Review Article, J Pharm Sci Emerg Drugs Vol: 10 Issue: 4
Therapeutic Significance of Pyrrole in Drug Delivery
Kasturi Pawar*
Department of Pharmaceutics, Auburn University, Auburn, USA
*Corresponding Author:Kasturi Pawar, Department of Pharmaceutics, Auburn University, Auburn, USA, Tel: 16504402316;E-mail: kasturipawar@gmail.com
Received date: 16 February, 2022, Manuscript No. JPSED-22-54563; Editor assigned date: 21 February, 2022, PreQC No. JPSED-22-54563 (PQ); Reviewed date: 01 March, 2022, QC No. JPSED-22-54563; Revised date: 15 April, 2022, Manuscript No. JPSED-22-54563 (R); Published date: 25 April, 2022, DOI: 10.4172/2380-9477.1000162
Citation: Kasturi Pawar (2022) Therapeutic Significance of Pyrrole in Drug Delivery. J Pharm Sci Emerg Drugs 10:4.
Abstract
Heterocycles encompass an important section in the discovery field of medicinal chemistry. Several commercial products have one or more heterocycles in their structure, underlining the importance of these moieties for therapeutic applications. Pyrrole is one such heterocycle which has been part of natural compounds existing in nature as well as commercial drugs in the market. It has changed the paradigm of drug delivery in bringing key therapeutic molecules to the market in variety of therapeutic areas. Further, pyrrole group is also part of other industries such as agriculture, paint, chemicals, dyes, and plastic. This highlights the multitude of advantages pyrrole moiety has in varied industrial areas. In drug discovery, pyrrole holds a special place due to its contribution in the history of therapeutics. Researchers have been working towards synthesizing and optimizing novel pyrrole-based molecules through Structural Activity Relationships (SARs) to lead to pharmaceutically viable New Chemical Entity (NCE) for various indications. This review is focused on applications of pyrrole in various therapeutic areas and discussing the current research that is been undertaking towards development of novel therapeutic moieties containing pyrrole group.
Keywords: Heterocycle; Pyrrole; Drug delivery; Drug discovery
Introduction
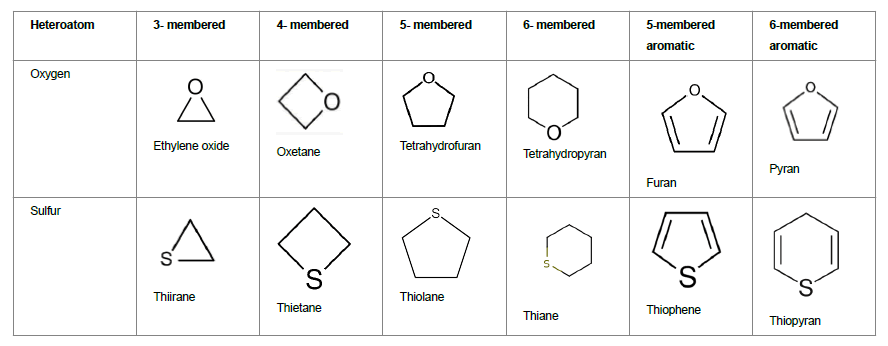
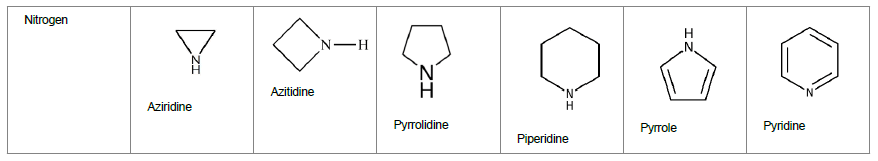
With the scientific progressions, medicinal chemistry field has expanded the realms of novel therapeutic molecules extensively for finding a cure for difficult to treat indications. These molecules include small molecules as well as biologics that were once discovered as NCE. While traditional dosage forms including oral dosage form such as tablets and capsules [1-3] continue to exist, extensive research has also been done in liposomes, micro particles, nanoparticles/Nano emulsions [4-8], and in novel route of delivery such as nose-to-brain delivery [9-10] in delivering the therapeutics to human body. Further, active techniques such as iontophoresis [11], ultrasound [12], microneedles [13-17], chemical enhances etc. [18] are also being utilized to improve drug delivery. However, the basis of drug delivery lies in the discovery of the novel molecules through chemistry and structural activity relationship studies, to further enhance the library of molecules. These molecules can further be developed into drug products by fine tuning their physicochemical properties. One such group of compounds that are highly utilized for discovery of novel therapeutics is heterocyclic compounds. Heterocyclic compounds belong to the largest class of medicinal chemistry amongst all. A heterocycle is a ring containing at least one atom that is not carbon. Heterocyclic compounds are of great importance in terms of industrial as well as therapeutic applications. Majority of the existing drugs and new chemical entities include one of the heterocycles in their structure. Much of literature is available on applications of heterocycles in drug delivery and other industrial areas, such as, pesticides, dyes, and plastic industry [19]. Heterocycles are divided into various classes depending on the heteroatom and ring structure. Heterocycles could be three-, four-, five- or six- or more membered rings containing one or more heteroatom such as, Nitrogen (N), Oxygen (O) or Sulfur (S). Nomenclature for these heterocyclic compounds is tabulated in Table 1 Out of these, the best known of the simple heterocyclic compounds are pyridine, pyrrole, furan, and thiophene.
Literature Review
History of heterocycles in medicinal chemistry
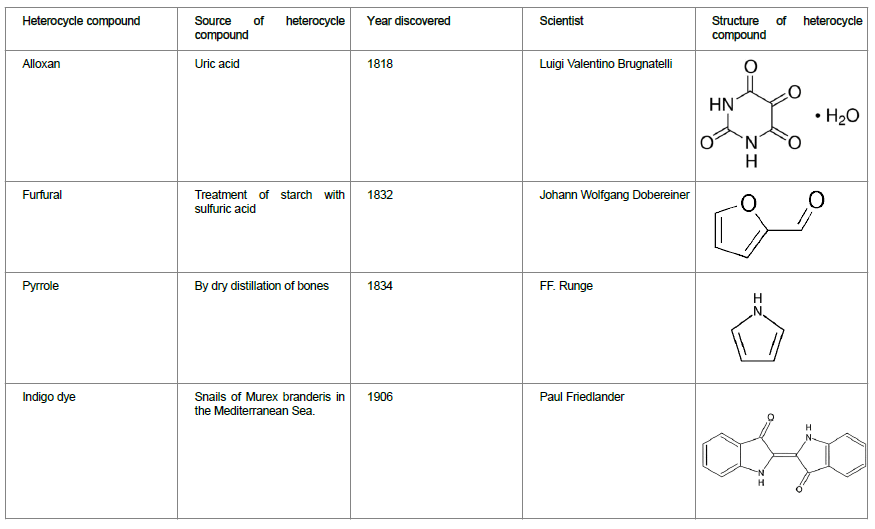
According to literature survey, the history of heterocycles in medicinal chemistry dates to 1800s. Scientists have discovered and reported the key chemicals containing heterocycles that found applications not only in therapeutics but also other industries such as dye/paint industry and oil industry, etc. Table 2 provides a snapshot of various heterocycles that were discovered in early days along with the scientist who first discovered/reported it.
Therapeutic applications of heterocycles
Heterocycles have proven to show the therapeutic activity for various indications, such as fungal infections [20-25], pain/inflammation [26-33], bacterial infections [34-39], neurological complications [40-45], allergies [46,47], cancer [48-56], cardiovascular complications [57-60], etc.
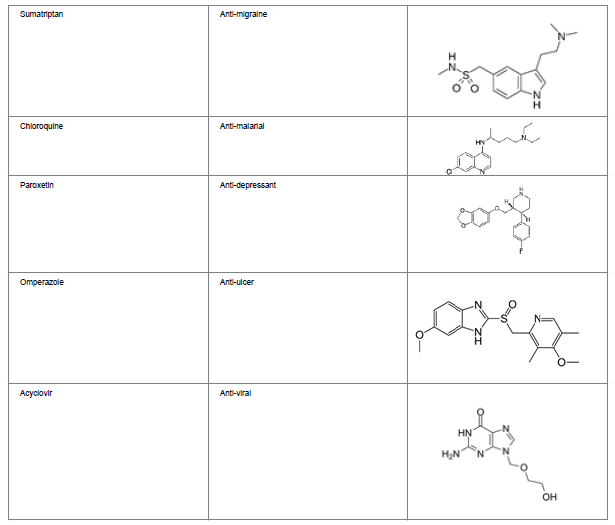
Several key drugs have heterocycle moiety in their structures, which imparts the therapeutic activity to the molecule. Table 3 depicts some of the medicinal drugs containing heterocyclic moiety.
This review focuses on the therapeutic properties and applications of pyrrole in drug delivery and literature spanning the last decade will be covered.
Pyrrole
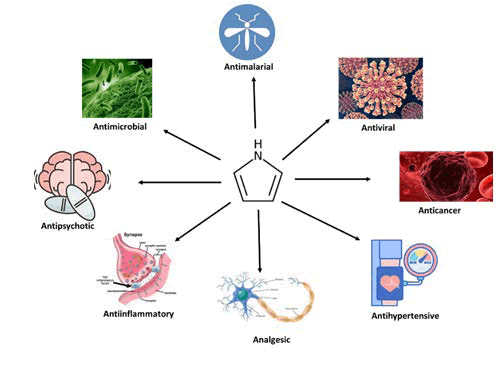
Pyrroles make an important class of organic compounds and were first isolated in 1857 during bone pyrolysis as a by-product and identified as clinically relevant when it was recognized as a structural part of heme and chlorophyll [61]. Apart from these two, pyrroles are also part of other naturally occurring compounds such as porphyrinogens, bile and melanin pigments, and vitamin B12 [62]. Pyrrole and its derivatives are also widely used as intermediates in a variety of industries such as pharmaceuticals, agriculture, dyes/paints, photographic chemicals, perfumes, and other organic compounds. They also find application as a catalyst for polymerization process, corrosion inhibition agents, preservative, solvent for resins and terpenes [63]. In pharmaceuticals, pyrrole and its derivatives have found applications in various indications as depicted in Figure 1.
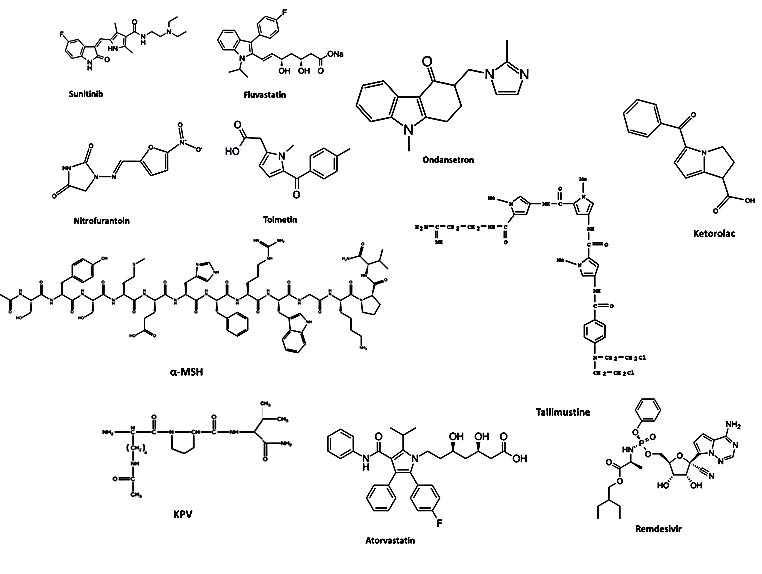
The scientific community got their attention towards pyrrole mainly after the discovery of the blockbuster drug atorvastatin. This followed utilization of pyrrole in many other top selling pharmaceutical drugs such as, Sunitinib, Fluvastatin, Nitrofurantoin, Tolmetin, Ketorolac, Ondansetron, Remdesvir, Tallimustine, etc. [64].
Apart from this, natural hormone, alpha-melanocyte stimulating hormone (α-MSH) which has a pyrrole moiety in its structure and displays an excellent anti-inflammatory property for body’s natural defense [65]. KPV is a α-MSH derivative containing this key structure of pyrrole, and thus without rest of the motif, is still able to elicit the anti-inflammatory effect [14].
Figure 2 shows chemical structures of various commercial drugs as well as natural compounds such as α-MSH and KPV that contain pyrrole moiety. Table 4 discusses the various pharmacological classes, molecular target, and manufacturing companies for the commercially available drugs containing pyrrole.
| Drug name | Pharmacological application/area | Molecular target | Manufacturing company |
|---|---|---|---|
| Ketorolac | Anti-inflammatory | Cyclooxygenase (COX) | Lemmon Company |
| Atorvastatin | Anti-lipidemic | HMG CoA reductase | Pfizer Ireland Pharmaceuticals) |
| Sunitinib | Antitumor | Platelet-Derived Growth Factor (PDGF-Rs) and Vascular Endothelial Growth Factor Receptors (VEGFRs) | Pfizer, Inc. |
| Ondansetron | Antiemetic | Serotonin receptors of the 5-HT3 type | Glaxo Wellcome Inc. |
| Remdesivir | Antiviral | Viral RNA-dependent RNA Polymerase (RdRP) | Gilead Sciences Inc. |
| Fluvastatin | Anti-lipidemic | HMG CoA reductase | Novartis Pharmaceuticals Corporation |
| Tolmetin | Anti-inflammatory | Cyclooxygenase (COX) | Mylan Pharmaceuticals Inc. |
| Nitrofurantoin | Antibacterial | Staphylococcus aureus, Enterococcus species, Escherichia coli |
Casper Pharma LLC. |
Table 4: Representative pyrrole-based drugs on market.
Following sections will discuss in detail on various novel pyrrole derivatives that have been investigated for their applications in some of the key areas of indications.
Anti-inflammatoryactivity
Xustudied2-substituted-1,4,5,6-tetrahydrocyclopentab)pyrroleforskin inflammation and were assessed by 12-O-Tetradecanoylphorbol-13-Acetate (TPA)-induced skin inflammation in mice [66]. Synthesis of various compounds in the series was undertaken, and compound 4 displayed 3.2-fold anti-inflammatory property as compared to celecoxib. Immunohistochemically analysis indicated that compound 4 suppressed the TPA-induced IL-1b, IL-6, TNF-a, and COX-2 by blocking IKK/NF-κB signaling pathway.
Yuan and group synthesized and evaluated 1H-pyrrole-2,5-dione derivatives for treatment of atherosclerosis as cholesterol absorption inhibitors by suppressing the foam cell formation and inflammatory response. Multiple compounds were synthesized and compound 20 seemed to be the most potent inhibitor. Cellular assays for studying inhibitory activity of cholesterol absorption indicated compound 20 exhibiting stronger in vitro cholesterol absorption activities than ezetimibe. Cytotoxicity and partition coefficient studies indicated LC50 values of ezetimibe to be 57.58 µM against HEK293 and 44.20 µM against RAW264.7, respectively. While compound 20 didn’t show any cytotoxicity in both, compounds 22a-c with the best in vitro potency displayed LC50 values of 31.5–47.3 µM against HEK293 and RAW264.7, along with cytotoxicity in both cell lines. Further, polarity evaluation of the compounds showed values ranging between 2 and 4 for compound 20 and others (similar to ezetimibe), except 22a-c that exceeded 5.0. Based on overall performance, compound 20 proved to be the best candidate amongst all the compounds [67].
A series 1,3,4-thiadiazole compounds containing pyrazole and pyrrole nucleus were made in the lab and evaluated for anti-inflammatory properties. Carrageenan-induced acute paw edema in Wistar albino rats was used as a model for studying the biological properties of these compounds. All the tested compounds, 3c, 3d, 4c, 3f, 4d, 3b and 3e displayed significant anti-inflammatory activity (77.27, 75.89, 76.24, 68.55, 63.72, 57.41, 53.05% and 81.00, 80.55, 78.62, 71.45, 68.95, 61.89, 56.32% inhibition in paw edema at 3 h and 5 h respectively) in comparison to reference, indomethacin (74.82 and 80.32% at 3 h and 5 h). Leads with pyrazole nucleus seem to have higher anti-inflammatory activity as compared to the leads with pyrrole nucleus [68].
Claudio synthesized and investigated the bio-pharmacological properties of a class of pyrrole derivatives featuring a small appendage fragment (carbaldehyde, oxime, and nitrile) on the central core. Inhibitory activity towards both cyclooxygenases (COX-1 and COX-2) was evaluated in the murine monocyte/macrophage J774 cell lines for these compounds. Carbaldehyde and nitrile fragments proved to be the most potent derivatives towards J774 COX-2, with IC50 values of 9.5 nM, and 2.2 nM, respectively and displaying 8- and 36-fold higher activity as compared to celecoxib. Oxime and nitrile fragments seemed to have less inhibitory activity towards J774 COX-2 [69].
Harrak and group synthesized and studied the 1,4-benzodioxine and/or pyrrole system for their anti-inflammatory properties. Carrageenan rat paw oedema assay and inhibitory activity of the rat liver 3a-hydroxysteroid dehydrogenase (3a-HSD) were studied with these compounds. Ibuprofen served as reference standard. Ibuprofen at 1000 µM for comparison and its IC50 was 100 µM. Compound 14 and Compound 17 showed enzyme inhibitory activity at IC50 values of 5.8 µM (~ 17 times higher than that of ibuprofen) and 34 µM, respectively. Further, compound 17 showed sustained anti-inflammatory activity in-vivo until after 4 h, whereas ibuprofen activity decreased rapidly [70].
Kumar et al investigated the effect of substituent at the N-1,2 and 5 positions of thiophene [3,2-b] pyrrole as potential anti-inflammatory agents [71]. Standard anti-inflammatory drugs such as Tenidap sodium, Diclofenac sodium and Piroxicam were used as reference. Anti-inflammatory effects were measured using carrageenan paw edema method in male/female Wister rats. None of the leads show any significant activity at a dose of 10 mg/kg, hence was tested at higher dose of 100 mg/kg. The anti-inflammatory properties displayed by the derivatives of bioisosteres were found to be significantly lower than the standards, Tenidap Sodium, Diclofenac Sodium and Piroxicam studied at 10 mg/kg, thus indicating an important role that substituents play at the N-position.
Anti-inflammatory and anti-arthritic effects of 3-(4-hydroxyphenyl)-4-(4-thiomethoxyphenyl)-1H-pyrrole-2,5-dione (1, HMP) were evaluated on LPS-induced RAW 264.7 macrophages and rats with carrageenan-induced paw edema and Adjuvant-Induced Arthritis (AIA). Ibuprofen (50mg/kg) was used as a reference standard. Oral administration of HMP (25 or 50 mg/kg, Per Os (po) reduced paw swelling, and PGE2 release and Myeloperoxidase (MPO) activity in tissue. A dose of 25 or 50 mg/kg, po of HMP significantly reduced paw swelling, arthritic indices and plasma PGE2 concentrations in rat with AIA. The attenuation of PGE2 production by HMP was result of inhibition of Cyclooxygenase-2 (COX-2) activity, and not COX-1 activity, in which HMP suppressed the release and expression of interleukin-1b (IL-1b) and IL-6 in LPS-induced macrophages. In addition, downregulation of the protein and mRNA expressions of Inducible Nitric Oxide Synthase (iNOS) lead to reduction in LPS-induced Nitric Oxide (NO) production. The results suggest HMP as a potential therapeutic agent for further evaluation [72].
Analgesic activity
Redzicka and group [73] synthesized a series of pyrrolo [3,4- c]pyrrole Mannich bases (7a-n) and evaluated for analgesic activity using COX-1/COX-2 inhibition activities and molecular docking study. Meloxicam (COX-2 inhibitor) was used as a reference compound. All the synthesized compounds (except for the 7e) suppressed both the COX-1 and the COX-2 enzymes. For COX-1 enzyme, showed IC50 values for the synthesized compounds were 7i-7k (99.6 µM, 104.3 µM, 94.3 µM, respectively), 7f (104.4 µM) and 7h (103.0 µM) as compared to Meloxicam (85.8 µM), whereas for COX-2 enzyme, all the compounds displayed higher (ranging 42.5 µM–53.9 µM) activity than Meloxicam (57.3 µM). Further, in the conducted test, compounds 7n, 7m, 7d proved to be most effective (the most selective towards COX-2) with a selectivity ratio (COX-2/COX-1) of 0.24, 0.26 and 0.28, respectively than Meloxicam, (selectivity ratio of 0.55), thus displaying nearly twice of selectivity for COX-2 enzyme.
WieslawsynthesizedaseriesofN2-{2-[4-aryl(benzyl)-1-piperazinyl(piperidinyl)] ethyl}pyrrolo [3,4-d]pyridazinones 4 and related derivatives 5 as potential analgesic agents. Phenylbenzoquinone induced writhing and hot plate tests in mice along with radioligand binding assay were done to evaluate the analgesic properties of these compounds. Writhing test indicated that all the synthesized compounds were more active than acetylsalicylic acid (reference standard, ED50 for ASA is 39.15 mg/kg) with ED50 values ranging from 0.04 to 11 mg/kg (i.p.). Hot plate test showed that only three compounds 4c, e,f displayed the analgesic activity at a dose 3-5 times higher than morphine (ED50-3.39 mg/kg). Only compound 4f exhibited affinity for the m-opioid receptors similar to that of Tramadol, as suggested by Radioligand binding assay results [74].
Same group of researchers also synthesized N-(substituted-ethyl) pyrrole-3,4-dicarboximides and tested for analgesic properties [75]. Phenylbenzoquinone-induced writhing test showed that all the compounds, except for 3d (ED50=47.7), were ~1.5–5 times more active than Acetylsalicylic acid (reference standard, ED50=39.15) and ~3–10 times less active than morphine. Compound 4c displayed most potent effect (ED50=7.6 mg/kg), which was significantly active up to a dose of 1/320 LD50.
Another group of researchers extended their previous work and decorated the central framework of previously synthesized 2-phenyl-1H-pyrrole-3-carboxamide with diverse alicyclic amines at the 3-carboxamide fragment [76]. Neuropathic pain alleviating properties of compound 33 was evaluated in Sprague-Dawley male rats subjected to unilateral spinal nerve (L5) ligation. Intra-peritoneal (i.p.) injection of either compound 33 (5 and 25 μmol/kg), PZ-1388 (25 µmol/kg), or vehicle (water for injection, 5 mL/kg) was administered. Compound 33 (25 µmol/kg) significantly improved the 50% threshold at 30 to 90 min after i.p. injection and reached a maximal level at 30–60 min after injection and showed higher results than PZ-1388 (25 µmol/kg), resulting in a total abolition of SNL-induced tactile allodynia.
Burgart investigated the effect of methylation of 3-trifluoromethyl-1H-pyrazol-5-ol on the analgesic activity of the parent compound [77]. Mono-Me-substituted N1 - and O-isomers as well N1, N2-, N1, O- and N2, O-disubstituted isomers were synthesized. Analgesic activity using hot plate test was evaluated in rats at the dose of 15 mg/kg for compounds 1-4, 6, 8 with diclofenac (latent period prolongation, % (at the dose of 15 mg/kg) at 60 min. was 58.8 ± 8.5) being the reference drug. Except MeO-pyrazole 2, all the tested compounds elicited anti-nociceptive properties higher than the reference (ranging from 17.7 to 52.6%). Further, they were more active at 120 min after the administration as compared to activity at 60 min.
Anti-bacterial/Anti-microbial
Adamovich and group studied the antibacterial activity of new silatrane pyrrole-2-carboxamide hybrids for their antibacterial properties [78]. These compounds, 3a-d was also evaluated for their drug-likeness properties, and seems to pass Lipinski’s rules, with low skin permeation but high water-solubility and gastrointestinal absorption indicating a high bioavailability. Broth microdilution method was used to determine the antimicrobial activity and Minimal Inhibitory Concentration (MIC) for these compounds. The microbes tested were gram-positive Enterococcus durans B-603, Bacillus subtilis B-407 and gram-negative Escherichia coli B-1238. Control drug used was gentamicin. For E. durans, only compound 3a (3.1 µg/mL) seem to show an MIC value much lower than gentamicin (25 µg/mL), while other compounds had MIC values above 50 µg/mL. For B. subtilis too, compound 3a (6.2 µg/mL) seem to be more effective than gentamicin (50 µg/mL), while other compounds showed MIC values above 500 µg/mL. For E. coli, compound 3d had much lower MIC values (62.5 µg/mL) than gentamicin (100 µg/mL) while other compounds showed MIC values above 125 µg/mL.
Moorthy reported a one-step, microwave-assisted synthesis of polypyrrole grafted with chitosan (PPy-g-CS) and poly (pyrrole-N-(1-naphthyl) ethylenediamine, a Copolymer, (COP), using Carbon Dots(C-Dots) as initiators [79]. The compound was tested for its antibacterial activities against E. coli and S. aureus. PPy-g-CS completely inhibited the growth of E. coli as supported by Scanning Electron Microscopy (SEM). A delayed effect of COP on the growth of E. coli was observed, however, the antibacterial effect of COP against E. coli was minimal as compared to PPy-g-CS. The interaction of positively charged polymers with the negatively charged microbial cell surface lead to the aggregation and cell death, as indicated by SEM. Similar behavior was observed against S. aureus, where PPy-g-CS completely abolished the bacteria after 1 h, while COP was ineffective for 6 h, thereafter leading to complete bacterial death at 24 h.
Novel isoxazolo [4,3-e] indazole derivatives (5a–5g) were synthesized using novel Pyrrole-2-carboxylic acid functionalized magnetic Fe3O4 as novel nanocatalyst. For bacterial activity assessment, these compounds were tested against four bacterial species for growth of inhibition using Kirby–Bauer disc diffusion method. Examine organisms such as E. coli PTCC 1330, P. aeruginosa PTCC 1074, S. aureus ATCC 35923, and B. subtilis PTCC1023. Gentamicin (10 μg/disc) and chloramphenicol (30 μg/disc) as positive control and DMSO (20 mm3/disc) as negative control. Compounds 5a–5g displayed moderate to good growth inhibitory effect against most microorganisms, such as E.coli (ranging 7.0 0 ± 0.7 mm to 13.0 ± 0.7 mm as compared to gentamicin with 19.6 ± 1.1 mm and chloramphenicol with 20.7 ± 1.5 mm), P. aeruginosa (ranging from 11.5 ± 0.7 mm to 8.5 ± 0.7 mm as compared to gentamicin with 15.6 ± 0.5 mm), S. aureus (ranging from 7.5 ± 0.7 mm to 12.0 ± 1.4 mm as compared to gentamicin with 20.3 ± 1.5 mm and chloramphenicol with 21.7 ± 0.6 mm) and B. subtilis (ranging from 8.5 ± 0.7 mm to 13.5 ± 0.7 mm in comparison to gentamicin with 26.0 ± 1.7 mm and chloramphenicol with 22.3 ± 1.2 mm) except for compounds 5f that showed no activity against P.aeruginosa and 5g against P. aeruginosa and B. subtilis. Amongst all, highest antibacterial activity was observed against B. subtilis for these compounds [80]. Poonam and group [81] synthesized and evaluated Ethyl-4-{[-(1-(2-(4-nitrobenzoyl) hydrazono) ethyl]}-3,5-dimethyl-1H-pyrrole-2-carboxylate. Ethyl-4-acetyl-3,5-dimetyl-1H-pyrrole-2-carboxylate (EADMPC), Para-Nitrobenzohydrazide (PNBHz),ethyl-4-{[-(1-(2-(4-nitrobenzoyl)hydrazono)ethyl]}-3,5-dimethyl-1H-pyrrole-2-carboxylate ENBHEDPC) were evaluated for the antibacterial, antifungal, and antituberculosis properties against S. aureus, C. albicans and H37Rv using MIC values. For both S.aureus and C.albicans, EADMPC, PNBHz and ENBHEDPC seem to show an 8-, 4-, and 2-fold higher MIC values as compared to ceftriaxone and miconazole. For antituberculosis activity against H37Rv, EADMPC displayed almost a 14-fold higher MIC value as compared to ethambutol, while rest of the compounds did not show any significant difference in the activity.
Antiviral
A series of phenyl-1H-pyrrole-carboxamide entry inhibitors (NBD compounds) were synthesized using structure-based lead optimization for targeting at HIV-1 gp120 [82]. HIV-1 mediated cell-cell fusion inhibition assay for the three lead compounds (NBD-14136, NBD-14168, NBD-14189) was performed and compared with HIV-1 entry inhibitor, NBD-556 as the control. Results indicated that NBD-14189 prevented HIV-1 mediated cell-cell fusion with an IC50 of 9.4 ± 0.9 µM which is similar to the IC50 we detected for NBD-556 (9.1 ± 0.8 µM). However, NBD-14168 (~15 µM) and NBD-14136 (~28 µM) were active at slightly higher concentration than NBD-556 and NBD-14189. Test if infectivity of Cf2 Th-CCR5 cells by CD4-dependent HIV-1ADA indicated CC50 values of NBD-556: >60 µM; NBD-14136: 37.7 ± 1.7 µM; NBD-14168: >51 µM and NBD-14189: 34 ± 1 µM, thus indicating an enhanced HIV-1 infectivity. Further, neutralization activity of these NBD compounds against a panel of HIV-1 Env pseudo viruses (selection of 56 HIV-1 clones of clinical isolates of different subtype including primary and transmitted and early founder HIV-1 isolates (NIH#11563 and 11578), and 13 recombinant HIV-1 clones.) was done. NBD-14107 was used as reference standard. Overall mean of the IC50s for NBD-14107 was 0.5 ± 0.02 µM (IC50s in the range of 0.27 - 0.89 µM) with Selectivity Index (SI) of 79 whereas the overall mean of the IC50s determined for NBD-14136 was 0.29 ± 0.01 µM (IC50s in the range of 0.14 - 0.54 µM) and the calculated SI was 146.2, an increase of ~1.7 and 1.8 folds, respectively. NBD-14168 showed a 2.8- fold higher value in the overall mean of the IC50s relative to NBD-14107 (0.18 ± 0.008 µM with the IC50s in the range of 0.094 - 0.35 µM) and a ~3 fold higher value of the SI. Finally, the overall mean of the IC50s determined for NBD-14189 was 0.11 ± 0.004 µM (IC50s in the range of 0.063 - 0.18 µM) indicating a 4.5-fold increase as compared to NBD-14107, with 2.5-fold higher SI.
Antiviral Polyamides (AVPs) were synthesized from method described in the literature [83]. The AVPs, PA1 and PA25 were tested for their antiviral activities against HPV16, SV40, and BKPyV at concentrations ranging from 1 nM to 10 µM. Both AVPs reduced viral copy numbers to very low levels (Ct values of 35–40), specifically at highest doses while eliciting an IC50's of 7 nM and 437 nM against BKPyV-Dun, and 1 nM and 62 nM against BKPyV-Tu, for PA25 and PA1, respectively. The IC50 values for PA1 against HPV16 and SV40 were 100 nM and 218 nM whereas for PA25, the values were 218 nM and 32 nM, respectively. The mechanism is hypothesized to be bought by alteration and subsequent elimination due to AVP binding to the circular ~7.9 kb HPV genome, which is otherwise protected from cell surveillance mechanisms. In another study reported by same group of scientists, these novel polyamide leads were studied and found to control and decrease the HPV episome content of keratinocytes for three high-risk viruses, HPV16, HPV18, and HPV31[84]. Q-PCR results of HPV16 levels indicated that the single treatment with PA1 caused a dose-dependent decrease in HPV16 episome levels within the W12E cell-derived epithelium, with an 80% loss of viral DNA with highest dose of 100 µM, and 35% and 65% losses of HPV16 DNA from the rafts, with 10 and 50 µM doses, respectively. Polyamide (PA25) potently suppressed HPV18 episomes in Ker 4–18 cell monolayers with an IC50 of 56 ± 5 nM. Q-PCR confirmed that both the 1X and 2X treatments resulted in significant reductions of HPV18 episome levels of approximately 70% and 80%, respectively, with 1mM of PA25.
Mosaad and group [85] synthesized a series of novel pyrrolo [2,3-d] pyrimidines and pyrrolo [3,2-e][1,2,4] triazolo [4,3-c]pyrimidine derivatives with antiviral activity against Hepatitis C Virus (HCV). Five derivatives (25a-e) out of fifty-seven compounds showed considerable antiviral activity, causing a reduction in the virus titer of 90, 76.7, 73.3, 70, and 63.3%, respectively. SAR studies indicated that the 5,6-diphenyl-7-(3- or 4-chlorophenyl)pyrrolo[2,3-d]pyrimidines, suitably substituted at position 4 with a chlorine or an arylamino group (25a-c), may prove to be useful scaffold for anti-HCV agents.
Same group of researchers further reported the evaluation of these compounds against viral strains, Rotavirus Wa and Coxsackievirus B4 by using molecular docking against the homology models of viral polymerase enzymes of these viruses. The selected compounds, 5d, 7n and 14c showed the highest activity against Rotavirus Wa strain, with 80% reduction in the viral titer, whereas compounds 2d, 5d, 7b and 14c showed the highest activity against Coxsackievirus B4, with 90% reduction in the viral titer. This indicates that pyrrolo [2,3‑d]pyrimidine analogues exhibited significant anti-viral activity against the gastro enteric viruses [86].
Travis et al evaluated GS-5734, the single Sp isomer of the 2-ethylbutyl l-alaninate phosphoramidate prodrug, which effectively bypasses the rate-limiting first phosphorylation step of the Nuc (1-cyano-substituted adenine C-nucleoside ribose analogue that exhibits antiviral activity against several RNA viruses). GS-5734 was investigated for its antiviral activity against Ebola virus. While the parent Nuc displayed the half-maximum effective concentration, EC50 values of 0.77 to >20 μM, GS-5734 suppressed EBOV replication in multiple relevant human cell types including primary macrophages and human endothelial cells with EC50 values of 0.06 to 0.14 μM [87]. This molecule was further evaluated in a focused screening and lead optimization effort as 4b (GS-5734) with anti-EBOV EC50=86 nM in macrophages as the clinical candidate [88].
Anti-proliferative
Mostafa et al reported synthesis and evaluation of novel pyrrole and pyrrolo [2,3-d] pyrimidine derivatives bearing sulfonamide moiety as anti-proliferative agents. These derivatives were tested in human liver and breast (HEPG2 and MCF7) cell lines using doxorubicin as reference drug. Evaluating the pyrrole derivatives 5, 6, 11–18 in the liver cell line (HEPG2), 5 and 6 showed increased activity (IC50 =5.36 and 5.3 µM) when compared to compounds 3 and 4 (IC50=6.78 and 6.75 µM) and they were found to be nearly as active as doxorubicin (IC50=5.23 µM). A further increase in the anticancer activity observed in compounds 14 (IC50=3.49 µM), 15 (IC50=3.75 µM), and 16 (IC50=3.75 µM) upon substitution on the amino group at position 2, thus proving as the most potent compounds in this series. Among the pyrimidine derivatives namely 7–10, 19–24, 19 (IC50=3.39 µM), 20 (IC50=3.15 µM), and 21 (IC50=3.49 µM) showed highest potency in the series. When testing these compounds in the breast cell line (MCF7), compounds 5 and 6 belonging to O-amino carbonitriles displayed most potency (IC50=3.49 and 4.6 µM, respectively) and higher than doxorubicin (IC50=3.22 µM). In the pyrimidine series of derivatives, 22 (IC50=3.9 µM), 23 (IC50=3.12 µM), and 24 (IC50=3.8 µM) were found to be most effective as compared to the reference drug [89].
Ultrasound-assisted bismuth nitrate-induced green synthesis of novel pyrrole derivatives were reported by Debasish and group [90]. N-substituted pyrroles were synthesized using 2,5-dimethoxytetrahydrofuran (1) and various amines in the presence of catalytic amounts (5 mol%) of bismuth nitrate pentahydrate under solvent-free conditions. To evaluate the anti-proliferative activity, liver cancer cell lines (HEPG2 and HePA1-6), colon cancer cell lines (HT-29 and Caco-2), a cervical cancer cell line (HeLa) and NIH3T3 cells were used. Compounds 5-(1H-pyrrol-1-yl)-1,10-phenanthroline (9) and 1-(phenanthren-2-yl)-1H-pyrrole (10) showed good anticancer activity as compared to cisplatin in certain cancer cell lines. Compound 9 showed an IC50 value of 3.0 ± 1.6 µM in HEPG2 cells as compared to cisplatin (7.0 µM), 3.4 ± 0.4 µM in Hepal1-6 cells than cisplatin (4.0 µM), and in NIH3T3, 2.1 ± 1.3 µM as compared to cisplatin (8.5 µM). Compound 10 on the other hand showed high potency only in Hepal1-6 cells with an IC50 value of 3.4 ± 0.4 µM as compared to cisplatin (4.0 µM).
Discussion
Nishant and group investigated a molecular hybridization approach to synthesize a series of 2,4-dimethyl-1H-pyrrole-3-carboxamide derivatives bearing benzimidazole moiety for their anticancer activities. NCI-60 cell lines were used to test the anti-proliferative activity in leukemia, melanoma, lung, colon, CNS, ovarian, renal, and prostate and breast cancer at a single dose of 10 µM. While most of the compounds did show good anticancer activities in these cell lines, as indicated by the Growth Percentages (GP), 5-(1H-benzo[d]imidazol-2-yl)-N-(1-cyclohexylethyl)-2,4-dimethyl-1H-pyrrole-3-carboxamide, 8f displayed promising anticancer activity against many cell lines, such as MDA-MB-435 cancer cell line of melanoma with 37.54% GP, MDA-MB-468 of breast cancer (59.76% GP), K-562 of leukemia cancer (68.68% GP), SR of leukemia cancer (68.98% GP), HCT-15 of colon cancer (69.0% GP), and MDA-MB-231/ATCC of breast cancer (70.28% GP). Another most potent compound was 8n that displayed MDA-MB-468 (70.24% GP) and T-47D (72.93% GP) for breast cancer cell lines [91].
A series of Aroyl-Diaryl-Pyrrole (ARDAP) derivatives were synthesized by introducing one or two amino phenyl groups at positions 1 and 4 in pyrrole structure [92]. These derivatives were evaluated for their utility in Glioblastoma Multiform (GBM) and Chronic Myeloid Leukemia (CML). Various cancer cell lines such as, U87MG and U343MG cell lines, KBM5-wt, KBM5-T315I and KU812 leukemia cell lines, and Human colon cancer SW480 and SW6220 cell lines were evaluated. Further inhibition of Inhibition of human topoisomerases I and II was also investigated for these compounds. CSA4 was used as a reference compound yielded with IC50's in the 0.54 - 0.73 µM range. Presence of the thiophen-3-yl (7) or pyridin-4-yl (22, 25) group at position 4 of the pyrrole led to potent compounds such as 7, 22 and 25 against MCF-7 cell lines with IC50 values of 4.0, 9.0 and 9.0 nM, respectively. Several other compounds in the series also showed good anti-proliferative activity such as compounds 5, 9, 10, 12-14 and 21 with IC50 values in the 10-20 nM range, and compounds 3, 4, 8, 15, 20 and 24 with IC50 values in the 21-50 nM range, in the MCF-7 cell lines. Presence of 1-(3-methylphenyl) group improved the potency of the compounds with IC50's close to 20 nM (ARDAPs 3-6). 1-(4-Fluorophenyl) derivatives with a 4-thiophenyl or 4-furanyl group (12-14, IC50 of 15-17 nM) or pyridin-4-yl ring (compound 25, IC50 of 9.0 nM) also showed high potency against the cell lines. Alejandro and group reported two series of novel chiral hexahydro-2H-furo[3,2- b]pyrroles, 4-(7,8-dimethoxyquinazolin-4-yl) termed as series A and 4-(6,7- dimethoxyquinazolin-4-yl) termed as series B. These compounds were evaluated for their inhibitory activity against phosphodiesterase 1 (PDE1) and phosphodiesterase 4 (PDE4) enzyme inhibitors as well as on cell proliferation in A375 melanoma and 3T3 fibroblast cells in vitro. It was observed that hexahydro-2H-furo[3,2-b]pyrroles were highly effective against PDE1 isoenzyme than against PDE4 isoenzyme, and 18A was the most potent compound with an IC50 of 4.7 µM against PDE1A, 2.8 µM against PDE1B, and 0.6 µM against PDE1C. It also displayed a potent inhibitory activity in A375 melanoma cells at 1 μM, and even higher activity at 10 μM, and highest potency with overall lower survival at 50 μM [93-102].
Conclusion
Evaluation of heterocycles and their application in therapeutic drug delivery is ongoing since 1800s and is still of great interest. Based on the numerous research publications in the literature along with the existing blockbuster drugs on market containing pyrrole, it indicates that pyrrole is an important therapeutic moiety amongst the heterocycle family. While the discovery of new molecules containing pyrrole group is continuing, it is also crucial to conduct in-vivo studies of these molecules to further understand the mechanistic and pharmacological behavior for the potential lead molecules. Further, evaluating the safety and toxicity of these molecules in animals and humans is of utmost importance to further test their viability as potential drug molecule. Optimizing the molecule to fit into Lipinski’s rule as well as classifying under BCS category is warranted and would be critical in turning these leads into drug-like molecules ready for pharmaceutical processing. Current landscape of research towards pyrrole molecules provides a path for success for development of future blockbuster commercial products.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Alqahtani MS, Kazi M, Alsenaidy MA, Ahmad MZ (2021) Advances in Oral Drug Delivery. Front Pharmacol 12:1-21.
- Homayun B, Lin X, Choi HJ (2019) Challenges and Recent Progress in Oral Drug Delivery Systems for Biopharmaceuticals. Pharmaceutics 11:3-29.
- Pawar K, Render D, Rangari V, Lee Y, Babu RJ (2018) Evaluation of non-crystalline cellulose as a novel excipient in solid dose products. Drug Dev Ind Pharm 44:1512-1519.
- Santos AMD, Carvalho SG, Meneguin AB, Sabio RM, Gremiao MPD, et al. (2021) Oral delivery of micro/Nano particulate systems based on natural polysaccharides for intestinal diseases therapy: Challenges, advances and future perspectives. J Control Release 334:353-366.
- Render D, Rangari V, Samuel T, Ramapuram J, Jeelani S, et al. (2013) Development of drug delivery system using bio-based calcium carbonate nanoparticles. Technical Proceedings of the 2013 NSTI Nanotechnology Conference and Expo, NSTI-Nanotech 3:312-314.
- Pawar KR, Babu RJ (2014) Lipid materials for topical and transdermal delivery of Nano emulsions. Rev. Ther Drug Carr Syst 31:429-458.
- Pawar KR, Babu JR (2010) Polymeric and lipid-based materials for topical nanoparticle delivery systems. Crit Rev Ther Drug Carr Syst 27:419-459.
- Pawar K (2018) Recent Developments on Conquering Zika. J Pharm Drug Deliv Res 6:1-2.
- Babu R, Dayal PP, Pawar K, Singh M (2011) Nose-to-brain transport of melatonin from polymer gel suspensions: a microdialysis study in rats. J Drug Target 19:731-740.
- Pires PC, Fazendeiro AC, Rodrigues M, Alves G, Santos AO (2021) Nose-to-brain delivery of phenytoin and its hydrophilic prodrug fosphenytoin combined in a micro emulsion-formulation development and in vivo pharmacokinetics. Eur J Pharm Sci 164:1-10.
- Li Y, Yang J, Zheng Y, Ye Rui, Liu B, et al. (2021) Iontophoresis-driven porous micro needle array patch for active transdermal drug delivery. Acta Biomater 121:349-358.
- Daftardar S, Neupane R, Sai HSB, Renukuntla J, Tiwari AK (2019) Advances in Ultrasound Mediated Transdermal Drug Delivery. Curr Pharm Des 25:413-423.
- Pawar K (2017) Microneedles-Based Devices: Regulatory Insights. J Pharm Drug Deliv Res 6:1-2.
- Pawar KR, Kolli CS, Rangari VK, Babu RJ (2017)Transdermal iontophoretic delivery of lysine-proline-valine (KPV) peptide across microporated human skin. J Pharm Sci 106:1814-1820.
- Pawar KR, Smith F, Rangari VK, Babu RJ (2013) Effect of lipophilicity on microneedle-mediated iontophoretic transdermal delivery across human skin in vitro. J Pharm Sci 102:3784-3791.
- Pawar KR, Mulabagal V, Smith F, Kolli CS, Rangari V, et al. (2015) Stability‐indicating HPLC assay for Lysine–Proline–Valine (KPV) in aqueous solutions and skin homogenates. Biomed Chromatogr 29:716-721.
- Waghule T, Singhavi G, Dubey SK, Pandely MM, Gupta G, et al. (2019) Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed Pharmacother 109:1249-1258.
- Ng KW (2018) Penetration Enhancement of Topical Formulations. Pharmaceutics 10:1-3.
- Ruirui L, Bo L, Hongpeng Z, Cheng-Wei J, Ying Q, et al. (2021) A ring expansion strategy towards diverse azaheterocycles. Nature Chemistry 13:1006-1016.
- Xuesong W, Sumei G, Jian Y, Yang G, Ling W, et al. (2016) Synthesis and antifungal activity evaluation of new heterocycle containing amide derivatives. Nat Prod Res 30:682-688.
- Pablo RD, Martin GV, Eduardo AC, Michael F, Julio C (2007) QSAR analysis for heterocyclic antifungals. Bioorg Med Chem 15:2680-2689
- Nívea PS, Caroline ML, Cleudiomar IL, Paulo JSB, Ludmila MB, et al. (2017) Heterocycle thiazole compounds exhibit antifungal activity through increase in the production of reactive oxygen species in the Cryptococcus Neoformans-Cryptococcus Gattii species complex. Antimicrob Agents Chemother 61:1-8
- Xuesong W, Sumei G, Jian Y, Yang G, Ling W, et al. (2016) Synthesis and antifungal activity evaluation of new heterocycle containing amide derivatives. Nat Prod Res 30:682-688.
- Zhinan G, Min L, Qin L, Hui X (2015) Synthesis of heterocycle-attached ethylidenebenzenesulfonohydrazones as antifungal agents. Bio org Med Chem Lett 25:5092-5096.
- Zhigan J, YanW, Wenya W, Shengzheng W, Bo X, et al. (2013) Discovery of highly potent triazole antifungal derivatives by heterocycle-benzene bioisosteric replacement. Eur J Med Chem 64:16-22.
- Abdel-Hafez SH (2008) Selenium containing heterocycles: synthesis, anti-inflammatory, analgesic and anti-microbial activities of some new 4-cyanopyridazine-3(2H) selenone derivatives. Eur J Med Chem 43:1971-1977.
- Sobhi MG, Zeinab AM, Hatem MG, Mohamed MA (2017) synthesis of some novel heterocycles bearing thiadiazoles as potent anti-inflammatory and analgesic agents. J Heterocycl Chem 54:2708-2716.
- Kinga S, Andrzej M, Tadeusz L (2013) Nitrogen, oxygen or sulphur containing heterocyclic compounds as analgesic drugs used as modulators of the nitroxidative stress. Mini Rev Med Chem 13:335-352.
- Ahmed AOA, Gehad AAJ, Mohamed SKY (2019) Novel heterocyclic hybrids Based on 2-Pyrazoline: synthesis and assessment of anti-inflammatory and analgesic activities. Curr Org Synth 16:921-930.
- Omar Abd El-Fattah MF, Manal MA, Mogeda EH, Salwa MN (2019) Synthesis of novel tetrahydronaphthalen-2-yl heterocycles for analgesic, anti-inflammatory and antipyretic evaluation. Acta Pol Pharm 66:259-270.
- Cohen N, Banner BL, Lopresti RJ (1978) Analgesic activity of novel Spiro heterocycles. 2-Amino-7-oxa-3-thia-1-azaspiro[5,5]undec-1-enes and related compounds. J Med Chem 21:895-900.
- Leite LF, Ramos MN, da Silva JB, Miranda AL, Fraga CA, et al. (1999) Synthesis and analgesic profile of novel N-containing heterocycle derivatives: arylidene 3-phenyl-1,2,4-oxadiazole-5-carbohydrazide. Farmaco 54:747-757.
- Indumathi S, Karthikeyan R, Nasser AJA, Idhayadhulla A, Kumar RS (2015) Anticonvulsant, analgesic and anti-inflammatory activities of some novel pyrrole and 1,4-dihydropyridine derivatives. J Chem Pharm Res 7:434-440.
- Cai-Feng D, Hong-Xia M, Jing Y, Xu-Jie Q, Guy S S N, et al. (2018) Antibacterial Indole Alkaloids with Complex Heterocycles from Voacanga Africana; Synthesis, characterization and antibacterial activity of novel heterocycle, coumacine, and two of its derivatives. Org Lett 20:2702-2706.
- Yasser FM (2018) Synthesis, characterization and antibacterial activity of novel heterocycle, coumacine, and two of its derivatives. Saudi Pharm J 26:870-875.
- Arpad MR, Mihaela B, Lavinia LR, Ileana CF, Catalin M, et al. (2020) Copper(ii) complexes with mixed heterocycle ligands as promising antibacterial and antitumor species. Molecules 25:1-22.
- Vladimirova SP, Danalev DL, Marinkova DA, Raykova RN, Manova DS, et al. (2017) Synthesis and antibacterial activity of amino acids modified with specifically substituted pyrrole heterocycle. Bulg Chem Commun 49:86-89.
- Vladimir NK, Olga VP, Stepan VV, Ludmila VI (2020) Synthesis, redox properties and antibacterial activity of hindered phenols linked to heterocycles. Molecules 25:1-12.
- Pietro B, Turchetti B, Ieri F, Goretti M, Branda E, et al. (2007) Catechins and Proanthocyanidins: naturally occurring o-heterocycles with antimicrobial activity. Bioactive Heterocycles 10:239-263.
- Chandra BM, Shikha K, ManishaT (2015) Thiazole: A promising heterocycle for the development of potent CNS active agents. Eur J Med Chem 92:1-34.
- Gourav G, Rajarshi N, Rohit B, Md Jawaid A (2020) Synthetic and therapeutic perspectives of nitrogen containing heterocycles as anti-convulsants. Bioorg Med Chem 28:1-59
- Michal M, Lucie S, Wim D, Martina H, Michaela R, et al. (2021) Heterocyclic cathinones as inhibitors of kynurenine aminotransferase ii—design, synthesis, and evaluation. Pharmaceuticals 14:2-20.
- Khadidja B, Solène G, Emmanuelle L, Ludovic P, et al. (2021) Design and microwave synthesis of new (5Z) 5-Arylidene-2-thioxo-1,3-thiazolinidin-4-one and (5Z) 2-Amino-5-arylidene-1,3-thiazol-4(5H)-one as new inhibitors of protein kinase DYRK1A. Pharmaceuticals 14:1-31.
- Zahira T, Mohammed L, Jabrane J, Ismail H, Mümin AE, et al. (2021) Pyrido[2,10:2,3]imidazo[4,5-c]isoquinolin-5-amines as potential cytotoxic agents against human neuroblastoma. Pharmaceuticals 14:1-15.
- Chrisna MG, Legoabe TB, Janse van L, Rensburg HD (2021) Exploration of chalcones and related heterocycle compounds as ligands of adenosine receptors: therapeutics development. Mol Divers 26:1779-1821
- Venkatachalam TK, Qazi S, Samuel P, Uckun FM (2003) Substituted heterocyclic thiourea compounds as a new class of anti-allergic agents inhibiting IgE/Fc epsilon RI receptor mediated mast cell leukotriene release. Bioorg Med Chem 11:1095-105
- Kaur R, Kaur K, Arora R, Saini B, Arora S (2021) Natural fused heterocyclic flavonoids: potent candidates as anti- inflammatory and anti-allergic agents in the treatment of asthma. Curr Bioact Compd 17:28-40.
- Mohamed AA, Aya AES, Mohamed A, Hanem A, Marwa MAA, et al. (2020) Synthesis of novel naphthalene-heterocycle hybrids with potent antitumor, anti-inflammatory and antituberculosis activities. RSC Adv 10:42998-43009.
- Wen L, Jinyang Z, Min W, Ru D, Xin Z, et al. (2022) Pyrimidine-fused di nitrogenous penta-heterocycles as a privileged scaffold for anti-cancer drug discovery. Curr Top Med Chem 22:1873-4294.
- Sonakshi SA (2015) Comprehensive review on recent advances in synthesis and pharmacotherapeutic potential of benzothiazoles. Anti-Inflamm. Anti-Allergy Agents Med 14:98-112.
- Feng W, Yongfang Y, Hai-liang Z and Yinghui Z (2021) Nitrogen-containing heterocycle: A privileged scaffold for marketed drugs. Bio org Chem 21:439-441.
- Lu M, Dongyu M, Jung JL, Tao L, Yu C, et al. (2021) Synthesis and biological evaluation of heterocyclic ring-fused dammarane-type ginsenoside derivatives as potential anti-tumour agents. Bio org Chem 116:1-11.
- Pedro M, Joao J, Sofia S, Luis RR, Catarina RR, et al. (2015) Recent advances of heterocycle based anticancer hybrids. Molecules 20:16852–16891.
- Damanpreet KL, Rajwinder K, Rashmi A, Balraj S, Sandeep A (2020) Nitrogen Containing Heterocycles as Anticancer Agents: An Overview. Anticancer Agents Med Chem 20:2150-2168.
- Stephane BL (2019) Minireview: N-heterocyclic carbene platinum complexes: a big step forward for effective anti-tumor compounds. Eur J Inorg Chem 1-13.
- Renjing Y, Chunchun D, Ting C, Guanghui W, Xin J, et al. (2022) Synthesis and anti-hepatoma activities of u12 derivatives arresting g0/g1 phase and inducing apoptosis by pi3k/akt/mtor pathway. Pharmaceuticals 15:1-15.
- Glassman AH, Preud'homme XA (1993) Review of the cardiovascular effects of heterocyclic antidepressants. J Clin Psychiatry 54:16–22.
- Ram WS (2021) Novel heterocyclic compounds as delta-5-desaturase inhibitors for treating metabolic and cardiovascular diseases. CS Med Chem Lett 12:1218–1219.
- Xiu EF, Qin JW, Jie G, Shu RB, Qing SL (2017) Synthesis of novel nitrogen-containing heterocycle bromophenols and their interaction with keap1 protein by molecular docking. Molecules 22:1-22.
- Lorena C, Mircea VM, Octavia R, Ștefan CV, Andreea LR, et al. (2020) The effects of flavonoids in cardiovascular diseases. Molecules 25:1-18.
- Veronica E, Mercedes V, Menéndez JC (2010) Multicomponent reactions for the synthesis of pyrroles. Chem Soc Rev 39:4402-4421.
- Mohammad A (2017) A mini review: biological significances of nitrogen hetero atom containing heterocyclic compounds. Bio org Chem 2:146-152.
[Crossref]
- Ramadeep K, Vibhuti R, Vikrant A, Yagyesh K, Debabrata K, et al. (2017) Recent synthetic and medicinal perspectives of pyrroles: An overview. J Pharm Chem Chem Sci 1:17-32.
- Kumar TB, Sumantha CH, Vaishaly S, Srinivasa MR, Chandra Sekhar KB, et al. (2012) Pd-mediated functionalization of polysubstituted pyrroles: Their evaluation as potential inhibitors of PDE4. Bio org Med Chem Lett 22:5639-5647.
- Auriemma M, Luger T, Loser K, Amerio P, Tulli A (2009) The antinflammatory effect of alpha-msh in skin: a promise for new treatment strategies. Antiinflamm Antiallergy Agents Med Chem 8:14-21.
- Xue-Tao X, Xue-Qing M, Qin-Mei X, Wei-Ting L, Wen-Feng L, et al. (2016) Anti-inflammatory activity effect of 2-substituted-1,4,5,6- tetrahydrocyclopenta[b]pyrrole on TPA-induced skin inflammation in mice. Bio org Med Chem Lett 26:5334-5339.
- Xinrui Y, Yineng X, Peng L, Lijuan Z, Yuejiao Z, et al. (2018) Synthesis and evaluation of 1H-pyrrole-2,5-dione derivatives as cholesterol absorption inhibitors for suppressing the formation of foam cells and inflammatory response. Bio org Med Chem 26:1435-1447.
- Maddila S, Gorle S, Sampath C, Lavanya P (2016) Synthesis and anti-inflammatory activity of some new 1,3,4-thiadiazoles containing pyrazole and pyrrole nucleus. J Saudi Chem Soc 20:S306-S312.
- Claudio B, Giovanna P, Salvatore A, Giulio CP, Sara C, et al. (2013) A class of pyrrole derivatives endowed with analgesic/anti-inflammatory activity. Bio org Med Chem 21:3695-36701.
- Harrak Y, Rosell G, Daidone G, Plescia S, Schillaci D, et al. (2007) Synthesis and biological activity of new anti-inflammatory compounds containing the 1,4-benzodioxine and/or pyrrole system. Bio org Med Chem 15:4876-4890.
- Kumar PR, Raju S, Goud PS, Sailaja M, Sarma MR, et al. (2004) Synthesis and biological evaluation of thiophene [3,2-b] pyrrole derivatives as potential anti-inflammatory agents. Bioorg Med Chem 12:1221-1230.
- Shin JS, Noh YS, Yoo MS, Lee JY, Cho YW, et al. (2012) Anti-inflammatory and anti-arthritic effects of new synthetic 3-(4-hydroxyphenyl)-4-(4-thiomethoxyphenyl)-1H-pyrrole-2,5-dione. Bio org Med Chem Lett 22:2221-2225.
- Aleksandra R, Lukasz S, Andrzej K, Benita W, Katarzyna G, et al. (2019) COX-1/COX-2 inhibition activities and molecular docking study of newly designed and synthesized pyrrolo[3,4-c]pyrrole Mannich bases. Bioorg Med Chem 27:3918-3928.
- Wieslaw M, Aleksandra R, Magdalena JW, Barbara F, Malgorzata D, et al. (2011) Derivatives of pyrrolo[3,4-d]pyridazinone, a new class of analgesic agents. Eur J Med Chem 46:4992-4999.
- Malinka W, Kacmarz M, Redzicka A, Filipek B, Sapa J. (2005) Synthesis and analgesic action of N-(substituted-ethyl)pyrrole-3,4-dicarboximides. Farmaco 60:15-22.
- Marcin D, Florian J, Vittorio C, Severine CD, Maria W, et al. (2021) Neuropathic pain-alleviating activity of novel 5-HT6 receptor inverse agonists derived from 2-aryl-1H-pyrrole-3-carboxamide. Bio org Chem 115:1-12.
- Burgart YV, Agafonava NA, Shchegolkov EV, Borisevich SS, Khursan SL, et al. (2019) The competitive N1 -, N2 -, O- and C-methylation of 3-trifluoromethyl-1H-pyrazol-5-ol for synthesis of analgesic compounds. J Fluor Chem 218:1-10.
- Sergei NA, Evgeny KS, Igor AU, Elizaveta N, Oborina L, et al. (2021) Antibacterial activity of new silatrane pyrrole-2-carboxamide hybrid. Mendeleev Commun 31:204-206.
- Moorthy M, Kusha S, John HTL, Aharon G (2020) Antibacterial activities of microwave-assisted synthesized polypyrrole/chitosan and poly (pyrrole-N-(1-naphthyl) ethylenediamine) stimulated by C-dots. Carbohydr Polym 243:116474.
- Nazanin P (2021) Synthesis and characterization of a novel Pyrrole-2-carboxylic acid functionalized magnetic Fe3O4 as a nanocatalyst for synthesis and antibacterial activities of novel isoxazolo[4,3-e]indazole derivatives. J Mol Struct 1227:1-11.
- Poonam R, Anshu G, Singh RN (2022) Synthesis, spectral, structural and antimicrobial activities of Ethyl-4-{[-(1-(2-(4-nitrobenzoyl)hydrazono)ethyl]}-3,5-dimethyl-1H-pyrrole-2-carboxylatephenyl-1H-pyrrole-carboxamide entry inhibitors. J Mol Struct [Crossref]
- Francesca C, Dmitry SB, Young DK, Ranjith R, Anna MF, et al. (2018) Structure-based lead optimization to improve antiviral potency and ADMET properties of phenyl-1H-pyrrole-carboxamide entry inhibitors targeted to HIV-1 gp120. Eur J Med Chem 154:367-391.
- Edwards TG, Fisher C (2018) Antiviral activity of pyrrole-imidazole polyamides against SV40 and BK polyomaviruses. Antiviral Res 152:68-75.
- Terri GE, Kevin JK, Urszula S, Kam F, Michael H, et al. (2011) HPV episome levels are potently decreased by pyrrole–imidazole polyamides. Antiviral Res 91:177-86.
- Mosaad SM, Amira IS, Mohammed AK, Sameh HS (2016) Design, synthesis, assessment, and molecular docking of novel pyrrolopyrimidine (7-deazapurine) derivatives as non-nucleoside hepatitis C virus NS5B polymerase inhibitors. Bioorg Med Chem 24:2146-2157.
- Mosaad SM, Amira IS, Mohammed AK, Shahira N, Sameh HS (2019) Evaluation of novel pyrrolopyrimidine derivatives as antiviral against gastroenteric viral infections. Eur J Pharm Sci 127:102-114.
- Travis KW, Robert J, Michael KL, Adrian SR, Richard LM, et al. (2016) Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature 531:381-385.
- Dustin S, Hon CH, Edward D, Michael OC, Kwon C, et al. (2017) Discovery and Synthesis of a Phosphoramidate Prodrug of a Pyrrolo[2,1‑f ][triazin-4-amino] Adenine C‑Nucleoside (GS-5734) for the Treatment of Ebola and Emerging Viruses. J Med Chem 60:1648-1661.
- Mostafa MG, Fatma AR, Helmy IH, Hanan AY, Marwa GEG (2010) Synthesis of novel pyrrole and pyrrolo[2,3-d]pyrimidine derivatives bearing sulfonamide moiety for evaluation as anticancer and radiosensitizing agent. Bioorg Med Chem Lett 20:6316-6320.
- Debasish B, Sanghamitra M, Jose CG, John DS, Bimal KB (2012) Ultrasound-assisted bismuth nitrate-induced green synthesis of novel pyrrole derivatives and their biological evaluation as anticancer agents. Eur J Med Chem 50:209-215.
- Nishant KR, Rahul BS, Sangeeta VJ (2020) Potential 2,4-dimethyl-1H-pyrrole-3-carboxamide bearing benzimidazole template: Design, synthesis, in vitro anticancer and in silico ADME study. Bioorg Chem 97:1-8.
- Michela P, Hongliang S, Ruoli B, Antonio C, Marianna B, et al. (2021) Discovery of pyrrole derivatives for the treatment of glioblastoma and chronic myeloid leukemia. Eur J Med Chem 221:1-18.
- Alejandro C, Ramon B, Jose AD, Claus TC, Lars KR, et al. (2020) Debenzylative Cycloetherification as a Synthetic Tool in the Diastereoselective Synthesis of 3,6-Disubstituted Hexahydro‑2H‑furo[3,2‑b]pyrroles, PDE1 Enzyme Inhibitors with an Antiproliferative Effect on Melanoma Cells. J Org Chem 85.
- Rodica O, Mihaela B, Mihaela B, Mina R, Lavinia LR, et al. (2021) Antiproliferative and antibacterial properties of biocompatible copper(II) complexes bearing chelating N,N-heterocycle ligands and potential mechanisms of action. BioMetals 34:1155-1172.
- Yale HL, Beer B, Pluscec J, Spitzmiller ER (1970) Novel polycyclic heterocycles. Derivatives of 5,11-dihydrodibenz[b,e][1,4]oxazepine and 5,11-dihydrodibenzo[b,3][1,4]thiazepine. J Med Chem 13:713-722.
- Jin Z, Bohong L, Yinggui D, Xiaowen G, Jialan Z, et al. (2018) Design and synthesis of imidazo-fused heterocycles derivatives and their anti-tumor activity against breast cancer in mice. Nan Fang Yi Ke Da Xue Xue Bao 38:1052-1060.
- Lu M, Dongyu M, Jung JL, Tao L, Yu C, et al. (2021) Synthesis and biological evaluation of heterocyclic ring-fused dammarane-type ginsenoside derivatives as potential anti-tumor agents. Bioorg Chem 116:1-11.
- Richard KA, Patrick OS, Dorcas OS, Ivan AM (2021) Synthetic and Naturally Occurring Heterocyclic Anticancer Compounds with Multiple Biological Targets. Molecules 26.
- Thiault GA, Guen YL, Boucherle A, Walrant P (1984) N-aryl pyrrole derivatives with analgesic and anti-inflammatory activity 2. Pharmacologic modulation of the 1-arylpyrrole model. Farmaco Sci 39:765-80.
- Dovile M, Birute G, Rita V, Giedrius V, Birute SB, et al. (2021) Synthesis of Novel Thiazole Derivatives Bearing β-Amino Acid and Aromatic Moieties as Promising Scaffolds for the Development of New Antibacterial and Antifungal Candidates Targeting Multidrug-Resistant Pathogens. Molecules 27:74.
- Pawar K (2022) Recent Advances in KPV Peptide Delivery. J Pharm Drug Deliv Res 11:1-4.
- Brindusa T, Alina P, Radu T, Cristina N, Ioana I, et al. (2013) New anti-inflammatory thiazolyl-carbonyl-thiosemicarbazides and thiazolyl-azoles with antioxidant properties as potential iNOS inhibitors. Arch Pharm Res 36:702-714.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi