Biochemical relapse, PET with 18F-choline on 30th September 2016 with serum PSA 0.79 ng/ml with no pathologic uptake (Figure 9). Second PET with 68Ga-PSMA on 26th October 2017 with seric PSA 1,56 ng/ml showed pelvic lymph node with strongly increased PSMA uptake (Figure 10).
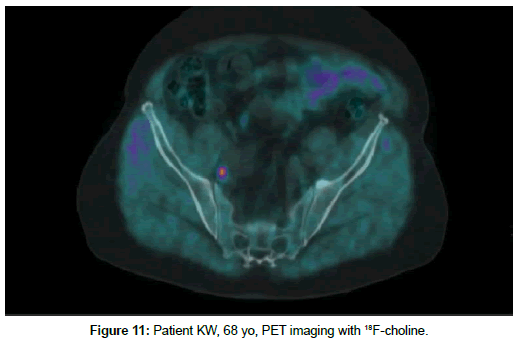
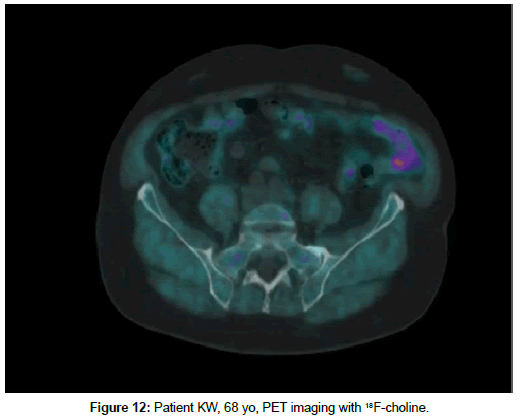
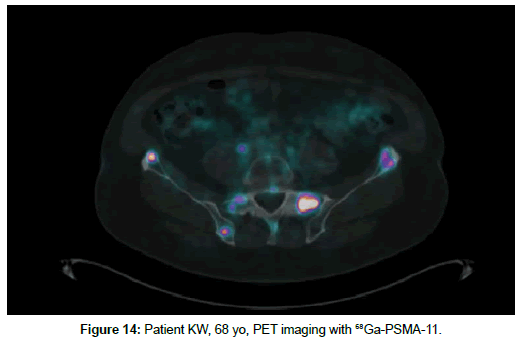
Patient KW, 68 yo, prostate cancer treated with radiotherapy in 2014, biochemical relapse during suppressive therapy with PSA 0,94 ng/ml on 29th December 2016. First PET scan 21th April 2017 positive at pelvic lymph node, with no bone uptake (Figures 11 and 12). Due to rising seric PSA to 31, 88 ng/ml a second PET with 68Ga- PSMA was carried out on 15 September, with confirmation of lymph node metastasis and multiple pathologic bone foci not present at choline imaging (Figures 13 and 14).
Conclusion
Synthesis of radiopharmaceuticals: 68Ga-PSMA and 18F-fluorocholine (18F-FCH) used in prostate cancer diagnostic procedures are completely different. In case of 18F-fluorocholine synthesis run in numerous steps with many chemicals compounds such as dibromomethane (DBM) or dimethylaminoethanol (DMAE) which are quite toxic and tough chemical during laboratory work. Some of 18F-FCH synthesis by-products are in gas phase [14], this triggers the need to use radioactive gases anti-contamination systems. The Fluorine-18 used in the 18F-FCH synthesis is produced in a liquid target of cyclotron [15]. The construction and use of such an accelerator is quite expensive, however the amount of Fluorine-18 isotope produced for a single synthesis, it allows producing a radiolabelled compounds for impressive number of patients (up to 15, using IBA Synthera modules). Such a connection high activity final product and long half-life of Fluorine-18 (110 minutes) allows the 18F-FCH could be transported to the PET diagnostic departments, distant from the place of production [16]. In case of second radiopharmaceutical 68Ga-PSMA, which are used to diagnostic prostate cancer, isotope of Gallium-68 has shorter halflife (68 minutes). This isotope is obtained from 68Ge/68Ga generator during column elution to receive Gallium-68 chloride. Synthesis of 68Ga-PSMA is a simple process with no complicated steps resulting from reaction mechanism [17]. 68Ge/68Ga generator can be used for several months, which could be eluted even two hundred times. Short half-life of Gallium-68 allows frequently elution of the generator on the same day, but compare to 18F-FCH, single synthesis of 68Ga- PSMA provide to PET diagnosed at most 3 patients. Compounds which are used during 68Ga-labeled PSMA ligand are easy to operate, and do not require special conditions. Labeling process is characterized by high yield. Finally, product of radiopharmaceutical PSMA is stable and in form of injectable formulation. An important parameter to both radiolabeled compound is time of synthesis. Synthesis of 18F-FCH run approximately 90 minutes, compared to 68Ga-PSMA only 15 minutes. Moreover, it is difficult not to mention in view of radiological protection about lower activity of 68Ga-PSMA than 18F-FCH during patient injection [18].
References
- Daniyal M, Siddiqui ZA, Akram M, Asif HM, Sultana S, et al. (2014) Epidemiology, etiology, diagnosis and treatment of prostate cancer. Asian Pac. J. Cancer Prev 15: 9575-9578.
- von Eyben FE, Kairemo K (2014) Meta-analysis of (11)C-choline and (18)F-choline PET/CT for management of patients with prostate cancer. Nucl Med Commun 35: 221-230.
- Afshar-Oromieh A, Haberkorn U, Eder M, Eisenhut M, Zechmann CM (2012) [68Ga] Gallium-labelled PSMA ligand as superior PET tracer for the diagnosis of prostate cancer: comparison with 18F-FECH. Eur J Nucl Med Mol Imaging 39: 1085-1086.
- Mease RC, Foss CA, Pomper MG (2013) PET imaging in prostate cancer: focus on prostate-specific membrane antigen. Curr Top Med Chem 13: 951-962
- Martin R, Jüttler S, Müller M, Wester HJ (2014) Cationic eluate pretreatment for automated synthesis of [68]CPCR4.2. Nucl Med Biol 41: 84-89.
- Gallium (68Ga) Edotreotide (monograph 2482) In: European Pharmacopoeia, European Directorate for the Quality of Medicines 1: 1062-1064.
- Eder M, Neels O, Müller M, Bauder-Wüst U, Remde Y, et. al. (2014) Novel Preclinical and Radiopharmaceutical Aspects of [68Ga]Ga-PSMA-HBED-CC: A New PET Tracer for Imaging of Prostate Cancer. Pharmaceuticals 7: 779-796.
- Hine GJ (1967) Sodium Iodine. Academic Press Inc. 1: 95-119.
- Table of physical characteristic of radionuclides mentioned in the European Pharmacopoeia. In: European Pharmacopoeia, European Directorate for the Quality of Medicines 1: 665-673.
- Serway RA, Jewett JW (2008) Physics for scientists and engineers with modern physics, Chapters 39-46 (7th edtn) Nuclear Structure. Cengage Learning.
- Ethanol content (monograph 20910) In: European Pharmacopoeia, European Directorate for the Quality of Medicines 1: 301-303.
- Gas chromatography (monograph 20228) In: European Pharmacopoeia, European Directorate for the Quality of Medicines 1: 43-45.
- Szydło M, Jadwiński M, Chmura A, Gorczewski K, Sokół M (2016) Synthesis, isolation and purification of [(11)C]-choline. Contemp Oncol (Pozn) 20: 229-236.
- Krasikova R (2007) Synthesis modules and automation in F-18 labeling. Ernst Schering Res Found Workshop 62: 289-316.
- Clark JC, Silvester DJ (1966) A cyclotron method for the production of fluorine-18. Int J Appl Radiat Isot 17: 151-154.
- DeGrado TR, Baldwin SW, Wang S, Orr MD, Liao RP, et al. (2001) Synthesis and evaluation of (18)F-labeled choline analogs as oncologic PET tracers. J Nucl Med 42: 1805-1814.
- Nanabala R, Anees MK, Sasikumar A, Joy A, Pillai MR, et.al. (2016) Preparation of [68Ga]PSMA-11 for PET-CT imaging using a manual synthesis module and organic matrix based 68Ge/68Ga generator. Nucl Med Biol 43: 463-469.
- Giesel FL, Hadaschik B, Cardinale J, Radtke J, Vinsensia M, et.al. (2017) F-18 labelled PSMA-1007: biodistribution, radiation dosimetry and histopathological validation of tumor lesions in prostate cancer patients. Eur J Nucl Med Mol Imaging 44: 678-688.
- Eppard E, Homann T, de la Fuente A, Essler M, Rösch F, et.al. (2017) Optimization of labeling PSMAHBED with ethanolâ€Âpostâ€Âprocessed 68Ga and its quality control systems. J Nucl Med 58: 432-437.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi